Abstract
Giardia duodenalis is considered a potentially zoonotic protozoan. Some animal species, including infected dogs, may play an important role in the spread of Giardia cysts through environmental contamination with their feces. In the present study, a commercial enzyme-linked immunosorbent assay (ELISA) was used to examine 143 samples of dog feces collected in urban areas as an indicator of the risk of field contamination. Using a Bayesian statistical approach, the ELISA showed a sensitivity of 88.9% and a specificity of 95.8% with positive and negative predictive values of 89.6% and 95.4%, respectively. The test affords the advantage of rapid processing of fecal samples without a complex technical structure and extensive costly labor. Moreover, the present results show that the assay provides public health veterinarians with a practical tool that can be used in screening programs, as a valid alternative or as an adjunct to other tests, in order to assess the biological risk of exposure to G. duodenalis cysts from dogs in human settlements. However, the test may not be completely accurate for human health risk evaluation, as it does not discriminate between zoonotic and non-zoonotic isolates.
Giardia duodenalis (syn. Giardia intestinalis and Giardia lamblia) is an intestinal flagellate infecting human beings and a wide range of mammals, including dogs. Currently, 8 distinct assemblages or genotypes (A–H) have been identified within this protozoan species. 11 In particular, dogs can be infected by host-specific assemblages C and D or zoonotic assemblages A and B.6,11 Infective cysts are present in the feces of infected hosts, and transmission occurs by accidental ingestion of Giardia cysts contaminating soil, coat, food, and mostly water. 11 Giardia duodenalis is one of the most common enteric parasites of dogs worldwide. 26 Because genetically identical, potentially zoonotic genotypes (predominantly assemblage A) may exist in human beings and dogs sharing the same environment, recent studies have focused on the role of dogs as a source of zoonotic transmission of G. duodenalis in human communities.20,22 The sampling of dog stools found in the environment could therefore be convenient and serve for screening studies in human settlements. 25 For this purpose, knowledge of appropriate tests for programs dealing with the risk of environmental contamination with G. duodenalis is important, as the effectiveness of these programs depends on the sensitivity (Se) and the specificity (Sp) of the tests used to identify contaminated dog feces in the environment. While Se and Sp are fixed test properties, they can be influenced by the prevalence of G. duodenalis and this influence thus affects test accuracy. 19
In veterinary medicine, the fecal flotation method with zinc sulfate is regarded as the best choice for the microscopic identification of Giardia cysts, with the test showing a Sp of 100%. 28 Due to the intermittent nature of cyst shedding,16,28 the Se of this procedure is 70% when a single sample is evaluated 16 and increases to 95% if at least 3 fecal specimens are examined within 5 days.16,28 Unfortunately, the possibility of multiple sampling from the same subject is often impractical and almost impossible when dog feces found in the environment are being tested.
Other diagnostic techniques have been developed for diagnosis of canine giardiasis in a single test and include identification of cysts by a direct fluorescent antibody test (DFAT), detection of coproantigens by traditional enzyme-linked immunosorbent assay (ELISA), and DNA amplification by polymerase chain reaction (PCR)-based methods. 28 However, such methods are costly and require sophisticated and expensive equipment as well as trained personnel. Therefore, the tests are best suited for well-equipped diagnostic centers. In frontline veterinary clinical laboratories, ELISA-based rapid diagnostic test kits are often used routinely as practical and time-saving assays. One particular commercial ELISA kit a has become available in Italy and many other countries for the detection of Giardia antigen in canine and feline feces.
Microscopic examination for diagnosis of canine giardiasis is an “alloyed” gold standard test (i.e., an imperfect reference test) with 100% Sp but lacking Se.13,28 A Bayesian model, which estimates the Se and Sp of 1 or more tests in the absence of a gold standard, can help in identifying the most suitable tests for routine diagnosis and screening programs. 4 This is particularly useful when none of the competing tests are treated as the gold standard, and the diagnostic error rates are estimated for all studied tests. 15 Prior information (such as that generated by previous research or expert opinions) is combined with the observed data to obtain posterior distributions of test parameters. 4 Knowledge of the true disease or infection status of the animal is therefore not necessary, and instead this unknown information is incorporated into the model as a latent variable. 10 Two studies have been published to date using a Bayesian model to estimate the performance of diagnostic techniques for canine giardiasis,13,30 and only 1 of these 13 included evaluation of the commercial kit evaluated in the current study.
