Abstract
Swine dysentery is classically associated with infection by the strongly beta-hemolytic Brachyspira hyodysenteriae; however, the proposed novel species “Brachyspira hampsonii” has also been isolated from clinical cases of dysentery in the United States and Canada. Microbial culture is highly sensitive for detecting Brachyspira in clinical samples but requires several days for completion and is often followed by molecular testing for speciation. Alternatively, in situ hybridization using molecular probes applied to sections of formalin-fixed tissue can provide rapid, culture-independent identification of agents observed histologically. Accordingly, a fluorescent in situ hybridization assay was developed for confirmation of a clinical diagnosis of swine dysentery associated with infection by “B. hampsonii.” An oligonucleotide probe (Hamp1210) targeting a specific 23S ribosomal RNA sequence of “B. hampsonii” was developed following sequence analysis and comparison of numerous Brachyspira spp. clinical isolates with reference sequences available in GenBank. The application of Hamp1210 and a previously published probe for B. hyodysenteriae (Hyo1210) to diseased colonic tissues successfully detected the target species in both experimentally infected pigs and naturally infected pigs from field cases, and the Hamp1210 probe consistently detected both clade I and clade II isolates of “B. hampsonii”; however, a strong positive signal was also observed in a single case where the Hamp1210 probe was applied to tissues infected with Brachyspira intermedia. In situ hybridization incorporating the Hamp1210 probe can reduce the delay from sample submission to pathogen identification in cases of swine dysentery associated with “B. hampsonii” infection where formalin-fixed tissues are available.
Swine dysentery (SD) is characterized by severe mucohemorrhagic diarrhea and uneven growth, most often observed in growing and finishing pigs, and can result in significant economic losses in affected production systems. 8 Swine dysentery is classically associated with infection by the strongly beta-hemolytic spirochete Brachyspira hyodysenteriae 9 ; however, experimental reproduction of SD following inoculation with clinical isolates of strongly beta-hemolytic spirochetes that are not identified as B. hyodysenteriae by polymerase chain reaction (PCR) assays has also been described.6,13 Strongly beta-hemolytic spirochetes identified as Brachyspira intermedia and “Brachyspira sp. SASK30446” by PCR were associated with clinical disease and gross and histologic lesions of SD in a study published in 2012, 6 and comparison of the reported nox gene sequences from those isolates with others available in GenBank reveals they are identical or nearly identical (≥99–100% identity) over the region compared to the clade I and clade II isolates, respectively, of the proposed novel species “Brachyspira hampsonii.” 7 Taken together, these findings establish that SD can occur following infection with “B. hampsonii.”
Clinical suspicion of SD is based on observation of characteristic diarrhea containing blood and/or mucus. Gross lesions of SD are limited to the cecum and colon and typically include variable degrees of mucosal thickening, hemorrhage, fibrinonecrotic surface exudation, and luminal mucus. 8 Laboratory confirmation of SD is often based on microbial culture, with the isolation of a strongly beta-hemolytic, ring phenomenon–positive spirochete followed by speciation through molecular methods such as PCR targeting the NADH oxidase (nox) gene2,16; however, as these assays are often applied to primary cultures, which can take 6 days or more to complete, there is often a significant delay between receipt of clinical samples and the reporting of Brachyspira culture results.
