Abstract
Since 2006, 3 new disease syndromes have emerged in farmed saltwater crocodiles (Crocodylus porosus) in the Northern Territory of Australia. We describe the syndromes through a retrospective study of laboratory findings from 187 diagnostic cases submitted to Berrimah Veterinary Laboratories between 2005 and 2014. The first syndrome was characterized by conjunctivitis and/or pharyngitis (CP), primarily in hatchlings. Herpesviruses were isolated in primary crocodile cell culture, or were detected by PCR directly from conjunctiva or pharyngeal tissue, in 21 of 39 cases of CP (54%), compared with 9 of 64 crocodiles without the syndrome (14%, p < 0.0001). Chlamydiaceae were detected by PCR in conjunctiva or pharyngeal tissue of 55% of 29 CP cases tested, and of these, 81% also contained herpesvirus. The second syndrome occurred in juveniles and growers exhibiting poor growth, and was characterized histologically by systemic lymphoid proliferation and nonsuppurative encephalitis (SLPE). Herpesviruses were isolated or detected by PCR from at least 1 internal organ in 31 of 33 SLPE cases (94%) compared with 5 of 95 crocodiles without the syndrome (5%, p < 0.0001). The third syndrome, characterized by multifocal lymphohistiocytic infiltration of the dermis (LNS), occurred in 6 harvest-sized crocodiles. Herpesviruses were isolated from at least 1 skin lesion in 4 of these 6 cases. Although our study revealed strong associations between herpesvirus and the CP and SLPE syndromes, the precise nature of the role of herpesvirus, along with the pathogenesis and epidemiology of the syndromes, requires further investigation.
Introduction
Crocodile farming is a growing industry in Australia, producing high-quality skins for the luxury leather market. Australian farmed crocodiles are almost exclusively saltwater crocodiles (Crocodylus porosus) obtained from either captive breeding or regulated sustainable egg harvesting from the endemic wild population. Crocodile farming is intensive as most farms contain thousands of crocodiles, incubating eggs and raising crocodiles from hatching to harvest at 2–4 years of age (Foster M. Emerging animal and plant industries: their value to Australia. Barton, ACT: Australian Government Rural Industries Research and Development Corporation Publication No. 14/069, 2014).
Three new disease syndromes have emerged in recent years among farmed crocodiles in the Northern Territory of Australia. The first syndrome to emerge was conjunctivitis and/or pharyngitis (CP). Chlamydiaceae bacteria have been noted in the literature as being associated with conjunctivitis in various Crocodylus spp.,15–17,32 and CP in saltwater crocodiles was initially attributed to this agent. However, the role of the bacterium became unclear when a survey on the prevalence of Chlamydiaceae in live saltwater crocodiles revealed a poor association between the syndrome and presence of the bacteria (Jerrett I, et al. Chlamydial infection in farmed crocodiles [final report to funding body]. Barton, ACT: Australian Government Rural Industries Research and Development Corporation Publication No. 08/188, 2008). The second syndrome to emerge was systemic lymphoid proliferation with nonsuppurative encephalitis (SLPE), and the third was lymphonodular skin lesions (LNS). Two novel herpesviruses, crocodyline herpesvirus 1 and crocodyline herpesvirus 2 (CrHV-1 and -2, respectively), were identified in 8 saltwater crocodiles affected with these 3 syndromes. Bayesian phylogenetic analysis of the amino acid sequences of a segment of the polymerase gene revealed the 2 viruses to be distinct and clustered within the subfamily Alphaherpesvirinae. CrHV-1 was isolated from conjunctiva of 2 crocodiles with CP syndrome, whereas CrHV-2 was isolated from conjunctiva of 1 crocodile with CP syndrome, internal organs from 3 crocodiles with SLPE syndrome, skin from 1 crocodile with LNS syndrome, and skin from 1 crocodile with ulcerative skin disease. 18
Herpesviruses are enveloped, large DNA viruses with intranuclear replication and generally very high host fidelity. 28 Transmission typically occurs following close contact, particularly mucosal, with an infected individual but it can also be via aerosol or vertical. Herpesviruses exhibit latency, commonly persisting in lymphocytes and neurons. Infection is considered to be life-long and is often subclinical. Clinical disease is typically associated with very young or otherwise immunocompromised individuals, concurrent disease, infection in an alternate host, or stressful conditions. 23
Diseases caused by herpesviruses in mammals and birds are often characterized by mucosal inflammation and ulceration (e.g., infectious rhinotracheitis and pustular vulvovaginitis caused by Bovine herpesvirus 1,6,23 rhinotracheitis and conjunctivitis caused by Felid herpesvirus 1, 10 and laryngotracheitis caused by Gallid herpesvirus 1 12 ). Herpesvirus infection may also be associated with lymphoproliferative disease (e.g., malignant catarrhal fever caused by alcephaline herpesvirus 1 or Ovine herpesvirus 2 39 and Marek’s disease caused by Gallid herpesvirus 2 33 ).
In reptiles, disease associated with herpesvirus infection includes chronic proliferative stomatitis in lizards42,43; acute and necrotizing, or chronic and proliferative, stomatitis and rhinitis in tortoises 27 ; conjunctivitis, necrotizing, or proliferative pharyngitis and tracheitis and proliferative bronchopneumonia in sea turtles 20 ; and papular to ulcerative skin lesions in sea turtles.30,36 Reports of herpesviral disease in crocodilians are limited. Mild skin disease associated with herpesvirus-like particles was reported in a 6-month-old saltwater crocodile that had been farm-hatched in the Northern Territory of Australia, and transported to a facility in Victoria, Australia for stress research. 25 There is also a report associating cloacal lymphoid nodules in juvenile farmed American alligators (Alligator mississippiensis) with herpesvirus infection. 11 However, the herpesvirus was subsequently determined to have likely been a contaminant (GenBank accession AY913769.1).
We provide a retrospective study analyzing the results of diagnostic laboratory testing in saltwater crocodiles with CP, SLPE, and LNS syndromes, including detailed descriptions of the pathology. We also establish the strength of association between herpesviruses and the syndromes by comparing virus isolation and generic herpesvirus PCR results from crocodiles with each syndrome to crocodiles not affected with any of the syndromes.
Materials and methods
Crocodile farms and animals
Saltwater crocodiles submitted for the study originated from 4 farms within 60 km of Darwin, in the Northern Territory of Australia. Crocodiles were reared in pens containing relatively small numbers of animals (<20) or hundreds of animals. Grading of pens occurred every few months to ensure each pen continued to contain similarly sized animals. Pens contained water maintained at ~32°C for hatchlings and juveniles or ambient temperatures for older animals, and at sufficient depth for crocodiles to submerge, with dry areas to allow crocodiles to leave the water. Pens were in sheds that were either only partly enclosed, or had windows that could be opened to provide ventilation. Crocodiles were fed 3–7 times per week. Hatchlings were fed finely minced red meat (horse or buffalo) supplemented with a vitamin and mineral mix, and larger crocodiles were fed whole or minced chicken heads, without any supplementation. The water in pens was emptied and replaced with fresh water either from a dam or bore on the day following feeding.
