Abstract
We performed a thorough fit-for-purpose evaluation of commercial ELISAs for the detection of bovine viral diarrhea virus (BVDV)-specific antibodies in serum and in milk by testing 2 panels of well-characterized serum and milk samples. Sixteen ELISAs from 9 different manufacturers, available on the Belgian market at the time of our study, were assessed for their diagnostic and analytical sensitivity (DSe and ASe, respectively), diagnostic specificity (DSp), and repeatability relative to the virus neutralization (VN) test considered to be the gold standard assay. Using serum as a matrix, DSe was much lower for competitive (c)ELISAs (min. 45%, max. 65%) than for indirect (i)ELISAs (min. 85%, max. 100%), partly because of the lower detection of positive samples from vaccinated animals included in the panel. ASe was also better for iELISAs; DSp was >95% for all but 2 ELISAs. Repeatability, expressed as coefficients of variation (CV) of optical densities, was generally good, although 3 ELISAs had a mean CV >10%. With milk samples, as observed for serum, DSe was lower for cELISAs (min. 57%, max. 75%) than for iELISAs (min. 61%, max. 89%), and DSp was high for all ELISAs (min. 94%, max. 100%). Both DSe and ASe were lower when testing milk samples compared to serum samples. These results confirm that serologic monitoring of BVDV-free herds should be performed using serum samples of unvaccinated animals to avoid interference of vaccination and to maximize the chance of detecting seroconversion linked to BVDV infection. Further investigations using a larger collection of field samples are recommended.
Keywords
Introduction
Bovine viral diarrhea (BVD) is one of the most important viral diseases in cattle, causing severe economic losses to the cattle industry worldwide. The causative agent of this disease is species Bovine viral diarrhea virus (BVDV; genus Pestivirus, family Flaviviridae). Two species or genotypes of BVDV are recognized, BVDV-1 and BVDV-2.4,25 Following the successful eradication of BVDV in Scandinavian countries, several European countries, such as Switzerland, Germany, Austria, Luxemburg, and Ireland, are undertaking efforts to control and eventually eradicate BVDV. Persistently infected (PI) animals are produced when fetuses are infected in utero between the second and fourth month of pregnancy of the dam, and they are the main source for the dissemination of the disease within and between herds. 18 The genome of BVDV constitutes single-stranded RNA coding for several structural proteins (capsid [C] and envelope [E]) and nonstructural proteins [NS]). Some of these proteins are antigens targeted directly or indirectly by laboratory tests. Erns (or E0) is an envelope glycoprotein with RNase activity that is secreted and involved in the immune interaction with host cells. 25 It can be detected by antigen ELISAs for the identification of BVDV PI animals. 7 NS3 (or P80) is a highly conserved nonstructural protein involved in the replication of the virus. 25 Animals previously exposed to BVDV harbor specific anti-NS3 antibodies detected by several commercial antibody (Ab)ELISAs (mostly competitive ELISAs). Other serologic tests recognize a wider range of antibodies, such as the virus neutralization (VN) test, detecting neutralizing antibodies, and most indirect ELISAs detecting total antibodies.
The Belgian national BVDV eradication program started in January 2015, based on the compulsory testing of all newborn calves for BVDV, to identify and eliminate all PI animals. In a second phase, planned to start mid-2017, a surveillance scheme to monitor BVDV-free herds will be set up. Progressively, certified BVDV-free herds will have the possibility to stop individual BVDV testing at birth, and serologic monitoring will be performed on a regular basis to ensure that these herds have not been re-infected. The detection of BVDV-specific antibodies in serum or milk samples is an efficient way to show the exposure to BVDV of individual animals or of a herd.12,15 This can be done at herd level by testing one or several groups of 5–10 young animals for the presence of BVDV antibodies (“spot test”), as described by several authors.6,12,22 However, vaccination against BVDV can also induce the production of antibodies and therefore can interfere with the interpretation of test results in serum samples1,8,10,19,20 as well as in milk samples.9,24 Vaccination against BVDV is allowed and commonly used by Belgian farmers; only inactivated vaccines were available at the time of our study.
Different questions were raised regarding the performance of the ELISAs available at the time of our study: the possible differences between the performances of competitive (c)ELISAs and indirect (i)ELISAs, and between ELISAs detecting total antibodies versus ELISAs detecting antibodies against specific BVDV antigens such as E0 (Erns) and NS3 (p80); the effect of short versus long incubation protocols; the possibility to certify or monitor BVDV-free herds by means of serology on serum and milk samples; and the impact of vaccination on seroconversion. Evaluations of several AbELISAs have been reported previously, but a broad study comparing most available ELISAs has not been reported, to our knowledge. To answer these questions, we submitted all BVDV AbELISAs available on the Belgian market to this fit-for-purpose evaluation, using 2 panels of well-characterized serum or milk samples. We assessed their diagnostic and analytical sensitivity (DSe and ASe, respectively), diagnostic specificity (DSp), and repeatability using the VN test as the reference assay.
Materials and methods
Antibody ELISAs
We evaluated, on serum and/or milk matrices, 16 commercial ELISAs produced by 9 manufacturers, available on the Belgian market at the time of our study. Nine ELISAs were tested both on serum and on milk, 6 were tested only on serum, and 1 only on milk, following assay specifications. Eleven ELISAs were cELISAs, all of them targeting anti-NS3 antibodies, except 1 targeting anti-E0 antibodies; the remaining 5 assays were iELISAs, all of them detecting total antibodies, except 1 targeting anti-NS3 antibodies. Short and long incubation protocols were performed, and all results were analyzed according to the assay manufacturer’s guidelines.
The following ELISAs were evaluated:
Monoscreen AbELISA BVDV (NS3)/blocking; Monoscreen AbELISA BVDV (E0)/blocking; Monoscreen AbELISA BVDV (NS3)/indirect, double wells (Bio-X Diagnostics, Rochefort, Belgium);
CIVTEST Bovis BVD/BD p80 (HIPRA, Amer, Spain);
IDEXX BVDV p80 Ab; IDEXX BVDV Total Ab (IDEXX Laboratories, Westbrook, MA);
ID Screen BVD p80 Antibody Competition (IDvet, Grabels, France);
INgezim Pestivirus Compac (INGENASA, Madrid, Spain);
LSIVet Ruminant BVD/BD p80 Serum; LSIVet Ruminant BVD/BD p80 Milk; PrioCHECK BVDV Antibody (Thermo Fisher Scientific, Waltham, MA);
SVANOVIR BVDV p80-Ab; SVANOVIR BVDV-Ab (confirmation and screening formats) (SVANOVA, Boehringer Ingelheim, Ingelheim am Rhein, Germany);
SERELISA BVD p80 Ab Mono Blocking (Zoetis, Parsippany, NJ); and
BVD-MD IgG ELISA (TestLine Clinical Diagnostics, Brno, Czech Republic).
