Abstract
Avian leukosis virus (ALV) is associated with tumor development and growth retardation in poultry. Eradication of virus infection at the primary breeder level is the principal method for controlling ALV infection in chickens. An indirect enzyme-linked immunosorbent assay (iELISA) method that utilized the prokaryotically expressed and affinity-purified viral capsid protein antigen p27 was developed for the detection of ALV-specific antibodies in chicken sera. The protocol of iELISA was validated and resulted in a higher agreement value than fluorescent antibody test (FAT) and was shown to be more sensitive and specific compared to the commercial ALV antibody test kit when FAT was used as a reference test. The main advantage of this method is the use of a single immunogenic protein to detect antibodies against all ALV exogenous subgroups. The results show that the developed iELISA is an inexpensive alternative and can potentially be used as a confirmatory test for the presence of anti-ALV antibodies on a large scale.
Keywords
Avian leukosis virus (ALV; family Retroviridae, subfamily Orthoretrovirinae, genus Alpharetrovirus) is known to cause significant economic losses in the poultry industry worldwide due to virus-associated neoplasia and reduced productivity in commercial layer and breeder flocks. Based on antigenic characteristics of the envelope glycoproteins, ALV is categorized into 6 subgroups, A–E and J.2,6 The exogenous ALVs are horizontally and vertically transmitted in chickens, 4 but the endogenous subgroup E is transmitted through Mendelian inheritance.3,6 Conventional diagnostic tests for detection of ALV include virus isolation, fluorescent antibody test (FAT), enzyme-linked immunosorbent assay (ELISA), and reverse transcription polymerase chain reaction (RT-PCR) assays. 1 Of these, ELISA is most widely used because it is sensitive, convenient, and easy to perform. 5 The ALV antigen ELISA is widely accepted as a screening test for detection of group-specific antigen p27 protein, which is common to all subgroups of ALV. 8 Detection of antibody to ALV subgroups A and B has been used extensively by poultry breeders and growers. 7 Moreover, molecularly cloned, baculovirus-expressed envelope glycoprotein gp85 of ALV-J are now being used in commercial ELISA kits that are specific for detection of antibody to ALV-J. 9 The purpose of the current study was to develop an indirect ELISA (iELISA) that utilized the recombinant ALV capsid protein antigen p27 for the diagnosis of ALV infection in chickens and to compare it with commercial ELISA kits and FATs.
For ELISA coated antigen preparation, the p27 gene was amplified with the following reagents in a total volume of 50 µl: 1 µl of genomic DNA (100 ng/µl), which was extracted from the Doug Foster chicken fibroblast-1 (DF-1) cells inoculated with ALV-J strain JS-nt a (HM235667), 1 µl of each primer (25 pmol/µl), 4 µl of 10 mM deoxyribonucleotide triphosphate mixture, 5 µl of 10× LA PCR Buffer II (magnesium plus), b 0.5 µl of LA Taq polymerase, b and 37.5 µl of sterile water. The primer pair was as follows: forward primer (5′-GCGGATCCATGCCTGTAGTGATTAAGACAG-3′), to which the BamHI restriction site was added to the 5′ end, and the reverse primer (5′-GCCTCGAGTTAGGCCGCGGCT ATGCCT-3′), to which the XhoI restriction site was added to the 5′ end. The cycling profile consisted of a denaturation step at 95°C for 3 min, followed by 35 cycles at 94°C for 1 min, 52°C for 1 min, 72°C for 50 sec, followed by final extension at 72°C for 10 min. The sequence of the p27 protein of the ALV-J JS-nt strain had an identity of 98.6% to the HPRS103 strain. The p27 fragment was subcloned into the pGEX-6P-1 vector; the expression protein was purified using a glutathione (GST) resin pre-packed column c and confirmed by Western blot analysis (Fig. 1).
