Abstract
A 2-mo-old pet chicken (Gallus domesticus) was presented because of lameness and a hind limb mass of 1 mo duration. Radiographs revealed a soft tissue mass extending from the mid-femur into the body wall. Cytology of a sample obtained from a fine-needle aspirate was unremarkable. The bird was prepared for surgical investigation of the mass but died acutely at the time of pre-medication. Autopsy revealed an extradural mesenchymal neoplasm that focally compressed the lumbar spinal cord, extended into and along the adjacent nerve structures, and displaced the kidneys, skeletal muscle, and body wall. Histopathology was consistent with a perineurioma, which was positive for alcian blue and negative for S100, GLUT1, and neurofilament proteins on immunohistochemistry. PCR testing of tumor tissue was negative for gallid herpesvirus 2 (Marek’s disease virus) and avian leukosis virus. Although the etiology of the mass was not determined, our case highlights a severe manifestation of an uncommonly diagnosed tumor in the chicken. Though uncommon, perineurioma should be considered a differential diagnosis for lameness in the chicken.
Keywords
A 2-mo-old male domestic chicken (Gallus domesticus) was presented because of lameness and a mass of 1 mo duration on the right leg. The chicken had been purchased from a local producer as a chick and was segregated from other birds when the mass was first noted. He was housed in a large dog crate and fed commercial chicken starter pellets and small amounts of fruit. His appetite and water intake were noted to be good and had not changed since the mass was noted. The chicken had been non-ambulatory for several weeks at the time of presentation.
Radiographs performed at the referring veterinary hospital noted a large soft tissue mass over the proximal right femur and involving the coelomic wall (Fig. 1). A fine-needle aspirate of the mass by the same veterinarian showed low cellularity, with moderate hemodilution and rare extracellular rods and cocci. No neoplastic cells or evidence of inflammation were noted.
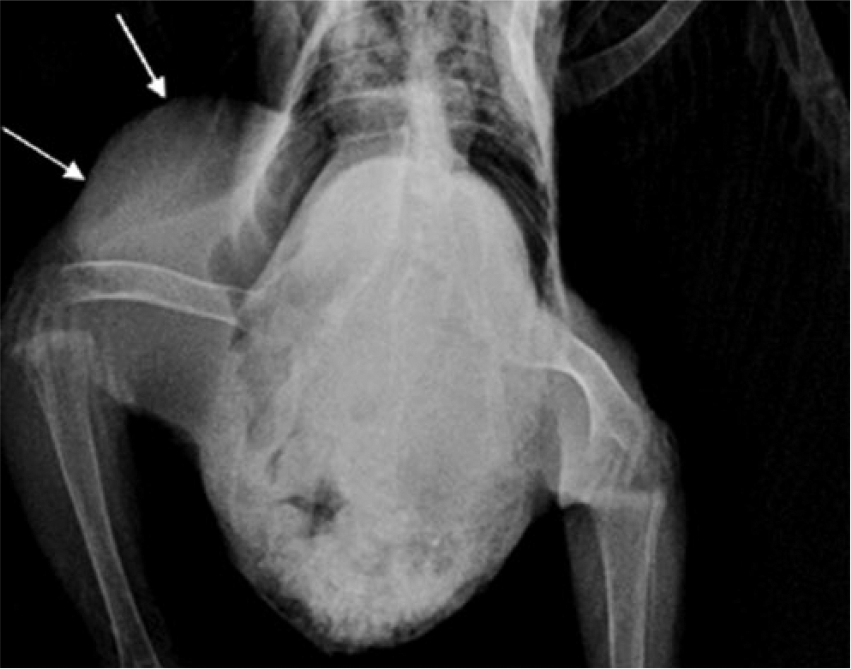
Radiographic image of the right pelvic limb, where a soft tissue density mass (arrows) has replaced most of the femoral muscles, and obscures the pelvic viscera.
Physical examination at presentation revealed a bird in good body condition. A 5 × 5 cm firm mass was noted on the right pelvic limb, over the craniomedial surface of the mid-to-proximal femur. The bird did not exhibit pain on palpation, but there was a significant decrease in the range of motion of the right leg, and the bird was unable to stand or ambulate. When not supported, the bird was in sternal recumbency with the right leg splayed caudally. No other abnormalities were noted on physical examination. The bird was bright, alert, and eating well in hospital. Primary differentials at this time included neoplasia, granuloma, and abscess. Given financial constraints, repeat radiographs under sedation with more ideal positioning were declined, and the owner elected surgical investigation and possible removal of the mass. The chicken was administered subcutaneous fluids (50 mL/kg, 2.5% dextrose in 0.9% sodium chloride) and crop fed (1% body weight; Emeraid Omnivore Diet, Emeraid, Cornell, IL). In preparation for surgery the following morning, food was removed from the cage on the night of presentation.