Bulk tank milk samples were evaluated using corresponding cutoffs if this distinction was made by the manufacturer (CIVTEST Bovis BVD/BD p80, LSIVet Ruminant BVD/BD p80 Milk, and IDEXX BVDV Total Ab). For each matrix, samples were distributed on 2 ELISA plates per evaluated assay, except for 2 that required 4 plates because their protocol prescribed 2 wells per serum or milk sample tested (Monoscreen AbELISA BVDV (NS3)/indirect, double wells; and SVANOVIR BVDV-Ab, confirmation format).
Virus neutralization test
In-house VN tests against BVDV-1 and BVDV-2 were performed on individual serum samples to detect antibodies against BVDV-1 strain NADL 11 and against BVDV-2 strain 3534. 17 The VN tests were performed in microtiter plates. Two-fold dilutions of heat-inactivated sera (56°C for 30 min) were tested in duplicate, starting at 1:5, against ~100 TCID50 of virus. After 2 h of pre-incubation at 37°C, Madin–Darby bovine kidney (MDBK) cell suspensions were added to each well. Cytopathogenic effect was determined after 3 d directly by optical microscopy (BVDV-1) or after immunolabeling with an anti-pestivirus polyclonal serum (BVDV-2). Each well was scored as positive or negative for viral growth. The inhibition of viral growth by virus neutralizing antibodies was determined by calculating the 50% endpoint titer of 50% effective (neutralizing) dilution (TCID50). 21 Titers were expressed as the reciprocal of the highest serum dilution yielding virus growth neutralization. Samples were considered positive if their VN titer was ≥1:10 according to World Organization for Animal Health (OIE) recommendations at the time of the study (as of 2017, the cutoff recommended by OIE is 1:8; https://goo.gl/owUd28). Samples with a VN titer = 1:5 or 1:7.5 were therefore considered negative. All sera included in the panel were tested in both BVDV-1 and -2 VN tests, whereas serum samples coupled to milk samples were tested in a BVDV-1 VN test only. Samples were considered positive when at least 1 of the 2 assays showed a positive result.
Panel of serum and milk samples
VN testing of serum was regarded as the gold standard assay for determination of the (positive or negative) status of sera and of corresponding individual milk samples. Most of the field samples were collected between October and December 2013, and the vaccination status of the animal and the history of BVDV circulation in the herd were recorded at the same time (Supplementary Table 1). Sera from 2 VN-positive and 3 VN-negative PI animals were also included. Some of the positive and negative samples were collected from unvaccinated animals involved in an experimental infection study (infected or negative control). 23 The design of the panels tested allowed evaluation of DSe and DSp, relative to VN test, and the ASe and repeatability of each ELISA for each incubation protocol and each matrix.
Serum samples included in the serum panel
The serum panel consisted of 20 positive samples, 60 negative samples, 2 different 2-fold dilution series of 2 strong-positive samples (BVDV-1 and BVDV-2), and 20 repeats of a strong-positive, a weak-positive, and a negative sample. All samples were distributed on 2 or 4 plates, depending on the number of wells needed according to the ELISA manual instructions.
Given that there is no BVDV AbELISA with true DIVA (differentiating infected from vaccinated animals) test properties, nor commercial vaccine that allows this differentiation, 20 seropositive samples from previously BVDV-infected and/or BVDV-vaccinated animals (field samples and experimental infections) were included in the panel (Supplementary Table 2). All field samples were collected from adult animals or young stock >6-mo-old in order to avoid interference of maternal antibodies, except for 2 sera obtained from 2 seropositive young PI calves.
Two-fold dilution series (DIL1 and DIL2) were prepared, using 2 different strong-positive sera (VN titer >1:640), the first collected from an animal (E-2) present in an infected herd and the second from an animal (MAC-5) experimentally infected with a BVDV-2 strain. 23 Each of the dilution series consisted of 14 subsequent dilutions of the positive serum in a negative serum (starting from dilution 1:8 for E-2 and from the pure serum for MAC-5; Supplementary Table 3).
Sixty sera obtained from unvaccinated animals and tested negative by VN both for BVDV-1 and BVDV-2, were included as negatives in the panel, including 3 VN-negative sera obtained from 3 PI calves (data not shown).
Twenty aliquots (10 aliquots/plate on 2 plates) of 3 different samples collected from animals involved in an experimental infection 23 (infected or negative control) were used to assess the repeatability parameter: (1) 1 strong-positive sample (MAC-1, VN titer = 1:320 for BVDV-1 and = 1:80 for BVDV-2); (2) 1 weak-positive sample obtained after dilution 1:30 of a strong-positive serum (MAC-2, VN titer = 1:10 for BVDV-1; negative for BVDV-2); and (3) 1 VN-negative sample for both BVDV-1 and BVDV-2.
Milk samples included in the milk panel
Briefly, the milk panel consisted of 44 positive samples, 36 negative samples, two 2-fold dilution series of 2 strong-positive samples, and 20 repeats of a strong-positive, a weak-positive, and a negative sample. These samples were distributed on 2 or 4 ELISA plates, depending on the number of wells needed according to the assay instructions. All samples were collected in plastic containers pre-filled with a small amount of milk preservative (bronopol) provided by a laboratory in charge of routine milk quality control. To check that the milk preservative used had no impact on the ELISA results, 1 negative and 1 positive individual milk sample was evaluated with and without the addition of bronopol. These samples were simultaneously tested on 2 different plates with each ELISA. No significant differences were observed when comparing the results for these 2 samples (data not shown).
In total, 44 positive samples were included in the panel, including 38 individual milk samples obtained from dairy cows originating from infected herds (vaccinated or not) that had tested positive by VN on the corresponding serum sample, and 6 positive bulk tank milk samples (BM herds A–E, G7) obtained from herds in which several seropositive lactating cows were present and had contributed to the bulk tank (Supplementary Table 4).
Samples (n = 36) obtained from unvaccinated animals that were VN-negative on the corresponding serum were included in the panel (data not shown).