Case material for the study consisted of 187 crocodiles that were submitted to Berrimah Veterinary Laboratories for postmortem examination between 2005 and 2014. Crocodiles were either submitted dead, after being found dead at a farm, or submitted alive if they were moribund, weak, exhibited poor growth, had unusual lesions, or had findings suggestive of the syndromes described herein. Crocodiles submitted live were euthanized with 80 mg/kg of intravenous pentobarbital. a Total length (snout to tail tip) was measured for each case. Exact ages were not known, but animals were categorized as hatchling (1–6 months of age, mean total length 39.5 cm, standard error [SE] 0.9), juvenile (6–12 months of age, mean total length 52.3 cm, SE 2.0), grower (12–24 months of age, mean total length 93.6 cm, SE 8.6), or harvest-size (24–48 months of age, mean total length 150.3 cm, SE 12.8).
Pathology, histology, and syndrome classification
A full gross postmortem examination was performed on each crocodile in the study, with the exception of 2 crocodiles with LNS in which only the skin was examined. Histologic examination of selected tissues (based on those that were deemed necessary for a case diagnosis) was also performed in most cases of CP and all cases of SLPE and LNS. Tissues collected for histologic examination were fixed in 10% neutral buffered formalin, processed using standard techniques, and stained with hematoxylin and eosin. To enhance visualization of gram-negative and -positive bacteria, acid-fast bacteria, and fungal elements, Gram–Twort, Ziehl–Neelsen, and periodic acid–Schiff stains, 4 respectively, were used to examine lesions in several cases from each of the syndromes
Based on gross and/or histopathological findings, crocodiles were categorized as being affected with 1 of the 3 syndromes or as being a nonsyndrome control animal. Crocodiles demonstrating grossly reddened and swollen conjunctivae with or without fibrinonecrotic exudate and/or pharyngeal fibrinonecrotic exudate were diagnosed as having CP syndrome (39 cases). SLPE syndrome was diagnosed based on the histological findings of systemic lymphoid proliferation and nonsuppurative encephalitis (33 cases). If a case met the inclusion criteria for both SLPE and CP, it was placed in the SLPE category. LNS syndrome was characterized by multiple grossly evident dermal nodules underlying intact epidermis, confirmed histologically as being comprised of primarily lymphocytes and macrophages (6 cases). The remaining 109 cases in the study did not meet the criteria for any of the syndromes, and in most cases were diagnosed with another disease. Consequently, these animals served as nonsyndrome control animals.
Bacteriology and PCR screening for Chlamydiaceae
Based on gross and/or histopathological findings, ancillary testing was performed to diagnose the cause of the morbidity or mortality. Aerobic bacterial culture of liver, spleen, kidney, and/or brain was used to screen for bacterial septicemia or meningitis, which are commonly associated with morbidity or mortality in farmed crocodiles.5,21 Aerobic bacterial culture and speciation were performed using standard veterinary bacteriology phenotypic and biochemical techniques. Briefly, samples were homogenized and plated onto sheep blood agar b and MacConkey agarb and incubated at 35°C for 48 h. The bacterial isolates were initially characterized by gram staining, colony morphology, and oxidase and catalase tests. The appropriate commercial kits were then used to speciate the isolates.c,d Aerobic bacterial culture of organs was performed in 13 CP cases, 25 SLPE cases, and 78 nonsyndrome control cases. Aerobic bacterial culture was performed on conjunctival swabs in 8 CP cases and 9 samples of affected skin from 4 LNS cases. Testing for Chlamydiaceae was performed on conjunctiva and/or pharyngeal tissue or swabs in 29 CP cases using PCR targeting the 16S gene of Chlamydiaceae as previously described. 8
Virus isolation and electron microscopy of isolates
Isolation of cytopathic viruses was attempted from conjunctiva and/or pharyngeal swabs or tissue, at least 1 internal tissue (liver, brain, spleen, kidney, lung, or thymus), or skin. For virus isolation, primary cell lines were derived from saltwater crocodile hatchlings less than 24 h old. For each sample, virus isolation was attempted on 2 cell lines: 1 of epithelial morphology (derived from kidney) and 1 of fibroblast morphology (derived from liver, subcutaneous connective tissue, heart, or trachea). 18 Cultures showing cytopathic effects, seen as loss of confluence of the cell monolayer, rounding up of cells, and, in some cases, syncytia formation, were categorized as virus isolation positive (VI+) and were stored at −70°C for later analyses. If no cytopathic effect was observed at any time during 2 passages, further testing was not pursued and the culture was considered negative for cytopathic viruses (VI–).
Initial investigations involved electron microscopy of VI+ cultures. After the media were removed, cells were fixed in 2.5% glutaraldehyde in phosphate-buffered saline and then processed. Ultrathin sections were double-stained with uranyl acetate and lead citrate, as previously described, except Sorenson phosphate buffer was used (300 mOsm/kg, pH 7.2). 41 VI+ cell cultures were analyzed from 11 cases: conjunctiva and/or pharynx from 6 CP cases; liver or kidney from 3 SLPE cases; skin from 1 LNS case; and pharynx from 1 nonsyndrome control case.
Herpesvirus PCR
Based on the preliminary findings of electron microscopy, either tissue homogenates or frozen-thawed cell culture homogenates from VI+ cultures were tested for herpesvirus using a pan-Herpesviridae nested PCR. 40 We determined that this PCR was suitable for screening samples for crocodyline herpesviruses by sequencing the PCR product from 8 isolates (representing 3 cases of CP, 3 cases of SLPE, 1 case of LNS, and 1 case of ulcerative skin disease) and confirming that every pan-Herpesviridae PCR product that we sequenced was a crocodyline herpesvirus. 18 DNA was extracted from a 200-µL aliquot of each cell culture or tissue homogenate using a commercial kit according to the manufacturer’s instructions. e Bovine herpesvirus 2 was used as a positive control, and tissue culture medium from uninfected crocodile cell culture was used as a negative control. Virus isolation with subsequent detection of herpesvirus by PCR in VI+ cultures was used to determine if a sample was herpesvirus positive or negative in 91 conjunctiva and/or pharyngeal samples, 140 internal organ samples, and 18 skin samples. PCR performed directly on tissue homogenates was used to detect herpesvirus in 30 conjunctiva and/or pharyngeal samples, 9 internal organ samples, and 4 skin samples. Uninfected cell lines were tested by PCR to ensure the cell lines were not contaminated with herpesvirus and were negative.