On the morning following presentation, the chicken was bright, alert, and responsive; normal droppings were noted in the cage. Physical examination remained unchanged from the day before, and temperature, heart rate, and respiratory rate remained normal. Drinking water was removed, and subcutaneous fluids (50 mL/kg, 2.5% dextrose in 0.9% sodium chloride) were administered. Several hours later, and, as pre-medication for surgery, the bird was administered butorphanol tartrate (2 mg/kg intramuscularly [IM] once; Torbugesic, Zoetis, Florham Park, NJ) and midazolam (1 mg/kg IM once; Midazolam, Akorn, Livonia, MI) in the pectoral muscle. Immediately following the injection, and, while still being handled, the bird became acutely unresponsive. Thoracic auscultation revealed an absence of cardiac and respiratory sounds. Despite attempted cardiopulmonary resuscitation, the bird did not recover.
At autopsy, there was an expansive multilobular mass, 6 × 3 × 3 cm, which extended from the coelomic cavity through the abdominal wall into the right thigh muscles (Fig. 2). The white-tan, multilobular mass replaced most of the architecture of both kidneys. On cut section, the mass encircled the lumbar vertebrae and replaced the adjacent skeletal muscles and nerves of the pelvis and right leg. Tissue samples were fixed in 10% neutral-buffered formalin solution. Trimmed tissue sections were routinely processed, embedded in paraffin, and sectioned at 5 μm. All tissue sections were stained with hematoxylin and eosin. Selected sections of the neoplasm were stained with alcian blue (pH 2.5)–periodic acid-Schiff.
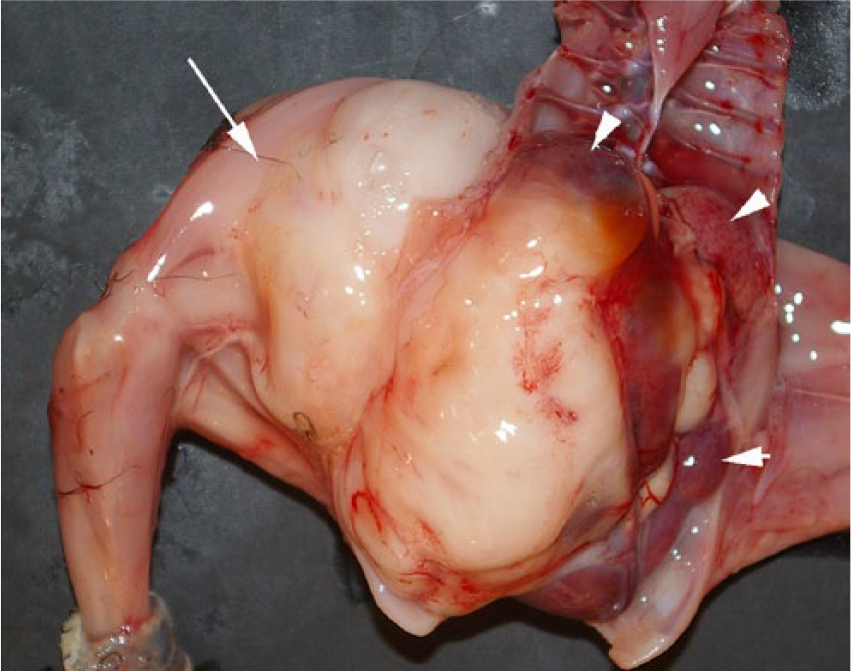
Gross autopsy image of the right pelvic limb, where a white-tan mass (arrow) has replaced most of the femoral muscles, extended through the abdominal wall, and replaced the kidneys (arrowheads).
Immunohistochemical staining of the mass was performed for S100, glucose transporter protein 1 (GLUT1), and neurofilament (Table 1). S100 was used to detect peripheral nerve sheath cells; GLUT1 was used to detect mucin-producing cells and perineurial cells 6 ; neurofilament was used to detect axonal filaments. Immunostaining with other antibodies, including vimentin, epithelial membrane antigen (EMA), and glial fibrillary acid protein (GFAP) was attempted on normal chicken tissues but was not successful in the laboratory. The substrate-chromogen system used was 3,3’-diaminobenzidine (DAB; Dako North America, Carpinteria, CA). Tissue sections were counterstained with Gills II hematoxylin and bluing, dehydrated in alcohol levels of 70%, 95%, and 100%, cleared in xylene, and mounted with xylene-based mounting medium.
