Abstract
Bovine viral diarrhea virus (BVDV) is an economically important disease of domestic cattle that is capable of infecting cervids. A first step in the formulation of a regional BVDV management plan is a local assessment of the likelihood of pathogen transmission from wildlife to domestic livestock. To achieve this, blood samples were collected from hunter-harvested white-tailed deer (Odocoileus virginianus) throughout New York State in the fall of 2009 and 2010. The SVANOVIR BVDV p80-AB enzyme-linked immunosorbent assay (ELISA; Svanova Biotech AV, Uppsala, Sweden) was used to screen sera for anti-BVDV antibodies. Because this ELISA is not validated for use in white-tailed deer, sera that tested positive were tested again using serum neutralization to verify the presence of antibodies. Spatial data describing the geographic location of BVDV antigen–positive cattle and camelid herds and BVDV-seropositive white-tailed deer were analyzed via the dual kernel density estimation method. In white-tailed deer, 7.48% (80/1,069) were BVDV-seropositive, whereas 3.43% (144/4,195) of tested herds were positive for BVDV antigen. An exploratory cluster analysis revealed 1 significant cluster of BVDV antigen–positive herds and 2 significant clusters of BVDV-seropositive deer. There was no spatial overlap between the clusters. The spatial point pattern and exploratory cluster analyses suggest that BVDV is maintained independently in domestic livestock herds in the western part of the state and in the white-tailed deer population in the northwestern part of the state.
Introduction
Infectious diseases are a serious threat to American livestock productivity and health, and the management of pathogens commonly diagnosed in domestic livestock has historically been considered independent of disease management in wild species. Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) is an economically important disease of domestic cattle that is capable of infecting cervids.20,26,31 Because the estimated persistence of BVDV in the environment ranges from an average 9 hr at 21°C to 67 hr at 4°C, 8 indirect interspecies viral transmission may occur. Persistently infected (PI) individuals consistently shed the virus in all bodily fluids throughout their entire life, 4 but transiently infected animals typically only remain viremic for up to 15 days.4,10 Thus, the main routes of both intraspecies and interspecies horizontal transmission are indirect and direct spread from PI animals. 18 Over 60% of northeastern U.S. dairy operators report definitive contact between cattle and wild white-tailed deer (USDA National Agriculture Statistics Service: 2007, Census of agriculture. Available at: http://www.agcensus.usda.gov/Publications/2007/Full_Report/usv1.pdf. Accessed on January 22, 2013), therefore interspecies BVDV transmission is possible.
Although BVDV is most commonly diagnosed in cattle, it has been recognized as an emerging infectious disease of concern in alpacas,2,5,6,12,14 and has also been isolated from free-ranging American elk (Cervus elaphus nelsoni), mule deer (Odocoileus hemionus), and white-tailed deer (Odocoileus virginianus).7,28,31 Under experimental conditions, it appears that the pathogenesis of BVDV in PI white-tailed deer is similar to that of PI cattle. 21 Additionally, fawns with immunohistochemistry staining comparable to that of a PI calf have been reported to consistently shed large amounts of virus in nasal secretions, suggesting that such white-tailed deer may be efficient sources of BVDV.21,23 Thus, wild white-tailed deer may be able to independently maintain BVDV without periodic input from domestic livestock and may serve as a viral reservoir population. 22
It is important to determine if the virus is able to be maintained in wildlife via an independent viral cycle in order to assess the likelihood of interspecies BVDV transmission and the possibility that particular white-tailed deer populations may be reservoir hosts for BVDV. However, direction of pathogen transmission cannot be elucidated. The spatial point pattern distribution of BVDV-positive livestock herds in New York has never been compared to the spatial point pattern distribution of BVDV-seropositive white-tailed deer. Thus, the objectives of the current study were to determine the seroprevalence of BVDV in white-tailed deer in New York and report the spatial patterns of apparent BVDV prevalence in cattle and camelid herds and the apparent BVDV seroprevalence in wild white-tailed deer in New York.
Materials and methods
Sample collection
Hunter-harvested white-tailed deer were surveyed throughout New York State in November 2009 and October–December 2010. No samples were collected from New York City or Long Island, therefore this portion of the state was excluded from the spatial analyses. The sex and township of harvest for each animal was recorded. Blood was collected either from the thoracic cavity or as it drained from the nasal and oral cavities 22 at New York State Department of Environmental Conservation (NYSDEC) deer check stations and at private deer processing facilities. The blood was collected in 10-ml glass serum tubes a and stored at 4°C prior to centrifugation at 1,300 × g for 10 min. Serum was frozen at −80°C.
