Abstract
A species-independent indirect enzyme-linked immunosorbent assay (iELISA) based on chimeric protein A/G was established for the detection of anti-
Bacterial species of the genus
Many brucellosis serological tests have been used for wildlife species. 13 The slow agglutination of Wright (SAW) is a sensitive test prone to detect immunoglobulin (Ig)M. The specificity is increased by an acid environment, chemical reduction or precipitation of IgM, or pretreating the serum with ethylenediamine tetra-acetic acid (SAW-EDTA). 30 The Rose Bengal test (RBT) and the buffered antigen plate agglutination test are agglutination tests using acidified antigen. Both may give false negatives. 30 The complement fixation test (CFT) generally has a high specificity and is used as a confirmatory test in eradication programs. 30 The fluorescence polarization assay is a sensitive and specific test directly measuring Ab binding to the antigen. 25 The competitive enzyme-linked immunosorbent assay (cELISA) is species unspecific and has a high specificity and a lower sensitivity. 30 The indirect ELISA (iELISA) typically performs with high sensitivity and sometimes restricted specificity, making it suited for screening purposes. 30
Some serological methods rely on species-specific Abs, making them unsuited for screening wildlife.
13
This can be overcome by using the species-unspecific protein A (PrA) or protein G (PrG) as conjugates.
36
Protein A is a 42-kDa polypeptide cell wall constituent of
The aim of the current study was to establish a species-independent PrA/G iELISA for the detection of anti-
Materials and methods
Hooded seal serum samples (
The iELISA was performed as previously described,22,32 with minor modifications. Tween-20,
b
Tween-80,
b
phosphate-citrate buffer,
b
o-phenylenediamine dihydrochloride,
b
PrA,
c
PrG,
d
and PrA/G
e