Spontaneous spinal plexiform perineurioma in a juvenile chicken (Gallus domesticus): summary of the immunostaining conditions used.

Sources: Avidin–biotin block, Dako North America, Carpinteria, CA; biotinylated anti-mouse IgG and biotinylated anti-rabbit IgG, Vector Labs, Burlingame, CA; mouse monoclonal anti-neurofilament IgG1 (clone N14), citrate (pH 6), and power block, BioGenex, San Ramon, CA; mouse monoclonal anti-S100 IgG (clone 4C4.9), Cell Marque, Sigma-Aldrich, St. Louis, MO; rabbit polyclonal anti-GLUT1 IgG, Abcam, Cambridge, MA.
On histologic examination, an extradural mesenchymal neoplasm compressed the lumbar spinal cord (Fig. 3), extended along nerve structures outside the vertebrae, and obliterated the adjacent kidneys, body wall, and skeletal muscle. The multinodular, densely cellular neoplasm was composed of mildly pleomorphic spindle cells arranged into interlaced bundles with widely spaced, mucin-producing (alcian blue-positive) plexiform structures (Figs. 4–6). The supporting fibrocollagenous stroma was scant. Spindle cells had moderately abundant eosinophilic fibrillar cytoplasm with indistinct cell borders that blended with the stromal matrix. The normochromatic, vesicular nuclei varied from round-to-oval-to-fusiform, with mild anisokaryosis and indistinct, often multiple, nucleoli. Mitoses in 10 random fields at 400× were zero, with no evidence of vascular invasion. No significant tissue alterations or evidence of metastases were detected in the heart, lung, liver, spleen, adrenal gland, or testes. Brain, cervical or thoracic spinal cord, and spinal nerves were not examined. Immunohistochemical detection of S100, neurofilament, and GLUT1 within the neoplasm was negative.
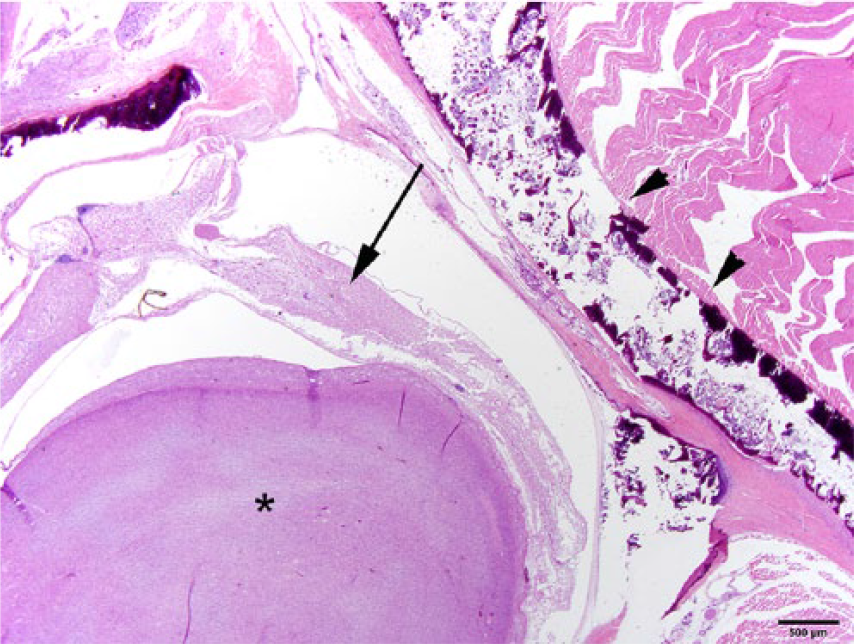
The neoplasm (*) has compressed the adjacent spinal cord (arrow). Arrowheads depict the dorsolateral lamina of the vertebral body. H&E. Bar = 500 µm.
