Abstract
The incursion of highly pathogenic avian influenza (HPAI) into the United States during 2014 resulted in an unprecedented foreign animal disease (FAD) event; 232 outbreaks were reported from 21 states. The disease affected 49.6 million birds and resulted in economic losses of $950 million. Minnesota is the largest turkey-producing state, accounting for 18% of U.S. turkey production. Areas with concentrated numbers of turkeys in Minnesota were the epicenter of the outbreak. The first case was presumptively diagnosed in the last week of February 2015 at the Minnesota Veterinary Diagnostic Laboratory (MVDL) and confirmed as HPAI H5N2 at the National Veterinary Services Laboratories on March 4, 2015. A total of 110 farms were affected in Minnesota, and the MVDL tested >17,000 samples from March to July 2015. Normal service was maintained to other clients of the laboratory during this major FAD event, but challenges were encountered with communications, staff burnout and fatigue, training requirements of volunteer technical staff, test kit validation, and management of specific pathogen–free egg requirements.
Introduction
Highly pathogenic avian influenza viruses (HPAIVs; family
During 2014, the H5N8 subtype of H5 HPAIV originating from Eurasia (EA) rapidly spread along the wild bird migratory pathways including the Pacific flyway. Introduction of this virus into the Pacific flyway, sometime during 2014, allowed its mixing with North American (AM) origin viruses and generated new (novel) combinations of reassortant viruses with genes from both EA and AM origin viruses.1,6,10,12 In November 2014, outbreaks of a novel HPAIV H5N2 were reported on chicken and turkey farms in the Fraser Valley, British Columbia, Canada.
7
From December 15, 2014 through January 16, 2015, the Animal and Plant Health Inspection Service (APHIS) of the U.S. Department of Agriculture (USDA) received 14 reports of birds infected with Asian-origin HPAI of H5N2 (
History of the 2015 H5N2 outbreak in Minnesota
The first farm affected with HPAI H5N2 was a 15,000-bird turkey breeder farm in Pope County, Minnesota. Affected birds were huddled, particularly quiet, and recumbent. In the terminal stage, the birds exhibited gasping, opisthotonus, and wing flapping followed by death. More than 70% of the birds on this farm died within 3–5 d (Yarnell J, pers. comm., 2015). Pools of tracheal swabs from these birds were submitted to the MVDL for testing for AIV by real-time reverse-transcription polymerase chain reaction (hereafter, qPCR). At the MVDL, these samples were tested in duplicate by qPCR targeting the matrix gene of AIV and were found to be presumptive positive. Further testing by qPCR specific for H5 and H7 subtypes confirmed that the samples were presumptive positive for H5. Presumptive positive samples for H5 were sent to the National Veterinary Service Laboratories (NVSL) in Ames, Iowa for confirmation. On March 4, 2015, the NVSL confirmed that the samples were positive for H5N2.
There were no further cases of HPAI in Minnesota until March 25, 2015 when a second flock in Lac Qui Parle County was confirmed positive by qPCR. From March until early June, birds on 110 turkey and commercial chicken farms in Minnesota were diagnosed with HPAI, and >9 million birds (turkeys and laying chicken hens) either died or were depopulated to control the outbreak. These outbreaks were reported from farms in 23 counties, with Kandiyohi County reporting the highest number of positive flocks (
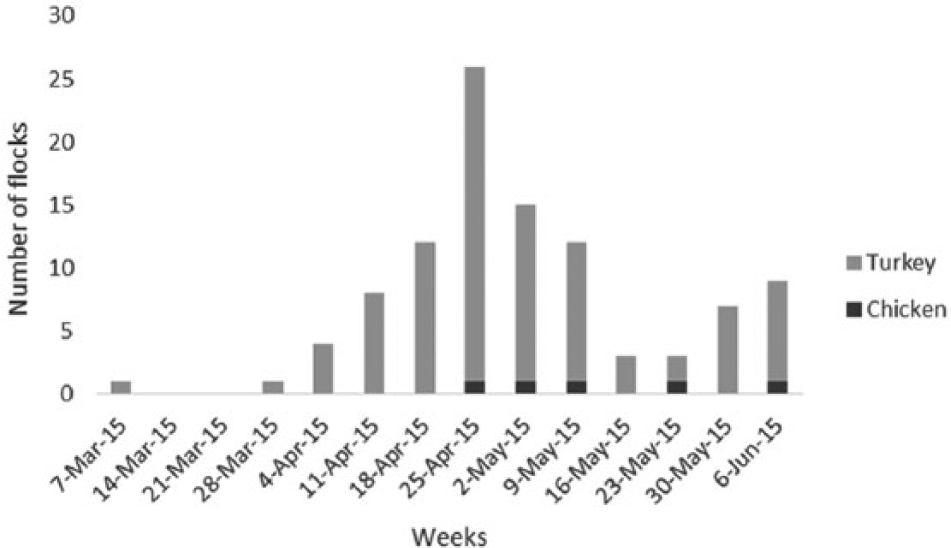
Number of highly pathogenic avian influenza virus H5N2–positive flocks on a weekly basis in Minnesota during the outbreak.