Statistical analyses
If either the conjunctiva or pharyngeal swab, or any internal organ, was positive for herpesvirus, the animal was considered to be infected with herpesvirus. Chi-square tests were used to test for statistical differences in herpesvirus detection rates between cases with the disease syndromes and nonsyndrome control animals. For the internal organ testing, the numbers of internal organs tested varied between syndrome animals and the nonsyndrome control animals. To remove any bias this may have caused, the number of organs tested in a case was introduced as a covariate in a multiple logistic regression analysis. To remove the potential bias of some internal organs being more likely than others to be herpesvirus positive, statistics were also compiled for liver samples only, as this was the most commonly tested internal organ. If the chi-square test revealed a significant difference between groups, the odds ratio (OR, also known as the cross-products ratio) was calculated to compare the likelihood of a crocodile having a syndrome if it was herpesvirus positive versus herpesvirus negative, using nonsyndrome cases as the case control group. 24 If one of the values used for calculating the OR was 0, 0.5 was added to each of the 4 values in the calculation. In order to refine the comparisons, and control for any effect of age category on herpesvirus-positive rate, statistics as described above, were also calculated for the age groups that were most commonly affected by a syndrome: hatchlings for CP and juveniles and growers for SLPE. A chi-square test and OR were used to compare herpesvirus and Chlamydiaceae detection rates in samples that were tested for both.
Results
Animal information
CP syndrome was first recognized by crocodile farmers in mid-June 2006. On the most extensively affected farm, nearly 50% of more than 2,000 hatchlings were noted to have some degree of conjunctivitis and/or pharyngitis. Over the next 6 weeks, the farm lost an estimated 70% of their hatchlings, presumably mainly a result of starvation, because they were unable to see the food, or suffocation because of occlusion of the larynx with pharyngeal exudate. At the time of writing (2015), outbreaks of CP were still occurring in hatchlings approximately every 2–4 years. The 39 CP cases in this study were submitted from 2006 to 2013, and 27 of them were hatchlings (Table 1).
Summary of herpesvirus testing results by disease syndrome and for nonsyndrome control crocodiles.

Tests included virus isolation (VI) and subsequent confirmation of isolate by either herpesvirus PCR or electron microscopy, or herpesvirus PCR directly on tissue. Virus isolates were identified by cytopathic effect in primary crocodile cell lines. + = positive.
Statistical comparison was performed between a disease syndrome category and the nonsyndrome controls category; p = level of statistical significance (significant comparisons in boldface), χ2 = chi-squared statistic. Odds ratio (OR) was calculated for statistically significant comparisons. For internal tissue statistical comparisons, number of internal tissues tested by VI for a case was entered as a covariate in multiple logistic regression.
Liver tested by VI and subsequent identification of isolate by herpesvirus PCR.
The first cases of SLPE were submitted for examination in October 2009. The syndrome was recognized only on 1 farm, mainly in association with outbreaks in 2 pens. The first outbreak was in a large pen composed of several smaller interconnected pens, in all containing ~1,400 juvenile crocodiles. Crocodiles in these pens had ill thrift and poor growth, and farmers reported a “moderately increased” mortality rate compared to similarly aged, unaffected crocodiles. Eighteen of the 33 cases of SLPE described in this study (Table 1) were crocodiles that were either from this pen or had been in contact with crocodiles from this pen. Of these 18 SLPE cases, 13 were submitted live in 2 groups for diagnostic postmortem examinations: 8 crocodiles in March 2010 and 5 crocodiles in August 2010. The 5 crocodiles in the latter group were judged by farmers to be clinically normal apart from exhibiting poor growth. Nine of the 33 SLPE cases were from a second pen outbreak that occurred in November 2013, involving a pen of 48 crocodiles. None of these animals died spontaneously, as they were in good body condition but exhibited poor growth. The majority had mildly reddened conjunctivae. One live crocodile with reddened conjunctivae from this pen was initially submitted for diagnostic postmortem examination, after which 8 more were randomly selected from the pen for the same purpose.
For the LNS syndrome, 6 harvest-sized crocodiles were submitted to the laboratory for assessment: 4 in 2009 and 2 in 2010 (Table 1). Crocodiles with this syndrome were generally in good body condition, and lesions were detected only by close inspection of the skin prior to harvest.
There were 109 other cases from the farms in this study that were determined, by gross and histologic examination, to not be affected with any of the syndromes. These crocodiles were categorized as nonsyndrome control cases (Table 1). The majority of these crocodiles were hatchlings or juveniles and were routine diagnostic submissions to the laboratory during the same time interval as the crocodiles affected by the 3 syndromes described in our report. Virus isolation and/or herpesvirus PCR was performed on these individuals for survey and comparative purposes. Forty-two of these 109 cases were diagnosed with some form of bacterial infection involving (in descending order of frequency) septicemia, meningitis, wound infection, or yolk sacculitis. Of the remaining animals, a variety of diagnoses were made, including runts (18 animals; described previously), 34 cause of death not apparent (16), hepatic chlamydiosis (5), spinal trauma (4), ulcerative or erosive skin disease (4), and single cases of other conditions: systemic coccidiosis, drowning, irritant eye lesions, metabolic bone disease, and nephrosis. The remaining 15 of these 109 animals were in good health and had been euthanized for unrelated research projects from which stored samples were scavenged for this investigation. Regarding the ulcerative or erosive skin disease nonsyndrome control cases noted above, 2 of the crocodiles were growers and 2 were harvest-sized. The skin lesions in the 4 cases did not fit the inclusion criteria for LNS syndrome and were sufficiently grossly and histologically distinct from each other that they could not be combined into a separate syndrome.
Gross pathology and histopathology
CP syndrome
Gross autopsy findings in CP syndrome were usually limited to the eyes, pharynx, and larynx. Eye lesions were typically bilateral. In relatively mild cases, there was thickening, edema, and reddening of the nictitating membrane and palpebral conjunctiva associated with scant mucopurulent exudate (Fig. 1A). In more severe cases, the exudate was copious, fibrinocaseous, and often adherent to the conjunctiva (Fig. 1B). In most cases, there was also concurrent mild to marked opacity of the cornea, and in the most severe cases, rupture of the cornea and copious, fibrinocaseous exudate filling the ocular chambers. Multifocal to regionally extensive mucosal erosion or ulceration with associated scant to abundant lightly adherent fibrinocaseous exudate were present in the pharynx and/or larynx. The pharynx at the junction of the base of the tongue and the gular flap (Fig. 1A), and surrounding the larynx (Fig. 1C), were frequently the most severely affected regions.