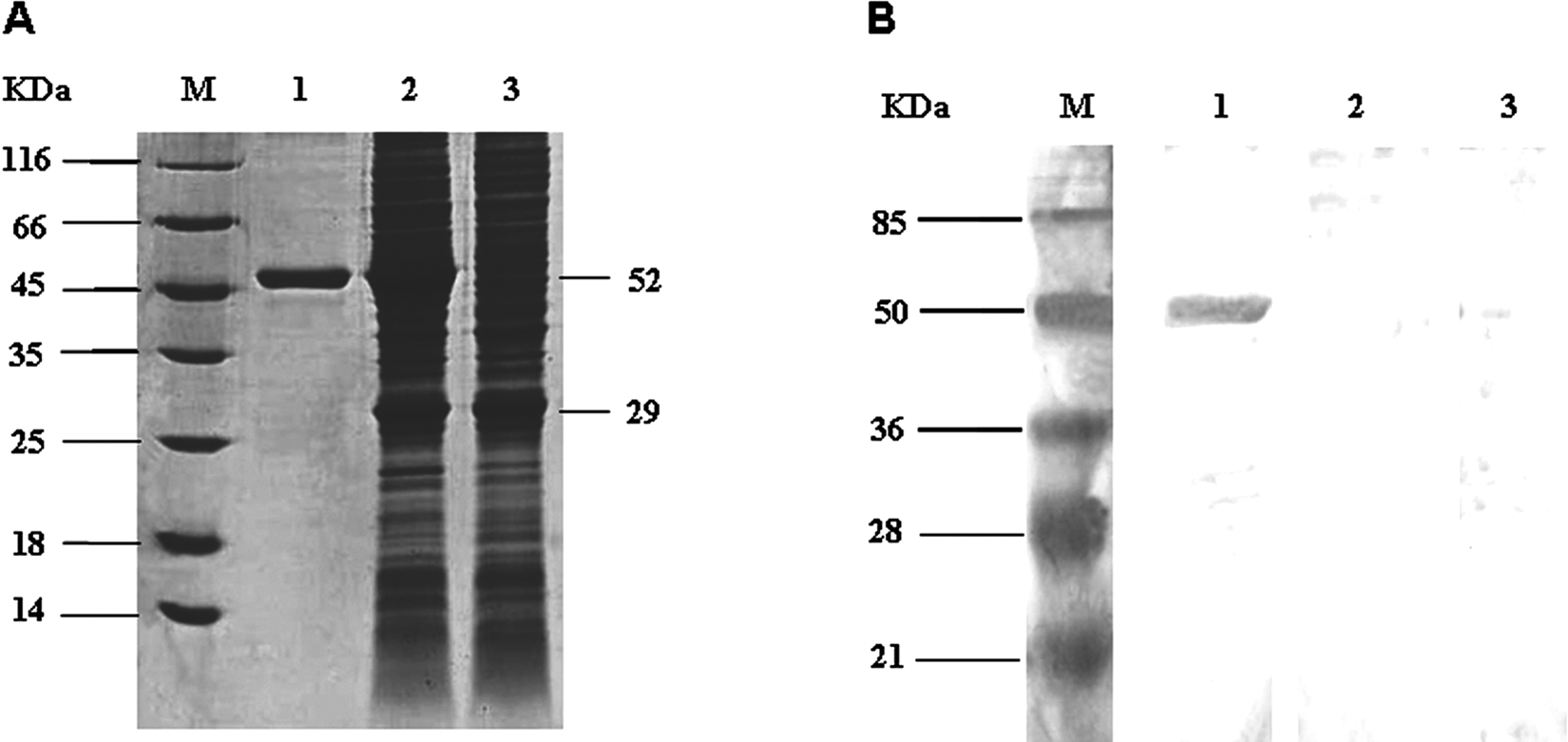
Characterization of expression of p27 recombinant capsid (rCap) fusion protein. A, sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis of the rCap fusion protein. Lane M: protein molecular weight marker; lane 1: purified fusion protein; lane 2: nonpurified soluble bacterial lysate; lane 3: vector control. B, Western blot analysis of the rCap fusion protein. Lane M: protein molecular weight marker; lane 1: purified fusion protein blot incubated with serum from Avian leucosis virus (ALV)-infected chicken; lane 2: purified fusion protein blot incubated with serum negative for ALV; lane 3: vector protein blot incubated with serum from ALV-infected chicken.
