Abstract
Verotoxigenic
Introduction
Since the beginning of the 1980s, verotoxigenic
In order to implement measures to reduce human infection by zoonotic pathogens, it is essential to understand the source and transmission routes. 4 Cattle are considered to be the main reservoir of VTEC O157:H7, 20 and several modes of transmission have been associated with human outbreaks, which include consumption of contaminated food and water, direct or indirect contact with cattle, and exposure to a contaminated environment. 2 Targeted interventions and large-scale field studies at herd level require cost-effective and reliable diagnostic methods.
Cattle shedding VTEC O157:H7 show a heterogeneous pattern in the number of excreted bacteria.6,26 There is also variation in the duration of shedding. On average, infected cattle shed the bacteria for 1–2 months; however, a few animals may shed for more than a year.13,25 It is also known that younger animals are more likely than adult cattle to shed the bacteria. 42 Moreover, the within-herd prevalence in infected herds shows a wide variability. 27 This heterogeneity has implications for diagnostic testing because sensitivity may vary in populations or subpopulations of animals. 19
Analysis of individual rectal samples has commonly been used to determine the VTEC O157:H7 herd status, both in outbreak investigations
3
and field studies.9,15 However, the use of individual fecal samples to determine the herd status is costly due to the time–consuming nature of the collection procedure and the large number of samples to analyze. A combination of environmental sampling methods consisting of pooled fecal material, overshoe, and dust samples is considered to be a practical and cost-effective method to detect
Material and methods
Study population
The criterion for inclusion in the present study was that VTEC O157:H7 had been previously detected in the herd or that an animal from the herd had sampled positive at slaughter. The farmers participating in the study voluntarily entered their herds, and 31 Swedish dairy herds were sampled continuously between autumn 2008 and spring 2010. The time period from the previous sampling occasion with a positive finding to the sampling in the current study ranged from 8 to 370 days with a median of 45 days. The number of animals in the herds included in the study ranged from 49 to 1,983, with a median of 181 animals.
Sampling
The animals in the herds were categorized into 3 different age groups: calves (6 weeks to 4 months), young stock (4–12 months), and adults (>12 months). Within each age category, both individual fecal samples and environmental samples consisting of dust, overshoe, and pooled pat samples were collected. The sampling was performed by staff from the regional livestock association serving the area where the herd was located. In total, 11 persons were engaged in sample collection. Two persons sampled 15 and 5 herds, respectively; the remaining persons sampled 1 or 2 herds each.
Sampling material, together with written sampling instructions, was provided by the National Veterinary Institute in Uppsala, Sweden (SVA). Samples were sent to SVA via the postal service at ambient temperature. To ensure that samples would reach the laboratory without a weekend delay all sampling was done on Mondays or Tuesdays. Bacterial analyses were initiated the day after sampling. The sampling was performed from November to May (i.e., during the housing season for the herd). Enclosed with the samples was also documentation with the unique animal identification of the sampled animals together with a description of the farm layout and where the environmental samples had been collected. All documentation was manually checked to detect deviations from the sampling instructions.
Individual fecal samples
Rectal samples were taken with disposable rectal gloves without lubricant and placed in 50-ml plastic containers. For herds with 140 or fewer animals, fecal samples from all animals were collected. If a farm held more than 140 animals, the number of individuals within each age category was divided with the total number of animals in the herd to obtain the percentage of animals within each age category. The percentage were rounded to the nearest 5 and multiplied with 140 to obtain the number of the individual fecal samples to be collected within the age category. The particular animals to sample within each age category were then selected by the sampler.
Overshoe samples
Overshoe sampling was performed by fitting gauze, moistened with phosphate buffered saline, to the outside of disposable boot protectors for each boot. The person wearing the boots then walked around in all areas where the animals (in each age category) were kept. Fecal material attached itself to the gauze, and the gauze was rotated during sampling to allow the whole surface to be used. The gauze from each boot was removed after sampling and placed into 1 stomacher bag for each age category.
