Abstract
Isolation of
Introduction
“Oral fluid” is a mixture of saliva and mucosal transudate. Saliva is produced by the parotid, submandibular, and sublingual salivary glands, as well as the minor salivary glands located on the lips, tongue, palate, cheeks, and pharynx. 14 Mucosal transudates originate from the gingival and buccal mucosa and contain serum-derived antibodies. 24
The presence of
Materials and methods
Experimental design
The level and duration of PRRSV and anti-PRRSV antibodies in serum and oral fluids was evaluated in 3 age groups of pigs (4, 8, or 12 weeks of age at time of inoculation). Each age group consisted of 16 pigs (12 PRRSV-inoculated, 4 negative controls) housed in pens of 4 pigs each, with the exception of 4–week-old PRRSV-inoculated pigs, which were housed in 2 pens of 6 pigs each. Pigs were randomly assigned to treatment groups. Serum samples collected 8 days before the start of the experiment and on day 0 were assayed by enzyme-linked immunosorbent assay (ELISA) to confirm the absence of PRRSV infection. The pigs were intramuscularly inoculated on day 0 with 2 ml of a preparation containing 1 × 101.7 50% tissue culture infective dose (TCID50) of PRRSV per ml. After PRRSV inoculation, serum, buccal swabs, and oral fluids were collected at regular intervals for 63 days. At the end of the experiment, all samples were randomized, relabeled, and submitted for testing. Samples were assayed for the presence of PRRSV by real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) and for the presence of anti-PRRSV antibodies by ELISA and indirect fluorescent antibody test (IFAT).
Animals and animal care
Pigs were obtained from a commercial swine herd known to be free of PRRSV. Pigs were received at 3, 7, or 11 weeks of age and housed in the Livestock Infectious Disease Isolation Facility at Iowa State University (Ames, IA). Animals were housed on the floor in pens that were cleaned daily. Feeder space, square footage per animal, ambient temperature, and room air exchanges all met or exceeded guidelines and requirements set forth in the
Porcine reproductive and respiratory syndrome virus
A PRRSV-ISU-P (type II [North American] prototype) isolate was used in this study. The isolate was initially recovered from a homogenate prepared from a pool of lung tissues collected in October 1990 from young pigs in a herd experiencing an acute outbreak of PRRSV in Illinois. As described elsewhere, 39 the virus was initially isolated on porcine alveolar macrophage (PAM) cultures. Subsequently, the isolate was cloned by 3 rounds of limiting dilutions in PAMs and twice by plaquing in MA-104 cells. The working stock of virus used in the present study represented the fourth passage in MA-104 cells.
Preparation of PRRSV inoculum
The PRRSV inoculum was prepared as previously described. 12 Each dilution was run in duplicate, and the means were used to calculate the virus titer using the Spearman-Karber method. 13 The titer of the inoculum was estimated at 1 × 101.7 TCID50 per ml.
Collection of biological samples
Blood samples were collected from all pigs twice weekly through 14 DPI, then weekly through 63 DPI. Samples were collected using a single-use blood collection system.
a
Blood samples were centrifuged at 1000 ×
Pen-based oral fluid samples were collected twice weekly through 61 DPI by suspending a length of 3-strand twisted cotton rope c in each pen of 4 or 6 pigs. To facilitate oral fluid collection, each pen was equipped with a 90° bracket with a 2.54-cm hole in the center of the horizontal surface. Brackets were fixed in place by bolting the bracket to a back plate through the vertical bars of the pen. One bracket was located in each pen such that the rope could be placed and recovered without the need to enter the pen. For sample collection, 1 end of the rope (1.59 cm or 1.27 cm [5/8 inch or 1/2 inch]) was knotted and the opposite (unknotted) end threaded through the hole in the horizontal surface of the bracket. The rope was cut to length so that the end was at shoulder height to the animals. Ropes were left in place for 20–30 min. Pigs actively sought out and chewed the rope, leaving the strands moistened with oral fluids. To recover the oral fluid sample, the bottom 30.48 cm (12 inches) of the rope was inserted into a 17.78 cm by 30.48 cm (7 inches by 12 inches) stomacher filter bag b while still suspended from the bracket and cut from the upper portion of the rope. Oral fluids were extracted from the rope by mechanical compression (wringer), decanted into 5-ml snap-cap tubes, and stored at −20°C until assayed.