Role played by the MVDL
The MVDL is the official laboratory for the MBAH, the state agency charged with protecting animal health and responding to a foreign animal disease (FAD) such as HPAI in Minnesota. The United States primary control and eradication strategy for HPAI in domestic poultry, as recommended by the World Organization for Animal Health (OIE), is “stamping out.” The MVDL received samples from Minnesota and nearby states for AIV testing and played an important role in 5 major areas based on the OIE stamping-out program for HPAI: 1) disease detection and identification; 2) surveillance within control zones; 3) daily testing of healthy flocks to maintain product movement (business continuity); 4) environmental testing of previously AIV test–positive premises to assess the efficacy of cleaning and disinfection procedures; and 5) testing flocks 3 wk after restocking of previously depopulated poultry premises to ensure AIV test–negative status.
Disease detection and identification
Autopsy findings
The majority of Minnesota HPAI cases were diagnosed on-farm; however, in April 2015, three 15-wk-old tom turkeys were submitted to the MVDL for suspicion of
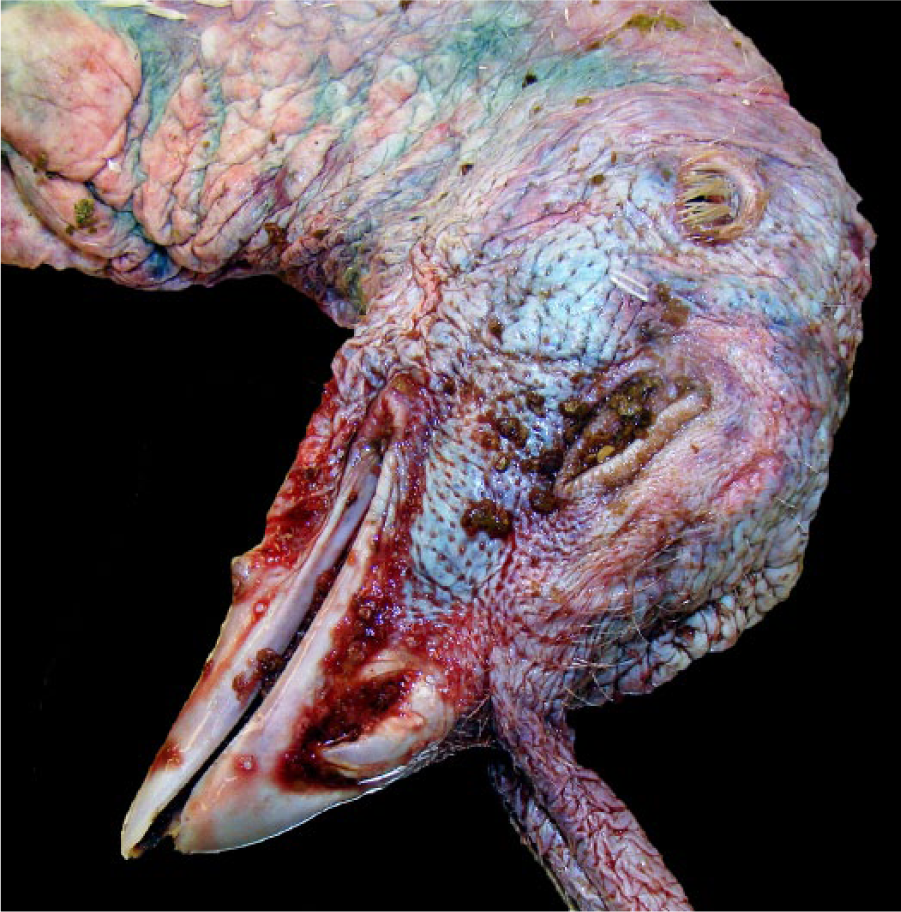
Perioral hemorrhage in 15-wk-old tom turkey with highly pathogenic avian influenza.