Pooled pat samples
From each age category, a pooled pat sample was taken. Each sample, consisting of approximately 50 g of fresh feces was taken from 10–15 different places on the floor where the animals were kept, and then placed in 100-ml plastic containers. The sampler was instructed to select places as widely distributed as possible throughout the floor space for each age category.
Dust samples
The dust samples taken for each age category were collected on 3 paper cloths a (23 cm × 23 cm) moistened with phosphate buffered saline. Samplers wore disposable gloves and collected the dust by wiping surfaces such as walls, gates, and water appliances where the animals were kept. Dust samples, grouped into each age category, were then placed in separate stomacher bags.
Bacteriological analysis
The individual fecal samples were pooled at the SVA laboratory. Each pool (
After preenrichment, automatic immunomagnetic separation (IMS) was performed according to the manufacturer’s directions.
c
Paramagnetic beads coated with antibodies to
After IMS, the beads were spread on sorbitol MacConkey agar plates
e
supplemented with 0.05 mg/l of cefixime and 2.5 mg/l of potassium tellurite. The plates were incubated 18–24 hr at 37° ± 1.0°C, then screened for VTEC O157:H7; up to 5 suspected colonies were subjected to a latex agglutination test.
f
For cases of positive agglutination tests, further biochemical confirmation was performed with a commercial test kit.
g
The biochemical confirmation was performed on a single colony and, if this colony was negative, all colonies with a positive agglutination test were subjected to confirmation. Polymerase chain reaction (PCR) was used to confirm for the presence of the genes coding for verotoxins 1 and 2 (
Combinations of environmental samples
In the current study, 7 combinations of environmental samples were evaluated. The combinations chosen were formed by combining the 3 types of environmental samples as follows: 1) pooled pat only, 2) dust only, 3) overshoe only, 4) dust and pooled pat, 5) overshoe and pooled pat, 6) dust and overshoe, and 7) dust, overshoe, and pooled pat. Combinations of environmental samples taken in each herd were interpreted in parallel to maximize sensitivity 18 (i.e., if any of the samples in the combination were positive, the result was considered positive).
Pool prevalence and statistical analysis
The within-herd pool prevalence, πh, was calculated as 100 × (number of positive pools/number of sampled pools) in the herd. Similarly, the within-group pool prevalence, πg, was calculated as 100 × (number of positive pools/number of sampled pools) in the group of animals in each age category. All statistical analyses were performed using R version 2.15.1. 31
Generalized linear mixed model
The probability for detection of VTEC O157:H7 in the environmental samples was estimated with a generalized linear-mixed model. Seven models were constructed, 3 for each environmental sample individually and 4 for each combination of environmental samples, as described above. The general model takes the form:
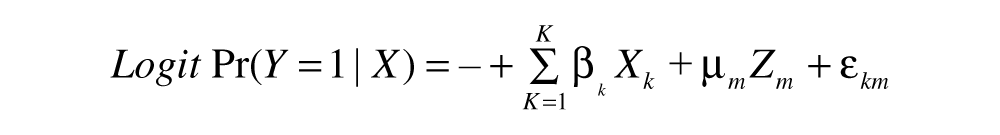
where the dependent variable
The data was analyzed with the R package lme4 (version 0.999999-0) using the g
Missing data
In some herds, environmental samples were not taken within all age categories for 3 reasons: 1) there were no animals in the age category in the herd (
Effect of age
Fisher exact test 1 was used to evaluate if the proportion of positive pools from calves was statistically different from those for young stock and adults. Comparison was also made between young stock and adults. The odds ratio and the 95% confidence interval (CI) were calculated using the method of Woolf. 43
Results
In total, 3,763 individual fecal samples were collected and analyzed for VTEC O157:H7 as 1,287 bacteriological cultures (Table 1). Of these bacteriological cultures, 179 tested positive, of which 171 were from pools containing fecal material from 3 animals, 4 from pools with 2 animals, and 4 from a single animal. Results of the environmental and pooled sampling are presented in Table 1. In 24 herds, at least 1 positive pool was found. In 20 of these herds (0.83, 95% CI: 0.63–0.95), 1 or more environmental samples were also positive (Table 1).