In cases of suspected SD where formalin-fixed tissues are available, characteristic spirochetes can often be readily visualized within affected colonic tissue by silver staining techniques such as the Warthin–Starry method. While observation of these spirochetes by histochemical methods is supportive of a diagnosis of SD, the imprecise nature of this staining procedure does not allow differentiation of highly pathogenic spirochetes from mildly pathogenic or commensal species such as Brachyspira murdochii and Brachyspira innocens, which may also be present in the porcine colon. Fluorescent in situ hybridization (FISH) using bacterial ribosomal RNA (rRNA)-targeted oligonucleotide probes can be an effective assay for specific molecular identification of pathogens noted histologically, and FISH assays have been described for detection of Brachyspira spp. at the genus level as well as at the species level for identification of Brachyspira pilosicoli, B. hyodysenteriae, and B. murdochii.4,10 Given the emergence of “B. hampsonii” associated with clinical SD in North America, 7 and the experimental reproduction of SD following inoculation with clinical isolates, 6 the development of rapid diagnostic tools for detection of this agent is a pressing need. Accordingly, a specific oligonucleotide probe targeting 23S rRNA was developed to detect “B. hampsonii” in formalin-fixed tissues after 6 hr of hybridization using modified buffers and environmental conditions relative to those previously reported for Brachyspira spp. where 16 hr of hybridization was described. 4
Clinical isolates of Brachyspira spp. from the culture collection of the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL; Ames, IA) were obtained for genetic sequencing and analysis. Historical information, phenotypic characteristics, and species identification for the specific isolates used in the current study are listed in Table 1. Brachyspira isolates were identified to species by nox-based PCR assays as previously described.15,16 For those strongly beta-hemolytic isolates not identified as B. hyodysenteriae by PCR, final identification as either clade I or clade II isolates of “B. hampsonii” was determined by PCR amplification and sequencing of the nox gene using previously described primers 14 followed by comparison with reference sequences available in GenBank. All clinical isolates had been stored at −80°C until use and were recovered on CVS selective agar containing colistin, vancomycin, and spectinomycin; and BJ selective agar containing pig feces extract, spiramycin, rifampin, vancomycin, colistin, and spectinomycin. Plates were incubated in an anaerobic environment provided by a commercial system a at 41 ± 1°C for up to 6 days with growth and strength of beta-hemolysis assessed every 48 hr. Once growth was deemed adequate, agar plugs were selected from hemolytic areas of each plate, and DNA was extracted by boiling the agar plugs in microcentrifuge tubes containing molecular biology-grade water followed by centrifugation at 20,000 × g for 5 min. The 23S rDNA of each isolate was PCR amplified and sequenced in 3 fragments using previously described 11 and modified primers (Table 2). The PCR amplicons were sequenced using a commercial analyzer, b and the sequences obtained were assembled and aligned using a commercial software package. c Comparison of the 23S rDNA sequences obtained from the clinical isolates in the present study with reference sequences available in GenBank revealed distinct clustering by species (Fig. 1); however, the overall sequence similarity between “B. hampsonii,” B. intermedia, and B. hyodysenteriae is very high with less than 1 nucleotide per 100 nucleotides difference. Analysis of the consensus sequence for Brachyspira spp. revealed a high degree of interspecies variability in the area of bases 1310–1328 of the 23S rRNA gene within the published whole genome sequence of B. hyodysenteriae WA1. 3 This area corresponds to the same region targeted by the previously described Hyo1210 probe, 4 and within this area the sequences of both clade I and clade II isolates of “B. hampsonii” were identical for all isolates tested. Only a single base difference was noted in this area between “B. hampsonii” and B. intermedia; however, as additional areas were evaluated in efforts to increase differentiation between these 2 species, differentiation from other species was often lost. For this reason, and given that B. intermedia is an uncommon clinical isolate in the ISU VDL, the 1310–1328 site was selected for “B. hampsonii” probe development and analysis. Analysis of the target site corresponding to the SER1410 probe revealed 100% sequence homology for all strains evaluated. Descriptions of the 3 oligonucleotide probes used in the present study are listed in Table 3. The specificity of the newly developed Hamp1210 probe was tested against available sequences in GenBank using BLAST analysis (http://blast.ncbi.nlm.nih.gov/Blast.cgi) with the closest match to other Brachyspira spp. being B. intermedia PWS/A (GenBank accession no. CP002874) and B. innocens (GenBank accession no. U72700) with 84% coverage each. Custom oligonucleotide probes were purchased from a commercial source d and were 5’-labeled with either green fluorescent e (Hamp1210) or orange fluorescent f (SER1410 and Hyo1210) dye.