Additionally, the body cavities were filled with abundant serosanguineous fluid, and there was marked pulmonary hemorrhage (Fig. 3). The digestive tract was mostly devoid of digesta, and the mucosa overlying the cecal tonsils was black and friable (Fig. 4). Histologic findings included marked pulmonary edema and hemorrhage, focal pancreatic acinar necrosis, and acute cecal tonsil mucosal necrosis.
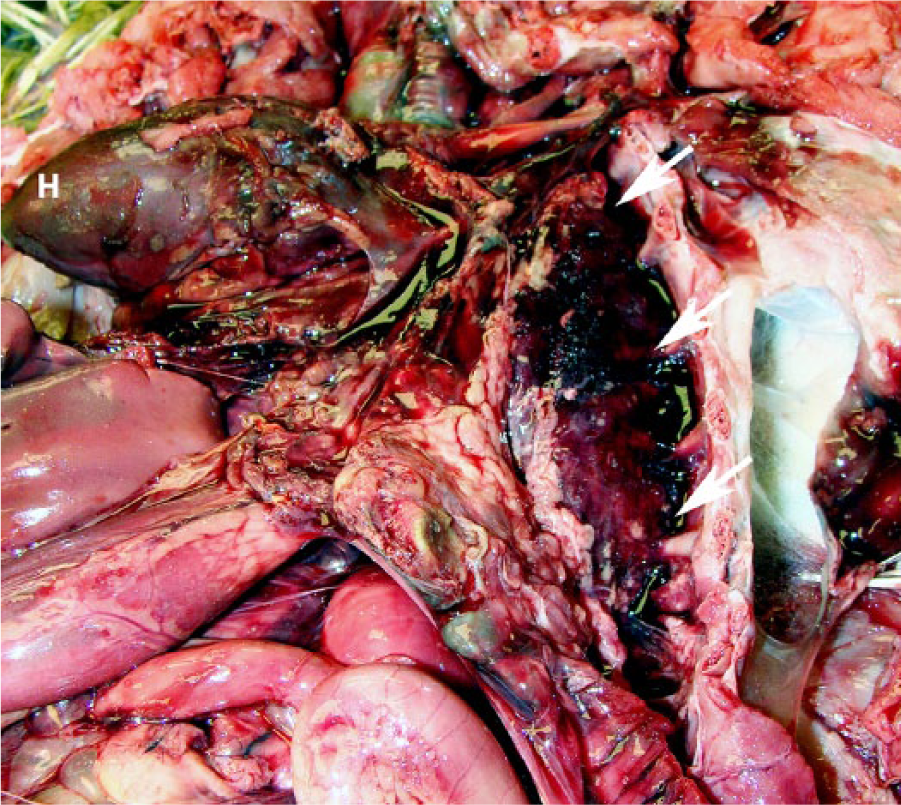
Marked pulmonary hemorrhages (arrows) in 15-wk-old tom turkey with highly pathogenic avian influenza. H = heart.