Blocking enzyme-linked immunosorbent assay
The SVANOVIR BVDV p80-AB blocking BVDV enzyme-linked immunosorbent assay b (ELISA) was used to screen the serum samples for the presence of anti-BVDV antibodies. The testing methodology was conducted according to the manufacturer’s instructions, but the optical density value (%) cutoff for positive samples was lowered to 19% based upon serum neutralization (SN) results of dual-tested samples. A subset of seronegative samples and all seropositive samples were dual-tested by SN for BVDV type 1 (Singer strain) and BVDV type 2 (strain 125). Serum neutralization tests were carried out following standard methods. 9 In brief, serum was inactivated (56°C for 30 min), then serially diluted two-fold in a 96-well cell culture microtiter plate with minimum essential medium with Earle salts (MEME) plus 10% fetal bovine sera, streptomycin, and penicillin. An equal volume of MEME containing 100 TCID50 of BVDV-1 or BVDV-2 was added to each well. Incubation was performed at 37°C for 4 days before examination for cytopathic changes. 9
Spatial point pattern analysis
Data was obtained from the New York Animal Health Diagnostic Center (Cornell University, College of Veterinary Medicine, Ithaca, New York) and the Nebraska Veterinary Diagnostic Laboratory (University of Nebraska-Lincoln, School of Veterinary Medicine and Biomedical Sciences, Lincoln, Nebraska) regarding the locations