Comparison of bacteriological culture of verotoxigenic
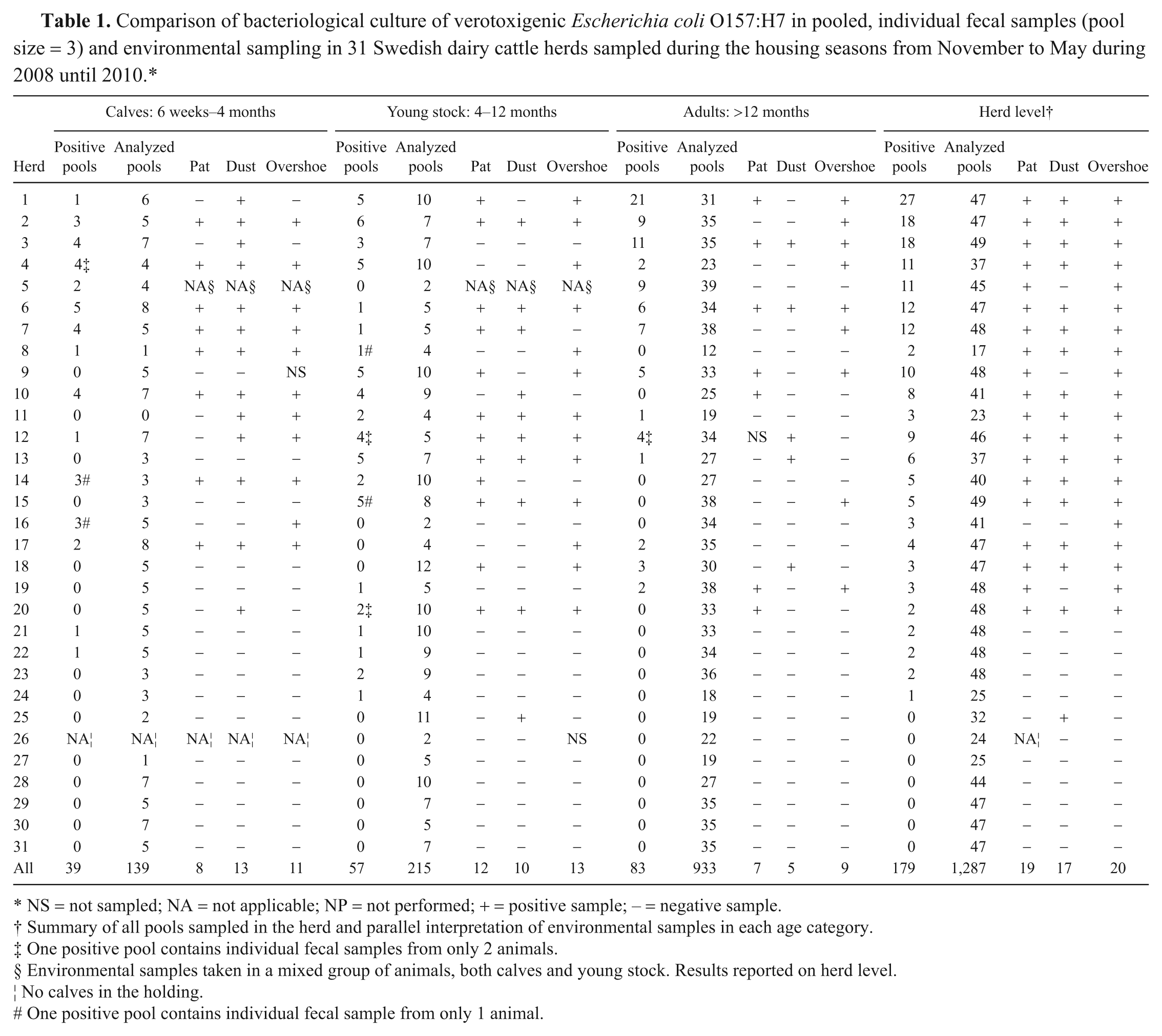
NS = not sampled; NA = not applicable; NP = not performed; + = positive sample; – = negative sample.
Summary of all pools sampled in the herd and parallel interpretation of environmental samples in each age category.
One positive pool contains individual fecal samples from only 2 animals.