Porcine reproductive and respiratory syndrome virus real-time quantitative reverse transcription polymerase chain reaction
For purposes of comparison, oral fluid samples were submitted to 2 laboratories (A and B) for PRRSV real-time qRT-PCR analysis. Serum samples were only submitted to laboratory A.
Oral fluid, serum, and buccal swab
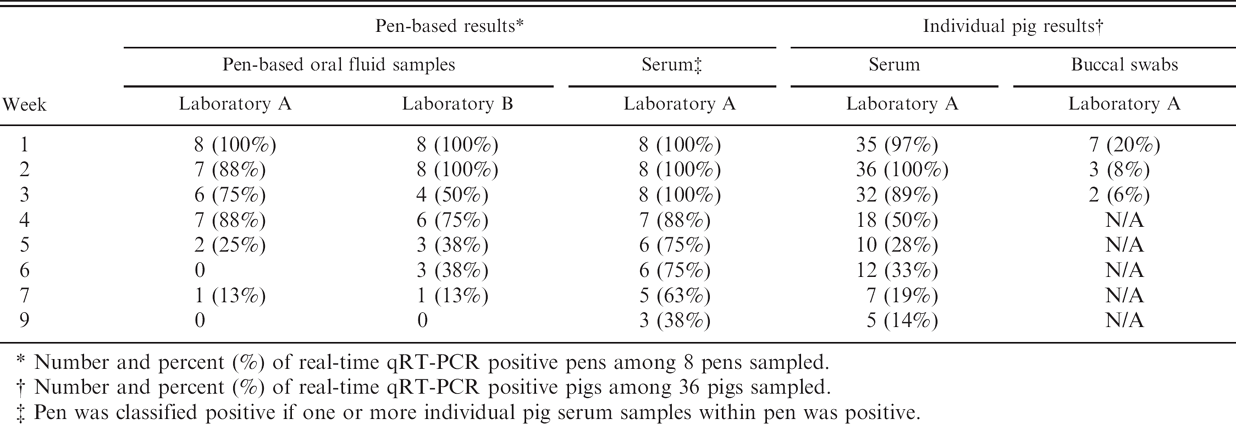
Number and percent (%) of real-time qRT-PCR positive pens among 8 pens sampled.
Number and percent (%) of real-time qRT-PCR positive pigs among 36 pigs sampled.
Pen was classified positive if one or more individual pig serum samples within pen was positive.
Indirect fluorescent antibody test
Oral fluid samples were assayed for the presence of specific anti-PRRSV IgG and IgA antibodies by IFAT using fixed MARC-145 cells infected with PRRSV as previously described, 39 with a few modifications. Known positive and negative controls were diluted 1:20 in 0.01 M PBS (pH 7.2) with 0.05% Tween 20 (PBST) while oral fluid samples were tested after being diluted 1:2 in PBST. For detection of anti-PRRSV antibodies, FITC-conjugated antiporcine IgG h and FITC-conjugated antiporcine IgA h were used as the secondary antibody for detection of anti-PRRSV IgG and IgA, respectively.
Enzyme-linked immunosorbent assay
Both oral fluid and serum samples were tested for the presence of antibodies against PRRSV using the HerdChek PRRS Antibody 2XR Test Kit.
i
Serum samples were diluted 1:50 and assayed according to the manufacturer's instruction. Oral fluids were assayed according to the manufacturer's instructions, with the exception that samples were centrifuged at 1000 ×
Statistical methods
All statistical analyses were performed using JMP 6.0.0.
j
Serum and oral fluids results for ELISA and real-time qRT-PCR were analyzed using multivariate analysis of variance (MANOVA), with time as the repeated measure and age as a model effect. When statistically significant (
Linear regression was used to evaluate the relationship between oral fluid samples and serum samples with respect to the quantity of viral RNA and, in a separate analysis, ELISA S/P values. Consolidation of data was necessary to conduct linear regression, despite its recognized effect on type 2 error rate. That is, oral fluid samples were collected from pens (not individual pigs) at 2- to 4-day intervals, whereas serum samples were collected from individual pigs twice a week through day 14, then weekly. Thus, the linear regression analyses (real-time qRT-PCR, ELISA) were based on a weekly average of oral fluid results for each pen and a weekly average of serum results for individual pigs within the same pen.