Two 2-fold dilution series (DIL1 and DIL2) were prepared starting from 2 strong-positive individual milk samples obtained from animals E-6 and C-1 present in herds infected with BVDV-1 (VN titer of the corresponding sera >1:640 and 1:480 respectively), both diluted in an AbELISA-negative milk sample (ultra-high temperature [UHT] processed skimmed milk). Subsequent dilutions (n = 13) were used, starting from the undiluted milk samples (1:1–1:4,096).
Aliquots (10 aliquots/plate on 2 plates) of 3 samples were used to assess repeatability: (1) 1 strong-positive individual milk sample from an animal present in an infected herd (E-2, BVDV-1 VN titer of the corresponding serum >1:640); (2) 1 weak-positive sample obtained after diluting 1:10 in negative UHT-processed skimmed milk, a strong-positive individual milk sample from an animal present in an infected herd (C-1, BVDV-1 VN titer of the corresponding serum = 1:480); and (3) 1 negative individual milk sample (corresponding serum BVDV-1 VN-negative).
Data analysis
For each evaluated ELISA, we determined the number of samples in the panel of positive samples (20 for serum and 44 for milk) and negative samples (60 for serum and 36 for milk) that were correctly identified as positive or negative, respectively. From these results, DSe and DSp were calculated as follows:
For DSe and DSp, 95% confidence intervals (“exact” method, based on binomial distribution) were calculated using EpiTools (http://epitools.ausvet.com.au). ASe or detectability was expressed by the limit of detection in the dilution series (i.e., the highest dilution at which the considered sample still tested positive [or doubtful]).
Results were interpreted according to the manufacturer’s instructions for each ELISA; “weak-positive” results were considered positive (CIVTEST Bovis BVD/BD p80 for serum, and LSIVet Ruminant BVD/BD p80 Serum), whereas “doubtful” results were reported as doubtful throughout the evaluation (not considered positive or negative; BVDV p80 Ab for serum; ID Screen BVD p80 Antibody Competition for serum; SERELISA BVD p80 Ab Mono Blocking; INgezim Pestivirus Compac; BVDV Total Ab for serum and milk; BVD-MD IgG ELISA).
The repeatability of each evaluated ELISA was assessed by calculating the coefficient of variation (CV) of the uncorrected optical density (OD) obtained for each of the 3 samples tested in 20 replicated aliquots (1 strong-positive, 1 weak-positive, and 1 negative sample). A mean CV was also calculated for each ELISA (average of the 3 CVs obtained for the 3 series of replicates).
Results
Serum samples
For DSe, total Ab iELISAs (DSe: 85–100%; average 94% for the total Ab iELISAs; Table 2) scored much better than cELISAs (DSe: 45–65%; average 57% for anti-NS3 cELISAs; Table 1). The anti-E0 cELISA (DSe: 75%) scored better than all anti-NS3 cELISAs. In contrast, the anti-NS3 iELISA scored much lower compared to total Ab iELISAs with a low DSe (55%) comparable to the worst cELISAs. Some of the positive serum samples of the panel frequently tested negative by most cELISAs and by the anti-NS3 iELISA (samples marked with an asterisk in Supplementary Table 2). Most of these samples were obtained from animals vaccinated against BVDV; some were from animals originating from BVDV-free herds and had low VN titers, but others were from animals with high VN titers from farms previously or currently infected with BVDV. For the 6 VN-positive sera from uninfected vaccinated animals, all iELISAs scored them as positive (6 of 6) except for the Monoscreen AbELISA BVDV (NS3)/indirect, double wells (2 of 6) and the SVANOVIR BVDV-Ab–screening format (5 of 6). For anti-NS3 cELISAs, the proportion of positive samples among these sera varied between 3 of 6 positive and 0 of 6 positive depending on the ELISA used and on the incubation protocol. The only ELISA that provided a negative result for the 6 samples was the Monoscreen AbELISA BVDV (NS3)/blocking.
Results for the competitive ELISAs using serum as matrix: number of compliant positive (doubtful) or negative results with corresponding diagnostic sensitivity (DSe) and specificity (DSp) and with highest dilution detected positive (doubtful) for the 2 series of dilution of strong-positive samples (DIL1 = E-2 and DIL2 = MAC-5).
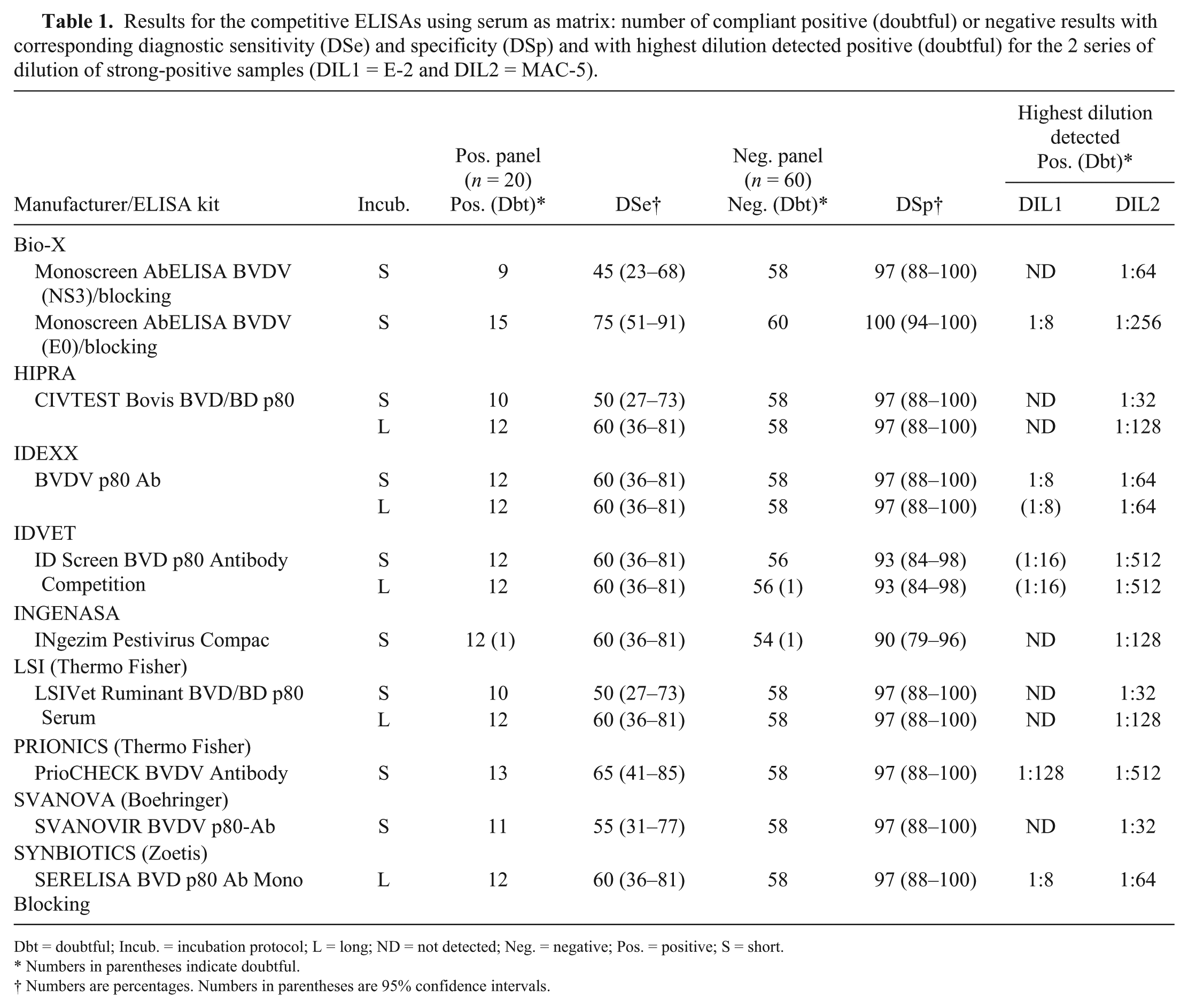
Dbt = doubtful; Incub. = incubation protocol; L = long; ND = not detected; Neg. = negative; Pos. = positive; S = short.