The optimal antigen dilution was established by titration against varying dilutions of a reference positive serum. The optimal serum dilution was chosen based on the lowest dilution of positive sera tested with control antigen, which showed absorbance or optical density (OD) values less than or equal to those presented by negative reference serum tested with purified antigen at the same concentration. Purified recombinant p27 protein was coated in 96-well microtiter plates with 5 μg/ml of 0.1 M carbonate/bicarbonate buffer, pH 9.6, overnight at 4°C and then blocked with 5% skimmed milk in 10 mM phosphate buffered saline solution, pH 7.4, containing 0.05% Tween-20 (PBST) for 2 hr at 37°C. The plates were washed 3 times with PBST before 100 µl of sera diluted 1:500 in dilution buffer (PBST with 1/100 volume of 3 mg/ml of Escherichia coli BL21 total lysate) was used for testing. Dilution buffer and negative and positive sera were used as blank and negative and positive controls, respectively. Plates were incubated for 1 hr at 37°C followed by a washing step. Then, 100 µl of the conjugate horseradish-peroxidase–labeled goat anti-chicken antibody (diluted 1:20,000 in the dilution buffer) was added to all wells and incubated at 37°C for 1 hr. After washing again, 100 µl of freshly prepared substrate solution (1 mg/ml O-phenylenediamine that contained 4 µl of 3% hydrogen peroxide) was dispensed into each well. Color development was in the dark for 15 min, and the reaction was stopped by addition of 50 µl of 2 M sulfuric acid. The absorbance values at 490 nm were measured using an ELISA reader. Each sample was analyzed in duplicate. The cut-off of the iELISA was defined as the mean OD value of 150 negative serum samples plus 3 times the standard deviation (SD). The average OD value obtained from these samples was 0.1930, while the calculated SD was 0.1025, which resulted in a cut-off of 0.5.
Testing 10 positive serum samples and 10 negative samples assessed the internal quality control. For each sample, the coefficient of variation (CV) was calculated between plates (interassay variation) and within the same plate (intra-assay variation). Each sample was tested in 4 different plates on different occasions to determine the interassay CV, and 4 replicates within each plate were used to calculate the intra-assay CV. The intra-assay CV ranged from 2.7% to 6.4%, and the interassay CV ranged from 2.5% to 7.2%. Also, there was no evidence of cross-reactivity with known positive sera to other avian viruses, and all assays gave values below the defined cut-off point.
The performance of the developed recombinant capsid protein–based iELISA (rCap-iELISA) was compared with the FAT and commercial antibody kits, d which are ALV antibody test kits for subgroup A, B, and J. The test was run according to the manufacturers’ instructions. The positive rate for the antibody ELISA d was a combined result of both kits. The positive serum samples in the 2 ELISA tests were confirmed by the FAT. Of the 269 serum samples collected from layer hens in Jiangsu Province, 67 were positive and 172 were negative by both ELISA tests, with an overall agreement of 88.8% between the 2 tests. Five of the 67 positive samples were negative by FAT. Thirty samples were discordant between the 2 ELISAs. Nine samples were negative by rCap-iELISA but positive by antibody ELISA, of which 1 was positive by FAT but 8 were negative. Twenty-one samples that were positive by rCap-iELISA were negative by antibody ELISA. Of these 21 samples, 4 were negative by FAT but 17 were positive. However, of the positive samples by rCap-iELISA (n = 88), 79 (89.8%) were positive by the FAT, which gave a false-positive rate of 10.2%. Of the 76 samples positive by antibody ELISA, 63 were positive by FAT, which gave a false-positive rate of 17.1% (Table 1). Results indicated that rCap-iELISA had a higher agreement than antibody ELISA when compared to FAT. In addition to the varying technologies of the tests used, a likely explanation for such results was that the commercial kits used concentrated on A, B, and J subgroups, whereas the rCap-iELISA and FAT could also detect serum samples that were positive for the other exogenous subgroups of ALV.