Clinical Brachyspira isolates used for 23S sequence analysis in the current study.
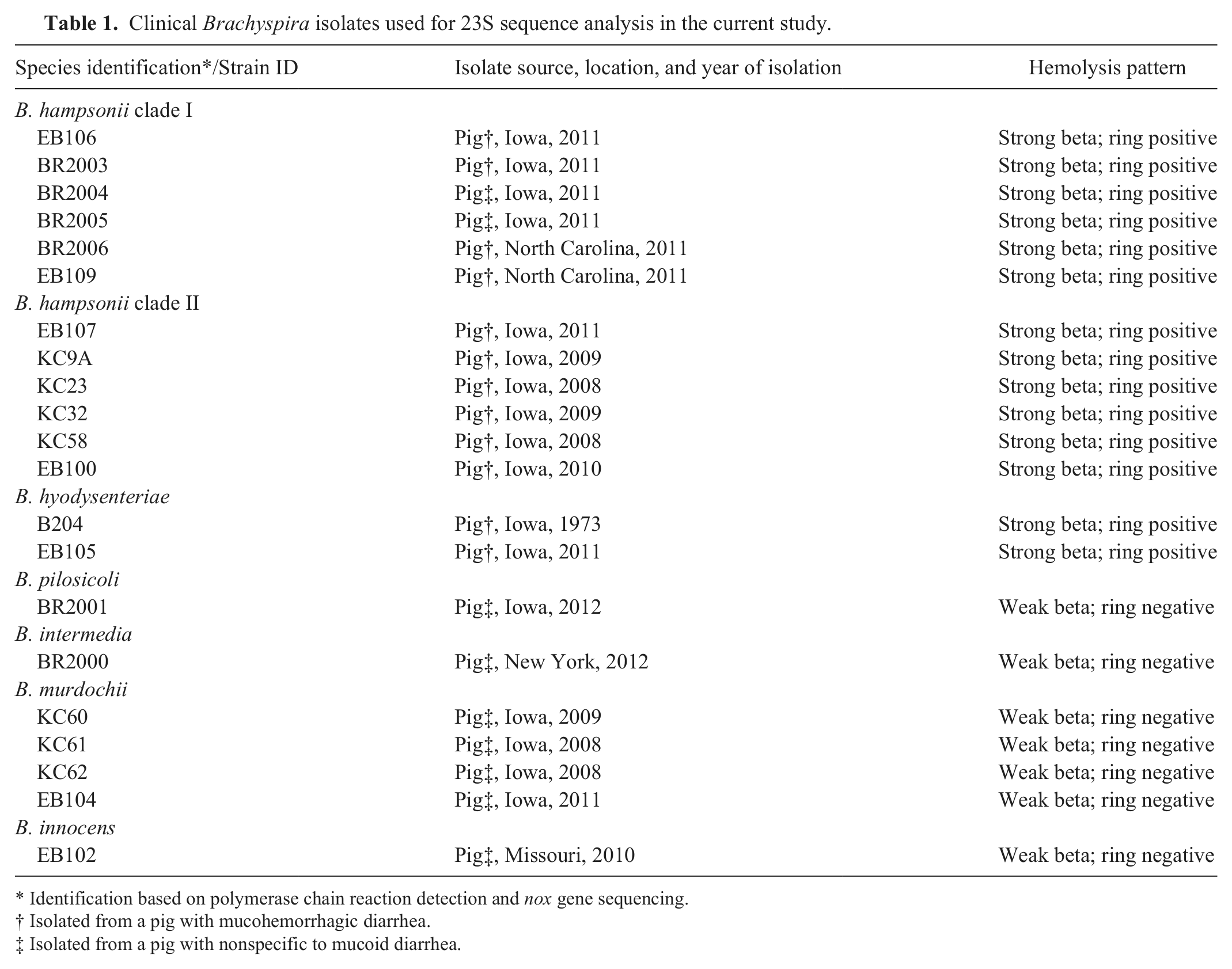
Identification based on polymerase chain reaction detection and nox gene sequencing.