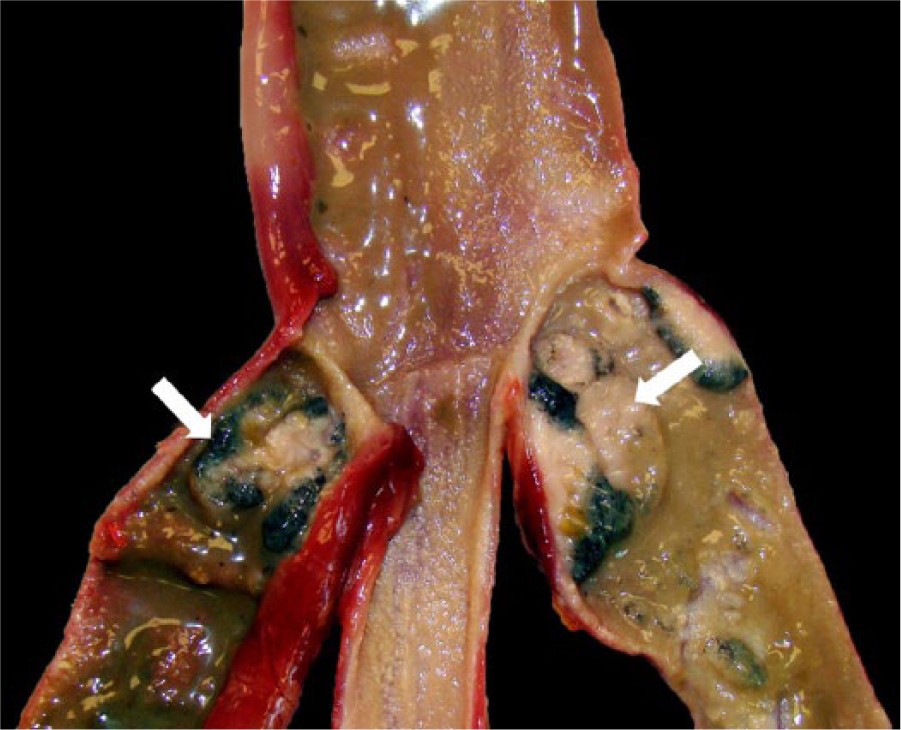
Acute cecal tonsil necrosis (arrows) in 15-wk-old tom turkey with highly pathogenic avian influenza.
Testing samples from infected and suspected flocks
Tracheal swabs from suspected flocks were submitted to the MVDL in person or through a courier service. All submitted samples were given an accession number by the laboratory receiving section of MVDL and then transferred to the molecular diagnostic section where they were processed for nucleic acid extraction and qPCR. All samples were first tested for AIV as described previously.
Because of the large number of samples received and because of urgency of the situation, the samples were tested in 2 or 3 runs per day. The MVDL was also notified of emergency or high-risk samples, defined as those samples collected from flocks showing clinical signs (e.g., dull, depressed birds, decreased feed and water intake, and high mortality) that were suspicious for AIV infection. As soon as they were received, these samples were tested in duplicate, by matrix, H5, and H7 PCR in order to verify and report results as quickly as possible. Results were reported to the field veterinarian, MBAH, USDA, and NVSL via the MVDL laboratory information management system (LIMS) immediately after completion of molecular tests.
A total of 17,053 samples were tested from March 1, 2015 to July 31, 2015 with maximum samples tested in April (Fig. 5). All H5-positive samples were sent to the NVSL for confirmation. All samples were negative for H7 subtype. At the NVSL, samples were tested for specific HA and NA subtypes by qPCR followed by sequencing of HA and NA genes. The NVSL also used a highly specific H5-icA assay 2 developed by the Southeast Poultry Research Laboratory, which targets the Eurasian H5 clade 2.3.4.4 gene, and conducted Sanger sequencing to generate partial HA/NA sequences directly from the sample for confirmation, pathotyping, and subtype determination. 9
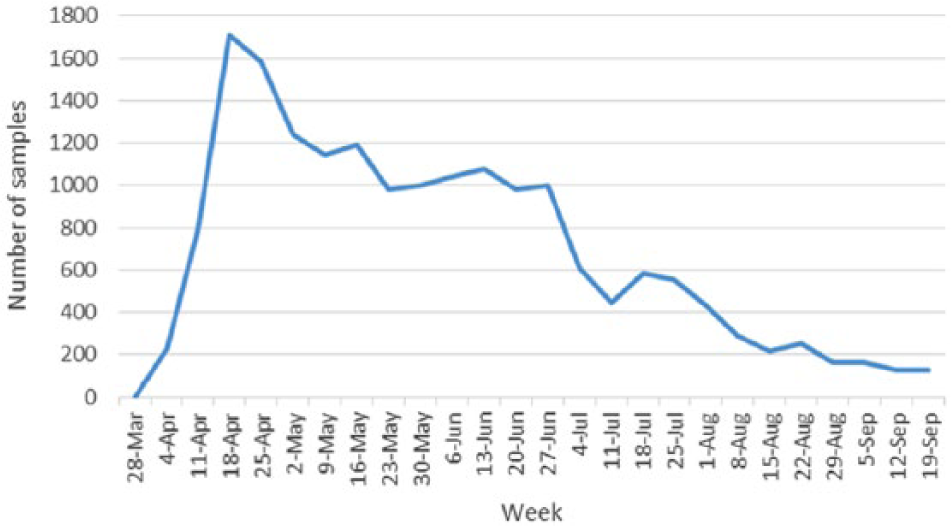
Number of samples tested by the Minnesota Veterinary Diagnostic Laboratory (MVDL) for avian influenza virus on a weekly basis (March 28–September 19, 2015).