Numbers in parentheses indicate doubtful.
Numbers are percentages. Numbers in parentheses are 95% confidence intervals.
All iELISAs scored well for detectability (ASe) on serum, always detecting the 20 replicates of the weak-positive sample from the panel (dilution of MAC-1; BVDV-1 VN titer = 1:10; Table 3) with a good ability to detect highly diluted positive samples (DIL1 = E-2 and DIL2 = MAC-5) in both dilution series (highest detected dilution 1:32–1:4,096 depending on the ELISA and the sample; Table 2). In contrast, the cELISAs did not detect consistently all replicates of the weak-positive sample (Table 3). Moreover, with cELISAs, low dilutions of the positive sample in the BVDV-1 dilution series (DIL1; 1:8, corresponding to a BVDV-1 VN titer >1:640) often tested negative, whereas the BVDV-2 dilution series (DIL2) was readily detected at higher dilutions (dilution 1:32 was always detected, corresponding to a BVDV-2 VN titer = 160; Table 1). All evaluated ELISAs scored the 20 repeated aliquots of the strong-positive sample (MAC-1; BVDV-1 VN titer = 1:320) and of the negative sample as positive and negative, respectively. When comparing short versus long incubation protocols for ELISAs that have both protocols proposed by the manufacturer, we observed that the long incubation protocol often resulted in an increased DSe and ASe.
Results for the indirect ELISAs using serum as matrix: number of compliant positive (doubtful) or negative results with corresponding diagnostic sensitivity (DSe) and specificity (DSp) and with highest dilution detected positive (doubtful) for the 2 series of dilutions of strong-positive samples (DIL1 = E-2 and DIL2 = MAC-5).
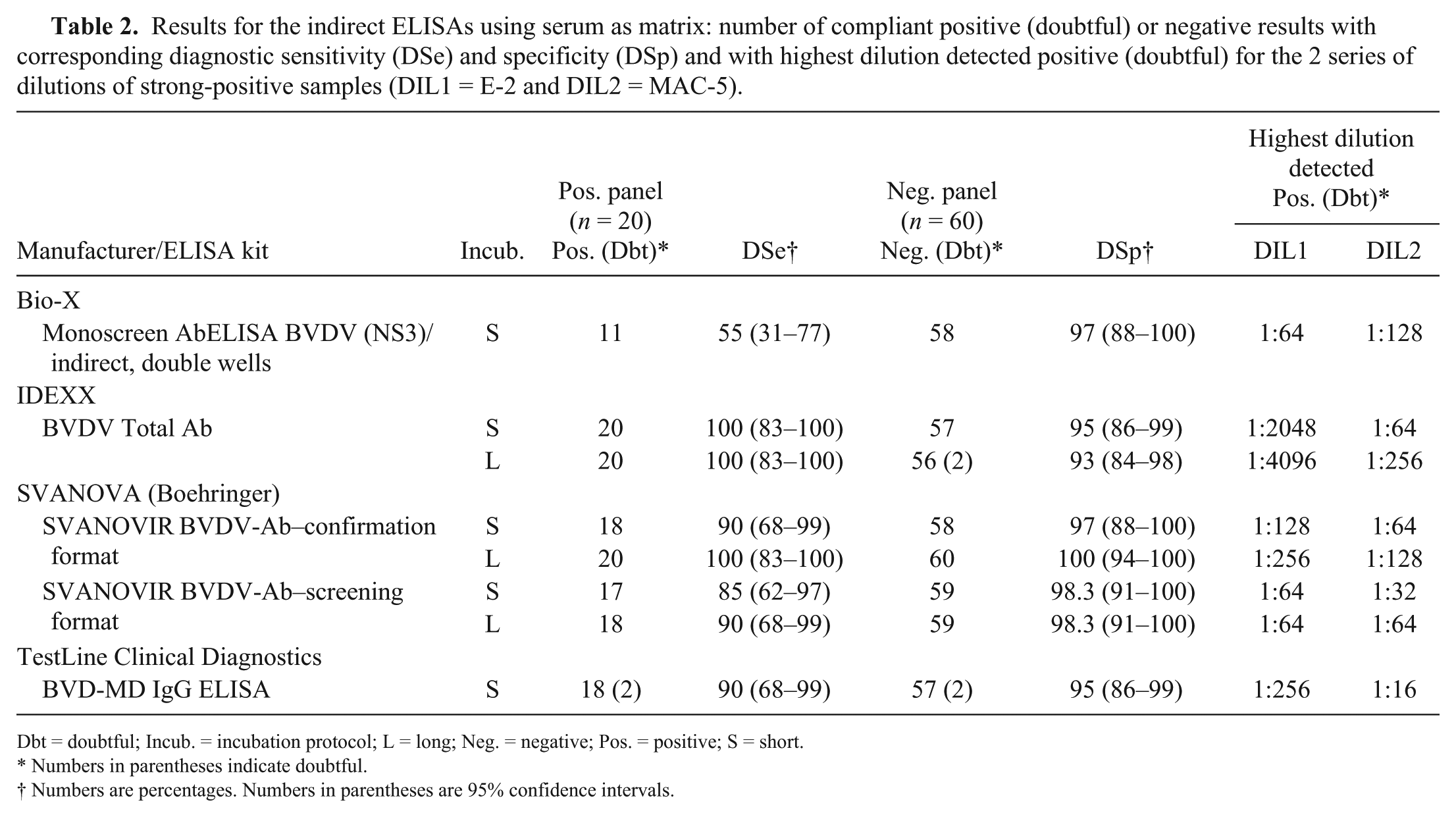
Dbt = doubtful; Incub. = incubation protocol; L = long; Neg. = negative; Pos. = positive; S = short.