Isolated from a pig with mucohemorrhagic diarrhea.
Isolated from a pig with nonspecific to mucoid diarrhea.
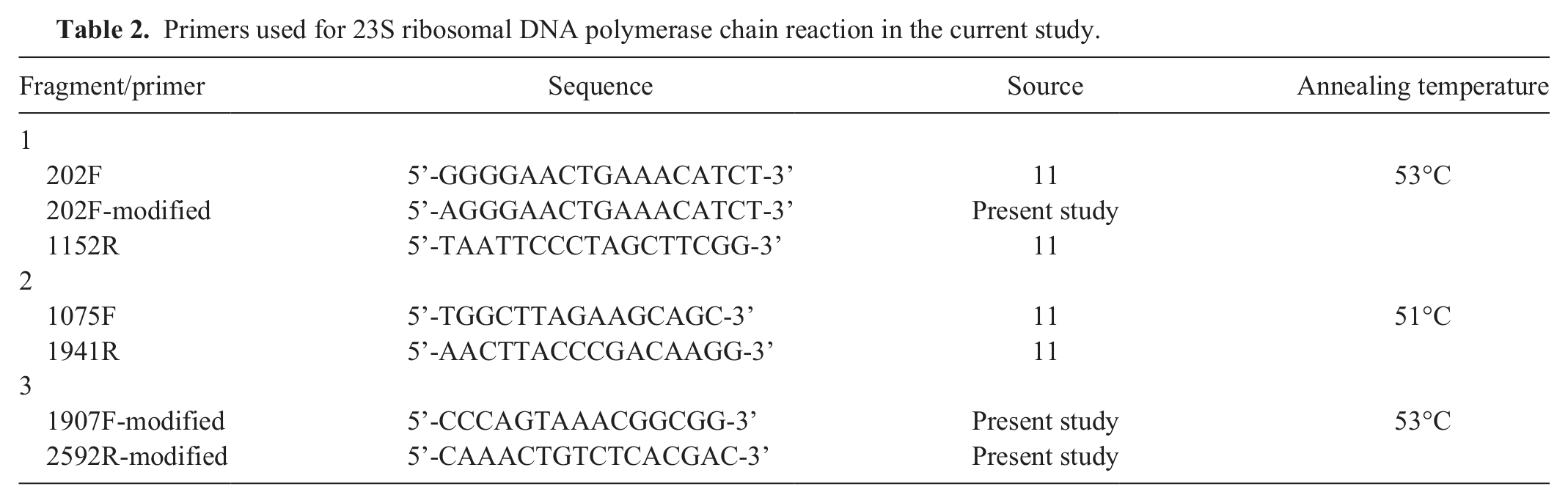
Primers used for 23S ribosomal DNA polymerase chain reaction in the current study.