of bovid and camelid herds that submitted samples for BVDV antigen testing between July 1, 2007 and December 31, 2010. Herds that submitted samples for BVDV isolation, polymerase chain reaction, immunohistochemistry, and/or antigen-capture ELISA were included in the dataset. Each herd was identified only by a randomly assigned number and was geographically localized to the township level in order to ensure anonymity. The township of each tested herd was entered into a commercial Geographic Information System (GIS) software. c The centroid of the township was used to symbolize the herd location, and a point pattern layer was generated (Fig. 1). In a similar manner, the township of each deer sample was entered into the GIS software, c and the centroid of the township was utilized to represent the location of harvest (Fig. 2).
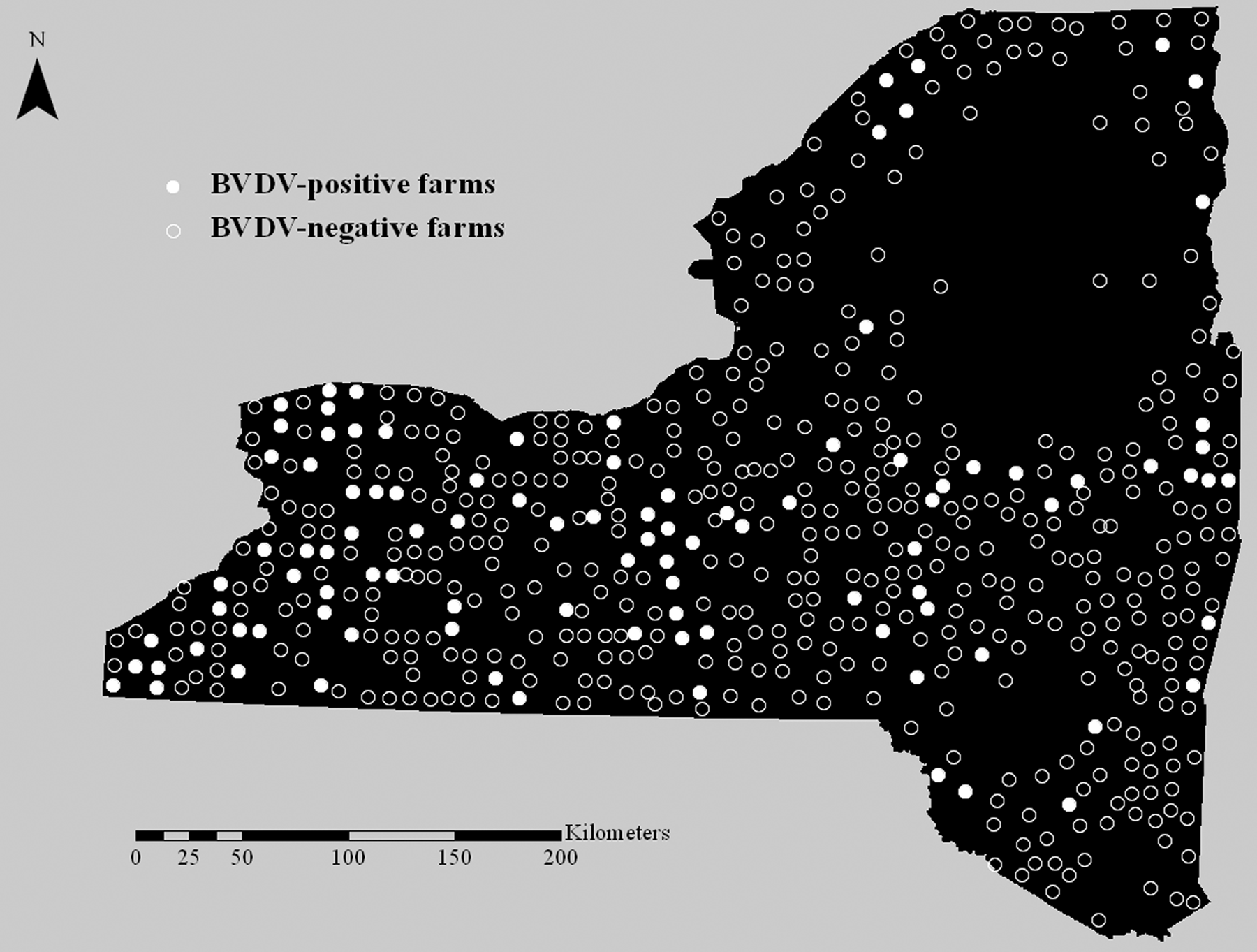
Locations of cattle and camelid herds sampled for Bovine viral diarrhea virus (BVDV) antigen in New York State, USA, from July 1, 2007 to December 31, 2010. Each circle represents the centroid of the township where the herd was located.
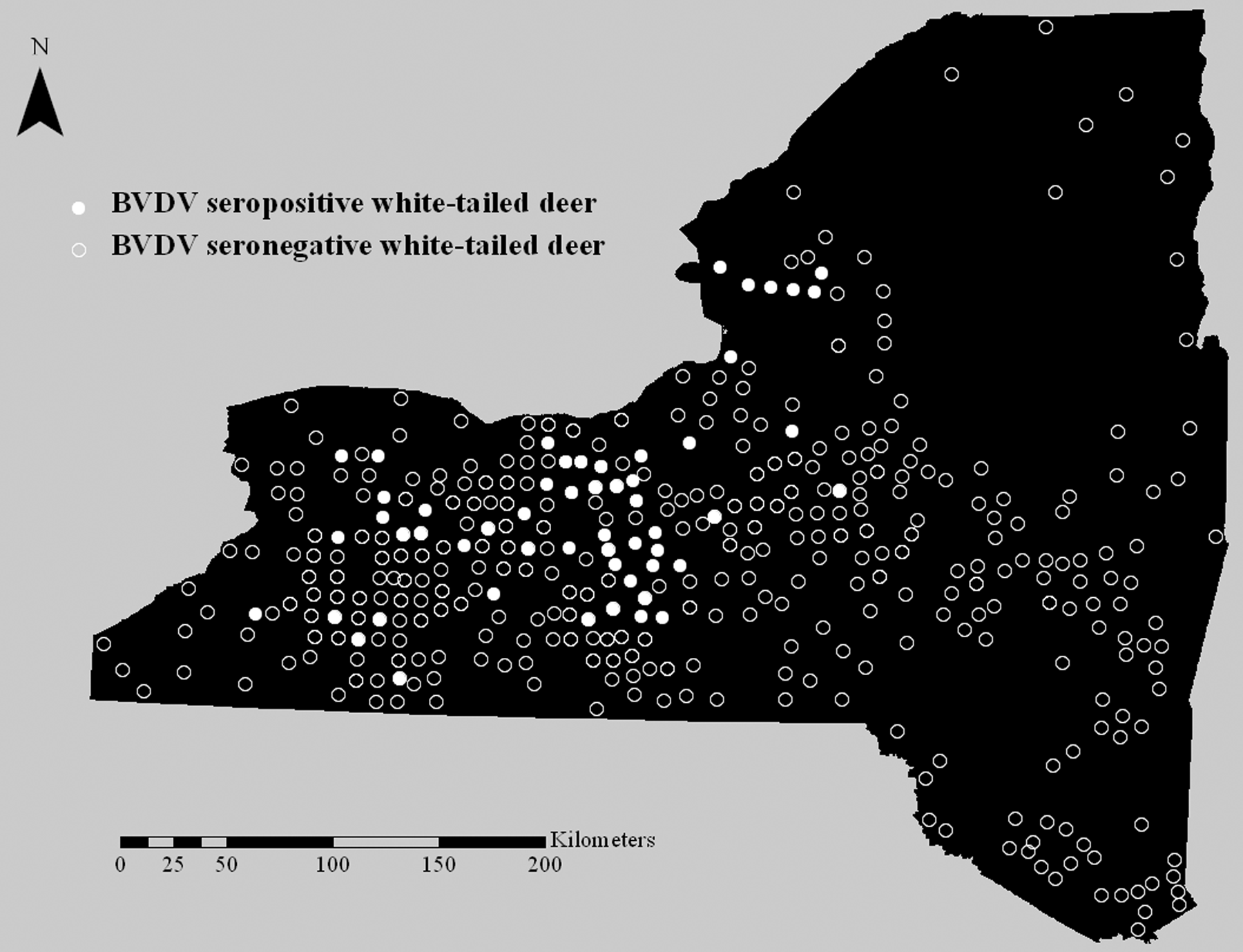
Locations of harvested white-tailed deer (Odocoileus virginianus) sampled for Bovine viral diarrhea virus (BVDV) antibodies in New York State, USA, in November 2009 and November–December 2010. Each circle represents the centroid of the township where the deer was harvested.
