Abstract
Classical swine dysentery is associated with the presence of the strongly beta-hemolytic Brachyspira hyodysenteriae. However, multiple Brachyspira spp. can colonize the porcine colon. Since 2008, several Brachyspira spp. not identified as B. hyodysenteriae by genotypic and/or phenotypic methods have been isolated from the feces of pigs with clinical disease typical of swine dysentery. In the current study, 8 clinical isolates, including 5 strongly beta-hemolytic and 3 weakly beta-hemolytic Brachyspira strains, and a reference strain of B. hyodysenteriae (B204) were inoculated into pigs (n = 6 per isolate) to compare pathogenic potential following oral inoculation. Results revealed that strongly beta-hemolytic isolates induced significantly greater typhlocolitis than those that are weakly beta-hemolytic, regardless of the genetic identification of the isolate, and that strongly beta-hemolytic isolates identified as “Brachyspira sp. SASK30446” and Brachyspira intermedia by polymerase chain reaction (PCR) produced lesions similar to those caused by B. hyodysenteriae. The results suggest that phenotypic culture characteristics of Brachyspira spp. may be a more sensitive indicator of potential to induce dysentery-like disease in pigs than molecular identification alone based on currently available PCR assays. Additionally, culture of mucosal scrapings obtained at necropsy was more sensitive than direct PCR on the same samples for detection of Brachyspira spp.
Keywords
Introduction
Swine dysentery (SD) is characterized by severe mucohemorrhagic diarrhea and often results in significant economic losses in affected swine. 9 The causative agent of SD was first named Treponema hyodysenteriae 11 before several reclassifications in the genera Serpula, 29 Serpulina, 28 and currently Brachyspira. 20 Brachyspira spp. are Gram-negative, oxygen-tolerant, anaerobic spirochetes, historically differentiated by their phenotypic growth characteristics, including strength of beta-hemolysis and ability to produce indole and hydrolyze hippurate. 27 When cultivated on blood agar, B. hyodysenteriae typically imparts a characteristic strong beta-hemolysis that is enhanced around slits or holes in the agar, in what is referred to as the “ring phenomenon.” 22 Additionally, isolates of B. hyodysenteriae typically have positive indole and negative hippurate reactions, although indole-negative isolates have also been described.7,31 Strength of beta-hemolysis has historically been considered sufficient to differentiate B. hyodysenteriae from B. innocens, B. intermedia, B. murdochii, and B. pilosicoli, all weakly beta-hemolytic Brachyspira spp. that may also be present in the porcine colon.
Clinical suspicion of SD is based upon observation of moderate to severe mucohemorrhagic diarrhea. 27 Gross lesions are limited to the large intestine and typically include variable amounts of mucosal thickening, multifocal hemorrhage, superficial necrosis with associated fibrinonecrotic exudate, and variable but often excessive luminal mucus. 9 Definitive laboratory confirmation of SD is based upon 1) appropriate microscopic lesions, which include variable suppurative infiltration of the lamina propria, mucosal crypt elongation, and goblet cell hyperplasia, 2) identification of spirochetes within lesions often by silver staining, 3) isolation of a characteristic strongly beta-hemolytic, ring phenomenon–positive spirochete via microbial culture, and, more recently, 4) identification of the spirochete by molecular methods such as polymerase chain reaction (PCR). 9 Brachyspira can be differentiated to species level by PCR targeting the NADH oxidase (nox) gene.1,33 While PCR brings increased precision and specificity to the identification of many pathogens, it has added a new level of complexity to the diagnosis and understanding of SD as a disease. Results of PCR testing have revealed that not all strongly beta-hemolytic isolates are identified as B. hyodysenteriae by assays described as specific for this species, and these findings are the basis of several recent reports of potentially novel, strongly beta-hemolytic Brachyspira spp. including “Brachyspira suanatina” 24 and “Brachyspira sp. SASK30446” (Harding J, Chirino-Trejo M, Vermette C, et al.: 2010, Detection of a novel Brachyspira species associated with haemorrhagic and necrotizing colitis. Proceedings of the Annual Western Canadian Association of Swine Veterinarians Conference, pp. 65–70, October 15–16, Saskatoon, SK, Canada). The identification of potentially novel pathogenic Brachyspira spp. warrants further investigation and is the subject of the current study. The objectives of the current study were 1) to determine if dysentery-like disease follows oral inoculation of pigs with Brachyspira spp. other than B. hyodysenteriae, 2) to compare the virulence potential of strongly beta-hemolytic and weakly beta-hemolytic clinical isolates, and 3) to compare culture and direct PCR for detection of Brachyspira spp. from necropsy samples.
Materials and methods
Animals
All procedures were approved by the Institutional Animal Care and Use Committee of Iowa State University (ID# 7131). Fifty-four 4-week-old crossbred pigs, with no known previous history of Brachyspira-associated disease, were obtained from a commercial source. Prior to arrival, multiple fecal samples from the source farm were negative for Brachyspira spp. by direct culture as described below. Upon arrival, pigs were ear tagged, randomly separated into groups of 6, and swabbed rectally to test for the presence of Brachyspira spp. by microbial culture. Each group of pigs was maintained in its own raised deck pen approximately 2 m × 4 m, with a nipple watering system and single ad libitum feeder; all pens were separated from one another to prevent any contact among groups. Pigs were acclimated to these groups and to the facility for 1 week prior to experimental inoculation and were fed a non-medicated, nutritionally complete, age-balanced diet for the duration of the study.
Bacterial strains, growth conditions, and preparation of inocula
Media used included trypticase soy agar with 5% defibrinated bovine blood (TSA); CVS selective agar containing colistin, vancomycin, and spectinomycin 13 ; and BJ selective agar containing pig feces extract, spiramycin, rifampin, vancomycin, colistin, and spectinomycin. 17 Plate media used in the present study were prepared in-house and passed the quality assurance standards of the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL). An anaerobic environment was provided by a commercial system, a and plates were incubated at 41 ± 1°C.
Isolates used in the current study (Table 1) were obtained from the culture collection of the ISU VDL. Isolates administered to groups 2–8 were recent field isolates, and the year and geographic location of origin are reported in Table 1. Each isolate was submitted by a different veterinarian and was representative of different groups of pigs with clinical diarrhea that varied from mild and nonspecific (KC60, EB102, and EB104) to mucohemorrhagic (KC35, KC58, EB106, and EB107); however, it has not been definitively ruled out if any of these strains are epidemiologically connected. The B. hyodysenteriae B204 isolate administered to group 9 had been previously recovered from an experimentally challenged C3H/HeN mouse with severe typhlitis; the isolate was at the eleventh in vitro passage level at the time of inoculation in the present study. All isolates had been subcultured 3 times from a single colony isolate to new plate media (TSA). Stock cultures of each cloned isolate were stored at −80°C. The isolates were at 8–11 passages in vitro at the time of challenge.