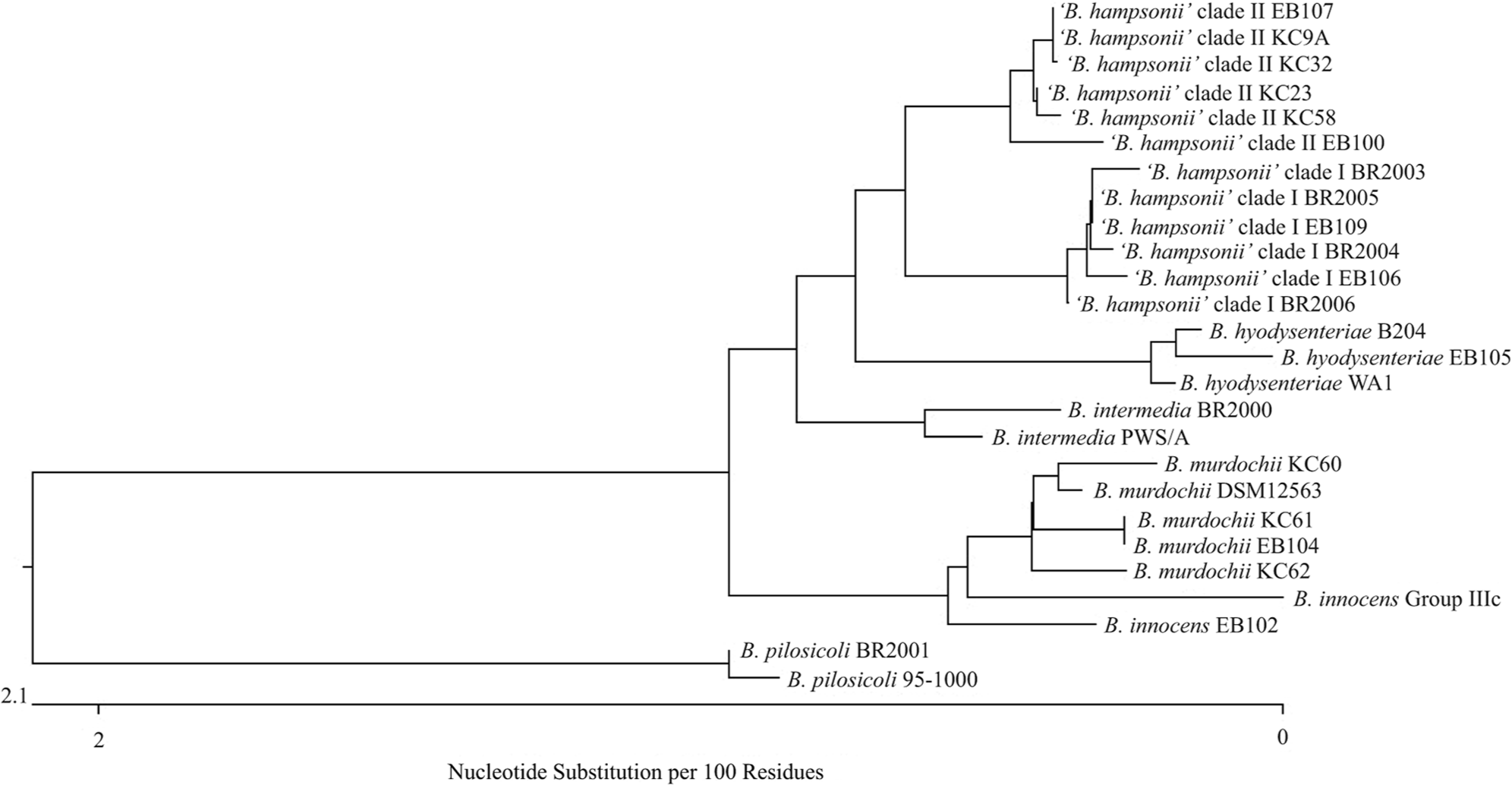
Sequence diagram representing 23S ribosomal DNA sequences from all clinical isolates and reference strains used in the development of a specific oligonucleotide probe for “B. hampsonii” (Hamp1210).
Sequences, target sites, and specificities of ribosomal RNA-targeted oligonucleotide probes used in the current study.