Numbers in parentheses indicate doubtful.
Numbers are percentages. Numbers in parentheses are 95% confidence intervals.
Repeatability assessment of ELISAs using serum as matrix: number of compliant positive (doubtful) or negative results and coefficients of variation (CV) of optical density values of the repeated aliquots for the strong-positive (SPOS = MAC-1), weak-positive (WPOS = MAC-2 diluted 1:30), and negative samples, and mean CV for the 3 series.
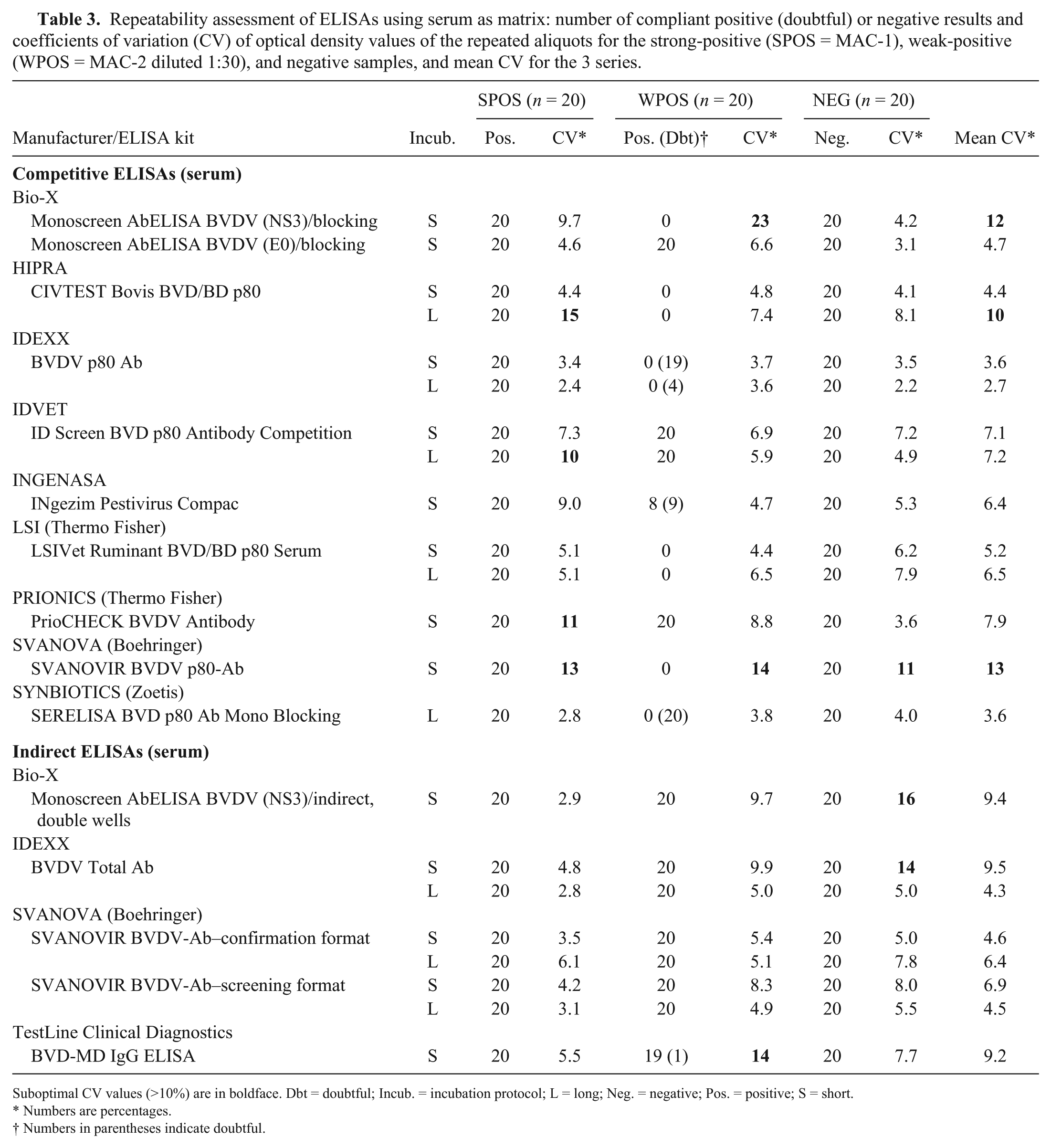
Suboptimal CV values (>10%) are in boldface. Dbt = doubtful; Incub. = incubation protocol; L = long; Neg. = negative; Pos. = positive; S = short.
Numbers are percentages.
Numbers in parentheses indicate doubtful.
In contrast to DSe results, most ELISAs had a good DSp (≥95%) except for 3 ELISAs (Table 5). Two of the 3 VN-negative sera obtained from PI calves consistently tested positive with all ELISAs except 2 (Monoscreen AbELISA BVDV (E0)/blocking, and SVANOVIR BVDV-Ab–confirmation format).
Regarding repeatability, whereas all iELISAs had mean CVs <10%, 3 (anti-NS3) cELISAs had a mean CV >10%. Five competitive and 3 iELISAs had CVs >10% for at least 1 of the 3 series of aliquots, and 3 of them had a CV > 5% for 1 of the 3 series, reflecting poor repeatability performances (Table 3).