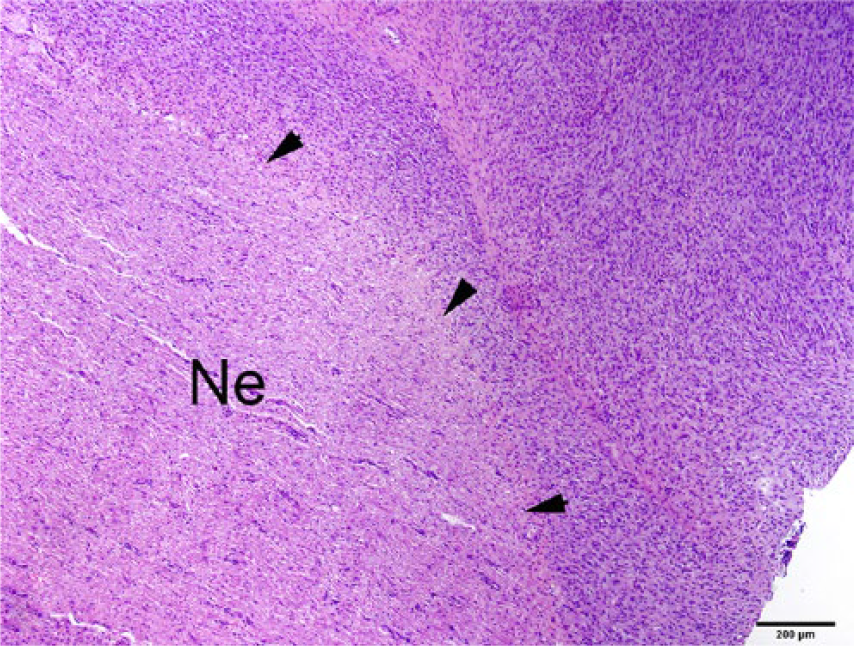
A densely cellular neoplasm (right side of the image) is closely associated with a peripheral nerve (Ne). Arrowheads depict the junction between the neoplasm and the nerve. H&E. Bar = 200 µm.
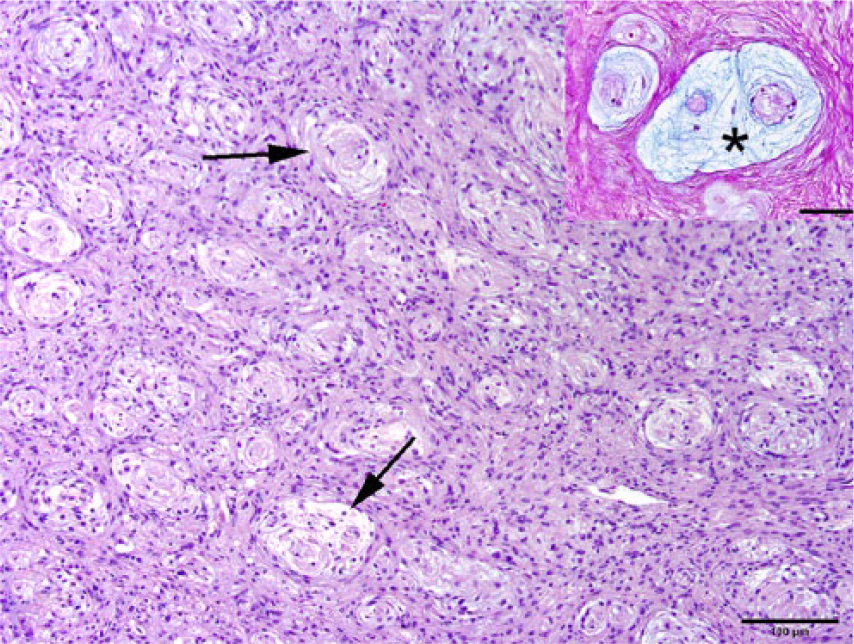
The densely cellular neoplasm is composed of closely apposed spindle cells and spaced plexiform structures (arrows). H&E. Bar = 100 µm. Inset: Representative image that depicts a cluster of plexiform structures. Individual structures consist of loosely arranged pale blue material or mucin (*), widely spaced reddened nuclei, contained by periodic acid–Schiff (PAS)-positive fibrils. PAS–alcian blue (pH 2.5). Bar = 100 μm.