were purchased commercially. The wells of 96-well polystyrene plates
f
were coated with
Mean optical density (OD) from a set of positive (positive with all tests) and negative (negative with all tests) samples from each species tested with protein A, protein G, and protein A/G in the indirect enzyme-linked immunosorbent assay.*
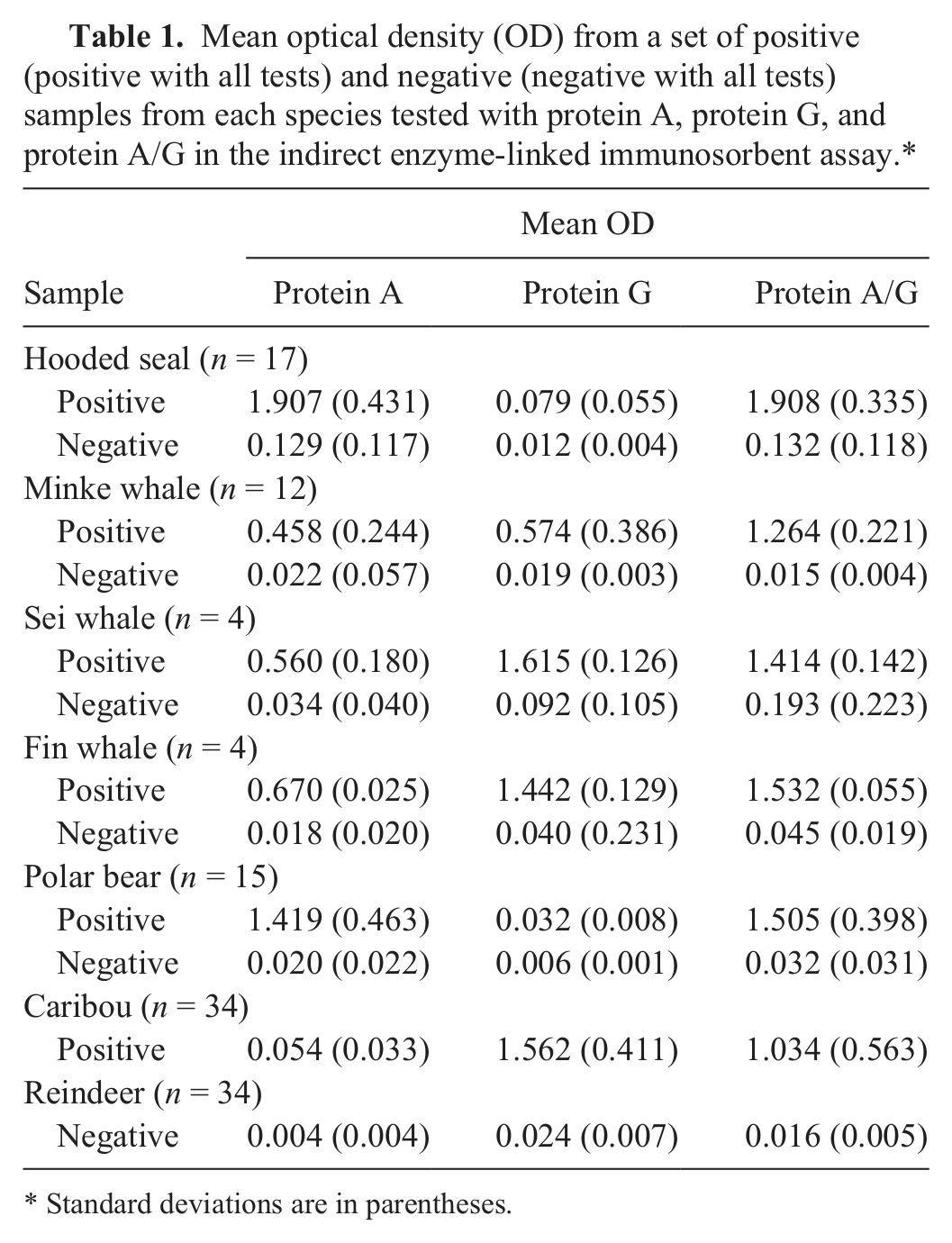
Standard deviations are in parentheses.
Statistical analysis was conducted using StataSE 11
h
and R (http://www.R-project.org/). The proportion of samples positive in each test was calculated with 95% confidence intervals (CI) using general linear models with a binomial distribution and logit link. To assess pairwise coherence among the different serological tests, Cohen kappa (κ) was calculated.
7
Receiver operator characteristic (ROC) analysis
12
was performed on the PrA/G iELISA results from the reindeer and caribou using the bacteriology-positive caribou as true positives.
2
The sensitivity (the proportion of positives in the PrA/G iELISA that were from the bacteriology-positive caribou) and specificity (the proportion of negatives in the PrA/G iELISA that were from the negative reindeer from Norway) of the PrA/G iELISA for reindeer and caribou was calculated. The Spearman rank correlation coefficient (
Results
Hooded seal
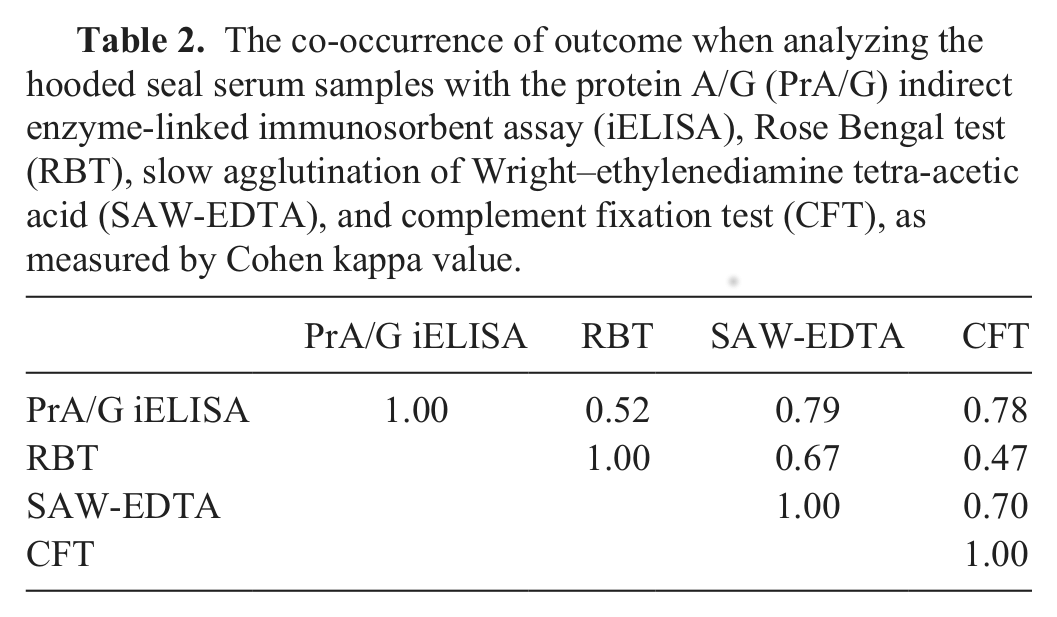
The mean value of the %P for samples negative in CFT, SAW-EDTA, and RBT (125/172) was 18.1 %P with a SD of 21.5 %P, providing a cutoff for the PrA/G iELISA for hooded seals of 73.6 %P. The PrA/G iELISA yielded a seroprevalence of 23.8% (41/172, 95% CI: 17.4–30.3), while the seroprevalence found with CFT was 26.2% (45/172, 95% CI: 19.5–32.8), followed by SAW-EDTA (31/172, 18.0%, 95% CI: 12.2–23.8) and RBT (17/172, 9.9%, 95% CI: 5.4–14.4). There was a good pairwise fit between the tests (Cohen kappa; Table 