Milk samples
Competitive ELISAs (DSe: 57–75%; average DSe = 67%) were found again to be less sensitive compared to total Ab iELISAs (DSe: 61–89%; average DSe = 77%), whereas anti-NS3 iELISAs (DSe = 61%) performed similarly to the worst cELISAs. Some of the positive milk samples of the panel frequently tested negative by several ELISAs (samples marked with an asterisk in Supplementary Table 4). These samples were obtained both from vaccinated and unvaccinated animals, including animals originating from farms previously or currently infected with BVDV, with high VN titers on the corresponding serum. The 6 samples derived from bulk milk tanks from different farms all tested positive by all evaluated ELISAs.
All ELISAs had a lower detectability (ASe) on milk compared to serum. Indeed, the highest dilution detected positive was usually 1:8 (min. 1:2, max. 1:32, depending on the assay; Table 4). Moreover, only 2 of 8 cELISAs and 4 of 6 iELISAs were able to detect all 20 repeats of the weak-positive sample from the panel (Table 5). Both the strong-positive and the negative sample were correctly identified by all ELISAs.
Results for ELISAs using milk as matrix: number of compliant positive (doubtful) or negative results with corresponding diagnostic sensitivity (DSe) and specificity (DSp) and with highest dilution detected positive (doubtful) for the 2 series of dilution of strong-positive samples (DIL1 = E-6 and DIL2 = C-1).
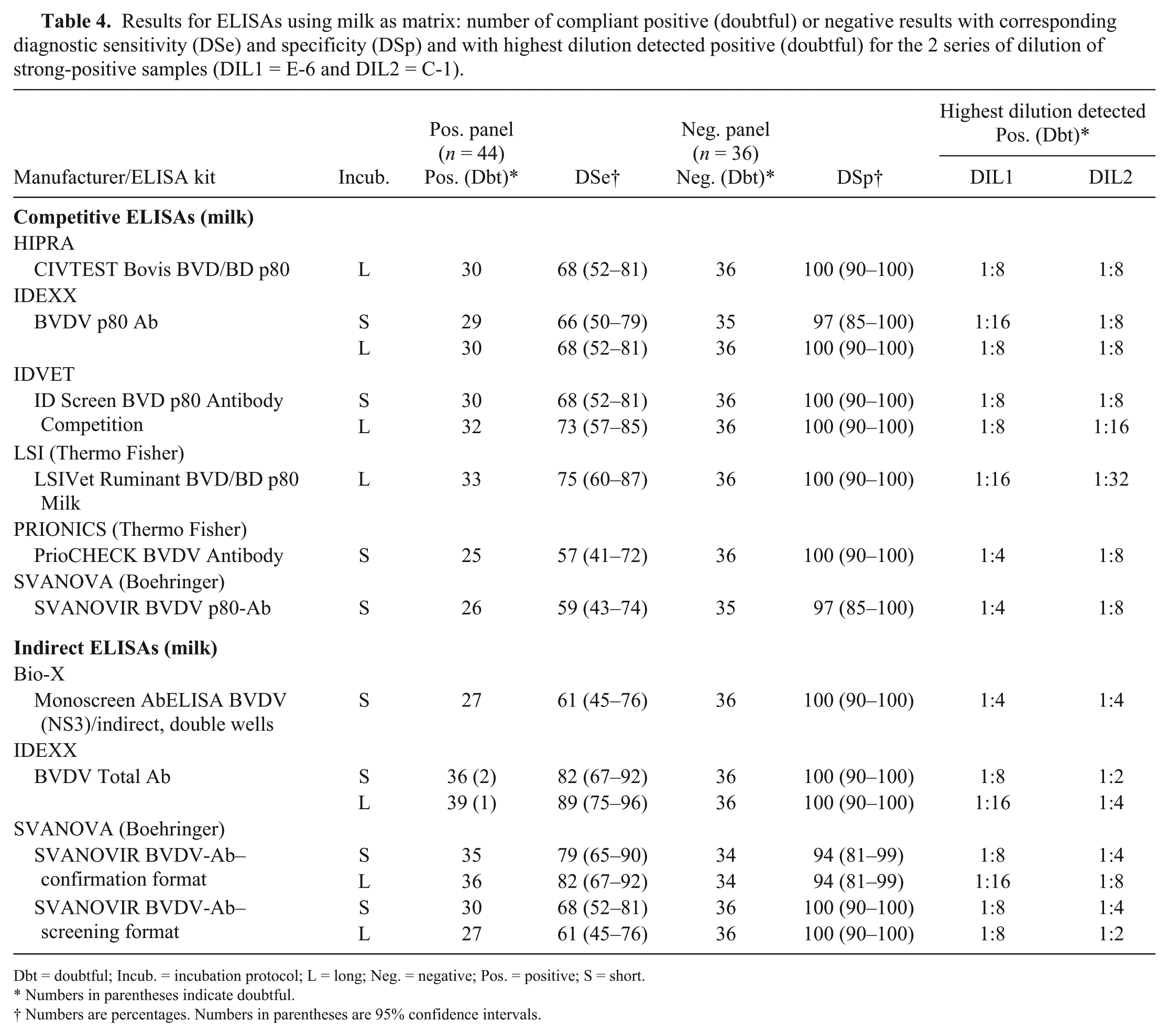
Dbt = doubtful; Incub. = incubation protocol; L = long; Neg. = negative; Pos. = positive; S = short.
Numbers in parentheses indicate doubtful.
Numbers are percentages. Numbers in parentheses are 95% confidence intervals.
Repeatability assessment of ELISAs using milk as matrix: number of compliant positive (doubtful) or negative results and coefficients of variation (CV) of optical density values for repeated aliquots of the strong-positive (SPOS = E-2), weak-positive (WPOS = C-1 diluted 1:10), and negative (NEG) samples, and mean CV for the 3 series.