The dual kernel density estimation method was used to compare both the density estimates of seropositive deer to the density estimate of sampled deer (population at risk) and the density estimate of positive farms to the density estimate of sampled farms using spatial statistics software. d The kernel density estimation method is an interpolation and smoothing technique used to generalize point locations to an entire study site. It functions by moving a three-dimensional function (kernel) of a prescribed or fluctuating bandwidth to each sample location and weights the area surrounding each point location according to the distance from the center of the kernel. In adaptive bandwidth smoothing, sub-areas where many samples are collected promote a kernel with a smaller bandwidth than areas where fewer samples are collected. The sum of the individual kernels is calculated for the entire study site, and a smoothed disease risk surface is obtained.13,27 The ratio of BVDV-seropositive deer to total deer tested provided a spatial distribution of estimated BVDV seroprevalence for wild white-tailed deer in New York State. The same analysis was conducted using the density estimates of BVDV-positive herds to total herds tested. For both analyses, the normal method of interpolation and an adaptive bandwidth (minimum of 10 points per kernel) were used. The results were represented using GIS software. c
Cluster analysis was performed using statistical software.e,17 A maximum population size window of 15% (approximately twice the seroprevalence determined in 2009 and 2010) was selected. The software utilizes a focal point (in this case, the centroid of each township) and employs circles of varying diameters to check for the presence of clusters of either BVDV-positive herds or deer. The Bernoulli method was used to compare the number of positive and negative deer or herds within each arbitrarily sized circle to the number of positive and negative deer or herds remaining outside the circle. 19 A likelihood ratio was calculated for each circle, and a cluster of positive deer or herds was considered to be significant if P ≤ 0.01. A chi-square test was used to compare seroprevalence in male and female deer (P ≤ 0.01).
Results
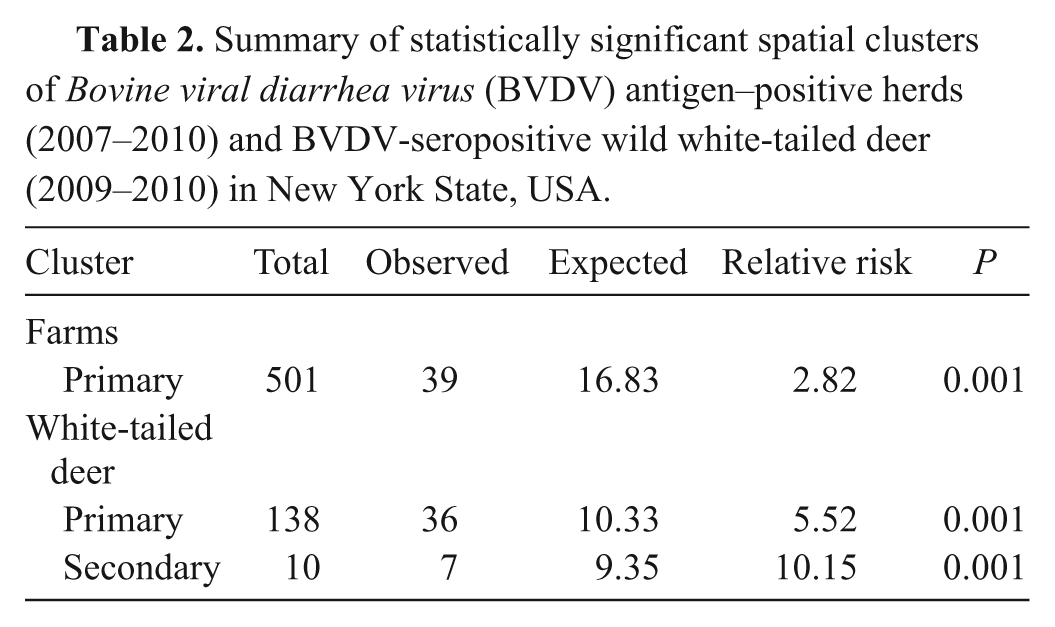
There was 100% agreement between the samples positive via the blocking ELISA and a follow-up SN test. Out of a total of 1,069 white-tailed deer samples, there were 80 BVDV-seropositive individuals (7.48%). There was no significant difference in prevalence between females (6.99%, 32/458) and males (9.21%, 48/521; P = 0.24; Table 1). Out of 4,195 tested camelid and cattle herds, 144 had at least 1 individual in the herd that tested positive for BVDV antigen (3.43%) between July 1, 2007 and December 31, 2010.