Brachyspira spp. isolates used in inocula in the current study.
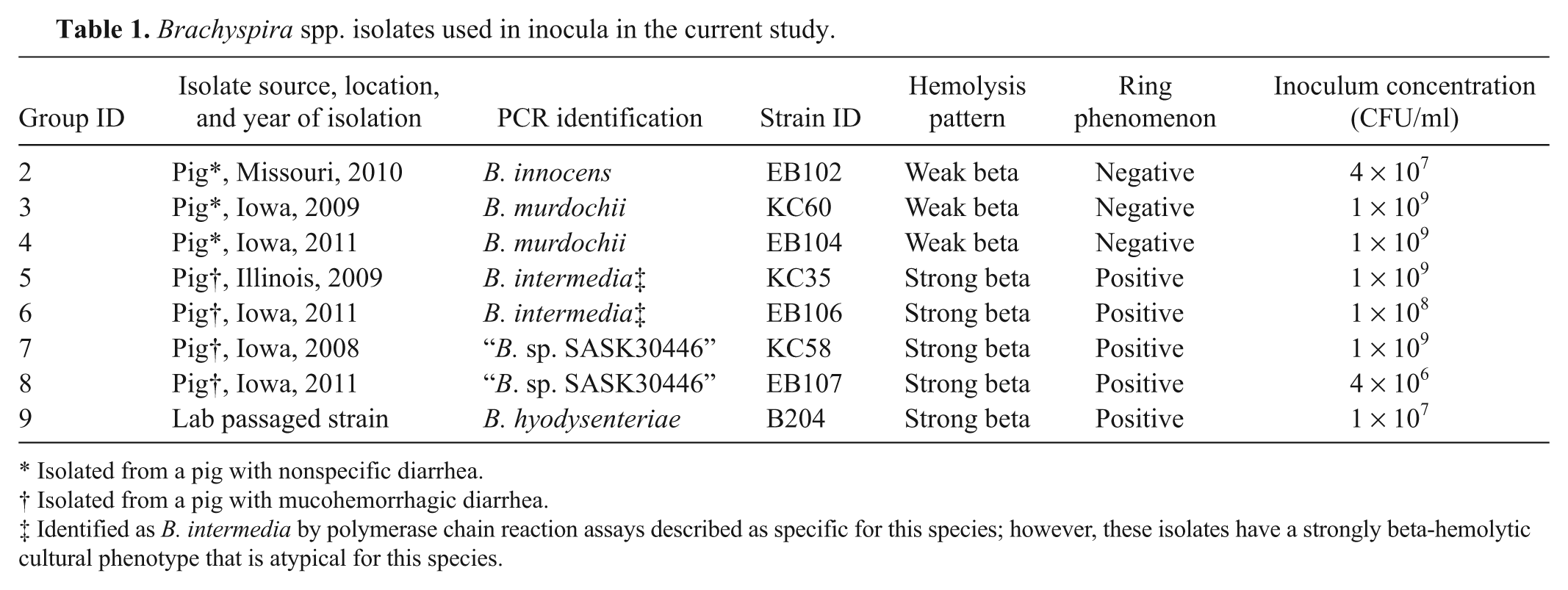
Isolated from a pig with nonspecific diarrhea.
Isolated from a pig with mucohemorrhagic diarrhea.
Identified as B. intermedia by polymerase chain reaction assays described as specific for this species; however, these isolates have a strongly beta-hemolytic cultural phenotype that is atypical for this species.
To prepare the inocula, isolates were thawed and plated onto several TSA plates and incubated anaerobically for 2–4 days. All plates were examined for contaminating colonies and were discarded and replaced if questionable. Thirty-five plates of each isolate were used to prepare the inoculum for each day. Agar was removed from plates with sterile wooden tongue depressors and placed in large plastic bags yielding approximately 630 ml of pooled agar per isolate. The agar was then homogenized by hand until a uniform consistency was obtained. Sterile saline (70 ml) was added to form a slurry for inoculation. Inoculation was performed within 1.5–3 hr of preparation of the inocula. A 5-g sample of each slurry was retained for titration of the inoculum in which dilutions were plated and colonies counted after 6 days incubation to obtain the inoculum titer in colony-forming units (CFU) per ml. A sham inoculum, consisting of non-inoculated TSA plates processed in the same fashion as the inoculated media, was also prepared.
Animal inoculation
Each pig received 2 doses of inoculum (100 ml/dose; Table 1) administered via gavage 24 hr apart, with each administration preceded by a 6-hr fast. For gavage, pigs were restrained briefly by hand, a rubber speculum was placed between their incisors, and an 18-Fr rubber feeding tube was passed into the esophagus. A 60-ml feeding syringe was used to administer the inocula in two 50-ml boluses, and the pigs were immediately returned to their respective pens. Feed was made available when all pigs in the group were inoculated. All pigs within each group received the same isolate prepared the same way on 2 consecutive days. Each group received the isolate corresponding to their group number (2–9), with group 1 receiving the sham inoculum.
Molecular identification
Each isolate was identified to species by PCR assays targeting nox gene sequences as previously described (B. hyodysenteriae, B. pilosicoli, and B. intermedia 26 ; B. murdochii and B. innocens 33 ), and a real-time PCR assay targeting the nox gene of “B. sp. SASK30446” was developed based on the nox gene region previously described 26 and utilizing as a probe 5′-/5Cy3/AGAAGGATTAAAACAAGAAGGTACTG/3IAbRQSp/-3′. A commercial software package b was used for sequence alignment and probe design. In addition, all strongly beta-hemolytic isolates were tested in another assay described as specific for B. hyodysenteriae. 18 All isolates were tested by each assay prior to inoculation and then again postinoculation against cultures isolated from rectal swabs or mucosal scrapings.
Sequencing of the nox gene of each isolate was performed using previously published forward and reverse primers, 25 and the sequences obtained were compared with those available in GenBank using a commercial software package b ; sequences of PCR amplicons generated from the 16S ribosomal RNA (rRNA) gene were also compared. BLAST analysis (http://blast.ncbi.nlm.nih.gov/Blast.cgi) of the sequences obtained by PCR from strongly beta-hemolytic isolates identified as B. intermedia was performed to determine percent similarity with previously published sequences.
Animal observations and necropsy
Following inoculation, animals were observed at least twice daily for feed and water consumption and signs of clinical illness. Each pen was evaluated daily for evidence of fecal blood and/or mucus, and pigs were weighed weekly. At 7 and 14 days postinoculation (DPI), the fecal consistency of each pig was determined, and each pig received a score based upon the following system: 0 if normal, 1 if soft but formed, 2 if unformed with a semisolid consistency, and 3 if severely liquid to watery with an additional 0.5 point added each for the presence of discernible mucus and/or blood. Animals were euthanized by barbiturate overdose 7 days after the first observation of fecal blood and/or mucus or at the termination of the study 16 DPI. At necropsy, the entire intestinal tract was examined for gross lesions, and the cecum and colon were evaluated for the presence or absence of excessive luminal mucus, mucosal hemorrhage, and fibrinous exudate on the mucosal surface. Samples of liver, jejunum, ileum, cecum, spiral colon, and descending colon were placed immediately in 10% neutral buffered formalin for 24 hr and then processed routinely for histopathology. Samples, including a rectal swab, feces from the spiral colon, and colonic mucosal scrapings, were also collected from each pig for Brachyspira culture.