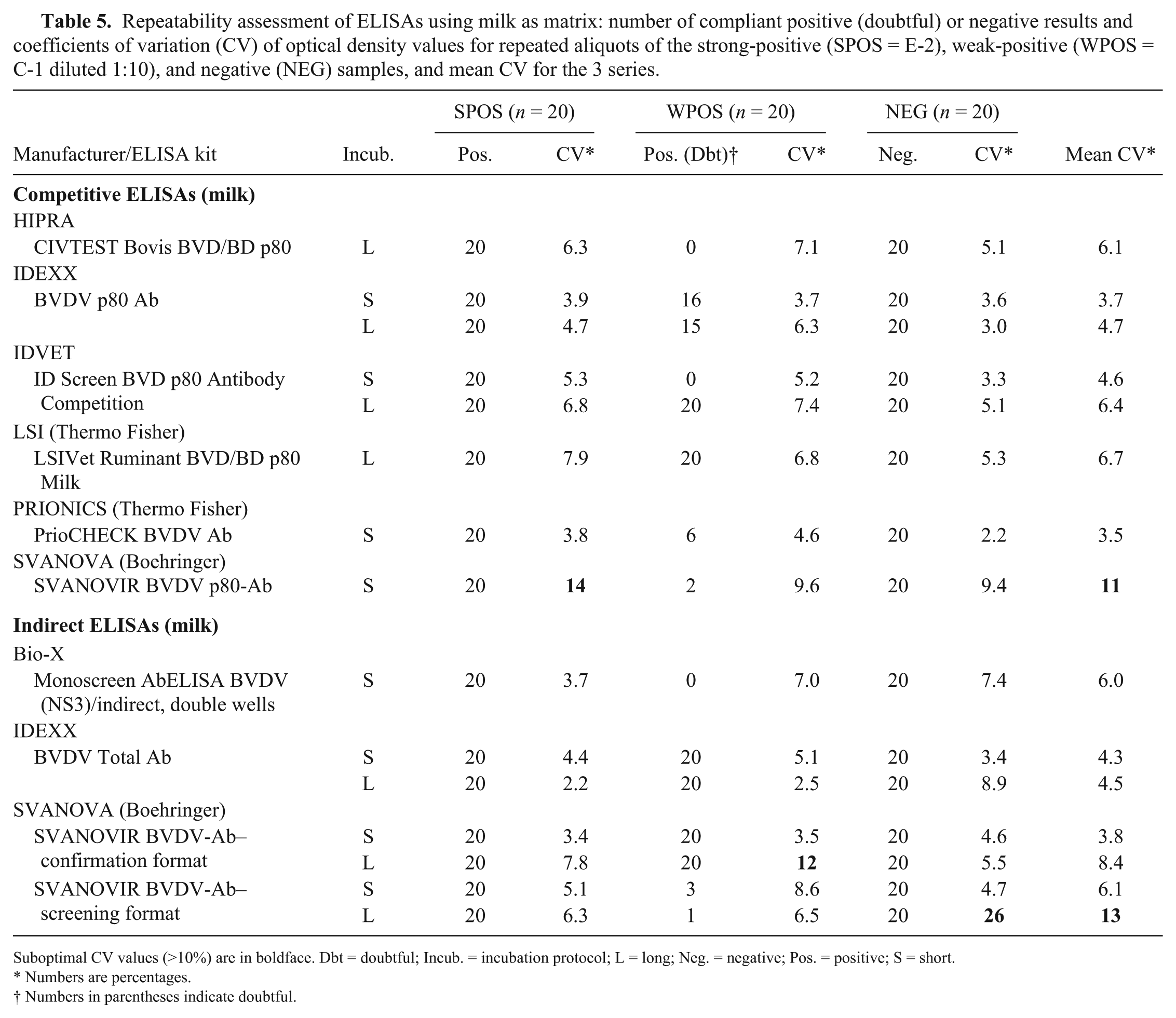
Suboptimal CV values (>10%) are in boldface. Dbt = doubtful; Incub. = incubation protocol; L = long; Neg. = negative; Pos. = positive; S = short.
Numbers are percentages.
Numbers in parentheses indicate doubtful.
The DSp of both cELISAs and iELISAs on milk samples was good, with an average value approaching 100% (cELISAs: DSp 97–100%, average DSp = 99%; iELISAs: DSp 94–100%; average DSp = 98%; Table 4). CVs calculated to evaluate the repeatability (Table 5) were generally satisfactory except for 2 ELISAs from SVANOVA that had mean CVs >10%, indicating poor repeatability.
Discussion
For both serum and milk matrices, anti-NS3 (competitive and indirect) AbELISAs were less sensitive compared to the total Ab iELISAs. Regarding detectability (ASe), we observed that iELISAs scored better than anti-NS3 cELISAs when using serum as a matrix, given that they always detected the weak-positive sample and were able to detect positive samples at higher dilutions. We did not find such a difference between competitive and indirect ELISAs with the milk panel, as some competitive and some indirect ELISAs had poor detectability compared to the other assays, being unable to detect consistently the weak-positive sample.
Specificity was excellent for both milk and serum matrices. However, with the panel of negative sera, 2 of the 3 sera from VN-negative PI calves consistently tested positive with all but 2 ELISAs. PI animals are in principle seronegative because they do not produce antibodies towards the BVDV strain with which they are infected. Yet, after colostrum ingestion, young PI calves remain seropositive for several months because of the presence of antibodies of maternal origin. PI calves can also produce antibodies after vaccination against BVDV or after infection with a BVDV strain antigenically different from the strains that established the persistent infection. It is possible that such antibodies were effectively detected by ELISAs although the sera were considered as negative according to the gold standard VN test.
Most ELISAs had good repeatability (low CVs) when using either milk or serum as matrix, but some had mean CV values >10%, which is usually considered excessive.