Surveillance within control zones
During the outbreak, the MBAH established a 10-km control zone around each infected premises, which included a 3-km infected zone and an additional 7-km buffer zone for HPAI surveillance. Within the control zone, MBAH and USDA officials worked to identify all premises with commercial and backyard poultry. Backyard flocks were placed under quarantine and could not move poultry or poultry products on or off the premises before completion of 2 rounds of surveillance testing (21 d apart), all of which must have been negative before quarantines were lifted. Tracheal swabs from control zones were submitted daily to the MVDL through a courier service. Samples were tested and results reported daily. Commercial flocks inside the control area underwent surveillance in accordance with USDA protocols (https://goo.gl/Khyycq).
Commercial poultry operations that fell within this area submitted samples from their flocks for AIV testing every 7 d in accordance with USDA protocols (https://goo.gl/Khyycq). Samples for premarket testing were also tested daily for product movement in the state. For our purposes, surveillance samples are defined as samples from the control zones as well as the premarket testing samples.
Maintaining product movement (business continuity)
Samples received at the MVDL for premovement testing were tested daily in the afternoon, and results were reported the same day in late evening. The flocks that were negative on 2 consecutive tests were issued a shipping permit from the MBAH, and owners were allowed to move turkeys, chickens, or eggs off the premises.
Testing of environmental samples from affected farms for the purpose of restocking
The first affected farm in Pope County was in the process of restocking their barns with turkeys in early June, ~3 mo after confirmation of infection on that farm. This event marked a new phase in HPAI response; the focus shifted from surveillance to recovery. The restocking plans were developed by turkey producers and animal health officials working together.
Environmental swab samples (
Testing after restocking
Once the environmental samples were verified as being virus-negative, the premises were eligible for restocking with the approval of the incident commander. After repopulation of the initial barn on the premises, the birds on the premises were tested at least 3 times for AIV with sampling at least 7 d apart. All birds sampled for testing were at least 21 d old and were in the barn(s) for at least 7 d. All barns with test-eligible birds were tested until the third test on the initial barn was complete. All samples were collected by an authorized poultry testing agent. The tracheal swab samples were collected from the 5 most recent dead birds with live birds used to supplement if <5 were dead in the barn. A 5-bird pooled sample (1 tube of BHI) was required for each barn with samples submitted to MVDL for AIV testing. If all testing was negative, the quarantine was released. In addition to the required surveillance, all postquarantine abnormal mortality events within 180 d of quarantine release were investigated by MBAH personnel.
Continuity of other MVDL services
The MVDL is the only full-service, AAVLD-accredited laboratory in the state of Minnesota that offers diagnostic service for a broad range of species including food animals, companion animals, poultry, fish, and wild animals. During fiscal year 2015, porcine samples made up 55% of all the procedures done at the MVDL, 26% were poultry, and 13% were bovine. The MVDL conducts an average of 1.3 million procedures annually. There is a published turnaround time for each test conducted, and the expectation is to meet this time for all tests. Although the H5N2 avian influenza outbreak increased the workload of the MVDL, the normal level of service was maintained to all of the MVDL clients during the outbreak. During the outbreak, the workload increased for the molecular diagnostics and the virology sections, as well as the laboratory receiving area and the administrative sections of the MVDL. Before the outbreak, the laboratory receiving section had 5 full-time equivalents (FTE), the administrative section had 6 FTE, the virology section had 5 FTE, and there were 14 AIV proficiency–tested qPCR technicians in the molecular diagnostics section. Normal business hours for the laboratory are from 7:45 am to 4:30 pm Monday through Friday. In order to meet testing demands, the laboratory operated 7 d a week from 7:00 am to midnight or later during the outbreak.