Microbial culture
Pooled fecal content from each pen, individual rectal swabs from each pig, and fresh mucosal scrapings obtained at necropsy were plated onto BJ and CVS agar within 6 hr of collection and incubated as described. Plates were observed for growth at 2, 4, and 6 DPI. Pooled feces collected from each pen on the last day of the study were used to inoculate MacConkey agar and brilliant green agar to screen for Salmonella spp.
Histopathology and histochemistry
Sections were cut to 4 µm and stained with hematoxylin and eosin. Sections of liver, jejunum, and ileum were evaluated for the presence of lesions unrelated to Brachyspira infection that might potentially have contributed to clinical diarrhea. Sections of cecum, spiral colon, and descending colon were evaluated for the presence of superficial necrosis, mucosal hemorrhage, goblet cell hyperplasia with excessive mucus production, crypt abscesses, and suppurative infiltration of the lamina propria. A suppurative inflammation score (SI) was determined for each section as follows: 0 if no neutrophilic infiltrates were identified, 1 for multifocal infiltrates of less than 10 neutrophils each, 2 for multifocal infiltrates of 10–20 neutrophils each, and 3 for multifocal infiltrates of greater than 20 neutrophils each. Superficial necrosis was scored as follows: 0 if no epithelial necrosis was observed, 1 for multifocal necrosis spanning less than 3 crypts, 2 for necrosis spanning 3–5 crypts, and 3 for necrosis spanning more than 5 crypts. A composite typhlocolitis score (TS) was then determined for each pig as the sum of cecal SI, spiral colon SI, descending colon SI, superficial necrosis score, 1 point for hemorrhage, 1 point for goblet cell hyperplasia, and 1 point for crypt abscesses if more than 15 crypts were affected. The maximum possible TS under this system is 15. Mucosal thickness was measured in each section using a standard eyepiece micrometer, and measurements were taken from an area where the crypts were perpendicular to the mucosal surface and where the epithelium was intact. Serial sections of spiral colon were stained with Warthin–Starry silver stain and evaluated for the presence of characteristic large spirochetes within colonic crypts and luminal contents. All histologic sections were evaluated by 2 separate pathologists (ER Burrough, BL Wilberts) blinded to the treatment groups, and the average of these 2 scores was used in all analyses.
Statistical analyses
A commercial statistical software package c was utilized to perform all analyses. A 1-way analysis of variance was used to detect differences in mean clinical diarrhea scores, average daily gain, mean typhlocolitis scores, and colonic crypt depth, with a Tukey adjustment for multiple comparisons. A Fisher exact test for binomial variables was used to compare the presence or absence of grossly observable colonic luminal mucus. In all circumstances, P values ≤ 0.05 were considered significant.
Results
Animal observations
Three pigs (1 each from groups 2, 7, and 8) developed severe respiratory disease within 72 hr postinoculation and were removed from the study and from all analyses. Diarrhea was first observed at 5 DPI in group 9. Diarrhea scores at 7 and 14 DPI are summarized by group in Table 2. At 7 DPI, the diarrhea scores for groups receiving strongly beta-hemolytic Brachyspira spp. (mean: 0.964 ± 0.210) were numerically higher than those from groups receiving weakly beta-hemolytic isolates (mean: 0.353 ± 0.119) or non-inoculated controls (mean: 0.167 ± 0.167); however, these differences were not significant (P = 0.0764 and P = 0.1261, respectively). At 14 DPI, diarrhea scores were considerably higher for the groups receiving strongly beta-hemolytic Brachyspira spp. (mean: 1.571 ± 0.246) than for groups receiving weakly beta-hemolytic isolates (mean: 0.441 ± 0.171) or non-inoculated controls (mean: 0.333 ± 0.211), and these differences were statistically significant (P = 0.0035 and P = 0.0351, respectively). Grossly detectable fecal blood and/or mucus (Fig. 1A, 1B) was first observed at 7 DPI in group 9, at 8 DPI in groups 5 and 7, and at 9 DPI in groups 6 and 8. No fecal blood or mucus was observed in any pig in groups 1–4 for the duration of the study. Average daily gain was lowest in pigs receiving strongly beta-hemolytic isolates (mean: 0.327 ± 0.023 kg/day) when compared with those receiving weakly beta-hemolytic isolates (mean: 0.418 ± 0.027 kg/day) and non-inoculated controls (mean: 0.378 ± 0.043 kg/day); however, this difference was not statistically significant (P = 0.0508).
Summary of clinical disease and diarrhea scores by group.
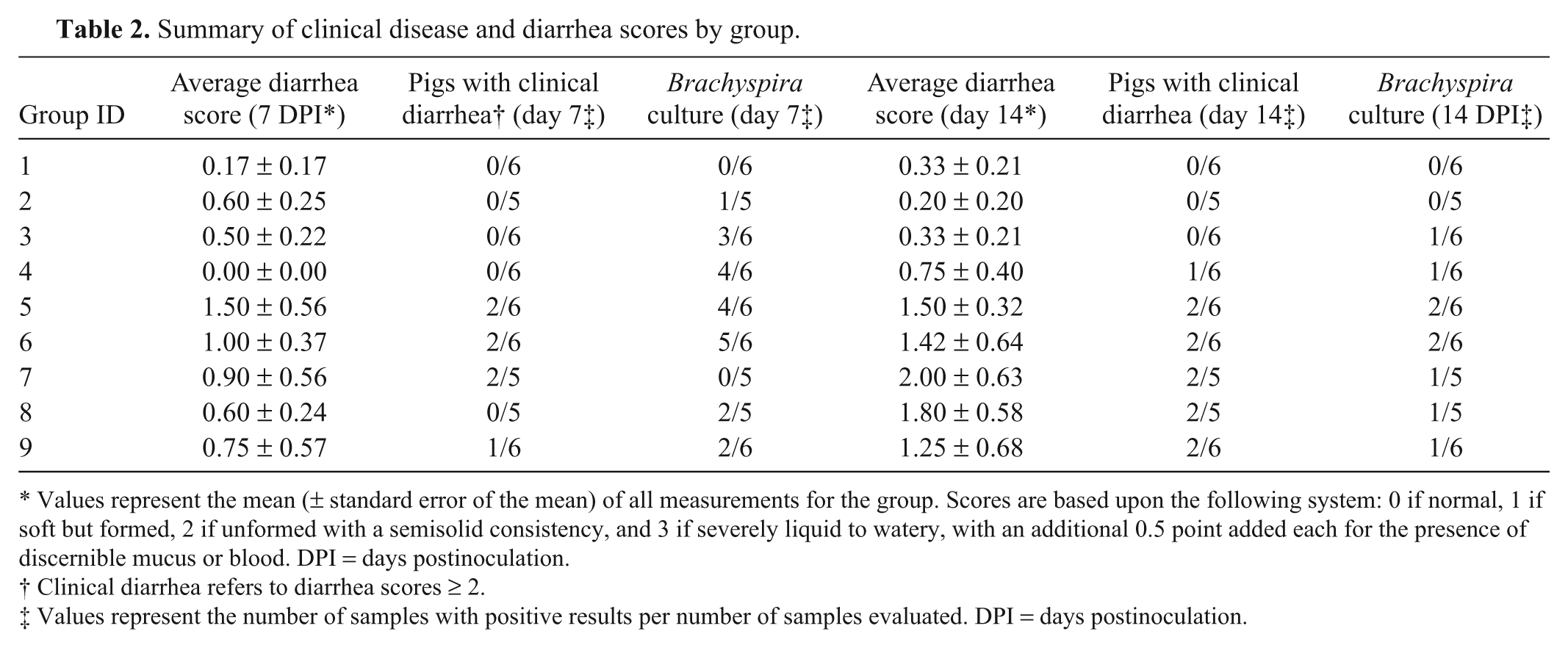
Values represent the mean (± standard error of the mean) of all measurements for the group. Scores are based upon the following system: 0 if normal, 1 if soft but formed, 2 if unformed with a semisolid consistency, and 3 if severely liquid to watery, with an additional 0.5 point added each for the presence of discernible mucus or blood. DPI = days postinoculation.