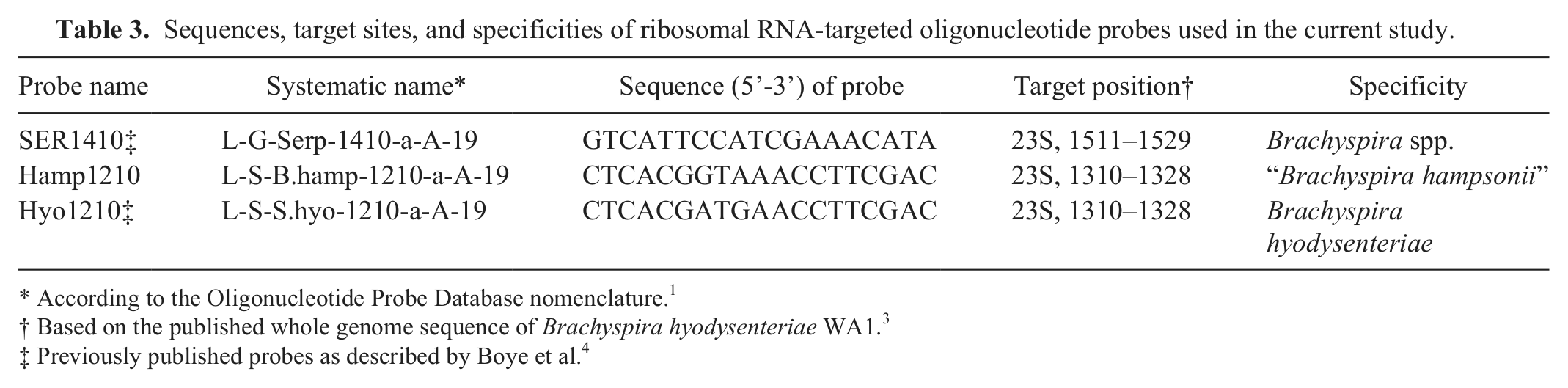
According to the Oligonucleotide Probe Database nomenclature. 1
Based on the published whole genome sequence of Brachyspira hyodysenteriae WA1. 3
Previously published probes as described by Boye et al. 4
Blocks of paraffin-embedded colonic tissue from pigs with clinical SD were obtained from clinical submissions to the ISU VDL and from a previous experimental study, 6 as well as blocks from pigs with catarrhal colitis from which a weakly beta-hemolytic Brachyspira sp. was isolated. Sections were selected where spirochetes with features typical of Brachyspira spp. were visible by Warthin–Starry silver staining, and in all cases samples were selected from tissues where a single Brachyspira sp. was isolated by culture. The following species were represented in this analysis, and the number of individual samples tested for each species appears in parentheses: B. hyodysenteriae (4), “B. hampsonii” clade I (4), “B. hampsonii” clade II (4), B. pilosicoli (4), B. intermedia (1), and B. murdochii (4). Tissue sections (4 µm) were mounted on glass slides and deparaffinized by passage through xylene (3 passages, 10 min), 100% alcohol (2 passages, 5 min), 95% ethanol (5 min), and 70% ethanol (5 min) and then allowed to air dry. The area of interest on each slide, as determined by silver staining, was circumscribed with a hydrophobic marking pen prior to application of the hybridization solution. The labeled probes were reconstituted using DNase- and RNase-free water and diluted to a working concentration of 5 ng/μl in hybridization buffer (20 mM of Tris, 0.9 M of NaCl, 20% sodium dodecyl sulfate [SDS] buffer, 40% formamide, 10% dextran sulfate [pH 7.2]). Sections were covered with 150 µl of the hybridization solution containing the desired probe and were placed in a moisture chamber. Hybridization was carried out for 6 hr at 42°C for both the Hamp1210 and Hyo1210 probes or overnight at 45°C for the SER1410 probe. Following hybridization, slides were washed in a wash buffer (hybridization buffer without SDS or formamide and prewarmed to the appropriate hybridization temperature) for 20 min. After washing, the slides were rinsed with sterile water, allowed to air dry, and mounted using a commercial antifade reagent. g A fluorescent microscope with filters specific for the fluorophores used in the current study was used for all analyses.
Sections were first screened using the SER1410 probe, and only those with positive signal by this probe were subjected to the species-specific probes. All sections tested had moderate to strong signal detected after hybridization with the SER1410 probe except 1 from a clinical case in which B. pilosicoli was recovered by culture and 1 in which B. murdochii was isolated.