Summary of Bovine viral diarrhea virus (BVDV) prevalence in camelid and cattle herds (2007–2010) and BVDV seroprevalence in wild white-tailed deer (2009–2010) in New York State, USA.

The prevalence density surface generated by the dual kernel estimation method for BVDV-positive camelid and cattle herds showed variation in prevalence ranging from 0% to 7.5% (Fig. 3). Discrete areas of higher prevalence were detected in the western and middle portions of the central region of New York. Software e analysis confirmed the presence of a significant cluster (P = 0.001) of positive herds in the western portion of the central region of the state (Table 2, Fig. 3). Apparent seroprevalence ranged from 0% to 23.9% in the white-tailed deer BVDV seroprevalence density surface. Focal areas of elevated prevalence were revealed in the middle part of the central region of New York and in the western portion of the northern aspect of the state. These areas nearly exactly corresponded to the locations of 2 significant clusters (P = 0.001 for cluster in central region; P = 0.001 for cluster in northern region) of seropositive deer revealed by software e analysis (Table 2, Fig. 4).
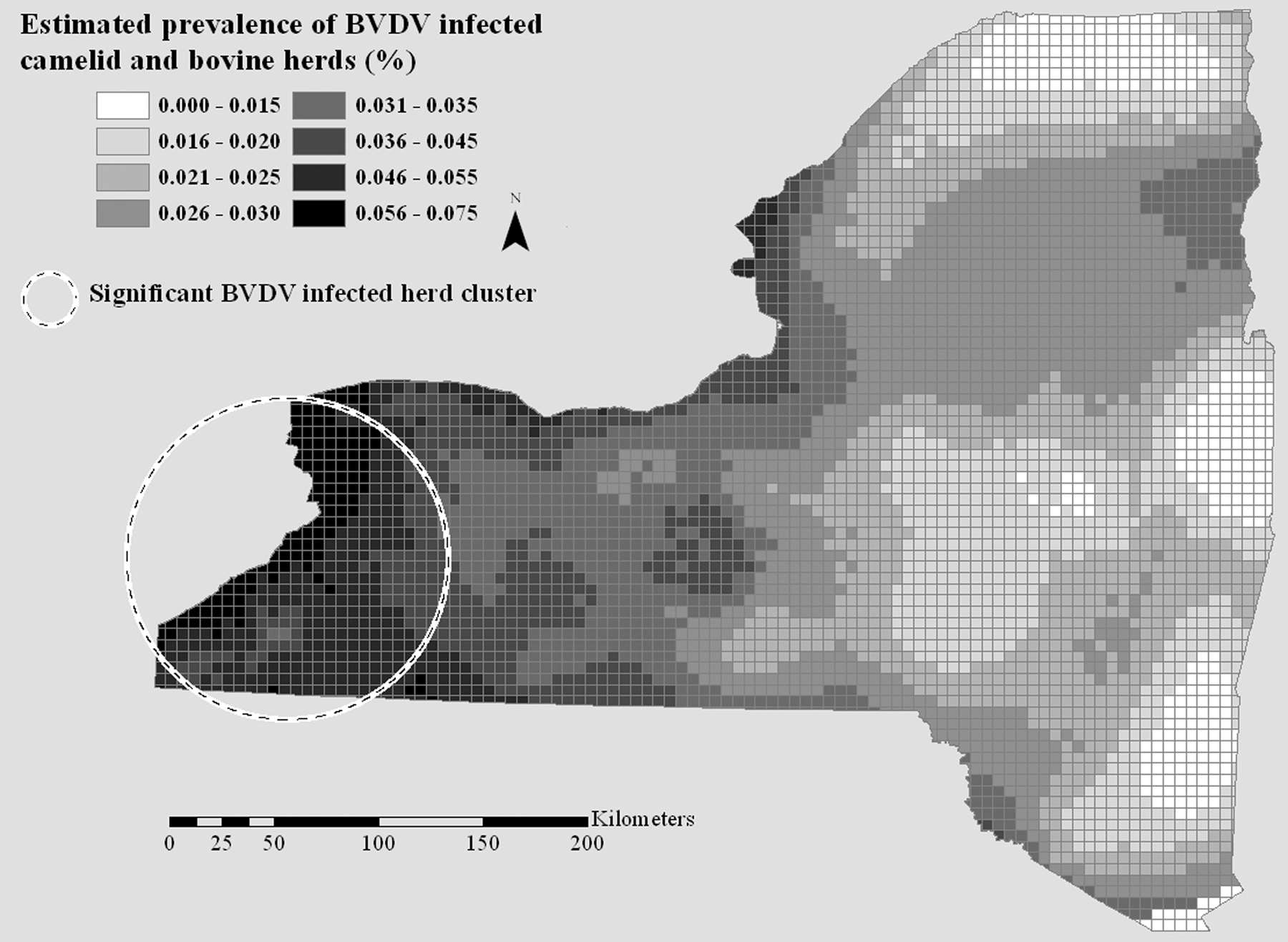
Bovine viral diarrhea virus (BVDV) prevalence disease risk surface and locations of significant clusters of BVDV-positive camelid and bovine herds in New York State, USA, 2007–2010.
Summary of statistically significant spatial clusters of Bovine viral diarrhea virus (BVDV) antigen–positive herds (2007–2010) and BVDV-seropositive wild white-tailed deer (2009–2010) in New York State, USA.
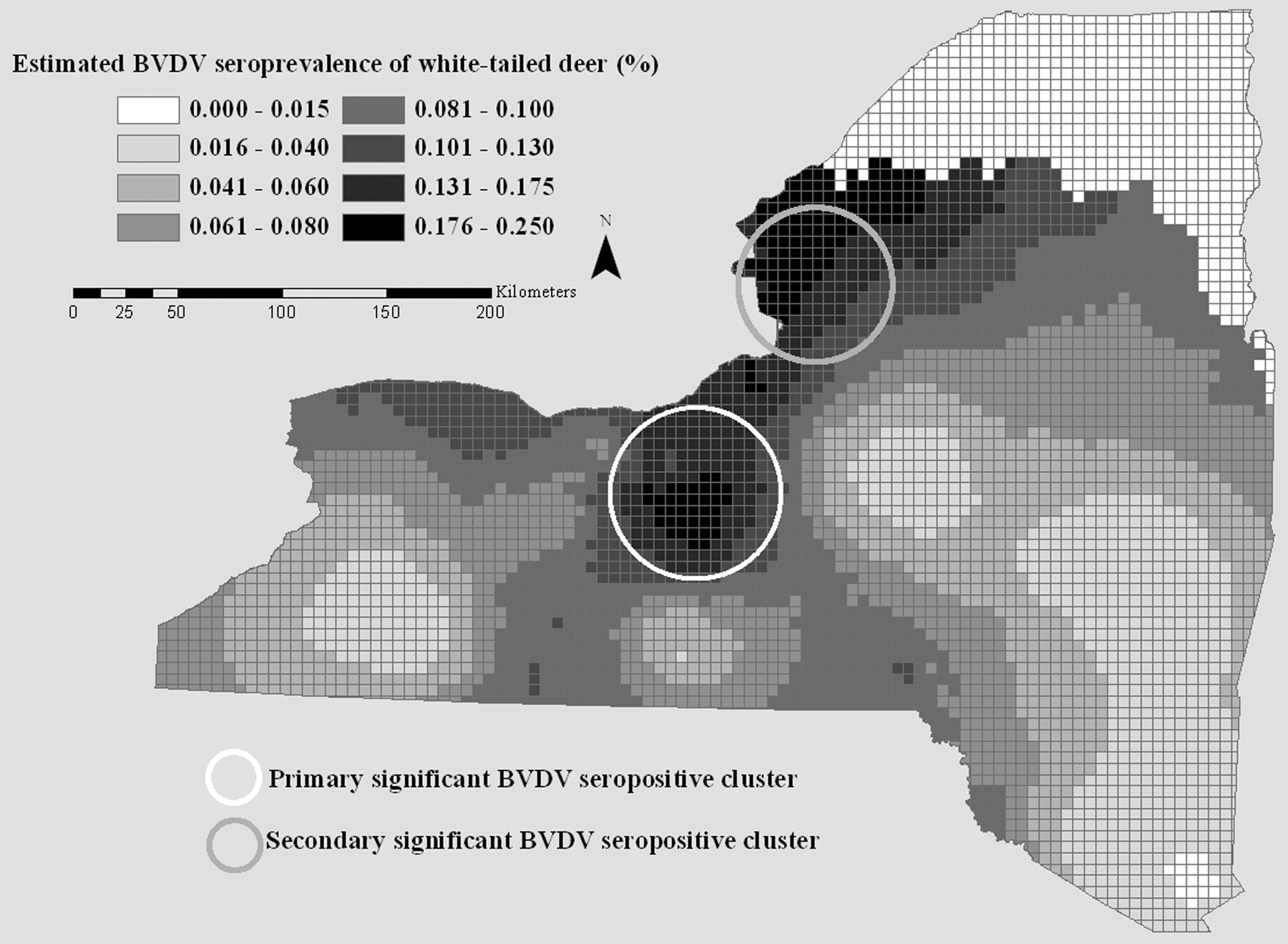
Bovine viral diarrhea virus (BVDV) seroprevalence disease risk surface and locations of significant clusters of BVDV-seropositive white-tailed deer (Odocoileus virginianus) in New York State, USA, 2009–2010.