The molecular diagnostics laboratory conducted 17,053 avian influenza qPCR tests during the outbreak period (March 1, 2015 to July 31, 2015). This was in addition to 81,139 routine tests carried out in this laboratory during this period. The additional AIV testing resulted in a 21% increase in the workload of this section. All these samples had also been processed through laboratory receiving and the administrative office.
Successes of MVDL
The MVDL staff successfully faced the challenges posed by the largest FAD outbreak in U.S. history. Of the 17,053 samples tested, none was reported false-positive during the outbreak period. Only 3 complaints were received regarding timeliness of the AIV results. This demonstrates the skill and efficiency of properly trained and proficiency-tested staff along with the availability of adequate supplies and equipment. Communication between poultry diagnosticians at the MVDL and the field veterinarians, poultry industry personnel, and the staff of MBAH and MVDL was, for the most part, efficient and effective. The results were reported immediately on test completion, and positive samples were forwarded to NVSL as soon as possible for further confirmation.
Challenges faced by the MVDL
Several challenges were faced by the MVDL during the outbreak as described below.
Communication challenges
During the course of the outbreak, there were several changes in courier delivery schedule, which required adjustment in testing schedules of the MVDL thereby affecting the timing of reporting results. Initially, in many cases, the samples either did not have a National Premises ID (NPID) number or had an incorrect NPID number on the submission form. This caused problems in coordinating the results among poultry industry personnel, designated company contacts, MVDL, MBAH, USDA, and NVSL. A NPID number is required for entering test results in the USDA/APHIS/VS Emergency Management Response System (EMRS). This web-based application is used for the reporting of routine investigations of suspected FAD surveillance and control programs, state and national animal health emergency responses, as well as permitted movements of poultry and poultry products.
Another communication challenge was to ensure that the results were forwarded to and received by the appropriate people. The MVDL has a web-based system that allows test results to be viewed online in real time. During the outbreak, as per laboratory protocol, results were reported by the MVDL pathologist directly to the submitting veterinarian or company by phone or text. Unless requested to do so, the expectation was that the submitting veterinarian or company would then contact the producers and convey the results to them. Unfortunately, there were some instances in which the producers had the expectation that they would be contacted by the laboratory directly as well, which resulted in some frustration. The MVDL online results system has the ability to allow access to additional interested parties to view results, which may help resolve this issue in the future. Eventually bar-coded submission forms, which included NPID, farm name, and farm address, helped solve most of the communications issues.
Staffing challenges
Normal business hours at the MVDL are from 7:45 am to 4:30 pm, Monday through Friday. During the outbreak, MVDL staff in the receiving, administration, and molecular diagnostics sections worked for extended hours during the week (from 8 am to midnight) and for additional hours on the weekends for several months. This resulted in staff burnout and fatigue and an increased need for sick and vacation time, which further reduced staffing levels. In order to maintain AAVLD accreditation status and quality assurance standards, MVDL has a very extensive training program, especially for employees in the molecular diagnostics and virology laboratories. This negated the option of hiring new employees to assist with AIV testing.
National Animal Health Laboratory Network resources
The National Animal Health Laboratory Network (NAHLN) is a network of state, university, and other laboratories in the United States that work together with the USDA to perform surveillance for specific diseases and to provide resources and the capacity to respond to FAD outbreaks. The NAHLN also ensures that methods and results are consistent between laboratories in the network by “proficiency testing” technicians who run the tests. Because of the availability of this network, routine surveillance samples from backyard flock testing were routed to other nearby NAHLN laboratories, namely South Dakota State University and North Dakota State University, which helped reduce some of the workload for the MVDL. However, it was not feasible to outsource samples from sick birds or other surveillance or movement samples in order to ensure the maintenance of poultry industry business continuity and prompt reporting of results.