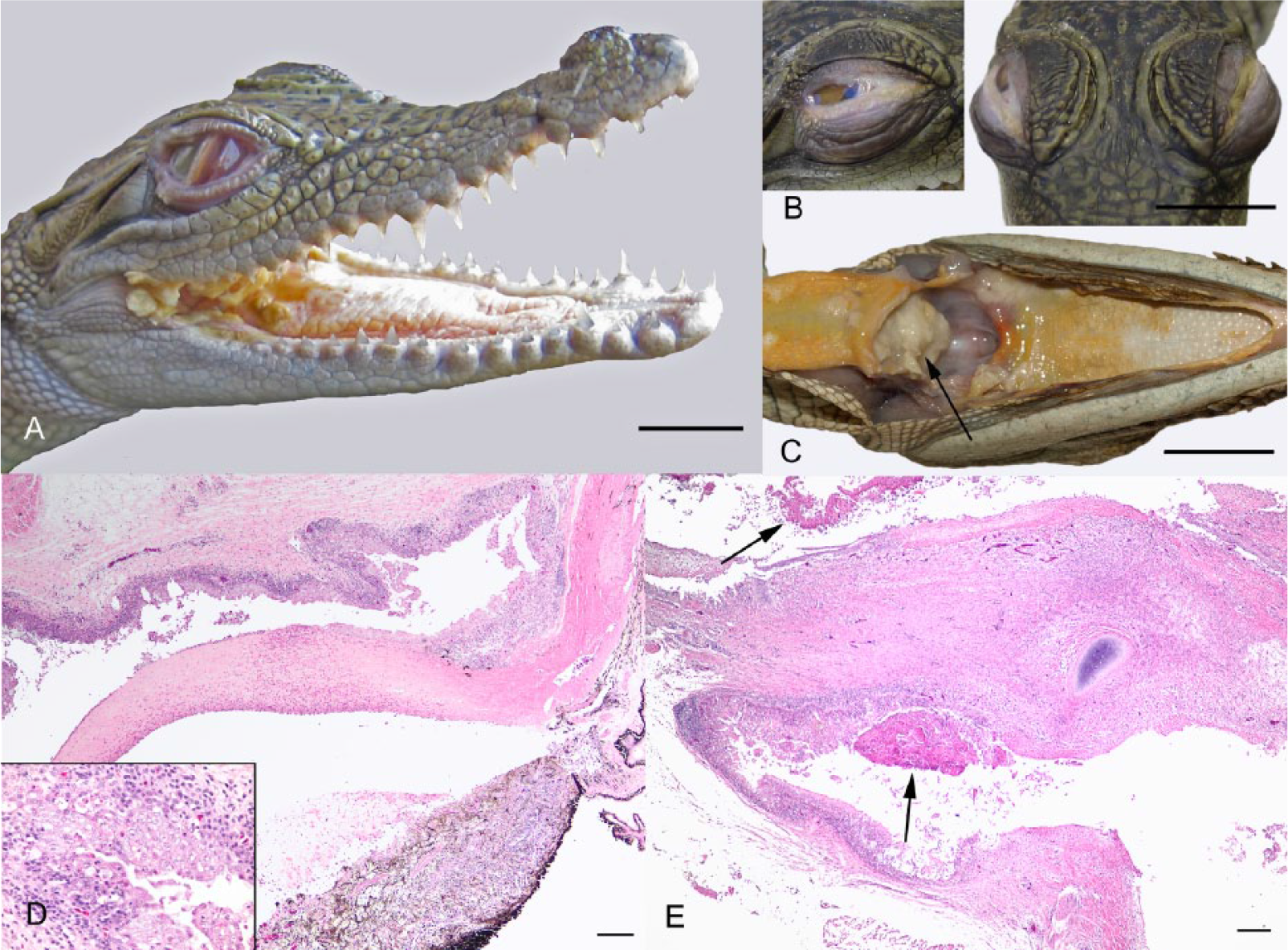
Typical gross pathology and histology of conjunctivitis and/or pharyngitis (CP) syndrome in hatchling saltwater crocodiles (Crocodylus porosus).
Histologically, eye lesions varied in appearance depending on severity, and presumably chronicity. In relatively acute lesions, there was moderate to severe edema and necrosis of the conjunctival epithelium resulting in erosion or ulceration with associated mild to marked epithelial and submucosal heterophil and macrophage infiltration and overlying coagula composed of fibrin, heterophils, and necrotic cell debris. In more chronic lesions, or often intermingled with lesions containing the above described acute features, there were segments of moderate to marked conjunctival epithelial hypertrophy and hyperplasia, with mild to marked epithelial and submucosal lymphoplasmacytic and macrophage infiltration. The cornea varied from normal to edematous, with epithelial erosion or ulceration and epithelial and stromal heterophil and/or lymphocyte infiltration. In many affected eyes, there was mild to marked heterophil, macrophage, and/or lymphocyte infiltration of the iris (Fig. 1D). The pharyngeal and laryngeal mucosal epithelium exhibited similar lesions as the conjunctiva (Fig. 1E). In the mucosal epithelia of both the conjunctiva and pharynx, where the epithelium had not yet ulcerated, superficial epithelial cells sometimes appeared to have condensed, dark nuclei, or enlarged nuclei undergoing karyorrhexis (Fig. 1D, inset). However, convincing intranuclear bodies suggestive of viral inclusions were not observed. In severe conjunctival and pharyngeal lesions, there were frequently mixed gram-negative and -positive bacterial rods and cocci and occasionally fungal hyphae visible histologically in the necrotic superficial exudate, although none appeared to be infiltrating viable tissue.
SLPE syndrome
Gross postmortem findings were minimal and limited to affected crocodiles being in poor body condition with variable splenomegaly and/or pulmonary edema. Six crocodiles with this syndrome (3 juveniles and 3 growers) had mild gross lesions of conjunctivitis and/or pharyngitis as described above for the CP syndrome.
Histologically, SLPE was characterized by lymphohistiocytic infiltration of a wide variety of tissues, including the pulmonary septae (Fig. 2A), hepatic periportal regions, pancreatic interstitium (Fig. 2B), gastrointestinal submucosa, pericardium, epicardium, and in severe cases, the wall of large blood vessels (Fig. 2C). There was abundant proteinaceous fluid in pulmonary air spaces (Fig. 2A). In the spleen, perivascular lymphoid cuffs were large and confluent, and the red pulp was expanded by abundant macrophages, many having vacuolated cytoplasm containing variegated fine pale eosinophilic material (presumably phagocytized cell debris). The tonsils were frequently markedly active, with obliteration of the tonsillar epithelium by lymphocytes and solid dense sheets of lymphocytes and macrophages expanding the tonsillar folds. In the brain, there was diffuse mild to moderate gliosis and lymphohistiocytic infiltration surrounding parenchymal vessels and in the meninges and choroid plexus (Fig. 2D). Histology of the eyes was conducted on 25 crocodiles with SLPE syndrome, and all had a mild to moderate degree of chronic hyperplastic lymphocytic conjunctivitis, with most crocodiles for which the iris was examined also having mild to marked lymphoid infiltration of the iris. No gram-positive or -negative or acid-fast bacteria, fungal elements, or protozoa were appreciable within lymphohistiocytic infiltrates in any of the tissues.