Clinical diarrhea refers to diarrhea scores ≥ 2.
Values represent the number of samples with positive results per number of samples evaluated. DPI = days postinoculation.
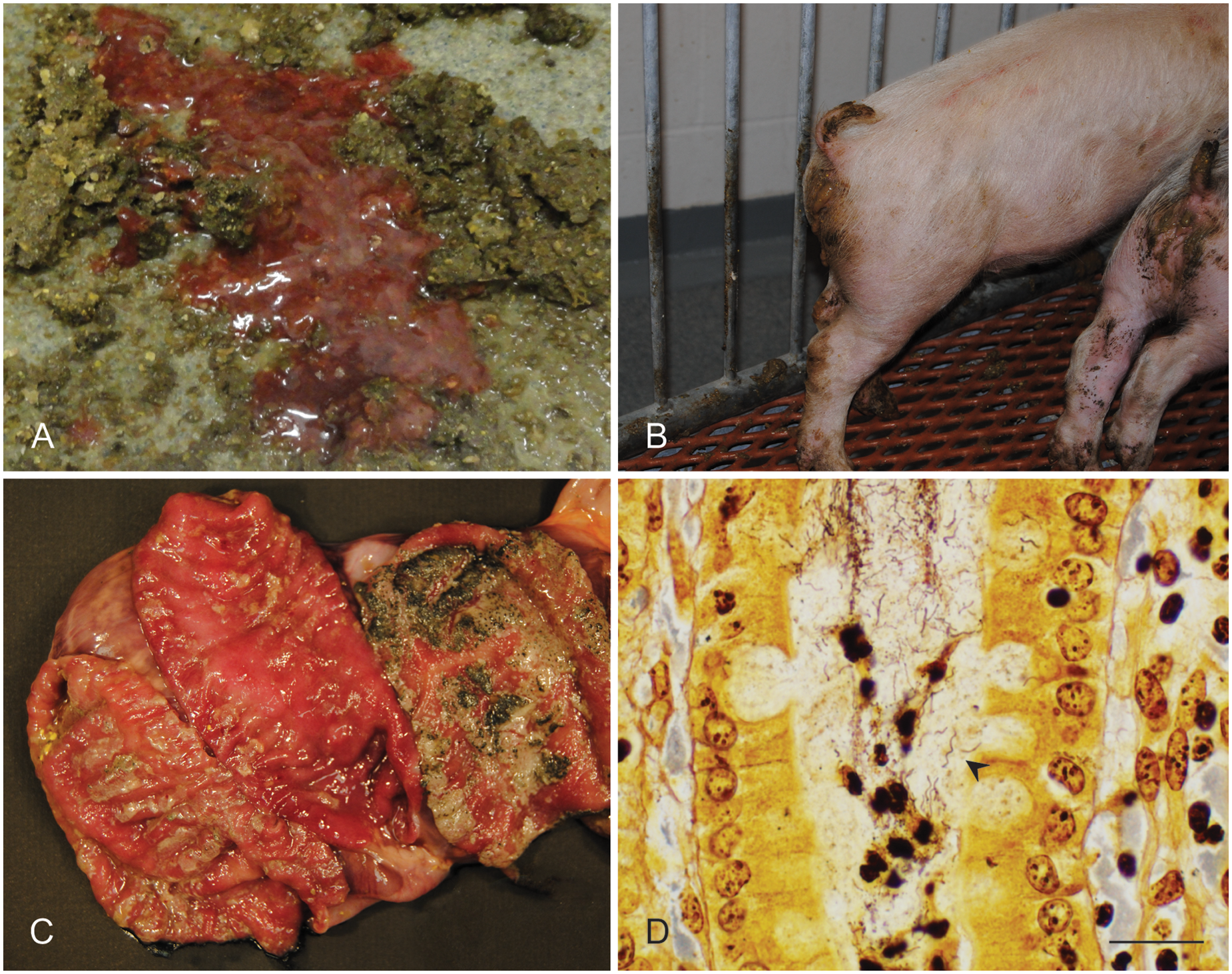
Clinical, macroscopic, histopathologic, and histochemical features of Brachyspira-associated colitis in 6 to 7-week-old pigs.
Gross and microscopic lesions
Gross and microscopic lesions observed in each group are summarized in Table 3. Groups were also combined by cultural phenotype or PCR identification of the inoculum for the purpose of statistical analyses.