For the Hamp1210 probe, moderate to strong signal was detected after hybridization in all cases where “B. hampsonii” was recovered by culture, and in none of the cases that were culture positive for B. pilosicoli or B. murdochii. Labeled spirochetes were visible within crypt lumens, epithelia, and luminal exudate (Fig. 2). In the single case that was culture positive for B. intermedia, a strong positive signal was detected following hybridization with the Hamp1210 probe, and spirochetes could be visualized within crypt lumens, goblet cells, and luminal exudate. In 1 of 4 cases where B. hyodysenteriae was recovered by culture, a weak signal was variably detected throughout the section following hybridization with the Hamp1210 probe.
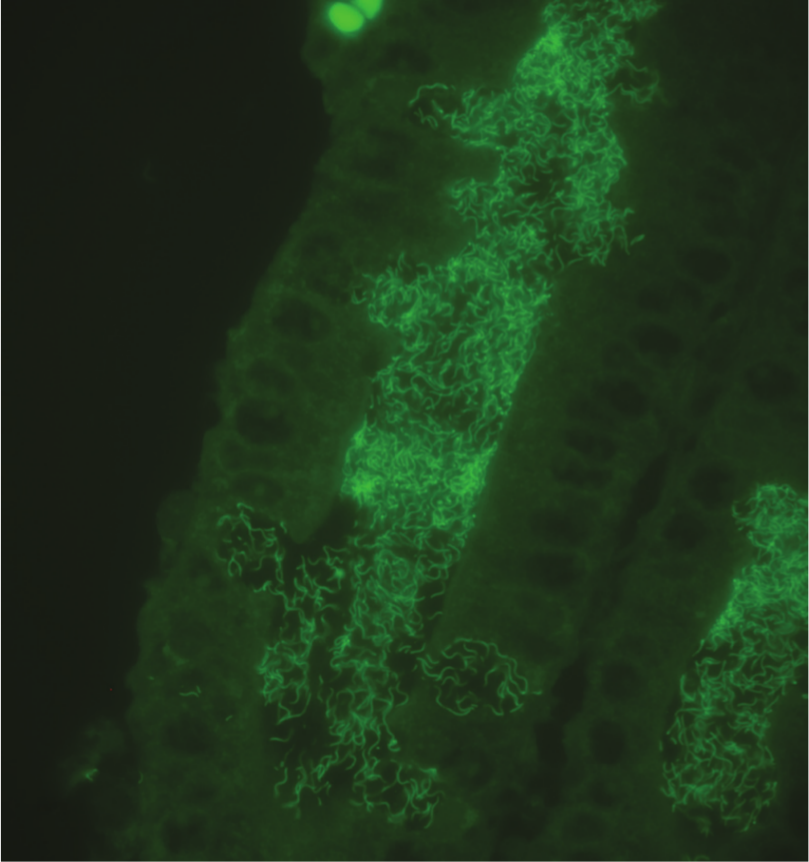
Histologic section of colonic mucosa from a pig infected with “Brachyspira hampsonii” clade II revealing strong positive signal labeling spirochetes within crypt lumens and within goblet cells. Fluorescent in situ hybridization using a green fluorescent probe (Hamp1210) specific for “B. hampsonii.”
For the Hyo1210 probe, moderate to strong signal was detected after hybridization in all cases where B. hyodysenteriae was recovered by culture, and in none of the cases that were culture positive for B. pilosicoli, B. murdochii, or B. intermedia. Labeled spirochetes were visible within crypt lumens, epithelia, and luminal exudate (Fig. 3). As with the Hamp1210 probe, in 1 case where “B. hampsonii” was recovered by culture, a weak signal was variably detected following hybridization with the Hyo1210 probe.

Histologic section of colonic mucosa from a pig infected with Brachyspira hyodysenteriae revealing strong positive signal labeling spirochetes within crypt lumens and enterocytes. Fluorescent in situ hybridization using an orange fluorescent probe (Hyo1210) specific for B. hyodysenteriae.