2). The proportion of samples positive in 1 or 2 of the CFT, SAW-EDTA, and RBT was 17.4% (30/172, 95% CI: 11.7–23.2). The proportion of samples positive in CFT, SAW-EDTA, and RBT simultaneously was 9.9% (17/172, 95% CI: 5.4–14.4). Boxplots show the distribution of the %P in the PrA/G iELISA for the groups “negative in CFT, SAW-EDTA, and RBT,” “positive in 1 or 2 out of CFT, SAW-EDTA, and RBT,” and “positive in CFT, SAW-EDTA, and RBT” (Fig. 1). The hooded seal sera reacted stronger with PrA and PrA/G, compared to PrG (Table 1). The %P obtained with PrA and PrA/G in the iELISA were well correlated (
The co-occurrence of outcome when analyzing the hooded seal serum samples with the protein A/G (PrA/G) indirect enzyme-linked immunosorbent assay (iELISA), Rose Bengal test (RBT), slow agglutination of Wright–ethylenediamine tetra-acetic acid (SAW-EDTA), and complement fixation test (CFT), as measured by Cohen kappa value.
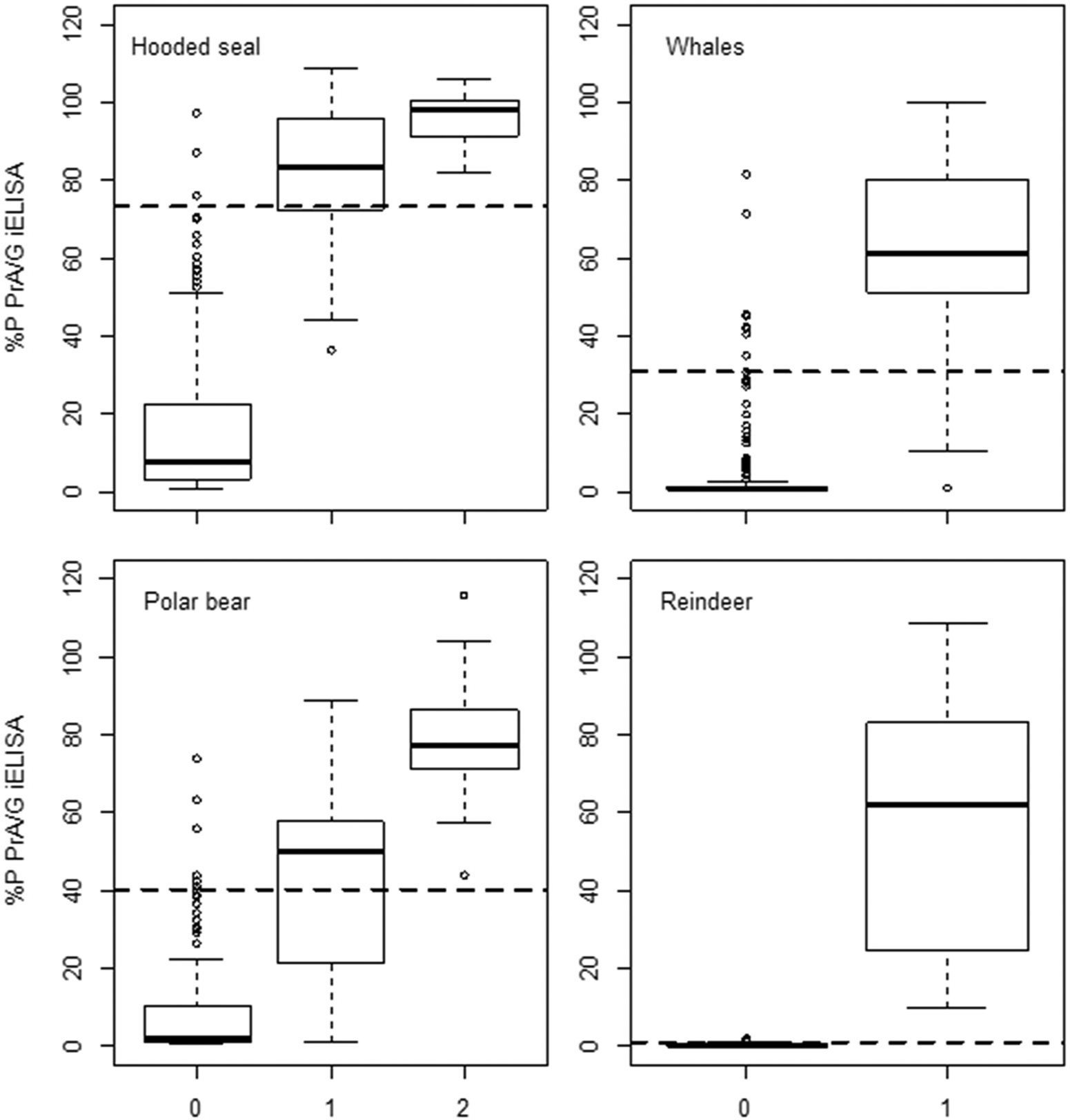
Boxplots showing the median, the 25% and 75% quartiles, whiskers (1.5 times the interquartile range), and the outliers for the protein A/G (PrA/G) indirect enzyme-linked immunosorbent assay (iELISA) percent positivity ([optical density sample/optical density positive control] × 100 = percent positivity [%P]) for each species. Categories for hooded seal: 0 = negative in complement fixation test (CFT), slow agglutination of Wright–ethylenediamine tetra-acetic acid (SAW-EDTA), and Rose Bengal test (RBT); 1 = positive in 1 or 2 out of CFT, SAW-EDTA, and RBT; and 2 = positive in CFT, SAW-EDTA, and RBT. Categories for whales: 0 = negative in the previous serological study
37
; and 1 = positive in the previous serological study.
37
Categories for polar bear: 0 = negative in SAW, SAW-EDTA, and RBT; 1= positive in 1 or 2 out of SAW, SAW-EDTA, and RBT; and 2 = positive in SAW, SAW-EDTA, and RBT. Categories for reindeer: 0 = bacteriology-negative reindeer from Norway; and 1 =