A previous study, carried out using a real-time PCR assay, revealed that 30.8% of dog feces left uncollected on soil in urban areas harbored G. duodenalis DNA. 25 The present study represents an extension of the previous survey as the same samples were re-examined using a commercial ELISA, a with the performance of the ELISA evaluated using a Bayesian model.
One hundred forty-three fecal aliquots stored at −20°C were examined. Each came from a single sample previously collected and tested by real-time PCR for a study on the prevalence and cyst burden of G. duodenalis in dog feces found in the urban environment. 25 Briefly, freshly voided dog fecal samples were collected from soil, immediately after defecation, in 4 green urban areas in the town of Pisa (latitude 43°42’42”48N, longitude 10°24’52”92E), refrigerated (2–7°C) in a cooler bag, brought to the laboratory on the day of collection, and stored at −20°C until thawed at room temperature (20–25°C) before DNA extraction. 25 Immediately after collection of the amounts needed for DNA extraction, the remaining amounts of all 143 samples were frozen again at −20°C and stored for an additional 25 months. After this further storage period, the remaining amounts of each sample were thawed at room temperature (20–25°C) and re-evaluated by the commercial ELISA. a The kit devices and test reagents were stored at 2–7°C until use. The tests were run at room temperature following the recommended manufacturer’s instructions as described previously.6,9 The cross-classified results obtained by the 2 tests are presented in Table 1.
Cross-classified test results obtained by the commercial enzyme-linked immunosorbent assay (ELISA) and a real-time polymerase chain reaction (PCR) test.*

The number of samples is presented, along with the number of samples for a particular combination of test results: (+) indicates a positive and (−) indicates a negative test result. The ELISA is a commercial kit.a The real-time PCR results have been published previously. 25