Environmental samples taken in a mixed group of animals, both calves and young stock. Results reported on herd level.
No calves in the holding.
One positive pool contains individual fecal sample from only 1 animal.
The within-group pool prevalence, πg, in the 3 age categories ranged from 0% to 100%. The distribution of results with complete cases for all 7 combinations of environmental samples, at 6 intervals of within-group pool prevalence, πg, is given in Table 2. As the within-group pool prevalence increases, there is a tendency of increased proportion of positive environmental samples, regardless of the sample combination.
Comparison of bacteriological cultures of verotoxigenic

The data is presented as the number of positive samples/total number of samples in interval. Within-group pool prevalence (πg) equals 100 × (number of positive pools/number of sampled pools) in the group of animals.
The within-herd pool prevalence, πh, ranged from 0% to 57%. In 4 herds, no positive environmental samples were detected despite positive pooled individual fecal samples. These herds had the lowest within-herd pool prevalence, πh (4.2%), among positive herds in the study.
The outcomes for the combinations of environmental samples at various levels of both within-group and within-herd pool prevalence are shown graphically in Figure 1. When comparing the plots for a single environmental sample, with parallel interpretation of all 3 environmental samples, there is a trend for a positive outcome, affected by both the within-group and within-herd pool prevalence for the combined samples.
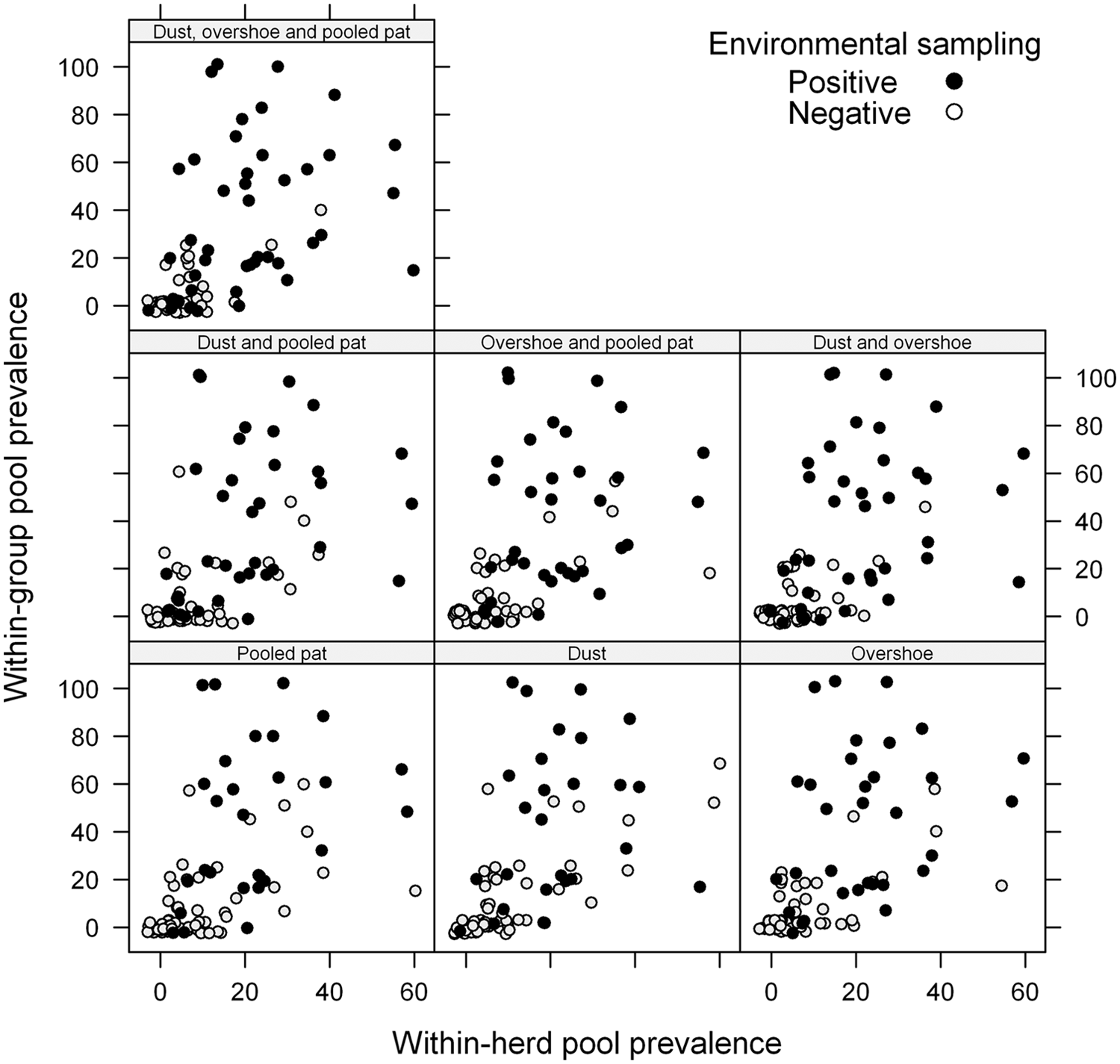
Graphical distribution of results from comparison of bacteriological culture of verotoxigenic
The within-group pool prevalence, as a single explanatory variable in the generalized linear mixed model with random effect on herd level, was significant for each of the 7 models (
Generalized linear mixed models with random intercept on herd level, estimating the probability for a positive outcome of bacteriological culture of verotoxigenic