Summary of lesions by group.*
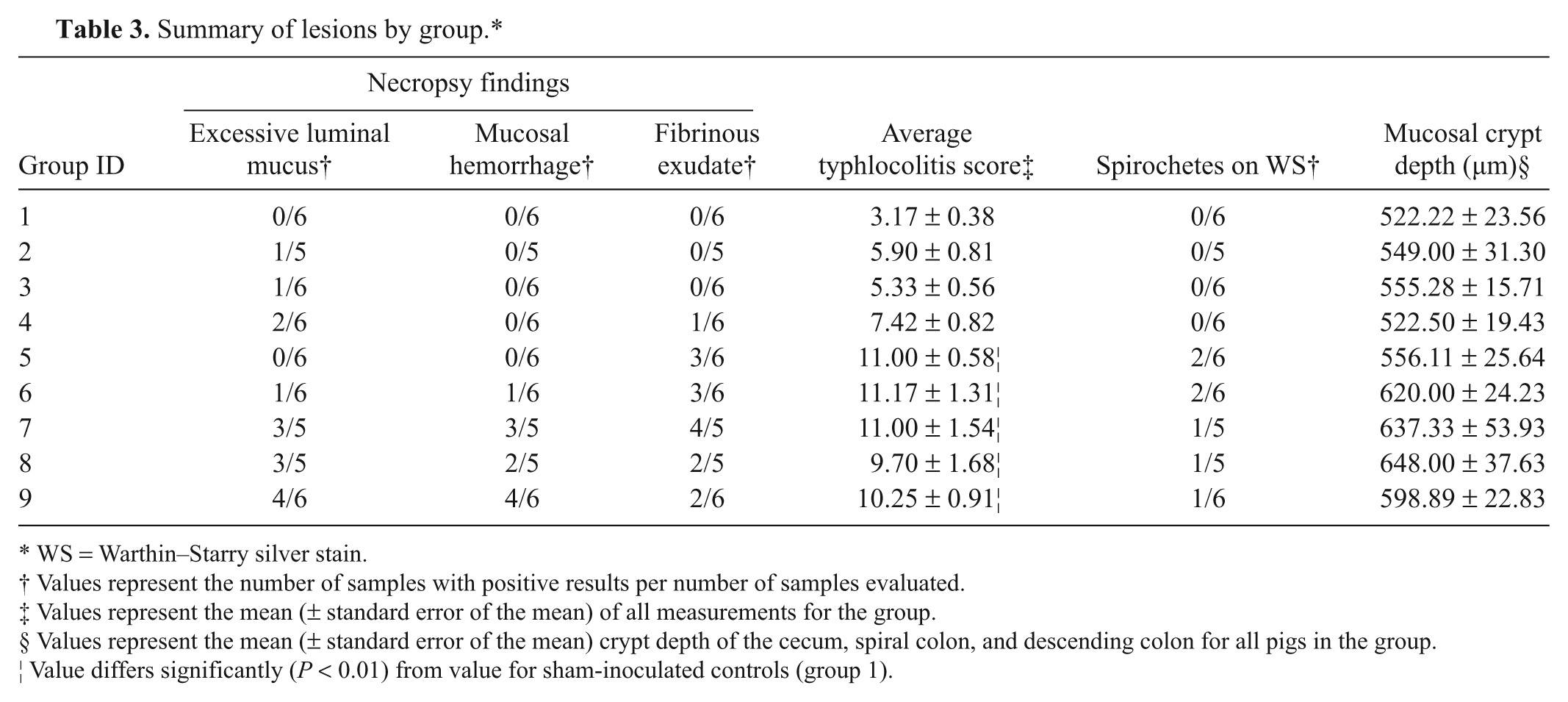
WS = Warthin–Starry silver stain.
Values represent the number of samples with positive results per number of samples evaluated.
Values represent the mean (± standard error of the mean) of all measurements for the group.
Values represent the mean (± standard error of the mean) crypt depth of the cecum, spiral colon, and descending colon for all pigs in the group.
Value differs significantly (P < 0.01) from value for sham-inoculated controls (group 1).
When present, gross lesions were limited to the ceca and large intestines and consisted of variable degrees of mucosal thickening, multifocal mucosal congestion and hemorrhage (Fig. 1C), fibrinous exudate on the luminal surface (Fig. 1C), and excessive luminal mucus. The incidence of excessive luminal mucus was significantly higher in groups inoculated with “B. sp. SASK30446” and B. hyodysenteriae than those inoculated with other isolates (P = 0.0149).
Microscopic lesions were limited to the ceca and large intestines and consisted of varying degrees of neutrophilic and lymphoplasmacytic infiltration of the lamina propria and variable luminal and interstitial hemorrhage. There was multifocal superficial mucosal necrosis, surface and luminal accumulations of neutrophils admixed with mucus, mucosal crypt elongation with variable goblet cell hyperplasia, and variable crypt ectasia with excessive mucus and/or neutrophils. Large delicate spirochetes with features typical of Brachyspira spp. were often noted within ectatic crypts and associated goblet cells by Warthin–Starry staining (Fig. 1D). Groups inoculated with strongly beta-hemolytic Brachyspira spp. had a significantly greater TS than those receiving weakly beta-hemolytic isolates or non-inoculated controls (P < 0.0001; Fig. 2A), and significant differences were also detected when the data was analyzed by molecular identification of the isolates (P < 0.0001; Fig. 2B). Weakly beta-hemolytic isolates as a group were associated with a higher TS than controls (P = 0.0209); however, when analyzed individually, these differences were not maintained (P ≥ 0.0960). No differences were detected between the TS of groups inoculated with any of the strongly beta-hemolytic isolates (P ≥ 0.9785, all analyses), or within groups inoculated with weakly beta-hemolytic isolates (P = 0.9990).
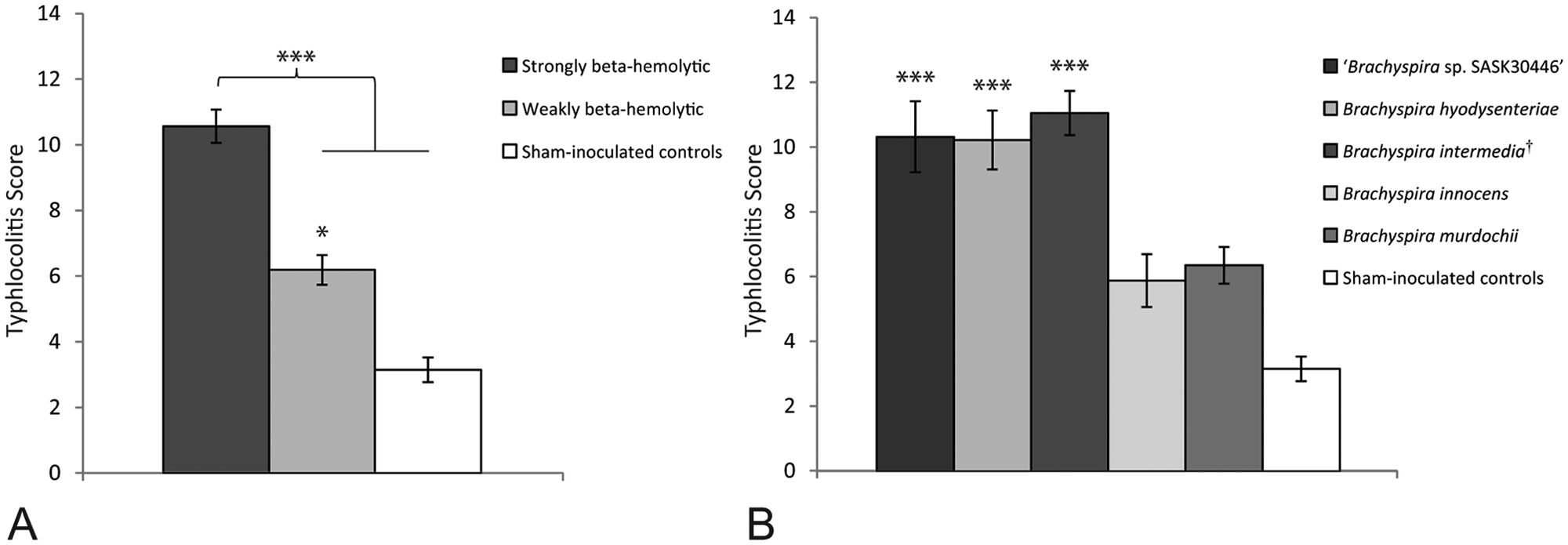
Typhlocolitis scores (TS) from pigs infected with various Brachyspira spp.