The lack of sensitivity of anti-NS3 cELISAs can partially be explained by the inclusion of several VN-positive samples in our panel, obtained from animals vaccinated with inactivated BVDV vaccines (Rispoval 3, Zoetis, Parsippany, NJ; or Bovilis BVD, MSD Animal Health, Kenilworth, NJ) originating from herds considered BVDV-free. Vaccination with inactivated vaccine usually induces the presence of antibodies detected by serologic tests such as VN. However, ELISAs targeting anti-NS3 antibodies do not consistently score positive when using samples from animals immunized with inactivated vaccines.1,8,10,20 The reason is that NS3 antigens, being nonstructural proteins, are produced in large quantities during virus replication after BVDV infection or after vaccination with a live attenuated vaccine, inducing the production of anti-NS3 antibodies. Inactivated vaccines, in contrast, contain a fixed antigen load and do not replicate so there is no additional production of nonstructural proteins such as NS3 in the vaccinated animal. This makes the immune response against NS3 weak or nonexistent, depending on the level of nonstructural proteins present in the inactivated vaccine. 20 Our study confirmed that some VN-positive samples obtained from vaccinated uninfected animals were more likely to test negative with the anti-NS3 ELISAs (competitive or indirect), but this was not systematic and depended on the assay and on the incubation protocol. This inconsistent negative reaction with most of the anti-NS3 ELISAs when using inactivated BVDV vaccines does not permit using these assays as DIVA tests at the individual level. Only 1 of the evaluated ELISAs (Monoscreen AbELISA BVDV (NS3)/blocking) scored all 6 samples from vaccinated uninfected animals as negative, but further investigation is needed to evaluate its true capacity to be used as a DIVA assay. The interest in such an evaluation is now limited given the increasing use on the European market of a live BVDV vaccine, which mimics natural infection. We therefore recommend serologic monitoring of BVDV-free herds only on unvaccinated animals, in which case any positive result should be regarded as possible seroconversion because of exposure to BVDV. For this reason, we decided to assess the sensitivity of ELISAs based on samples that tested positive with VN, regardless of the vaccination status of the animal. We can assume that if we had used only samples from unvaccinated animals in our positive panel, the DSe of anti-NS3 cELISAs would have scored better. But other observations indicated a lower sensitivity for anti-NS3 ELISAs: total Ab iELISAs could detect positive samples from animals experimentally infected with BVDV, at higher dilutions than anti-NS3 cELISAs. Moreover, iELISAs consistently gave a positive result for the weak-positive serum that was used to assess repeatability (obtained by diluting sera from experimentally infected animals), whereas this was not the case for some of the anti-NS3 cELISAs. We can conclude that total Ab iELISAs or cELISAs that are not targeting anti-NS3 antibodies appear to be more sensitive than anti-NS3 cELISAs, although the sensitivity of the latter has probably been underestimated in our study, given the use of samples from vaccinated uninfected animals in our positive panel.
Anti-NS3 cELISAs had a low DSe both on serum (average DSe = 57%) and on milk (average DSe = 67%), whereas, for total Ab iELISAs, the sensitivity was much better on serum (average DSe = 94%) than on milk (average DSe = 77%). Detectability (ASe) was much lower with milk than with serum samples given that dilutions of positive sera could still be detected at high dilutions (highest dilution detected 1:32–1:4,096 depending on the sample and on the assay), whereas positive individual milk samples could be detected by most ELISAs only at a maximum dilution of 1:8 (=c12.5% concentration). A bulk milk sample would therefore probably score positive only if a sufficient proportion of lactating cows has seroconverted (>10%). However, the interpretation of AbELISA results with bulk tank milk samples is usually based on different thresholds than with individual milk samples and is not restricted to a positive or negative result. Indeed some studies have shown the within-herd seroprevalence can be roughly estimated from the OD result of the bulk tank milk sample, a low percentage of inhibition being correlated with the absence or a low prevalence of positive cows in the herd. 2 Given that low DSe and ASe were observed when using milk as matrix, both with competitive and indirect ELISAs, individual milk samples should be considered as less appropriate to use than serum samples when monitoring BVDV-free herds.
A 2014 study found a high level of agreement (94.4%) and concordance (kappa = 0.865) between 125 paired serum and individual milk samples and suggested that individual milk was a good alternative to blood collection and serum testing. 16 However, it compared ELISA results between both matrices but did not evaluate performance based on the gold standard VN test. Our finding that ELISAs are less sensitive when used with individual milk samples does not exclude the use of bulk tank milk testing as a quick method of assessing herd exposure to BVDV and for the follow-up of farms having a history of BVDV infection as was shown in another study. 5 Indeed, we noted no difference between ELISAs concerning the 6 bulk milk samples that were correctly tested positive by all assays. Compared to serologic testing of individual animals, bulk tank milk testing has the advantage that it can be performed frequently and at low cost to monitor a dairy herd. This gives the possibility to compare historical data and to identify an increase of the OD sample-to-positive control value or of the inhibition percentage of a bulk milk sample, indicating a surge of antibody levels and possibly BVDV infection in the herd. Bulk tank milk testing is used to monitor BVDV-free herds in countries where BVDV eradication has been implemented or is at an advanced stage, such as Scandinavia and Switzerland.
Apart from test performance, when selecting which assay to use for a monitoring program, both price and ease of use should also be considered. Most ELISAs evaluated in our study follow similar test procedures and use similar sample volumes, except for the Monoscreen AbELISA BVDV (NS3)/indirect, double wells (serum and milk) and the SVANOVIR BVDV-Ab (confirmation format; serum and milk), which need twice the amount of time and consumables because each sample needs to be added twice on each plate (as a negative control for the Monoscreen ELISA or as confirmation for the SVANOVIR confirmation ELISA). Our results also show that, whenever possible, long incubation protocol is preferred to the short one given higher sensitivity.
Several previous studies have compared individual commercial BVDV AbELISAs with VN tests.3,13,14 These studies were not performed in similar conditions and with the same material, and they were evaluating a single commercial test at a time, therefore their results should be compared with care. Previous studies found a high sensitivity and specificity (>95%) for the evaluated assays, and they do not show major differences in terms of sensitivity between anti-NS3 cELISAs and total Ab iELISAs, in contrast to what we observed in our study. This is probably because these studies were performed on a larger number of field samples collected from endemic zones for BVD, whereas our study was done on a limited number of samples, including several positive samples from vaccinated uninfected animals, as mentioned above.
Our study reveals that the major differences in performance between ELISAs when testing serum samples are more related to the specific antibody detected (anti-NS3 vs. total Ab and anti-E0) rather than to the type of ELISA itself (cELISAs vs. iELISAs). The anti-E0 cELISA scored better than the anti-NS3 cELISAs. Similarly, the total Ab iELISAs showed better results than the anti-NS3 iELISA. For milk, the difference in performance between anti-NS3 and total Ab ELISAs was less pronounced.
Footnotes
Acknowledgements
We thank Sophie De Laet, Laurent Rosar, Muriel Verhoeven, Dennis Kozlowski, Sarah Matuyia, Virginie Colasse, and Celia Thoraval for their excellent technical assistance at the laboratory of CODA-CERVA.
Authors’ note
J-B Hanon and M De Baere contributed equally to this work.
Declaration of conflicting interests
Y. Van der Stede is currently employed with the European Food Safety Authority (EFSA) in the BIOCONTAM Unit that provides scientific and administrative support to EFSA’s scientific activities in the area of Microbial Risk Assessment. The positions and opinions presented in this article are those of the authors alone and are not intended to represent the views or scientific works of EFSA.
Funding
This research was financially supported by the Animal Health Funds of the Belgian Federal Public Service for Health, Food chain safety and Environment.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