Whales
Data showing which samples were positive in all tests simultaneously was obtained from a previous study
37
on whales (SAW, SAW-EDTA, RBT, CFT, PrG iELISA, and an anticomplement ELISA; 28/297, 9.4%, 95% CI: 6.1–12.8). The mean value of the %P of whales previously found negative (269/297) was 4.0 %P with a SD of 10.4 %P, providing a cutoff for the PrA/G iELISA for whales of 30.8 %P. The PrA/G iELISA yielded a seroprevalence of 12.2% (23/189, 95% CI: 7.5–16.9) for minke whales, 5.3% (4/76, 95% CI: 0.1–10.4) for fin whales, and 12.5% (4/32, 95% CI: 0.39–24.6) for sei whales. When comparing the PrA/G iELISA results with the previous results,
37
there was a good pairwise fit between the results (Cohen kappa); κ = 0.69 for minke whales (negative in both studies: 164/189; diverging results in the two studies: 11/189; positive in both studies: 14/189), κ = 0.64 for fin whales (negative in both studies: 68/76; diverging results in the 2 studies: 4/76; positive in both studies: 4/76), κ = 1.00 for sei whales (negative in both studies: 28/32; positive in both studies: 4/32), and κ = 0.72 for the whales as a group (negative in both studies: 260/297; diverging results in the two studies: 15/297; positive in both studies: 22/297). Figure 1 illustrates the distribution of the %P in the PrA/G iELISA for the groups “negative in the previous study” and “positive in the previous study” by boxplots. The sei and fin whale Abs reacted stronger with PrG than with PrA, while the minke whale Abs reacted equally with PrA and PrG. All whale species reacted with PrA/G (Table 1). The %P obtained with PrG and PrA/G in the iELISA were well correlated (
Polar bear
The mean value of the %P of samples negative in SAW, SAW-EDTA, and RBT
36
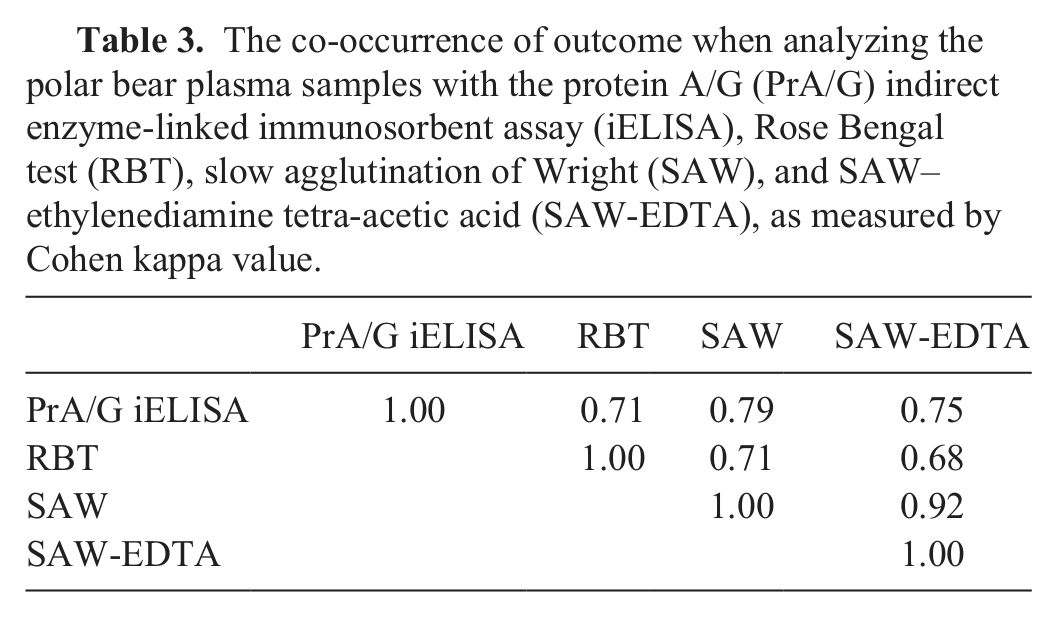