Mucosal crypt depth was significantly greater in large intestinal samples from groups inoculated with strongly beta-hemolytic Brachyspira spp. (mean: 609.88 µm ± 14.96) versus those from groups receiving weakly beta-hemolytic isolates (mean: 541.86 µm ± 12.41) or non-inoculated controls (mean: 522.22 µm ± 23.56; P = 0.0065 and P = 0.0183, respectively), whereas no difference was observed between groups receiving weakly beta-hemolytic isolates and controls (P = 0.8205).
Microbial culture
All preinoculation fecal samples were negative by bacteriologic culture for strongly beta-hemolytic Brachyspira spp.; however, 3 out of 54 samples (2 from group 5 and 1 from group 9) were positive for weakly beta-hemolytic Brachyspira spp., and PCR identified these isolates as B. murdochii. Culture results from 7 DPI and necropsy are summarized in Table 4. In general, there were fewer positive cultures at necropsy than at 7 DPI. The phenotypic growth characteristics of all positive necropsy specimens matched their respective inocula. Mucosal scrapings provided the highest yield of positive cultures from necropsy specimens, and there were no instances where an individual rectal swab was positive without a corresponding positive mucosal scraping. Direct cultures of pooled fecal samples collected at necropsy were negative for Salmonella spp. for all groups.
Summary of microbial cultures and polymerase chain reaction (PCR) results by group.*
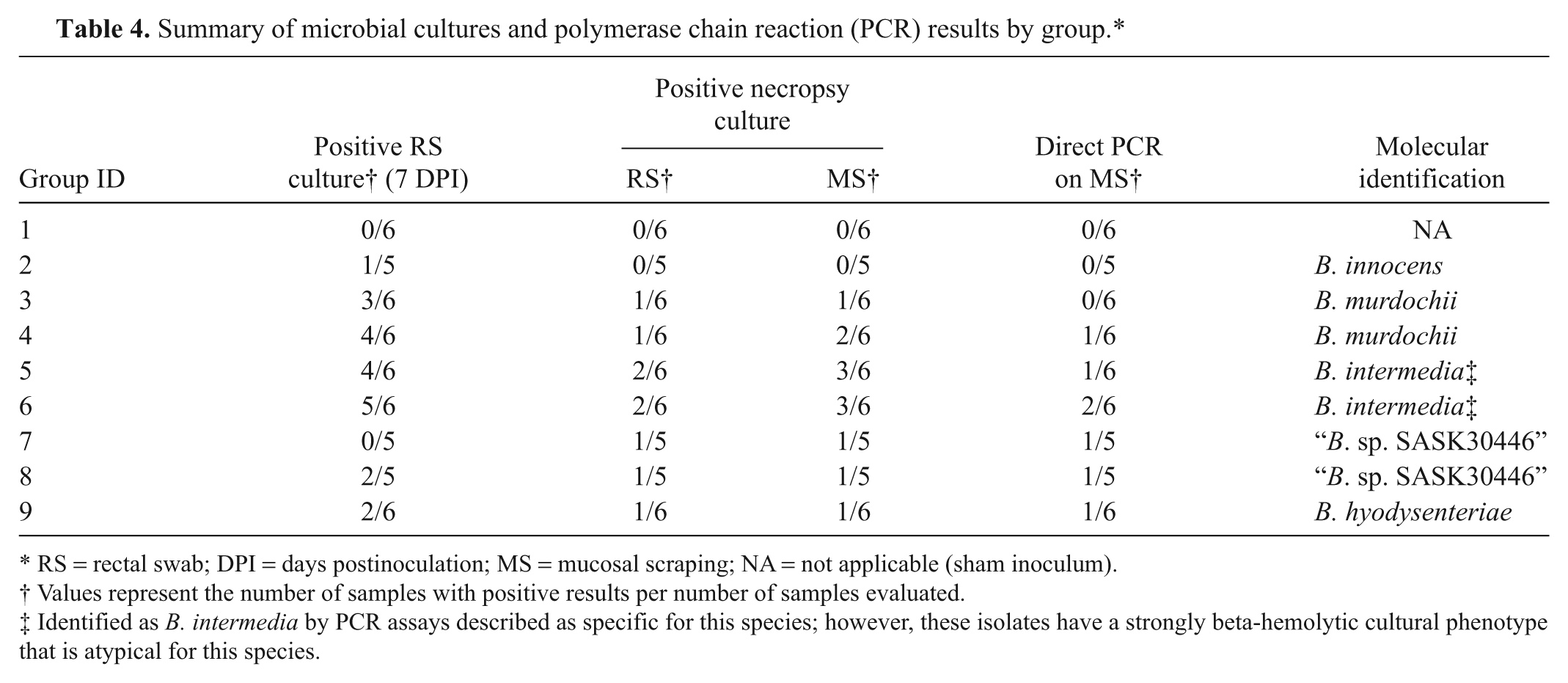
RS = rectal swab; DPI = days postinoculation; MS = mucosal scraping; NA = not applicable (sham inoculum).
Values represent the number of samples with positive results per number of samples evaluated.
Identified as B. intermedia by PCR assays described as specific for this species; however, these isolates have a strongly beta-hemolytic cultural phenotype that is atypical for this species.
Molecular identification
The strongly beta-hemolytic isolate administered to group 9 (B. hyodysenteriae B204) was identified as B. hyodysenteriae by 2 separate PCR assays described as specific for this species,18,26 whereas all other strongly beta-hemolytic isolates used in the current study were not identified as B. hyodysenteriae by either assay. Instead, these strongly hemolytic isolates were singly positive for either B. intermedia (KC35, EB106) or “B. sp. SASK30446” (KC58, EB107). All isolates used for inoculation were singly positive for 1 species in all testing, and no isolates tested positive for B. pilosicoli. For all necropsy specimens with positive culture results, a single Brachyspira sp. was detected by PCR from a representative culture from each group, and this molecular identity matched that of the respective challenge inocula for all groups. Direct PCR was also performed on all mucosal scrapings collected at necropsy and, when positive, revealed the presence of a single Brachyspira sp. (Table 4). In all instances, the direct PCR results were in agreement with the challenge inoculum for each group as well as the individual culture results for the same sample.