Centered by subtracting the within-herd pool prevalence.
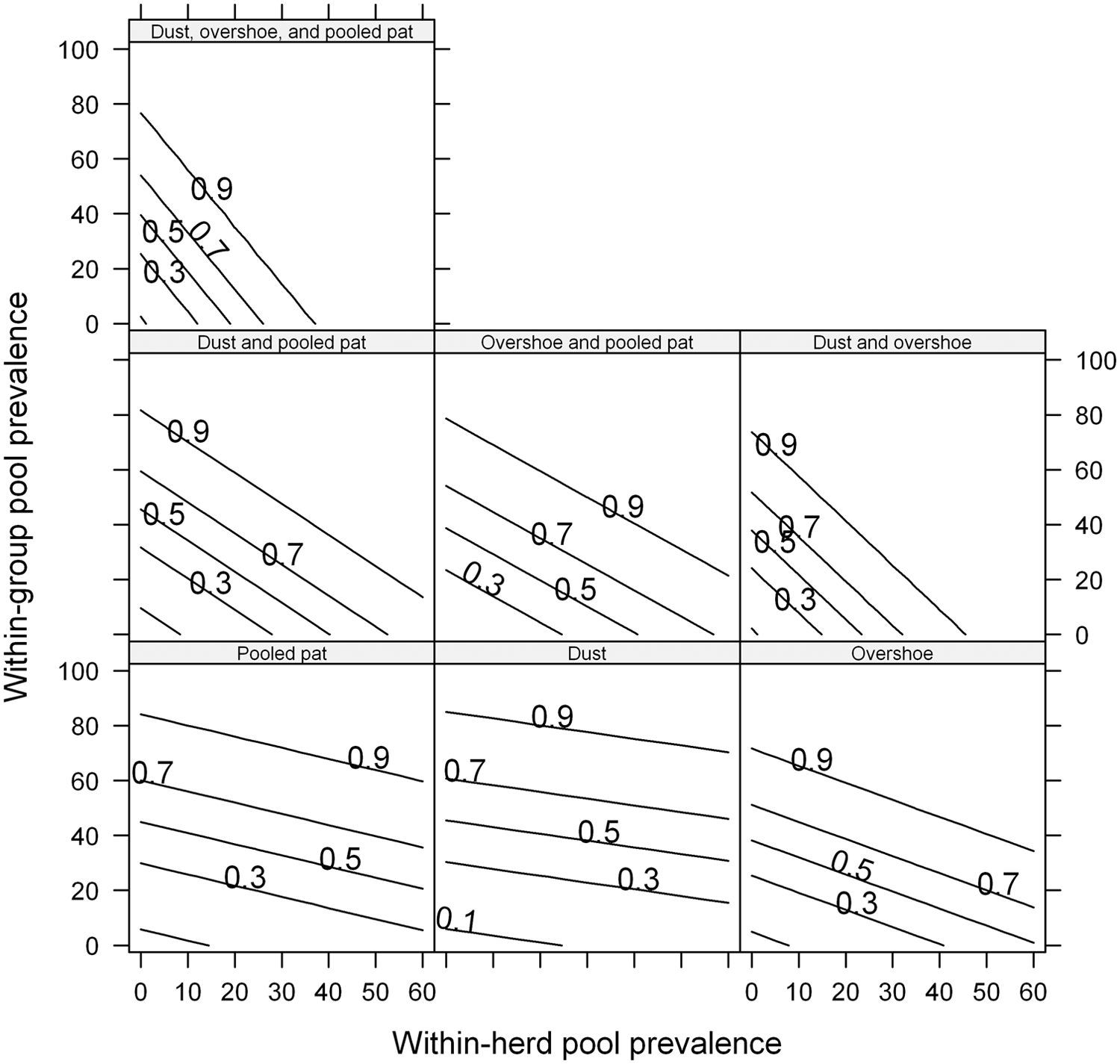
Estimated probability of detection using environmental samples of bacteriological culture of verotoxigenic
The proportions of positive pools were equal to 0.28, 0.27, and 0.09 in calves, young stocks, and adults, respectively. The proportions of positive pools from calves (
Discussion
In the current study, environmental sampling was compared to pooled, individual fecal sampling as a diagnostic method for the determination of VTEC O157:H7 herd status. The results show that environmental sampling reliably identified herds with naturally infected cattle shedding VTEC O157:H7 under field conditions.
Overshoe sampling, alone or in combination with dust and pooled pat sampling, correctly classified 20 of the 24 (0.83, 95% CI: 0.63–0.95) herds where at least 1 positive pool was detected. The usefulness of overshoe sampling to determine the VTEC O157:H7 herd status is in agreement with previous studies.7,8
The results show a statistically significant effect in the probability of isolating VTEC O157:H7 from environmental samples by increasing within-group pool prevalence. Moreover, a statistically significant contextual effect of within-herd pool prevalence was found in the probability of detection for the 2 combinations “dust and overshoe” and “dust, overshoe, and pooled pat” within each age category. Results suggest that, in a given group of animals, the probability of a positive outcome of the environmental sample depends both on the within-group pool prevalence and the average within-herd pool prevalence. One biological explanation for the effect of within-herd pool prevalence is that, with a higher bacterial load of VTEC O157:H7 in the herd environment, the probability of spread and transmission of the bacteria might increase with mechanical vectors such as personnel and equipment. Hence, even if there are few animals in the group that shed the bacterium, floors can still be contaminated. Overshoe samples are in some sense more like pooled pat sampling than dust sampling due to the fact that the sampler walks on pats on the floor. Moreover, larger areas of the environment are covered by overshoe sampling compared to pooled pat sampling because not only fecal material is sampled but also the area between pats due to the walking of the sampler.