Even though both the Hamp1210 and Hyo1210 probes had weak cross-reactivity with the opposite target species in a minority of sections evaluated, in all cases the hybridization signal was stronger for the target species and neither probe yielded a positive signal in tissues containing B. pilosicoli or B. murdochii, suggesting that these probes have a high degree of specificity under the hybridization conditions used in this study for detecting spirochetes capable of inducing clinical SD. The strong positive signal following hybridization with Hamp1210 in the single case of B. intermedia–infected tissues is not surprising given that there is only a single base substitution in the target site between this species and “B. hampsonii”; however, this does suggest that FISH should be used as a screening tool with cultural confirmation of hemolytic phenotype in index cases or in situations where B. intermedia infection is a concern. In the ISU VDL, B. intermedia is infrequently identified from porcine samples relative to other common weakly beta-hemolytic Brachyspira spp. including B. murdochii, B. pilosicoli, and B. innocens, which are isolated with decreasing frequency, respectively.
The similarity in 23S rDNA sequences between B. intermedia and “B. hampsonii” is not entirely unexpected given that many clade I isolates of the proposed “B. hampsonii” are identified as B. intermedia by PCR assays described as specific for this species,5,6 and Serpulina sp. P280/1, a proposed ancestral isolate of “B. hampsonii” from the United Kingdom, 7 is listed as an isolate of B. intermedia in a recent comparative analysis of strain diversity within this species. 12 Further investigation such as whole genome sequencing and analysis of isolates representing both clades of “B. hampsonii” is necessary to further understand the relationship, if any, between this proposed novel Brachyspira and the officially recognized species.
By utilizing the same target site for the Hamp1210 probe as that previously described for the Hyo1210 probe, laboratories already using FISH for detection of Brachyspira spp. can easily incorporate the new Hamp1210 probe into their existing assays and increase the overall sensitivity of those assays in confirming clinical cases of SD. In addition, the hybridization conditions described in the current report allow for shortening the time necessary for completion of the assay from overnight to 6 hr.
In conclusion, the results of the present study reveal that FISH applied to formalin-fixed colon from pigs with a clinical diagnosis of SD can serve as a rapid screening tool and provide a preliminary molecular identification within 24–48 hr of sample receipt at the laboratory. The application of the Hamp1210 and Hyo1210 probes in tandem to clinically affected tissues will allow for the detection and identification of the 2 most commonly identified, strongly beta-hemolytic spirochetes associated with SD in North America, and would provide rapid confirmation of an SD diagnosis thereby shortening the delay between sample submission and therapeutic intervention. Given the potential for cross-reactivity with these 2 probes related to the high degree of 23S rRNA sequence similarity between the target Brachyspira spp., additional testing and characterization of positive samples is indicated, including, but not limited to, cultural confirmation of hemolytic phenotype and nox gene sequencing of isolates for more definitive species identification.
Footnotes
Acknowledgements
The authors wish to thank Joann Kinyon and Hallie Warneke for their help with the Brachyspira culture work and maintenance of the isolate collection at the ISU VDL; Diane Gerjets, Jennifer Groeltz-Thrush, and Toni Christofferson for preparation of the histologic sections; and Todd Atherly for his help with the initial application of the fluorescent in situ hybridization protocols described in this work.
a.
BD GasPak EZ Anaerobe Container System, BD Diagnostic Systems, Sparks, MD.
b.
AB 3730xl DNA Analyzer, Applied Biosystems, Foster City, CA.
c.
Lasergene version 8.0, DNASTAR Inc., Madison, WI.
d.
Invitrogen Custom Oligos, Life Technologies, Carlsbad, CA.
e.
Alexa Fluor 488, Life Technologies, Carlsbad, CA.
f.
Alexa Fluor 555, Life Technologies, Carlsbad, CA.
g.
Invitrogen ProLong Gold, Life Technologies, Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared that they had no conflicts of interest with respect to the research, authorship, and/or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Iowa Pork Producers Association and the National Pork Board (NPB #12-192).