Comparison of the 16S rRNA gene sequences of the isolates used in the current study and 33 reference sequences available in GenBank was not discriminatory by species as no clustering of strains was noted by species and the maximum separation between strains was less than 4 nucleotides per 100 bases (data not shown). Conversely, comparison of the nox gene sequences was very effective in separating reference strains and study isolates into distinct clusters (Fig. 3). In this analysis, the “B. sp. SASK30446” and strongly beta-hemolytic isolates identified as B. intermedia by PCR are clearly separated from reference strains of both B. hyodysenteriae and B. intermedia as well as published sequences of “B. suanatina”.
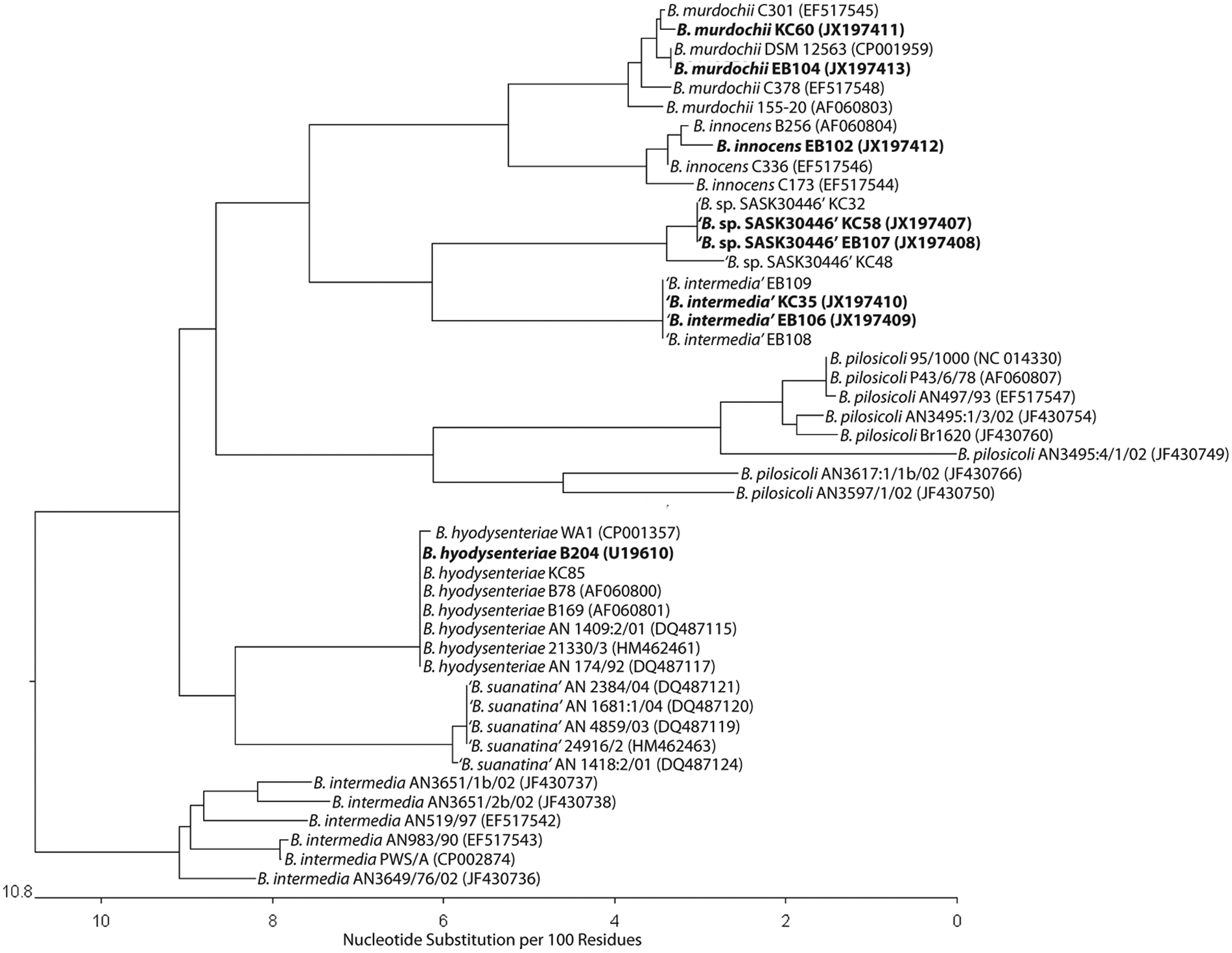
Comparison of nox gene sequences from multiple Brachyspira spp. representing 5 official species and 3 putative species. Isolates identified as “B. intermedia” have a strongly beta-hemolytic cultural phenotype as compared with the weakly beta-hemolytic phenotype typically associated with B. intermedia. Isolates used in the current study are designated in bold type. GenBank accession numbers are in parentheses.
BLAST analysis of the 16S rRNA sequences from the strongly beta-hemolytic isolates identified as B. intermedia by PCR (GenBank accession nos. JX197405 and JX197406) revealed greatest sequence similarity (99%) to B. intermedia PWS/A; however, the similarity of the nox gene sequences of these strains (GenBank accession nos. JX197409 and JX197410) and PWS/A was only 92%. The nox gene sequences of the strongly beta-hemolytic isolates identified as B. intermedia by PCR were most similar (96%) to a previously reported atypical strongly hemolytic spirochete identified as Serpulina sp. P280/1. 1
Discussion
In the current study, multiple lines of evidence reveal a strong association between the cultural phenotype of Brachyspira spp. and the ability of these isolates to induce dysentery-like disease in pigs. Strongly beta-hemolytic, ring phenomenon–positive isolates were associated with greater virulence including increased clinical diarrhea scores at 14 DPI, grossly detectable fecal blood and mucus, increased mucosal crypt depth, and increased severity of typhlocolitis. This is in agreement with previous work in CF1 mice 15 and C3H/HeN mice 3 where a correlation was noted between the strength of beta-hemolysis and enteropathogenicity.
Historically, cultural phenotype and biochemical testing were used for laboratory differentiation of Brachyspira spp. A 4-group system was proposed whereby group I included all strongly beta-hemolytic, indole-positive isolates. 6 Group I has historically included only 1 species, B. hyodysenteriae. Over time, subsets of strongly beta-hemolytic, indole-negative isolates were identified,7,31 and the system was amended to allow for variability in indole production. 7 This is particularly significant as dysentery-like disease has been induced in pigs by experimental inoculation with these indole-negative, strongly beta-hemolytic isolates. 31 Weakly beta-hemolytic isolates are further subdivided within this phenotypic classification system by biochemical means, and isolates of B. intermedia traditionally share all characteristics of B. hyodysenteriae except for the degree of beta-hemolysis. This is of particular note as, without the aid of PCR, any strongly beta-hemolytic isolates of B. intermedia, such as those evaluated in the present study and reported elsewhere, 5 would be classified as B. hyodysenteriae on the basis of phenotype alone. Furthermore, the recently described “Brachyspira suanatina”, 24 and other potentially novel Brachyspira spp. such as the “B. sp. SASK30446” evaluated herein, would be categorized as group I isolates due to their strongly beta-hemolytic phenotype and would be consistent with B. hyodysenteriae under this previously described system.