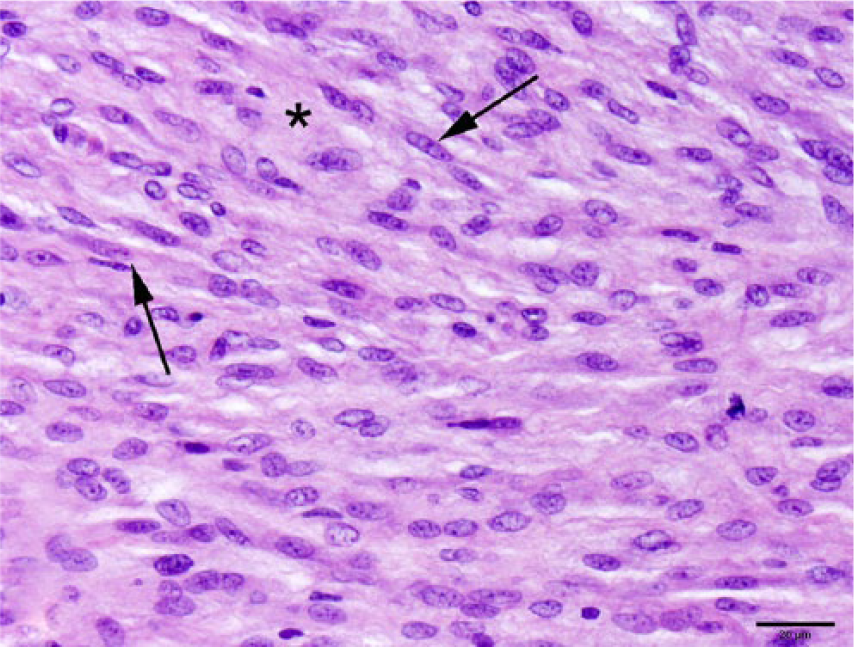
In other areas, the densely cellular neoplasm consisted predominantly of closely apposed spindle cells (arrows), within an eosinophilic fibrillar stromal matrix (*). The normochromatic nuclei varied from round-to-oval-to-fusiform, with mild anisokaryosis and indistinct, often multiple, nucleoli. H&E. Bar = 20 µm.
Immunohistochemistry findings contrast with those described in other reports, in which perineurial cells were immunohistochemically positive for GLUT1 and negative for S100 in humans 4 and chickens 8 with perineuriomas. Consistent with such reports4,8 is the negative immunohistochemical detection of S100 in the neoplasm described herein, which rules out Schwann cells as its origin. However, a specific cause for the discrepancy in immunohistochemical detection of GLUT1 could not be determined. Based on all immunohistochemical findings, histopathologic features, and results of alcian blue stain, the origin of the neoplasm is most consistent with a mucin-producing, plexiform peripheral nerve sheath neoplasm of likely perineurium origin.
Peripheral nerve sheath tumors (PNSTs), including perineuriomas, Schwannomas, and neurofibromas, are classified based on tumor origin and histologic features.9,12,13 In a 2010 review, 1 the term plexiform, as used in histopathology, was defined as an infiltrative growth pattern that looks like a plexus or a network typically associated with tumors of neural derivation. Prototypes of the plexiform pattern are the plexiform neurofibromas, in which the term implies a network-like growth involving multiple fascicles of a nerve. 1
Neurofibromas and schwannomas, the most common of the benign PNSTs, are differentiated based on histopathology, immunohistochemistry, and electron microscopy.4,5,7,10,11 The etiology of these tumors is poorly understood, and they remain an uncommon finding in poultry,9,11,14,15 although avian leukosis virus (ALV) had been isolated from a neurofibrosarcoma in a Japanese native fowl.9,14
Because the neoplasm described in our case was detected late in the disease process (the time the animal was first examined), the site of origin of the tumor could not be established nor whether it extended centrifugally or centripetally. Additional testing of formalin-fixed, paraffin-embedded tumor tissue did not reveal any molecular (polymerase chain reaction [PCR]) evidence of gallid herpesvirus 2 (Marek’s disease virus) or ALV infection. A specific cause of death was not determined.
Although multiple viral etiologies have been associated with lesions in the nervous system in poultry, signalment and gross, histopathologic, and laboratory findings did not support a specific etiology in our case 3 . However, findings suggest that an alternative, albeit undetermined, etiology for the tumor present in this bird exists.
Peripheral neuropathy, which is recognized as a separate entity from Marek’s disease, has also been documented in poultry. 2 Lesions include edema of peripheral nerves, with differentiation from Marek’s disease based on several factors, most notably the absence of visceral tumors. 2 Such characteristics makes peripheral neuropathy an unlikely cause of the clinical signs observed in our case. Age of the chicken, absence of bursal involvement, and negative PCR results for ALV do not support ALV as an etiologic agent in this case.
Spinal cord compression caused by the presence of the mass within the vertebral column was significant and explains the paresis observed clinically. The neoplasm extended widely and affected multiple organ systems. An earlier diagnosis may have allowed tumor resection and treatment, although the invasiveness of the tumor suggests that treatment would not have been curative. Our case highlights the presentation of an uncommonly diagnosed tumor in the chicken and the need for veterinary care of the pet chicken as an increasingly popular companion animal.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