(198/230) was 8.2 %P with a SD of 12.5 %P, providing a cutoff for the PrA/G iELISA for polar bears of 40.5 %P. The PrA/G iELISA yielded a seroprevalence of 13.5% (31/230, 95% CI: 9.0–17.9), while the seroprevalence found with SAW-EDTA was 13.0% (30/230, 95% CI: 8.7–17.4), followed by SAW (28/230, 12.2%, 95% CI: 7.9–16.4) and RBT (18/230, 7.8%, 95% CI: 4.3–11.3). There was a good pairwise fit between the tests (Cohen kappa; Table 3). The proportion of samples positive in 1 or 2 of the tests SAW, SAW-EDTA, and RBT was 6.5% (15/230, 95% CI: 3.3–9.7). The prevalence found when considering samples only as positive if they were positive in SAW, SAW-EDTA, and RBT simultaneously was 7.4% (17/230, 95% CI: 4.0–10.8). The distribution of the %P in the PrA/G iELISA for the groups “negative in SAW, SAW-EDTA, and RBT,” “positive in 1 or 2 out of SAW, SAW-EDTA, and RBT,” and “positive in SAW, SAW-EDTA, and RBT” are illustrated in Figure 1. The polar bear Abs reacted less with PrG as compared to PrA and PrA/G (Table 1). The %P obtained with PrA and PrA/G in the iELISA were well correlated (
The co-occurrence of outcome when analyzing the polar bear plasma samples with the protein A/G (PrA/G) indirect enzyme-linked immunosorbent assay (iELISA), Rose Bengal test (RBT), slow agglutination of Wright (SAW), and SAW–ethylenediamine tetra-acetic acid (SAW-EDTA), as measured by Cohen kappa value.
Reindeer
The mean value for the samples from the negative reindeer from Norway (
Discussion
The PrA/G iELISA offers a serological method for hooded seals, minke whales, sei whales, fin whales, and polar bears, yielding results consistent with established brucellosis serological tests. For reindeer and caribou, the PrA/G iELISA was validated as a sensitive and specific serological method in coherence with a bacteriological gold standard.