Results of the recombinant capsid protein–based indirect enzyme-linked immunosorbent assay (rCap-iELISA), the antibody ELISA, and the fluorescent antibody test (FAT) for the detection of Avian leukosis virus antibodies from 269 serum samples collected from layer hens in Jiangsu Province, China.*
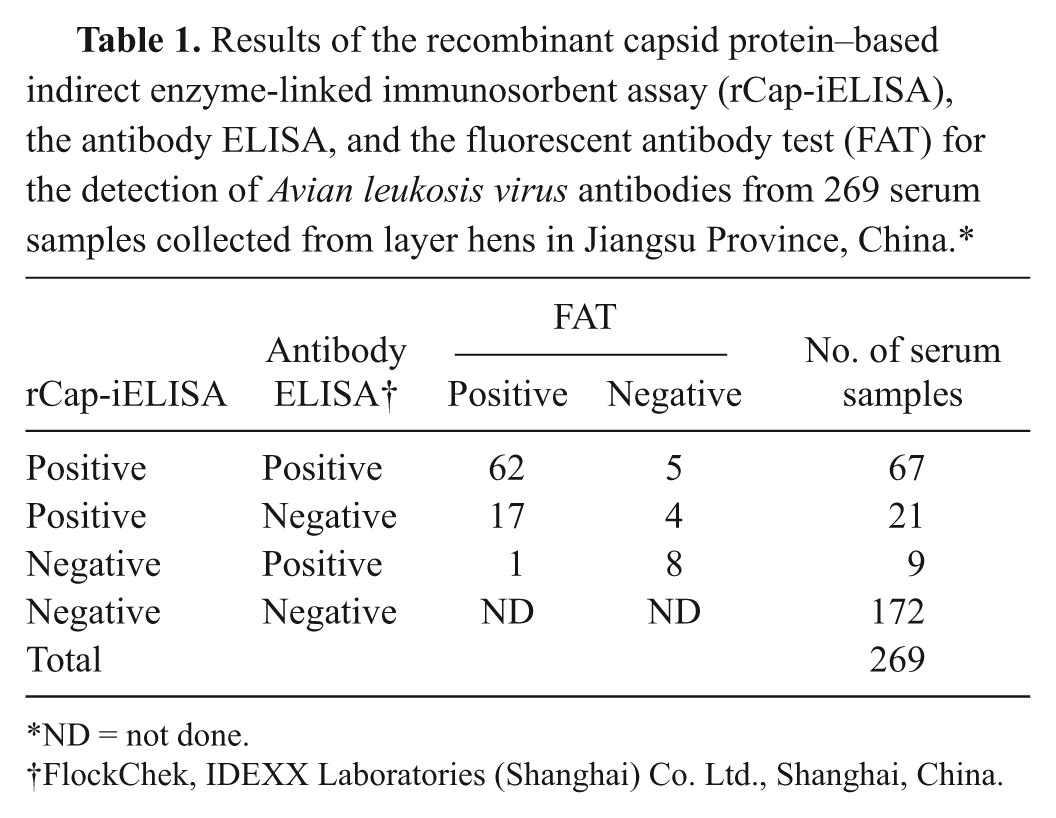
ND = not done.
FlockChek, IDEXX Laboratories (Shanghai) Co. Ltd., Shanghai, China.
The rCap-iELISA established in the current study was used in parallel with the antibody ELISA and FAT on 392 clinical samples from 5 flocks of layer breeders from Jiangsu Province, which were sent to the authors’ laboratory for ALV serodiagnosis. The positive rate of rCap-iELISA was 7.65% (30/392), whereas the positive rate of antibody ELISA and FAT was 5.88% (23/392) and 6.63% (26/392), respectively. Results indicated that rCap-iELISA had a higher sensitivity than the antibody ELISA in clinical samples. In conclusion, the rCap-iELISA developed in the current study serves as a potential tool for screening a large number of chicken sera for the presence of anti-ALV antibodies and that a combination of diagnostic tests is a more desirable approach for the routine examination of ALV infection.
Footnotes
a.
Key Lab of Jiangsu Preventive Veterinary Medicine, Yangzhou, China.
b.
TAKARA Inc., Dalian, Liaoning, China.
c.
Wsac Corp., Beijing, China.
d.
FlockChek, IDEXX Laboratories (Shanghai) Co. Ltd., Shanghai, China.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The Program for Changjiang Scholars and Innovative Research Team in University, the Joint Funds of the National Natural Science Foundation of China and Guangdong province (grant no. U0831002), and the Major Basic Research of Natural Science Foundation of the Jiangsu Higher Education Institutions of China (grant no. 07KJA2302) supported the current research.