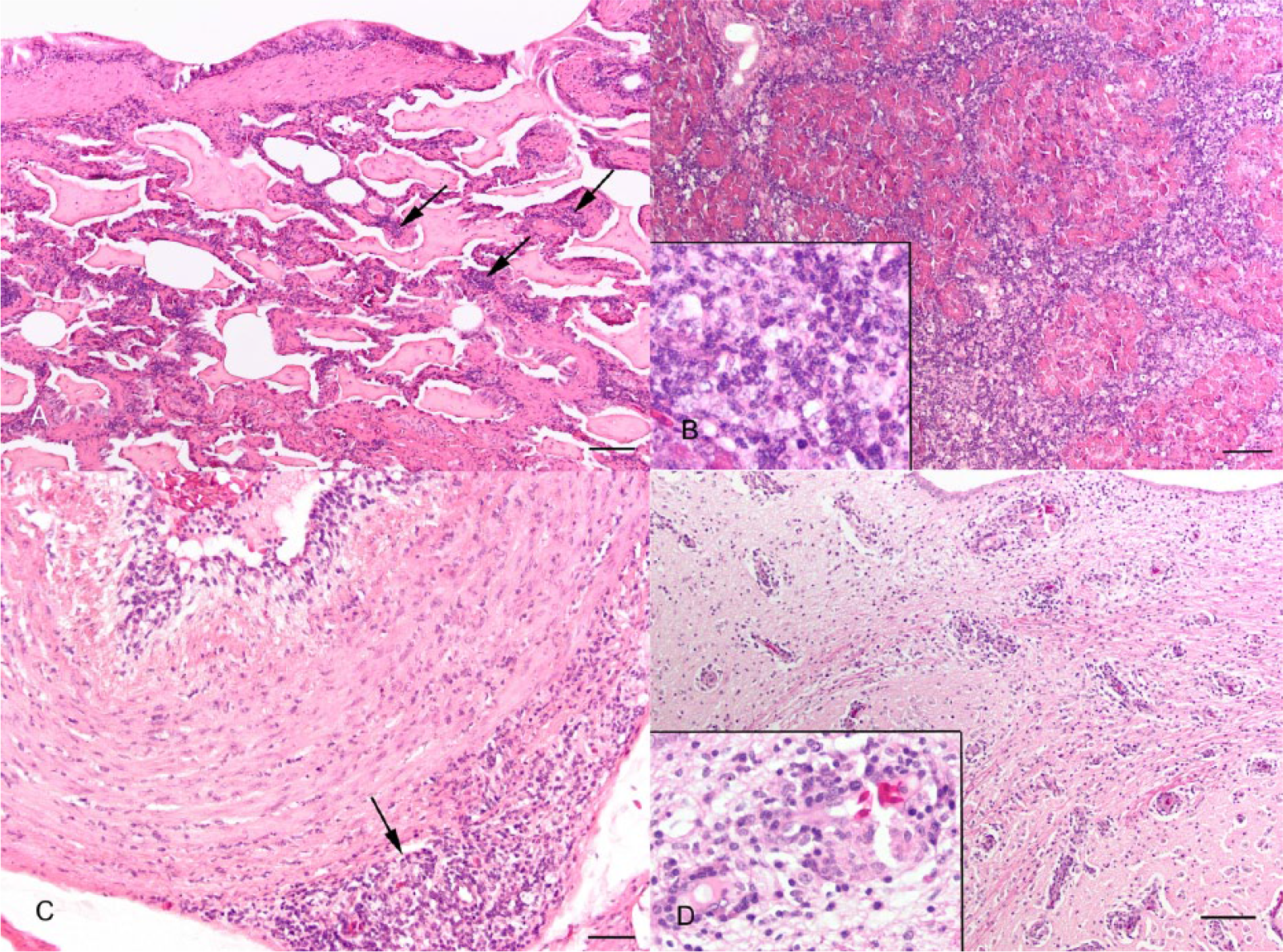
Typical histology of systemic lymphoid proliferation and encephalitis syndrome (SLPE) in juvenile saltwater crocodiles (Crocodylus porosus).
LNS syndrome
Grossly, skin lesions in this syndrome were characterized by multiple pale, soft, raised, well-delineated 4–20 mm foci most commonly involving the lateral abdominal scales. Lesions occurred either in the interscalar region, on part or all of a single scale, or a few contiguous scales (Fig. 3A). Lesions that involved only part of a scale were often centered on the integumentary sensory organ. Occasionally, large lesions were ulcerated and covered with caseous exudate. On cut section, lesions appeared as pale pink soft glistening tissue between the epidermis and deep dermal collagen (Fig. 3A, inset). In the 4 cases where a full autopsy was performed, there were similar soft raised foci involving the tongue or commissures of the mouth, at times with an ulcerated surface covered in caseous exudate. Tonsils were diffusely enlarged with a fine multinodular appearance. Other gross lesions present in 2 cases were discrete soft white foci present in subepithelial tissue of the conjunctiva, multinodular swelling of the cloacal mucosa, and a few scattered discrete white soft 1–3 cm foci expanding the parenchyma of the myocardium, liver, or kidney.
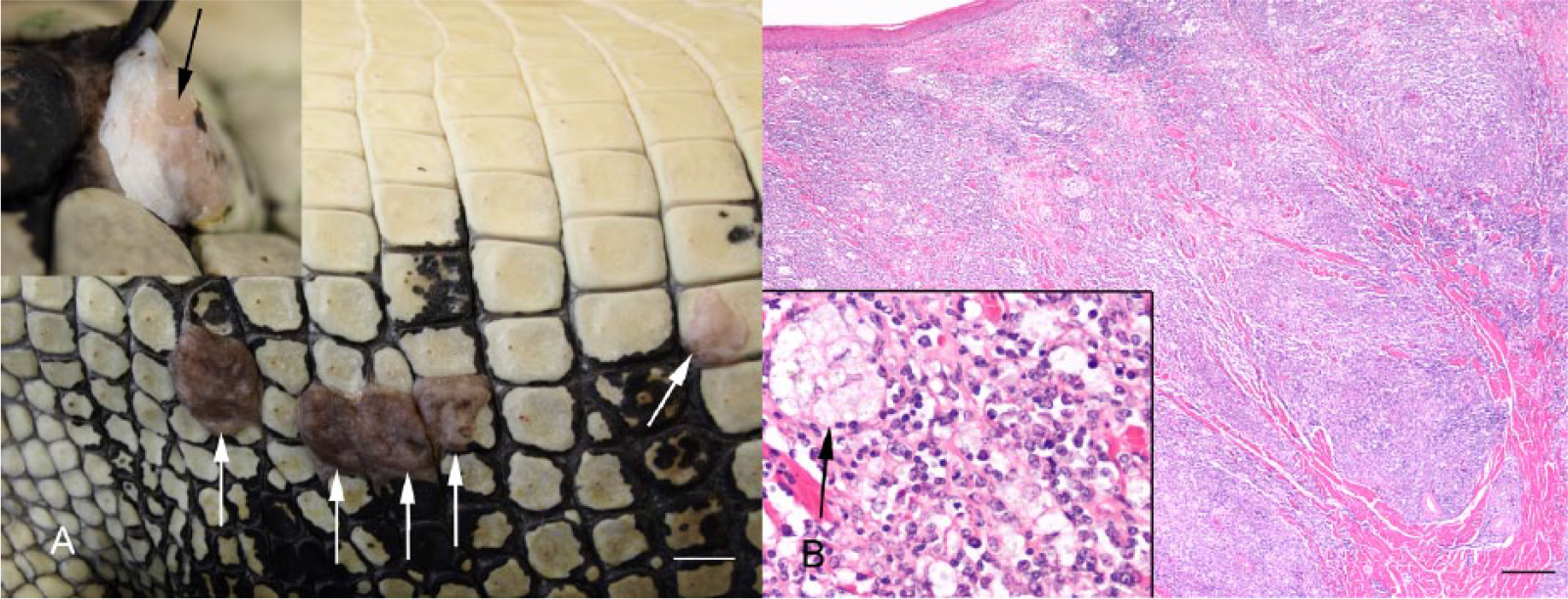
Typical gross pathology and histology of lymphonodular skin syndrome (LNS) in a harvest-sized saltwater crocodile (Crocodylus porosus).