The diagnostic performance of the ELISA was estimated within a Bayesian framework.1,17 Briefly, prior knowledge was incorporated to solve the statistically unidentifiable problem of estimating 5 parameters using the available data (3 degrees of freedom). Then, posterior distributions were generated, which contained updated beliefs about the values of the model parameters, after taking into account the information provided by the data.
In order to strengthen results of the present study, parameters for the real-time PCR were also included in the Bayesian statistical analysis, as shown in Table 2. Informative beta prior distributions were derived for Se and Sp of the 2 tests as well as for the prevalence. The beta α and β parameters for the prevalence were derived from the results of a previous study. 25 The parameters for Se and Sp were derived from fixed-effects meta-analysis of Se and Sp values reported in the package insert of the ELISA kit and previously published studies (Table 3).5,13,14,23,27,31 Confidence intervals for Se and Sp were computed using the score method incorporating continuity correction. 2 The fixed-effects meta-analysis was preferred to the random-effects meta-analysis because the true Se and Sp may be assumed to be the same in all studies. 3 For the analyses presented, posterior inferences were based on 100,000 iterations of the Gibbs sampler after a burn-in of 5,000 iterations was discarded. Convergence was assessed by running multiple chains from dispersed starting values. 12 A freeware program was used for all modeling. 21
Meta-analytical range (95%) for sensitivity and specificity of the commercial enzyme-linked immunosorbent assay (ELISA) and a real-time polymerase chain reaction (PCR) test and beta parameters for the prior distribution.*
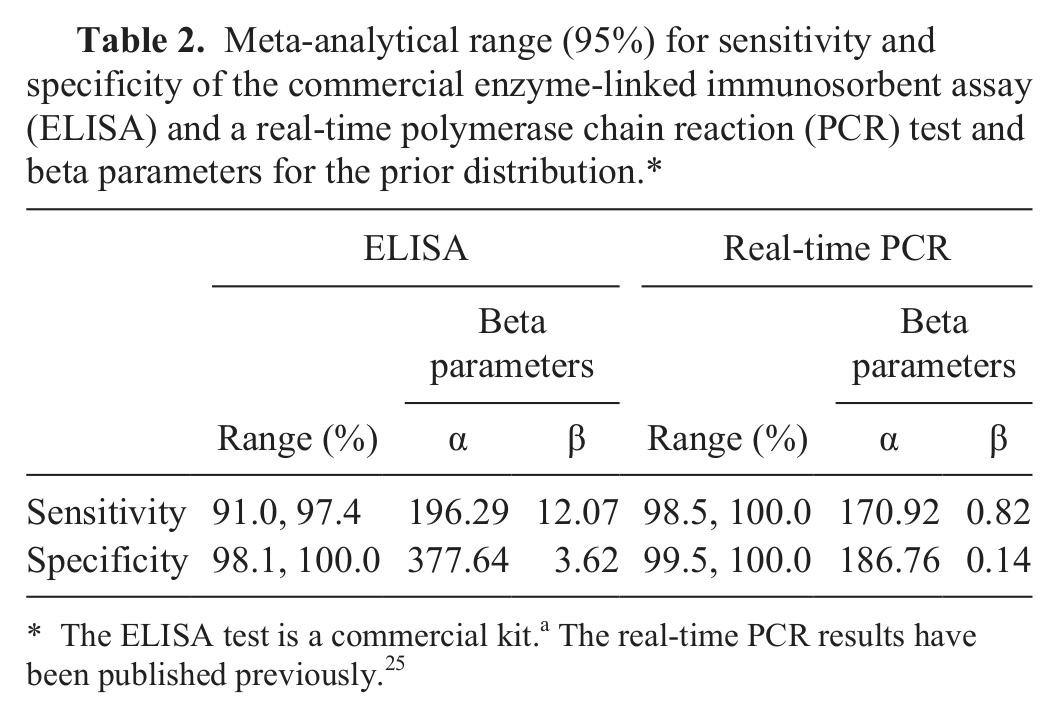
The ELISA test is a commercial kit.a The real-time PCR results have been published previously. 25
Sensitivity (Se) and specificity (Sp) values with the corresponding 95% confidence intervals (CIs) and references used in the meta-analysis.*
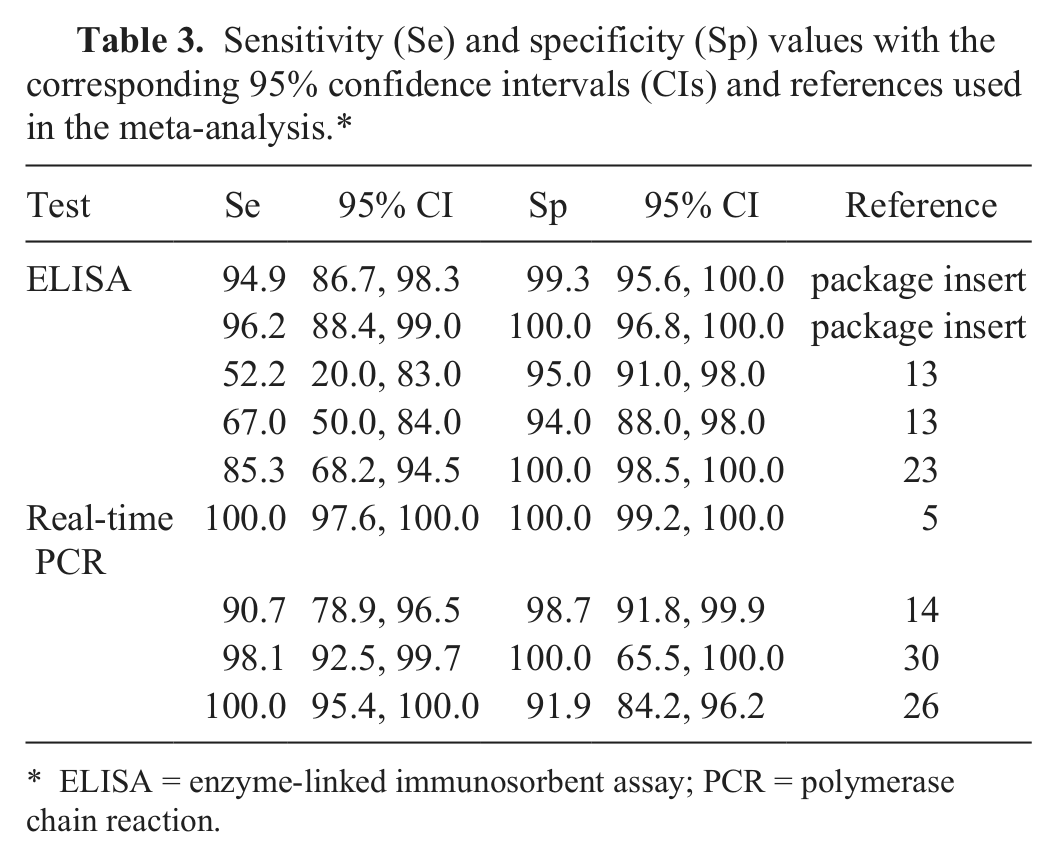
ELISA = enzyme-linked immunosorbent assay; PCR = polymerase chain reaction.
