Abstract
Retrobulbar neoplasms are not common in mammals and are even more infrequently seen in nonmammalian species. The current report describes a retrobulbar mass creating exophthalmia and neurologic signs in a red-lored Amazon parrot (Amazona autumnalis). A 27-year-old female parrot presented for a 3-day history of anorexia and a 2-week history of periocular soft tissue swelling and exophthalmia of the right eye. Physical examination revealed 9% dehydration and right eye exophthalmia with inability to retropulse the globe. A fine-needle aspirate was performed, and cytologic evaluation revealed necrotic debris with scattered clusters of epithelial cells, moderate numbers of macrophages, and few heterophils. Given the possibility of neoplasia and paucity of treatment options, the owners elected euthanasia and submitted the body for necropsy. A large, fluctuant, friable, red, retrobulbar mass with multiple areas of hemorrhage, on cut surface, was noted at necropsy. Histologically, the mass was composed of neoplastic, cuboidal to columnar epithelial cells, forming rosette-like glandular structures, admixed with abundant necrotic debris. The neoplastic cells were strongly positive for cytokeratin (AE1/AE3) by immunohistochemistry. Based on histopathology and immunohistochemistry, the mass was diagnosed as an adenocarcinoma.
Retrobulbar masses causing marked exophthalmos are very rare in avian species. Reported avian ocular tumors include lymphoma, rhabdomyosarcoma, lipoma, and osteosarcoma.5-7 Squamous cell carcinoma has been diagnosed in the infraorbital sinus causing exophthalmos in a Solomon Eclectus parrot. 4 Neoplasms previously reported within the retrobulbar space of birds include round cell sarcomas in multiple species, a liposarcoma in an African grey parrot, medulloepitheliomas in cockatiels, a teratoma in a great blue heron and lesser kestrel, a rhabdomyosarcoma in a budgerigar, a xanthoma in an Amazon parrot, and an adenoma in an African grey parrot.1,7,8,12-17 Other neoplasms, such as highly advanced cases of chromophobic tumors in the pituitary gland in budgerigars, can cause exophthalmia, although pituitary tumors are not technically retrobulbar masses.11,16 There is a brief mention of a ciliary adenocarcinoma in a budgerigar, but with limited discussion and information. 5 Retrobulbar adenocarcinomas in birds may be extensions from an adjacent primary site, such as the sinus cavities, oropharynx, ear canal, and nasolacrimal ducts. Such tumors may also arise from local glands, for example, the zygomatic gland, harderian gland, and lacrimal gland, with the exception of meibomian glands, as these are not present in avian species. 14
A 27-year-old female red-lored Amazon parrot (Amazona autumnalis) was presented to the University of Georgia, Veterinary Teaching Hospital, Zoological Medicine Service, with a 3-day history of anorexia and a 2-week history of periocular swelling. The bird had been housed in an outdoor enclosure with 9 other parrots. The diet consisted of seeds, vegetables, and occasional peanuts. External evaluation on presentation revealed an emaciated (body condition score of 2/9), dehydrated (approximately 9%), depressed, weak bird that had difficulty perching. The bird had a right-sided head tilt and exophthalmia of the right eye that resisted retropulsion. An intravenous catheter was placed in the bacillus vein for intravenous fluid administration (90 ml/kg/day, lactated Ringer solution). Nine milliliters of a liquid diet a was administered via gavage tube. The bird was given an injection of meloxicam b and trimethoprim–sulfamethoxazole c intramuscularly, and then placed in an incubator for thermal support. Once stable, the animal was sedated with 0.4 mg/kg of midazolam intramuscularly, and a fine-needle aspirate was performed. Cytologic evaluation of the aspirate revealed cuboidal to columnar cells exfoliating individually or in variably sized clusters with occasional acinar structures (Fig. 1A). These cells had a high nuclear-to-cytoplasmic ratio with scant, basophilic cytoplasm and a central, round to oval nucleus, with finely stippled chromatin. Anisocytosis and anisokaryosis were mild to moderate, and occasional pyknotic and karyorrhectic cells were noted. Moderate numbers of macrophages and fewer heterophils were scattered throughout, and a large amount of blood and necrotic debris were present in the background. Cytologic findings were suspicious of an epithelial neoplasm; however, histopathology was recommended for a definitive diagnosis. Due to the likelihood of neoplasia and the lack of improvement with supportive care, the owners opted for humane euthanasia and gave approval for necropsy evaluation.