The NAHLN was also willing to provide staffing assistance to MVDL. Three AIV proficiency–tested technicians from 3 different NAHLN laboratories were sent to the MVDL to assist in the Molecular Diagnostics section of MVDL for 3 wk (May 4–May 25, 2015). Once trained, the technicians provided scheduling relief for the laboratory staff, especially for evening and weekend shifts. However, some logistical issues within the MVDL limited the utility of these technicians. Despite being well-trained and proficiency-tested in AIV testing, the NAHLN technicians were unfamiliar with the MVDL’s high-volume PCR protocols and the various roles of the Molecular Diagnostics laboratory staff. They needed training on laboratory-specific equipment such as robotic extraction of nucleic acids. Training was done by a MVDL technician who was then not able to assist with AIV testing. In addition, as per MVDL policy, the NAHLN technicians were required to complete additional UMN training before starting work at the MVDL, such as site-specific safety training, quality assurance training, and result reporting (LIMS) training. To maintain quality standards, additional supervision of NAHLN technicians was necessary and hence the services of these technicians were not fully utilized. Despite the logistical challenges, we at MVDL are grateful for the work of these technicians and their willingness to spend time away from their homes to help the MVDL deal with this outbreak. We are also grateful to NAHLN for providing assistance when it was needed.
Challenges with testing of environmental samples
During the clean-up and restocking phase of the outbreak, the expectation of clients submitting environmental samples for testing (including qPCR and VI) was that all results would be available within 3 wk from the date of sample collection. This became a challenge for both molecular diagnostics and the virology sections of MVDL.
For molecular diagnostics, the biggest challenge was the need to use the detection kit b specified in the USDA procedure document that contained an internal control to account for any PCR inhibitors that could be present in environmental samples. This kit was not routinely used in the molecular diagnostics laboratory and hence required a NAHLN deviation, as well as test validation and training. Indeed, many internal control failures occurred, the samples of which were then required to be sent to the virology section for VI. The internal control feature also required separate procedures to be run, which increased the processing time and the workload.
The virology laboratory also experienced some challenges in testing environmental samples. Steps for VI included: ordering of appropriate numbers of SPF embryonated chicken eggs, incubating eggs to be ready for inoculation at 9–11 d, processing of samples followed by their inoculation in eggs, daily candling of inoculated eggs, harvesting of inoculated eggs, performing HA tests, and inoculation of eggs for a second passage. Initially, all samples with an internal control failure or a nonzero Ct value from the qPCR test were sent to the virology laboratory for VI. This was ~10% of all samples submitted for environmental testing. Later in the outbreak, internal control samples were retested by qPCR before being sent for VI. The incubator space for both inoculated and uninoculated embryos was limited; separate incubators had to be used for inoculated and uninoculated embryos because of biosafety and cross-contamination issues. The above issues were further complicated by the fact that it was difficult to predict the timing and number of VI tests that would be needed each week. If there was an increase in the number of tests needed, it would be a minimum of 9 d before the eggs were ready for inoculation even if enough eggs were on hand.
Problems with testing time and batch samples
Processing high volumes of samples or batch samples for qPCR doubled or tripled the test completion time, depending on the number of samples tested. The laboratory labeled all samples properly and took the necessary precautions to avoid cross-contamination and hence there was no false-positive result reported throughout the outbreak period. It was often difficult to explain to the producers and field veterinarians why it took 4–6 h to report a test that normally took 2 h to run. One solution to this communication challenge in the future is to include MVDL staff in producers’ meetings to explain some of the logistics of the laboratory testing.
Footnotes
Acknowledgements
We thank the staff of MVDL, MBAH, MPTL, NAHLN, and NVSL for rising to the occasion and dealing with this difficult situation.
Authors’ contributions
A Fitzpatrick, SK Mor, E Wiedenman, RE Porter, DP Patnayak, DC Lauer, and S Voss contributed to conception and design of the study, and to acquisition, analysis, and interpretation of data. M Thurn and SM Goyal contributed to conception and design of the study, and to analysis and interpretation of data. T Otterson contributed to design of the study and to analysis and interpretation of data. S Rossow contributed to conception and design of the study, and to acquisition and analysis of data. JE Collins contributed to design of the study. A Fitzpatrick, SK Mor, T Otterson, RE Porter, and DP Patnayak drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Ambion MagMax-96 viral RNA isolation kit, Thermo Fisher Scientific, Waltham, MA.
b.
Life Technologies VetMAX Gold AIV detection kit, Thermo Fisher Scientific, Waltham, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