In the current Bayesian evaluation, posterior mean for prevalence was 29.0% (95% Bayesian credible interval [BCI]: 22.1–36.3). The Se and Sp values for the assay were 88.9% and 95.8%, respectively. The estimated Se and Sp along with their positive predictive values, negative predictive values, and 95% BCI are summarized in Table 4.
Mean values and 95% Bayesian credible interval (BCI) for the prevalence, sensitivity, specificity, positive predictive values (PPV), and negative predictive values (NPV) of the commercial enzyme-linked immunosorbent assay (ELISA) and real-time polymerase chain reaction (PCR) test.*
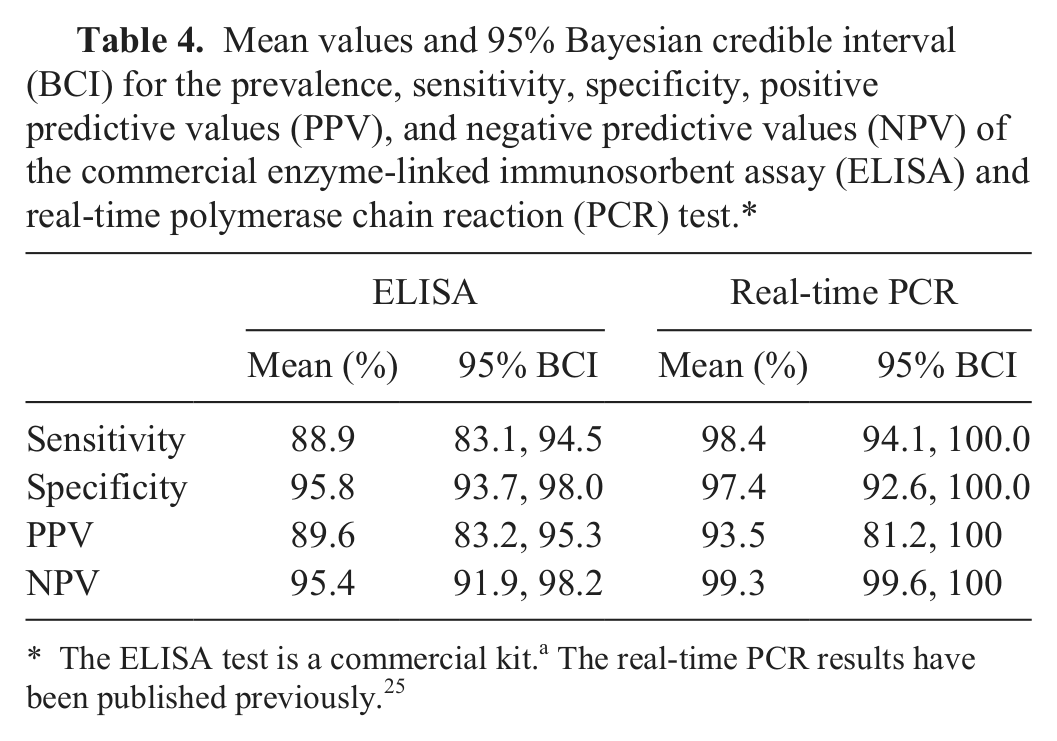
The ELISA test is a commercial kit.a The real-time PCR results have been published previously. 25
The Giardia fecal antigen has been reported to be stable indefinitely when samples are frozen at −20°C or for up to 1 week when samples are refrigerated at 2–7°C. 26 It has also been reported that multiple freezing and thawing episodes do not affect results of ELISAs. 18 However, it is possible that the color intensity of the reaction results, visually readable in the sample spot of the assay, may be reduced by multiple (2 or more) freeze–thaw events, mainly for samples with low levels of antigen (weak positives). Overall, the samples in the current study were first short-term refrigerated (less than 12 hr at 2–7°C) and then stored under constant freezing conditions (−20°C) until they were processed at different times for each of the 2 tests. The samples were also subjected to 2 freezing–thawing episodes. Therefore, the present storage conditions are unlikely to have had effects on the validity of the assay performance. However, it cannot be excluded that true weakly positive samples tested as false negatives due to effects of the 2 freezing–thawing episodes.