The current study shows that animals younger than 12 months are more likely than adult cattle to shed the bacteria, a finding which is in agreement with published work. 42 In modeling the outcome of environmental sampling, age was not found to be a significant predictor. It can therefore be concluded that environmental sampling per se does not work better in younger animals. However, younger animals on average shed more VTEC O157:H7 and therefore environmental sampling is more likely to be positive in a group of animals younger than 12 months. The current study also identified herds where only pools sampled from adults were positive. A similar finding can also be seen for environmental samples, were 1 herd was only positive in the adult age category. Thus, even if animals younger than 12 months are more likely to shed the bacteria, all age categories should be included in the sampling regardless of the sampling method.
The number of animals shedding and the concentration of VTEC O157:H7 excreted at any given sample occasion will vary due to the intermittent shedding pattern and fluctuating levels.11,34 Thus, sampling individual animals requires that many animals are included to establish the herd status with a high level of confidence. 3 Several studies show that VTEC O157:H7 has a good survival in the environment. 16 Therefore, sampling the environment may circumvent the complication with intermittent shedding; environmental sampling compensates for the fluctuations in the number of excreted bacteria by individual animals. Furthermore, if the environment is contaminated then individual animals are at risk of colonization that could multiply the bacteria and maintain the herd infection. 36 Such risk can be identified with environmental sampling.