Discussion
Persistence of BVDV may be a particular problem in central New York because it appears that the virus exists in multiple host species. Depending on the rate of interspecies transmission, bovids, camelids, and white-tailed deer in the area may function together as a host community for the pathogen. 3 Domestic livestock producers in this area that are considering eradication of BVDV from their herd should consider blocking tactics to prevent both indirect and direct contact between livestock and potentially infected white-tailed deer.
The spatial point pattern analyses also suggest that domestic livestock herds located within the significant cluster of infected herds in western New York do not need to include blocking tactics in their BVDV management or eradication herd plans. There is a comparatively low estimated BVDV seroprevalence in the spatially concurrent white-tailed deer population, and it appears that the chance for interspecies viral transmission is low.
The spatial point analyses also reveal a focus of increased BVDV seroprevalence in deer in northern New York in the absence of a geographically concurrent spike in BVDV domestic livestock herd prevalence. This suggests that BVDV is able to be maintained in this wild white-tailed deer population without periodic viral input from infected domestic livestock. Independent maintenance of BVDV in wild white-tailed deer populations is dependent upon the presence of PI deer in the population during the critical gestational period (approximately 50–67 days of gestation for white-tailed deer).22,25 In New York, the critical gestational period for does falls between mid-January and mid-February, meaning that PI fawns need to survive at least 8 months in order to be able to infect pregnant does and potentially cause the birth of more PI deer. In Illinois, high contact probabilities in white-tailed deer have been reported in forest cover during the gestation season, 16 leading to a greater chance for PI individuals to contact a pregnant doe. A single PI fawn born to a doe experimentally infected with BVDV was reported to survive to 10 months of age, 21 and wild PI deer have been diagnosed in Alabama and Indiana,22,24 but the average life span of PI deer in the wild is not known. The current study supports the theory that BVDV is able to maintain itself in particular white-tailed deer populations, suggesting that at least some PI deer live long enough to infect pregnant does and perpetuate the infection within the population.
The patterns of BVDV seroprevalence in deer and prevalence in domestic herds are unique in western and northern New York and overlapping in central New York. The predominate regional cattle operation (dairy vs. beef) may be one of the driving forces behind the presence or absence of interspecies viral transmission, but the dataset used for the herd prevalence point pattern analysis did not reveal whether the tested herds were dairy or beef. Beef cattle are typically believed to be more at risk for transmission of BVDV from wildlife due to the extensive management systems used to raise beef cattle in the United States. 30 If there are more beef herds in central New York than in the northern or western regions, then there would likely be a greater chance for viral transmission between deer and cows in the central area of the state.
Buck harvest density is often used as a relative index of deer density because of the difficulty and cost associated with gaining accurate estimates of deer density based on absolute counts. Buck harvest density is indicative of trends in a population and may provide some suggestion of population density. The lowest buck harvest density reported in New York in 2010 (NYSDEC, Division of Fish, Wildlife, and Marine Resources, Bureau of Wildlife: 2011, Management plan for white-tailed deer in New York State, 2012-2016. Available at: http://www.dec.ny.gov/docs/wildlife_pdf/deerplan2012.pdf. Accessed on January 22, 2013) was from the northern part of the state (0.2–1.2 bucks per square km), which includes the significant cluster of BVDV-seropositive deer. The central part of New York reported an intermediate buck harvest (0.8–1.5 buck per square km), and the highest buck density was reported in the western aspect of the state (0.8–2.0 bucks per square km). The only significant cluster of infected herds is located in the area of New York with the highest reported buck harvest densities. This suggests that interspecies viral transmission is not dependent upon deer densities and that intraspecies viral transmission among white-tailed deer may be more dependent upon the movement or social behavior of deer in a particular area.