It has been reported that G. duodenalis may have an impact on public and animal health through environmental contamination with feces of infected hosts.20,22,25 The present Bayesian evaluation provides evidence on the efficiency of the assay a for the detection of Giardia antigen in dog feces dispersed in the environment as an indicator of field contamination. Based on the results of the current study, the test is more likely to have false-negative results. In a clinical setting, the high Sp minimizes the risk of false positives and unnecessary antimicrobial treatment. On the other hand, a proportion of infected dogs may go undetected due to false-negative results, though to a limited extent. From a public health point of view, infected dogs may serve as reservoirs and be potential sources of infection for human beings and other animals, mostly when they remain undetected and thus untreated. It has been suggested that the Se of the assay can be increased by multiple samplings.11,20 This can be done in dogs but is unrealistic for studies on dog feces found in the environment. If used on canine feces contaminating the environment, as well as on symptomatic dogs with diarrhea, the assay could be performed as a first step for primary screening while another test could be carried out supplementary to the assay, to improve the likelihood of correctly identifying dog feces with G. duodenalis cysts. For instance, additional diagnostic testing with PCR-based methods could provide added sensitivity and would be indicated because molecular characterization of isolates could be performed to determine their assignment to genotypes. The present results show that the assay, which features rapid processing of fecal samples without a complex technical structure and extensive costly labor, is a specific and fairly sensitive technique for the diagnosis of Giardia in clinically affected dogs as previously reported. 13 As well, the current study has shown that the ELISA can also be an appropriate method and a practical alternative to other methods for large-scale environmental screening programs to assess the risk of exposure to G. duodenalis cysts of canine source in human settlements.
According to the ELISA kit insert, reported Se and Sp values are 95% (95% confidence interval [CI]: 87–98%) and 99% (95% CI: 96–100%), respectively, when compared to DFAT, or 96% (95% CI: 88–99%) and 100% (95% CI: 97–100%) when compared to a microplate ELISA. In a conventional comparison study in cats, this ELISA was equally sensitive (85.3%, 95% CI: 68.9–95.1%) and specific (100%, 95% CI: 99–100%) to fecal flotation when compared to DFAT. 23 In a previous Bayesian analysis using microscopy, DFAT, and the ELISA kit used in the present study for the diagnosis of giardiasis in symptomatic and asymptomatic dogs, the ELISA was found to have Se values of 52% (95% CI: 20–83%) or 67% (95% CI: 50–84%) and Sp values of 95% (95% CI: 91–98%) or 94% (95% CI: 88–98%) in epidemiological and clinical settings, respectively. 13 In the present Bayesian evaluation, this rapid test was found to have a Sp (95.8%) overlapping the value previously observed in dogs 13 but lower than the value reported in cats. 23 In addition, the present model provided a higher value for Se (88.9%) with respect to the results of both studies.13,23 Different Se and Sp values estimated in different studies might be due to differences in the prior information settings, but they may also be a result of differences in the performances of the assay between the animal populations examined.