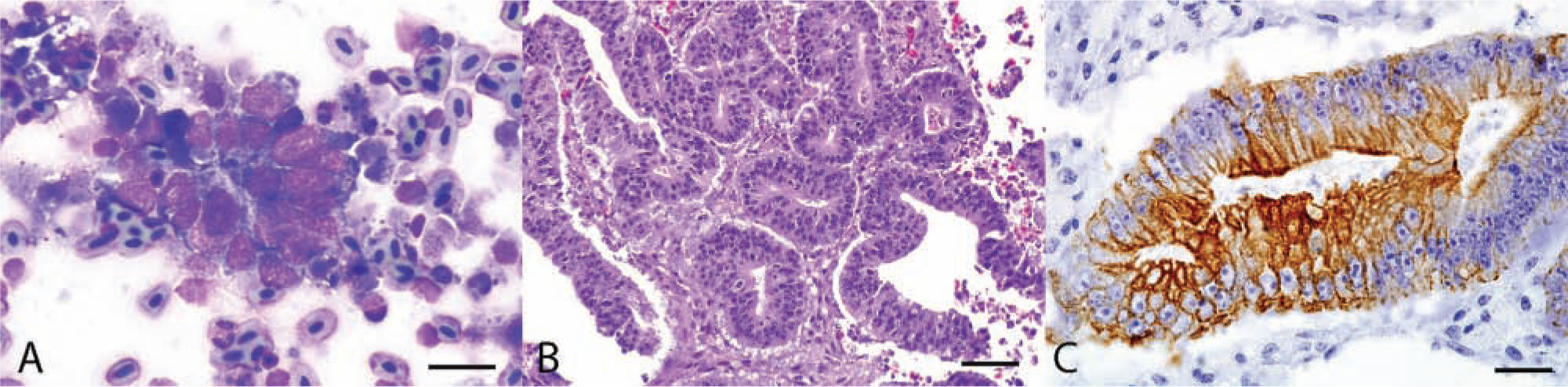
Amazon parrot (Amazona autumnalis); retrobulbar mass; adenocarcinoma.
Postmortem examination confirmed emaciation with a significantly prominent keel bone due to atrophied pectoralis muscles. The right eye was exophthalmic due to a fluctuant, friable, red, retrobulbar mass that was approximately the same size as the globe. The mass was soft, ill-defined, mottled dark red to tan to gray, granular, and extended through the caudal orbit and calvaria. The subjacent caudal right cerebral hemisphere was approximately 20% compressed due to extension of the mass through the right temporal bone. Grossly, the mass did not appear to invade the brain. The optic nerve was intact. The main additional significant gross findings were cloudy thoracic air sacs containing occasional yellow-gray, opaque, 1 mm in diameter plaques. Sections of the mass, brain, air sacs, heart, spleen, eye, liver, gastrointestinal tract, and thyroid and parathyroid glands were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 µm thickness, and stained with hematoxylin and eosin stains, by standard procedures, for evaluation by light microscopy.
Histologically, sections from the mass were unencapsulated, surrounded fragments of calvarial bone, and were composed of multifocal, loosely arranged, cuboidal to columnar cells forming glandular and rosette-like structures (Fig. 1B), supported by minimal fibrovascular stroma. The neoplastic cells were often pseudostratified and palisading around a central lumen that was either empty or contained necrotic debris. The neoplastic cells had indistinct cell borders, small amounts of microvacuolated to fibrillar eosinophilic cytoplasm, and often basally located nuclei. The chromatin was densely packed to vesicular, and there were 1–2 prominent nucleoli. Anisocytosis and anisokaryosis were mild. The mitotic rate was 10 per ten 400× fields. There was severe, multifocal necrosis, characterized by hypereosinophilic cytoplasmic remnants, karyorrhectic debris, and degenerate heterophils, admixed with fibrin and hemorrhage. Embedded within multiple sections of neoplasm were small fragments of bone, interpreted to be remnants of fractured calvarial bone. The mass did not infiltrate the meninges, and the leptomeninges between the neoplastic mass and the brain were lined by numerous degenerate heterophils, few erythrocytes, fibrin, and rare clumps of neoplastic cells. The neoplasm filled the retrobulbar space, but did not infiltrate the eye. Based on the compressive nature of the neoplasm, bone destruction, lack of a capsule, and extensive necrosis and hemorrhage, the tumor was considered malignant. The thoracic air sacs contained a few granulomas composed of epithelioid macrophages, fibroblasts, lymphocytes, and areas of dense collagen. Lillie–Twort Gram stain and Ziehl–Neelsen acid-fast stain were performed; however, no infectious organisms were identified. There were no other significant histologic lesions.