Histologically, the main feature of all LNS skin lesions was expansion of the superficial and mid-dermis by large dense aggregates of lymphocytes of various sizes and macrophages, sometimes arranged in clusters and containing variegated fine pale eosinophilic material, which was presumed to be phagocytized cell debris (Fig. 3B). In many lesions, the overlying epidermis was normal, whereas in a few lesions it was mildly infiltrated with lymphocytes. In the lesions in which the overlying epidermis was eroded or ulcerated, there was heterophil infiltration and an overlying crust of necrotic cell debris with superficial mixed bacteria and/or fungi. Oral lesions were histologically similar to skin lesions. Tonsils were markedly hyperplastic and contained dense solid sheets of lymphocytes and intermingled macrophages expanding the tonsillar folds and infiltrating the epithelium. The discrete white foci in internal organs, noted grossly in 2 cases, were histologically characterized by dense lymphohistiocytic aggregates, similar to those described in the dermis. No gram-positive or -negative or acid-fast bacteria, fungal elements, or protozoa were appreciable within lymphohistiocytic infiltrates in the skin or other tissues.
Bacteriology
Aerobic bacterial culture of the conjunctival swabs from the 8 CP cases that were tested yielded no significant growth in 2 cases and light to moderate growth of a variety of bacteria in 6 cases. Predominant organisms were Streptococcus sp. (3 cases), Aeromonas hydrophila (1 case), Morganella morganii (1 case), and Enterobacter sp., Staphylococcus sp., and Corynebacterium sp. (all from 1 case). For 13 CP cases in which there was a suspicion of concurrent bacterial septicemia based on history (found dead with relatively mild CP lesions) and gross and/or histological findings, bacterial culture was positive in 7 cases from liver, kidney, and/or spleen. Bacteria isolated were Streptococcus sp. (1 case) and gram-negative environmental species: Providencia rettgeri (2 cases), Aeromonas sp. (2 cases), Edwardsiella tarda (1 case), and Salmonella sp. (1 case).
Aerobic bacterial culture of internal organs from 25 crocodiles with SLPE was negative in all but 3 cases in which P. rettgeri, Salmonella sp., Aeromonas sp., and M. morganii were isolated (the latter 2 organisms both from the same case).
Aerobic bacterial culture that was performed on 9 skin lesions from 4 of the LNS skin cases showed no growth for 4 lesions and light to moderate mixed bacterial growth in 5 lesions, with predominant isolates being Dermatophilus sp. (2 cases), Nocardia sp. (2 cases), Streptomyces sp. (1 case), and Streptococcus sp. (1 case).
Of the 78 nonsyndrome control cases in which aerobic bacterial culture of internal organs was performed, bacteria were isolated in 47 (60%). Providencia rettgeri was the most common bacteria cultured (19 cases), with less common isolates (<10 cases each), including Salmonella sp., M. morganii, Streptococcus agalactiae, Aeromonas sp., Providencia stuartii, and E. tarda.
Virus isolation, electron microscopy, and PCR for herpesvirus and Chlamydiaceae
Electron microscopy of VI+ cultures revealed herpesvirus-like virus in 10 of the 11 cultures tested. There were no virus structures observed in 1 VI+ culture from the liver of an SLPE case. Seven of these VI+ cultures examined by electron microscopy were also tested by herpesvirus PCR and all were positive, including the VI+ culture in which no virions were observed.
Herpesvirus was detected in the conjunctiva and/or pharynx in significantly more CP cases (54%) than nonsyndrome control cases (14%; Table 1). The OR between these 2 groups indicated that the rate of CP syndrome in crocodiles with herpesvirus-positive conjunctiva and/or pharynx was 7 times greater than the rate of CP syndrome in crocodiles with herpesvirus-negative conjunctiva and/or pharynx. The strength of this relationship increased substantially if the age grouping was made more comparable by just including hatchlings, as 59% of 27 hatchlings with CP were positive for herpesvirus in the conjunctiva and/or pharynx, whereas none of 32 nonsyndrome control hatchlings were positive (p < 0.0001, χ2 = 24.8, OR = 93). Of the 29 CP cases in which the conjunctiva and/or pharynx were tested for both Chlamydiaceae and herpesvirus, 18 (62%) were positive for herpesvirus. Thirteen of these 18, plus 3 other cases that were negative for herpesvirus, were positive for Chlamydiaceae (55%). The odds of detecting herpesvirus in a crocodile with Chlamydiaceae were 6.9 times greater than the odds of detecting it in one without (p < 0.05).
For the SLPE syndrome, results for herpesvirus detection in any internal organ revealed that the odds of having the syndrome were over 350 times greater for crocodiles with detectable herpesvirus infection than for crocodiles with no detected herpesvirus (Table 1). The relationship between SLPE and herpesvirus remained highly significant when potential bias resulting from which organ(s) were tested was controlled for by calculating statistics for virus isolation and subsequent PCR confirmation of VI+ cultures from only liver samples (Table 1). These 2 comparisons (any internal organ and liver only) between SLPE cases and nonsyndrome control cases remained significant when the comparisons were made more age comparable by including only juveniles and growers from the nonsyndrome controls (all SLPE cases were juveniles and growers). For herpesvirus detection in any internal organ, 11% of 38 nonsyndrome control juvenile and/or grower cases were positive (significantly less than SLPE cases, p < 0.0001, χ2 = 42.0, OR = 125), whereas for herpesvirus detection in liver, 8% of 25 nonsyndrome control juvenile and/or grower cases were positive (significantly less than SLPE cases, p < 0.0001, χ2 = 18.8, OR = 18). Of the 21 crocodiles with SLPE from the 2 pen outbreaks that were submitted live for euthanasia and diagnostic autopsy, all were positive for herpesvirus from at least 1 internal organ.