The significant cluster of BVDV-seropositive deer in northern New York is located adjacent to the Adirondack Mountains region. However, the buck harvest density of the cluster is reported to be between 0.4 and 1.2 bucks per square km, whereas the buck harvest density in the Adirondacks is only 0.2−0.4 bucks per square km. Because of the geographic proximity of the cluster to the Adirondack region, it is likely that seasonal deer behavior and migration patterns in the area of the cluster are similar to reports from the Adirondacks, in spite of the higher estimated densities. White-tailed deer are reported to congregate in winter yards from December to April in the Adirondacks, primarily because of severe winter weather and deep snow. 15 Range fidelity is reported to be less pronounced in winter than in summer,1,29 suggesting that deer may have a greater chance for contact with new deer in winter when they congregate. The higher estimated deer densities on the western periphery of the Adirondacks, in combination with the proximity of the site to the Adirondacks and the likelihood that at least some yarding occurs, suggests that deer may congregate in higher densities in the winter and exhibit a greater chance for disease transmission. This may account for the significant cluster of BVDV-seropositive deer in the northwestern part of New York.
While opportunistic sampling is frequently the only method available to researchers to conduct disease surveillance in wildlife populations, it does have many potential disadvantages. Hunter-harvested collection regimes potentially lead to overrepresentation of adult animals, and certain geographic areas that may be pertinent in a spatial epidemiological investigation may lack enough samples to detect the virus even if it were present at a high level. 11 Similarly, the bovine and camelid BVDV dataset only includes farms that submitted samples for BVDV antigen testing between July 1, 2007 and December 31, 2010. This dataset is thus likely biased towards veterinarians recommending BVDV testing and herd owners that are financially capable of and interested in testing. Epidemiological gaps in the spatial point analysis are likely in areas where BVDV herd testing is infrequent. Consequently, the estimated BVDV prevalence surface applies only to the herds being tested, and may not necessarily represent the risk of BVDV infection to all cattle and camelid herds in New York.
Similarly, not all diagnostic tests validated for use in domestic livestock are equally sensitive and/or specific in wildlife. The use of the SVANOVIR BVDV p80-AB ELISA b as a screening test for white-tailed deer serum testing was explored in this project because of the ability of most researchers to perform ELISA testing, as compared to SN. While SN requires full cell culture facilities, ELISAs require only basic laboratory equipment. The SVANOVIR ELISA has not yet been validated for use in white-tailed deer but it exhibited 100% agreement with ELISA-positive samples and SN results. Potential causes of false-positive samples include interaction with a nonspecific antibody or interference from a blood component that may be present in white-tailed but absent in cattle. There is the chance that false-positive results may be clustered. Commercial availability of an ELISA determined to be appropriate for white-tailed deer samples will hopefully lead to expanded BVDV seroprevalence investigations in this species.
The research in the current study emphasizes the need to conduct a risk assessment for pathogen transmission between wildlife and livestock on a regional scale. Additionally, the spatial point pattern analysis technique described herein can provide information necessary for the epidemiological risk assessment that should precede the development of any regional BVDV management plan.
Footnotes
Acknowledgements
The authors thank Inda Mahler and LouAnn Pelowski for assistance with serum neutralization. The authors also thank all of the State University of New York College of Environmental Science and Forestry students, the New York State Department of Environmental Conservation, and Elizabeth Bunting for help with sample collection; and Isabelle Schweitzer and Roxane Ellis for help with BVDV herd database compilations.
a.
Vacutainer, BD, Franklin Lakes, NJ.
b.
SVANOVIR BVDV p80-AB ELISA kit, Svanova Biotech AV, Uppsala, Sweden.
c.
ArcMap GIS software version 10.0, Environmental Systems Research Institute Inc., Redlands, CA.
d.
CrimeStat software version 3.3, Ned Levine & Associates, Houston, TX, and the National Institute of Justice, Washington, DC.
e.
SaTScan version 9.0, Martin Kulldorff and Information Management Systems Inc., Silver Spring, MD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Edna Bailey Sussman Foundation, Sigma Xi (grant ID no. G20101015153851), and the SUNY-ESF Graduate Student Association.