In order to reduce costs of analyzing individual fecal samples, the samples were pooled at the laboratory. However, pooling reduces the sensitivity of the bacteriological analysis of VTEC O157:H7.3,35 The within-herd prevalence can be estimated using the results of bacterial culture of pooled, individual fecal samples, and several alternative methods exist.10,39 However, none of the methods can simultaneously account for variation in pool size or test sensitivity and specificity; therefore, the within-herd and within-group individual prevalence was not estimated, and the actual prevalence of positive pools was used instead.
For various reasons, missing data is often found in epidemiological studies. 38 In the present study, there were 4 herds where some environmental samples were not collected in each age category, leading to 3.3% missing data. Depending on the reason(s) for missing data, there is potentially a bias introduced in the analysis. 38 Consequently, both complete case analysis and multiple imputation were used when analyzing the data. The analyses of complete cases and those containing inputted data gave the same conclusions. The only difference found was a minor shift of the regression coefficients calculated from the inputted data compared to that in the complete case analysis. This is not surprising due to the relatively low proportion of missing observations.
The herds in the present study were not selected at random. Herds that were likely to have animals shedding VTEC O157:H7 were intentionally targeted. All herds included in the study were from the southern third of Sweden where the highest cattle density is found. 41 Within subregions in this area, the presence of dairy herds positive for VTEC O157:H7 has ranged from 1% to 23%. 15 All herds in the study population were dairy herds with a median herd size of 181 animals. The average size of a dairy herd in Sweden in 2011 was 65 animals (Statistics Sweden, http://www.jordbruksverket.se/download/18.50fac94e137b680908480004068/6_Husdjur.pdf. In Swedish).There are reports that the risk of testing positive for VTEC O157 increases with herd size. 15 This may be one explanation of why the median herd size of the study population is larger than an average herd size.
In 6 herds, neither fecal nor environmental sampling detected any VTEC O157:H7 and, in 1 herd, the bacteria were only detected in an environmental sample. However, all herds included in the study had a positive VTEC O157:H7 finding before entering the study. The transient appearance of an infection in dairy herds has been reported earlier. 32 Reasons for why the bacteria were not detected could be: 1) the animals might have cleared the infection, 2) imperfect tests, and 3) decay of VTEC O157:H7 to nondetectable levels in the environment.
Parallel interpretation of combined tests maximizes sensitivity.
18
However, identifying VTEC O157:H7–positive herds based on the combination of “dust, overshoe, and pooled pat” does not increase sensitivity greatly above identifying positive herds based on the combination of “dust and overshoe.” Therefore, dropping the complication of pooled pat sampling would be worthwhile for the relatively small reduction in positive detection likelihood. The combination of “dust and overshoe” has also been found useful for detection of
Footnotes
Acknowledgements
The farmers and personnel at the regional livestock associations and the SVA laboratory are thanked for their cooperation in this research. Helena Olofsson, Swedish University of Agricultural Sciences, Department of Clinical Sciences, identified several herds used in the current study via Swedish Farmers’ Foundation for Agricultural Research (grant number V0930039). The 2 anonymous reviewers are thanked for their helpful comments.
a.
Readi Wipes Plus, Allanda Ltd, Bedfordshire, UK.
b.
Modified Tryptone Soya Broth (mTSB), Oxoid Ltd., Basingstoke, UK.
c.
Dynabeads Anti-
d.
Dynal BeadRetriever, Dynal Biotech ASA, Oslo, Norway.
e.
MacConkey agar plates (CT-SMAC: cefixime tellurite sorbitol MacConkey agar), Oxoid Ltd., Basingstoke, UK.
f.
Oxoid DR 622, Oxoid Ltd., Basingstoke, UK.
g.
API 20 E, bioMérieux SA, Mercy l’Etoile, France.
Declaration of conflicting interests
The author(s) declare that there are no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Swedish Board of Agriculture funded this work.