There was also a significant relationship between the SLPE syndrome and detection of herpesvirus in the conjunctiva and/or pharynx (Table 1). If the comparison of herpesvirus-positive rates of conjunctiva and/or pharynx between SLPE cases and nonsyndrome control cases was made more age comparable by just including juveniles and growers, the relationship was weaker but still significant (26% of 31 nonsyndrome control cases were positive, p = 0.02, χ2 = 5.1, OR = 4.0).
In the LNS syndrome, virus isolation was attempted from all 6 cases on a total of 13 skin lesions. Four out of the 6 LNS cases had at least 1 herpesvirus-positive skin lesion. Across all skin lesions, there were 5 herpesvirus-positive skin samples and 8 herpesvirus-negative samples. Therefore, any relationship between LNS skin lesions and herpesvirus infection is equivocal. In the 4 ulcerative or erosive skin cases in the nonsyndrome control cases, herpesvirus was detected in skin samples from all 4 cases.
Discussion
Diseases are having a significant impact on the number of crocodiles that can be produced in commercial operations in the Northern Territory in Australia. The purpose of our study was to retrospectively examine the results of diagnostic laboratory investigations related to 3 emerging disease syndromes in farmed saltwater crocodiles and to determine the strength of association of these syndromes with the presence of herpesviruses.
The association between the CP syndrome and herpesvirus was not strong, as the virus was not detected in conjunctival and/or pharyngeal samples from 46% of crocodiles with the syndrome. However, given that there was a highly significant difference in the detection rate between crocodiles with and without CP, it seems likely that herpesvirus at least plays some role in the syndrome, or is more likely to infect animals with the CP syndrome, and is not simply present incidentally in the conjunctiva or pharynx.
The conjunctiva and pharynx lesions in crocodiles with CP were characterized by acute erosive or ulcerative necrotizing lesions with fibrinocaseous exudate. These lesions were consistent with herpesvirus infection, being similar to conjunctivitis, stomatitis, or pharyngitis associated with herpesvirus infection in a wide variety of other species, including sea turtles, 20 tortoises, 27 lizards,42,43 chickens (infectious laryngotracheitis), 12 and cats.2,45
The lack of histologically visible inclusion bodies in the crocodile CP lesions was unexpected, given that inclusion bodies are frequently seen in herpesvirus mucosal infections in some species of reptiles.19,27,42 However, in mucosal herpesvirus infections in other species (e.g., monitor lizards, 43 chickens, 12 bovids, 6 and cats 45 ), typical herpesvirus inclusions have either not been appreciable, or are transient, appearing only in the early stages of infection. Further evidence for rapid loss of herpesvirus from lesions comes from molecular studies of Gallid herpesvirus 1 and Felid herpesvirus 1 infections in which, even when using highly sensitive PCR assays, herpesvirus was frequently undetectable within 30 days of infection.38,47 Therefore, the absence of herpesvirus inclusions in CP-affected crocodiles does not rule out a causal association of the virus infection and disease, but could represent either herpesvirus infection without inclusion formation, or transient presence of herpesvirus.
In addition to transience, the incomplete association between CP syndrome and herpesvirus could arise from multifactorial causation. For example, in infectious bovine keratoconjunctivitis, Moraxella bovis is considered the primary etiological agent, but there is frequently concurrent infection with other agents, including Bovine herpesvirus 1 and Mycoplasma sp. as well as potential exacerbation by environmental factors (dust, ultraviolet radiation) that may contribute to lesion severity. 45 Another example of multifactorial conjunctivitis is in cats, in which the condition may be caused by Felid herpesvirus 1 or Chlamydophila felis, with a questionable minor role for concurrent infection with Mycoplasma sp.29,31,45 In the investigation of the earliest cases of CP that occurred during 2006, pan-Mycoplasma PCR was performed on 6 conjunctiva and 4 pharyngeal samples, with only a single conjunctiva sample returning a positive result (Jerrett et al. 2008, as referenced in their introduction), suggesting that any Mycoplasma spp. involvement in CP cases in saltwater crocodiles is perhaps sporadic or transient. However, the possible involvement of Mycoplasma spp. in CP syndrome warrants further investigation.
There remains uncertainty in the role that Chlamydiaceae plays in the pathogenesis of CP syndrome. In our study, cases tested for both Chlamydiaceae and herpesvirus failed to determine which organism had a stronger association with the presence of lesions. Similar percentages of the cases were positive for each agent (62% for herpesvirus and 55% for Chlamydiaceae), and 45% of them were positive for both, with herpesvirus being significantly (6.9 times) more likely to be detected in crocodile conjunctiva and/or pharynx if Chlamydiaceae was also present. The significance of the combined presence of herpesvirus and Chlamydiaceae is unknown, but a similar situation has been noted in a herpesvirus investigation of Australian marsupials in which the presence of Chlamydia pecorum was a significant predictor of herpesvirus presence in 68 koalas (OR = 60). 37 In the literature, there is allusion to conjunctivitis being caused by Chlamydiaceae infection in Crocodylus niloticus in Africa,15,16 C. porosus in Papua New Guinea, 17 and recently, C. siamensis in Thailand. 32 However, in all of these investigations, the diagnosis appears to be based primarily on identification of Chlamydiaceae in internal organs, most commonly the liver, because unlike the CP cases detailed in our study, the infections also involved internal organs. In a previous epidemiological investigation sampling live crocodiles from the same farms as the present study, there was a very poor association between PCR presence of Chlamydiaceae in combined conjunctival and pharyngeal swabs and presence of CP syndrome (as defined by a crocodile having eyelid swelling or discharge and/or pharyngeal fibrin; Jerrett I, et al., 2008). In that study, in 224 hatchlings up to 4 months of age, 26% were positive for Chlamydiaceae but only 4% had CP syndrome. In 476 crocodiles 1–3 years of age, 18% were positive for Chlamydiaceae, whereas 8% had CP syndrome. Within this group of 476 crocodiles, there were 120 crocodiles from one farm that were all positive for Chlamydiaceae despite none of the tested crocodiles having CP syndrome. Conversely, on another farm, there were 120 crocodiles sampled in which none were positive for Chlamydiaceae but 29 had CP syndrome.