When conducting brucellosis serology in wildlife, an important issue is to set, and confirm, a cutoff value. The cutoff value for the reindeer and caribou in the current study was established using the mean value of the %P of reindeer from Norway, utilizing samples that were very likely to be true negatives. ROC analysis of the reindeer and caribou PrA/G iELISA results, using as true positives the bacteriology-positive caribou 2 showed an AUC of 0.99, verifying the cutoff. In the absence of a gold standard, the cutoff for the hooded seal, polar bear, and whales was based on agreement with other serological tests. The present study found that the PrA/G iELISA results for these species, using the suggested cutoffs, were consistent with the results of the other serological tests, as shown by Cohen kappa values ranging from 0.47 to 0.92.
Brucellosis serological tests utilizing species-specific Abs designed for the detection of Abs in a given wildlife species have been developed. 17 For whales, it has been shown that Abs against IgG of toothed whales had a reduced ability to recognize IgG of baleen whales, 28 demonstrating the rationale of producing species-particular Abs against each host species of interest. This often has the drawback of necessitating the production of a finite amount of antispecies Abs in experimental animals that has to be renewed, which is not in line with the principles of reduction, refinement, and replacement of animal testing. 24
Protein A or PrG are good alternatives to species-specific Abs. The Fc-region, and thus the affinity of PrA and PrG to the Fc-region, differs between Abs from different species.
15
Carnivore Abs are shown to react mostly with PrA,
22
and PrA has been used to detect anti-
Protein A/G combines the binding features of PrA and PrG,
11
reacting with Abs from a number of species,
26
and has been used to detect anti-
Two (of the 304) samples from the reindeer from Norway were classified as weak positives (<2.0 %P) in the PrA/G iELISA, but were negative with RBT, CFT, and SAW-EDTA. 39 The samples were regarded as false positives, in agreement with the finding of no seropositive individuals during assaying for brucellosis of more than 5,500 reindeer from Finnmark County, Norway, in 1990–1994, 3 and given that Norway has been free of brucellosis in terrestrial animals for more than 6 decades (Hofshagen M, Lange H, Hauge K: 2011, Norway 2011).
Serological cross-reactions and false positives are major problems when detecting anti-
The aim of the current study was to establish a species-independent PrA/G iELISA for the detection of anti-
Footnotes
Acknowledgements
The authors would like to thank E. M. Breines and E. Hareide at the Norwegian School of Veterinary Science, Section for Arctic Veterinary Medicine, Tromsø, Norway for excellent laboratory work, and C. de Smedt and D. Fretin at the Veterinary and Agrochemical Research Centre, Brussels, Belgium, for helping with design and validation of the iELISA. The authors also acknowledge T. Haug, K. T. Nilssen, A. K. Frie, N-E Skavberg, andM. T. Poltermann at the Institute of Marine Research, Tromsø, Norway, for making the sampling of hooded seals possible; Ø. Wiig, A. Derocher, J. Aars, and M. Andersen for samples from polar bears; and M. Kjeld, A. Alfredson, and A. Arnason, for samples from fin and sei whales from Iceland. The authors greatly appreciate the help from A. Evans in correcting the language.
a.
Venoject tubes for biochemistry (serum; no additives, 10 ml), Terumo Medical Corp., Somerset, NJ.
b.
Sigma Aldrich, St. Louis, MO.
c.
Protein A-Peroxidase from Staphylococcus aureus, horseradish (1 mg/ml, 1:2,000 dilution, HRP:protein 1:2), Sigma Aldrich, St. Louis, MO.
d.
Purified Recomb Protein G, peroxidase conjugated (1 mg/ml, 1:2,000 dilution, HRP:protein 2:1), Thermo Fisher Scientific Inc., Waltham, MA.
e.
Purified Recomb Protein A/G, peroxidase conjugated (1 mg/ml, 1:16,000 dilution, HRP:protein 1:1), Thermo Fisher Scientific Inc., Waltham, MA.
f.
Nunc PolySorp 475094, Thermo Fisher Scientific Inc., Waltham, MA.
g.
Epoch Microplate Spectrophotometer, BioTek Instruments Inc., Winooski, VT.
h.
Stata: Release 11, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by Astri and Birger Torsteds grant, Norway.