Considerable strain diversity has been reported with B. intermedia and, given this degree of diversity, several authors have suggested that isolates currently defined as B. intermedia may actually represent multiple distinct species. 21 Brachyspira intermedia has traditionally been viewed as either nonpathogenic or only mildly pathogenic for pigs.6,16,19 However, the data of the current study suggests that isolates detected as B. intermedia by PCR with a strongly beta-hemolytic phenotype are highly virulent. These results support the notion that isolates identified as B. intermedia by currently available PCR assays are indeed diverse, and may reflect more than 1 species. Additional molecular investigation of these isolates by methods such as pulsed field gel electrophoresis, multilocus sequence typing, or full genome sequencing is needed to determine if the strongly beta-hemolytic isolates are actually B. intermedia or if they represent some other species.
It is clear that the advent of PCR has led to greater specificity in identification of infectious agents and that this assay is useful for detection and differentiation of pathogenic microorganisms at the nucleic acid level. For diagnosis of Brachyspira-associated disease, however, this increased specificity is not without pitfalls as the overall sensitivity of the assay, from a diagnostic perspective, depends upon the individual species targeted and may therefore not account for all potentially pathogenic species within the genus when applied to clinical specimens. Accordingly, PCR assays described as specific for B. hyodysenteriae were negative when applied to pure cultures of a strongly beta-hemolytic, indole-positive Brachyspira sp. isolated from pigs and mallards and prompted the proposal of a new species, “B. suanatina”. 24 Since the later part of 2008, PCR assays described as specific for B. hyodysenteriae also failed to detect multiple strongly beta-hemolytic Brachyspira spp. isolated from pigs with clinical disease typical of SD that presented to the ISU VDL. These isolates were ultimately identified as “B. sp. SASK30446” and B. intermedia by PCR, and a subset of these isolates were included in the study reported herein. PCR assays have been described for the simultaneous detection of B. hyodysenteriae and B. pilosicoli from porcine fecal samples.18,26 However, these assays do not specifically detect defined virulence genes, and the results of the current study and others 24 suggest that caution should be applied in the clinical interpretation of the results of such assays, as potentially virulent Brachyspira spp. may go undetected.
The results of the current study and others 23 suggest that direct PCR for certain Brachyspira spp. from clinical specimens may be less sensitive than culture. In the current study, direct PCR from mucosal scrapings yielded the lowest percentage of positive samples, fewer than both individual rectal swab cultures and the primary cultures from mucosal scrapings. Moreover, direct PCR was negative in 5 out of 12 samples that were positive by direct culture. In contrast, direct culture of these same mucosal scrapings detected spirochetes more frequently than any other assay, suggesting that direct culture of colonic mucosa is ideal for inclusion in the diagnostic approach for submissions from cases with lesions suggestive of SD.
The clinical significance of weakly beta-hemolytic Brachyspira spp. in swine is unclear, as they are commonly present in the colons of pigs with and without diarrhea 12 and often occur as mixed infections. 16 Brachyspira pilosicoli is a well-defined, pathogenic, weakly beta-hemolytic spirochete that is the cause of porcine intestinal spirochetosis, 32 and its virulence has been confirmed experimentally. 30 The potential virulence of other weakly hemolytic species is not as clear. Weakly beta-hemolytic isolates of B. intermedia have been recovered from pigs with diarrhea,2,6 but have not been confirmed as pathogens under experimental conditions. 19 Mild catarrhal colitis has been demonstrated in pigs following experimental challenge with B. murdochii, 14 and this species has been reported as the sole agent in cases of Brachyspira-associated colitis in swine 33 ; however, B. murdochii can also be recovered from pigs without diarrhea 8 and, along with B. innocens, is commonly regarded as a nonpathogenic commensal of the porcine colon. 10 In the present report, the weakly beta-hemolytic Brachyspira spp. evaluated did not appear to be significantly virulent in pigs over the course of this study.
The overall incidence of disease in the current study was somewhat lower than expected, particularly with the strongly beta-hemolytic isolate B. hyodysenteriae B204. It is unknown as to the precise reason for this lower than expected result, but potential causes may include the young age of the pigs (5 weeks at the time of inoculation), previous mouse passage of the B204 isolate which may have reduced its virulence, and housing of the pigs on raised decks which likely prevented significant horizontal spread from shedders throughout the study. While the results have confirmed that strongly beta-hemolytic Brachyspira spp. not detected as B. hyodysenteriae by PCR can induce clinical dysentery in pigs, further study is necessary to fully compare the specific lesions and clinical disease associated with these various strongly beta-hemolytic isolates versus typical B. hyodysenteriae strains. Additionally, the relatively close association of the nox gene sequences from the strongly beta-hemolytic isolates detected as “B. sp. SASK30446” and B. intermedia by PCR suggests these isolates may be related and may belong to the recently proposed novel species “Brachyspira hampsonii”. 4 Full genome sequencing or other molecular analysis is indicated to better determine the relatedness of these putatively novel species.
In summary, the results of the present study demonstrate a strong correlation between the degree of beta-hemolysis and the potential for virulence of Brachyspira spp. in swine, and that multiple strongly beta-hemolytic Brachyspira spp. not identified as B. hyodysenteriae by PCR are capable of inducing clinical disease and lesions similar to those associated with infection by typical B. hyodysenteriae isolates. Accordingly, the results further suggest that culture phenotype may be a more sensitive indicator of potential to induce dysentery-like disease than the molecular identity of the isolate as established by currently available PCR assays, and provide further support for the use of culture as part of any initial screening protocol to detect pathogenic Brachyspira spp. in diagnostic specimens.
Footnotes
Acknowledgements
The authors wish to thank Dorothy Murphy and Dr. Kris Clothier for their help with the Brachyspira culture work and cloning of the isolates, and Dr. Paulo Arruda, Kate Sawyer, Brent Pepin, Marissa Rotolo, and Drew White for their assistance with the animal inoculation and necropsy procedures.
a.
BD GasPak EZ Anaerobe Container System, BD Diagnostic Systems, Sparks, MD.
b.
Lasergene version 8.0, DNASTAR Inc., Madison, WI.
c.
JMP Pro 9.0.0, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared that they had no conflicts of interest with respect to the research, authorship, and/or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Iowa Pork Producers Association and the National Pork Board (NPB #11-178).