In our study, the preliminary results of electron microscopy focused our screening for herpesviruses. The possible involvement of other viruses was not explored. Examples of other viruses associated with conjunctivitis and/or pharyngitis are iridovirus in chelonians,35,44 paramyxoviruses in birds (e.g., Newcastle disease), 1 morbillivirus in mammals (e.g., canine distemper), 6 calicivirus in cats, 22 and adenoviruses in humans. 3 Future work should include efforts to examine the role that other viruses may play in CP syndrome. Unbiased molecular techniques, such as next-generation sequencing that are capable of detecting a number of infectious agents, including agents with fastidious culture requirements, should be considered.
We found a strong association between SLPE and the prevalence of herpesvirus in internal tissues, with SLPE-affected crocodiles having a substantially higher prevalence of infection (94%) compared to nonsyndrome control crocodiles (5%). The 100% prevalence of herpesvirus in SLPE-affected crocodiles that were euthanized for diagnostic postmortem examination from the 2 pen outbreaks suggests that chronic infection or latency may be common in some circumstances. Histologic examination of crocodiles with SLPE did not reveal lesions suggestive of a known disease, and aerobic bacterial culture of internal organs provided equivocal results.
The SLPE lesions are similar to those seen in other species with herpesvirus infections that have the ability to transform lymphocytes. Histologically, SLPE bears a strong similarity to Marek’s disease in chickens, which is caused by the cell-associated, lymphotropic alphaherpesvirus Gallid herpesvirus 2. Marek’s disease is characterized by lymphoproliferative disorders ranging from lymphoid infiltrations in various tissues, including the iris and other tissues of the eye, to lymphomatous masses in various internal organs. 33 Another herpesviral disease with some pathological similarities to SLPE is malignant catarrhal fever, caused by several ruminant gammaherpesviruses. In this lymphoproliferative disease, lymphohistiocytic infiltrates are widespread, including in the brain and blood vessels, and the virus causes panophthalmitis.23,39
Crocodiles with SLPE frequently had a mild degree of CP, and herpesvirus was detected in 58% of these tissues, suggesting that SLPE and CP syndromes may be related. Herpesvirus infections can manifest differently depending on various factors, including host age, immune competence, exposure circumstances, and latency status.10,13,23,33 Most of the CP cases were hatchlings, and all of the SLPE cases were in juveniles and growers. It is conceivable that CP and SLPE represent manifestations of herpesvirus infection at different ages or levels of immune competency. This supposition would benefit from more information regarding the specific identities of herpesviruses involved in the syndromes, a limitation of this study being that we did not go beyond generic PCR for herpesvirus identification. CrHV-1 was found in 2 of 3 cases of CP tested, and CrHV-2 was found in the third CP case tested and 3 of 3 SLPE cases tested. 18 Sequencing of greater numbers of herpesviruses from CP and SLPE cases is warranted to clarify the respective roles of these 2 crocodyline herpesviruses.
There are only 6 cases of LNS described in this study, and herpesvirus presence in affected skin was inconsistent. No alternate etiology for these lymphohistiocytic dermal nodules was evident histologically, and aerobic bacterial culture of intact and ulcerated lesions yielded several genera of bacteria, with none being convincing primary etiological agents. Herpesviruses have been associated with lymphocytic skin infiltrates in malignant catarrhal fever, 39 and in lymphocytic nodules associated with feather follicles in Marek’s disease. 33 However, given that the association of herpesvirus infection with LNS was weak, further investigation is required, including consideration of other viral etiologies. For example, retroviruses in birds and mammals cause lymphoproliferative syndromes.9,23 Endogenous retroviruses have been found in C. porosus in the Northern Territory of Australia 7 ; however, disease associations with retroviruses in C. porosus have yet to be investigated. In American alligators, proliferative lymphohistiocytic cutaneous lesions have been associated with West Nile virus infection. 26
In addition to the LNS cases, there were 4 cases in the nonsyndrome control cases that had erosive and/or ulcerative skin lesions. Although a detailed description of these cases was beyond the scope of this study, they bear mention because herpesvirus was detected in skin from all cases. Herpesvirus infection is capable of causing ulcerative or necrotizing skin lesions in several species, including turtles, 30 cats, 14 and wallabies. 46 Notably, the only published finding of a herpesvirus in a crocodilian relates to relatively mild, erosive skin lesions in a young crocodile from the Northern Territory of Australia that was involved in a stress experiment and in which herpesvirus-like particles were detected in superficial keratinocytes using electron microscopy. 25
Our study provides evidence of widespread herpesvirus infection in farmed populations of a crocodilian species and presents data correlating the infection with emerging disease syndromes on the farms. The associations at this stage are far from being established as causal. Further research should attempt to clarify the temporal and spatial association of viral presence with lesions using immunohistochemistry or in situ hybridization, and to experimentally reproduce the disease syndromes with transmission studies. If a causal relationship can be established between the virus and 1 or more of the syndromes, further epidemiological research to establish prevalence and latency should be pursued to determine how common these viruses may be in farmed saltwater crocodiles in Australia, to detect any insidious effects they may have on health (and therefore growth rates and skin quality), and to clarify any relationships among the syndromes. Most important to the farming industry, preventative measures would need to be established, including how to prevent exposure and transmission or any triggers that may exist for clinical expression of disease. Finally, the risk of these viruses to wild populations should be established.
Footnotes
Acknowledgements
We thank all the staff at the crocodile farms and Berrimah Veterinary Laboratories for assistance with this study. The electron microscopy was performed by Dr. Alex Hyatt, at the Australian Animal Health Laboratories, Geelong, Victoria, Australia. We thank the reviewers for their thorough editing and constructive comments regarding an earlier version of this manuscript.
Authors’ contributions
CM Shilton contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. IV Jerrett contributed to conception and design of the study; contributed to acquisition of data; and critically revised the manuscript. S Davis contributed to conception and design of the study; contributed to acquisition and interpretation of data; and critically revised the manuscript. S Walsh and S Benedict contributed to acquisition and interpretation of data, and critically revised the manuscript. SR Isberg, GJW Webb, and C Manolis contributed to acquisition of data, and critically revised the manuscript. TH Hyndman and D Phalen contributed to interpretation of data, and critically revised the manuscript. GP Brown contributed to analysis of data, and critically revised the manuscript. L Melville contributed to conception and design of the study, and critically revised the manuscript. All authors gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Lethabarb euthanasia injection (325 mg/mL), Virbac Animal Health, Milperra, New South Wales, Australia.
b.
Oxoid Australia, Thebarton, South Australia, Australia.
c.
Api 20 Strep and api Coryne, bioMérieux, Marcy-l’Etoile, France.
d.
Microbact gram-negative identification system, Oxoid Ltd., Basingstoke, Hants, United Kingdom.
e.
MagMAX viral RNA isolation kit, Ambion, Austin, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by the Australian Government Rural Industries Research and Development Corporation project no. PRJ-002461, and results summarized in a non–peer-reviewed publicly available report for the funding body.