Test evaluation is traditionally performed by contingency table analysis using results of a single assay or a combination of assays as a gold standard. Some studies focused on the evaluation of different assays for the diagnosis of G. duodenalis in dogs and, typically, estimated the Se and Sp of diagnostic tests for Giardia by comparing them with known microscopy positive and negative samples.7,9 In agreement with statements of other authors, 30 obtaining a gold standard for diagnosis of canine giardiasis in the field is not realistic at present. When an imperfect reference test is used as a gold standard, the estimates of test accuracy will be biased. This phenomenon is called imperfect gold standard bias. If the test and imperfect gold standard are highly correlated (i.e., a tendency to make the same errors), the test is credited for misclassifying the same results as the gold standard, thus overestimating the Se and Sp. Conversely, if they are independent, the accuracy of the test can be underestimated. This makes the determination of the Se and Sp of a diagnostic test in the absence of a gold standard ineffective with a traditional approach. The Bayesian approach has been proposed to deal with the situation when a gold standard does not exist. 4 This methodology has proven its potential in estimating the parameters of a single test 29 and two 8 or more tests13,30 for the diagnosis of various helminth and protozoa parasitoses in human beings and animals in the absence of a gold standard. For these reasons, the Bayesian evaluation was selected in the current study.
As the ELISA and the real-time PCR assay measure different biological parameters associated with Giardia in dogs (i.e., the ELISA detects the presence of Giardia coproantigen while the real-time PCR assay measures Giardia cyst DNA in fecal samples), the 2 tests can be considered as independent of each other. Therefore, the ELISA and the real-time PCR assay can be used in combination at the same time in a single animal to allow improved diagnosis. Parallel and series interpretation can be used to combine the information provided by the individual test results. The combined Se and Sp values for the 2 tests can be calculated. 24 With parallel interpretation, an animal is considered to have Giardia infection if 1 or both tests are positive. In this interpretation, the animal is considered Giardia-positive after the first test that produces a positive result, but if the first test is negative, the second test is still necessary. In the current study, parallel test interpretation increases Se from 88.9% for the ELISA and 98.4% for the real-time PCR assay to a combined level of 99.8%, thus cases of canine giardiasis are almost certain not to be missed. But on the other hand, it reduces Sp to 93.3%, hence false-positive results will be more likely. With serial test interpretation, an animal is considered Giardia-positive if both tests are positive. In other words, the animal is classified as negative as soon as 1 test is negative. The serial interpretation substantially improves Sp from 95.8% for the ELISA and 97.4% for the real-time PCR assay to a combined level of 99.8%. This means that any positive animal almost certainly will have the infection, so that it may become useful for clinical and therapeutic perspectives. However, it results in a drop in the Se value to a moderate 87.4%. As a consequence, although being considerably more expensive due to the high costs of reagents and equipment for molecular procedures, the simultaneous use of the ELISA and the real-time PCR assay could be more useful than the ELISA alone, as it will increase Se (with parallel test interpretation) or Sp (with serial interpretation). The use of both tests could make economic sense in specific situations of particular epidemiological and clinical utility, such as identification of risk factors associated with community outbreaks of giardiasis. For analysis of dog stools in the environment, parallel interpretation might be the preferred method because false positives are unlikely to have serious consequences.
The present report confirms the efficiency of the ELISA for the diagnosis of canine giardiasis and, in addition, provides evidence on its possible application to dog feces found in the field. The main advantages of the assay are that it is rapid, is relatively inexpensive, and employs user-friendly techniques without special equipment. This suggests that the assay could reasonably be included, as a valid alternative or as an adjunct to other techniques, in screening studies for the identification of areas exposed to risk of environmental contamination with Giardia cysts of canine source. Nonetheless, molecular procedures for detection and identification of Giardia species and genotypes cannot be replaced when the zoonotic potential of canine feces contaminating the environment has to be accurately assessed in human communities.
Footnotes
a.
SNAP Giardia test kit, IDEXX Laboratories, Milan, Italy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