Immunohistochemistry (IHC) specific for glial fibrillary acid protein d (GFAP; 1:800, monoclonal), neuron-specific enolase e (NSE; 1:400, monoclonal), synaptophysin d (1:400, monoclonal), vimentin d (1:600, monoclonal), and pancytokeratin d (AE1/AE3, 1:200, monoclonal) were performed to further characterize cytogenesis of the neoplasm. The neoplastic cells showed variable, apical cytoplasmic staining for pancytokeratin (Fig. 1C), which was relatively nonuniform with higher numbers of neoplastic cells in the periphery of the tissue staining as compared to the more centrally located neoplastic cells. The synaptophysin and vimentin IHC were negative; however, nonneoplastic tissues in the sections stained rarely and weakly with synaptophysin and vimentin, indicating that these 2 antibodies may not work as well in exotic avian species as compared to mammals. Additionally, neoplastic tissue showed no positive staining by GFAP and NSE IHC; however, these antibodies did not stain the nervous tissues of the bird used as an internal control, indicating that the antibodies used for mammalian GFAP and NSE IHC were unreactive with avian tissue, at least in this species.
The current report describes the clinical presentation, cytology, histopathology, and IHC of a previously undescribed neoplasm in parrots. Multiple types of neoplasms and inflammatory masses causing exophthalmos in birds have been described.1,3-8,11-17 The neoplastic mass described in the present study had histologic similarities to both adenocarcinoma and medulloepithelioma. Similarities to medulloepithelioma include the presence of glandular structures with a stratified appearance, similar to the Flexner–Wintersteiner rosettes described in medulloepitheliomas that are larger in size and consist of several rows of tumor cells. 1 Medulloepitheliomas are uncommon tumors of young animals, arising from the primitive neural tube and medullary plate, and can occur anywhere in the central and peripheral nervous system, including within the optic nerve and ciliary body.2,9 Those that arise around the optic nerve, from the optic cup, can become large masses distinct from the rest of the globe, similar to the gross findings in the current case.1,2 Medulloepitheliomas in human beings are often vimentin-immunoreactive, variably cytokeratin-immunoreactive, and often NSE and GFAP negative. 10 In reports of this neoplasm in cockatiels, cytokeratin IHC was not performed, and NSE, vimentin, and GFAP were variably positive in areas of the neoplasm. 1 In human beings, a cockatiel, and a llama, the neoplastic cells are most consistently positive for vimentin and NSE, but vary in GFAP staining.1,2,9 In the presently described tumor, pancytokeratin exhibited positive immunoreactivity while vimentin and synaptophysin were negative, supporting a diagnosis of adenocarcinoma. Retrobulbar adenocarcinomas in bird species can arise from the periocular glands, including the zygomatic gland, harderian gland, or other lacrimal glands. There was no evidence of tumor extension from the sinuses or globe of the eye, and there were no other masses identified at necropsy or by light microscopy.
Other nonneoplastic lesions that may cause exophthalmia in avian species should be kept on the list of differential diagnoses until cytology and/or histology can be pursued. Vitamin A deficiency has been associated with metaplasia of the ocular glands, which causes glandular proliferation and periocular swelling.15,16 Granulomatous diseases should also be considered, including mycobacteriosis, aspergillosis, and cryptococcosis. 16 Infection with Pasteurella multocida has been associated with swelling of the sinus infraorbitalis causing periocular swelling in pheasants. 3 Canaries and budgerigars with periocular Knemidokoptes spp. infection can have periorbital lesions, but these are more surface-oriented lesions that are not typically associated with exophthalmia. 16 Any inflammation or abscess formation in the periocular glands or ducts can also cause exophthalmia. 16
The friable nature of this neoplasm made it difficult to assess the extent of infiltration; however, the presence of neoplastic tissue surrounding small pieces of bone supports that the mass led to a pathologic fracture of the calvaria. Due to the diagnosis of neoplasia and owners’ election for euthanasia, debulking surgery was not pursued as a treatment; however, other retrobulbar neoplastic masses in birds have been treated surgically.1,15 The morphologic similarities between adenocarcinoma and medulloepithelioma necessitated IHC for diagnosis in this case, although the latter was less likely given the age of the bird. Retrobulbar masses in avian species can be due to neoplastic, inflammatory, or metabolic processes; all of these differentials must be included and can be confirmed via histopathology and/or cytology. Improved and more thorough ancillary testing for the definitive diagnosis of the cytogenesis of neoplasms in birds will allow for further studies regarding prognosis and treatment.
Footnotes
Acknowledgements
The authors would like to acknowledge the technical support of Ms. Abbie Butler, Ms. Patricia Howe, and the staff of the Histology Laboratory of the University of Georgia, College of Veterinary Medicine, for histology and immunohistochemistry. The authors would also like to thank Dr. Elizabeth Howerth for her guidance on this case and Dr. Raquel Rech for her assistance with editing the final figure image.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article from the Department of Pathology, College of Veterinary Medicine, University of Georgia.
a.
Emeraid Omnivore Care, Lafeber Co., Cornell, IL.
b.
Boehringer Ingelheim Vetmedica Inc., Saint Joseph, MO.
c.
Schering-Plough, Merck & Co. Inc., Whitehouse Station, NJ.
d.
BioGenex Laboratories Inc., San Ramon, CA.
e.
Invitrogen Corp., Carlsbad, CA.