Results
Porcine reproductive and respiratory syndrome virus real-time quantitative reverse transcription polymerase chain reaction
Oral fluid samples were assayed by real-time qRT-PCR at 2 laboratories (A and B). All oral fluid samples collected from negative control pens were negative in both laboratories (
Given that real-time qRT-PCR results from the 2 laboratories were comparable, buccal swabs and serum samples (reported below) were only assayed at laboratory A. Buccal swab samples were collected from individual pigs twice weekly through 21 DPI.
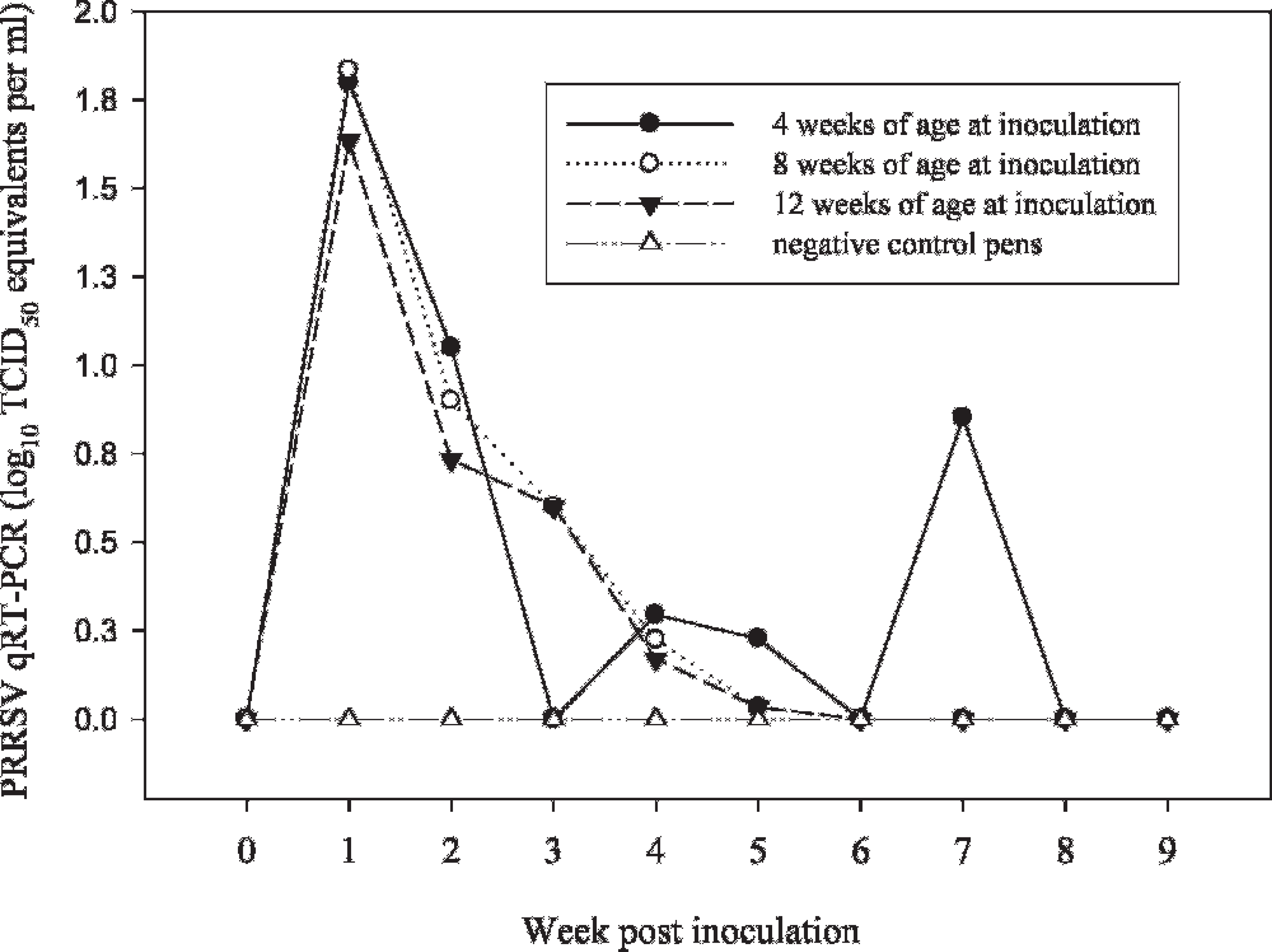
Laboratory A: Oral fluid
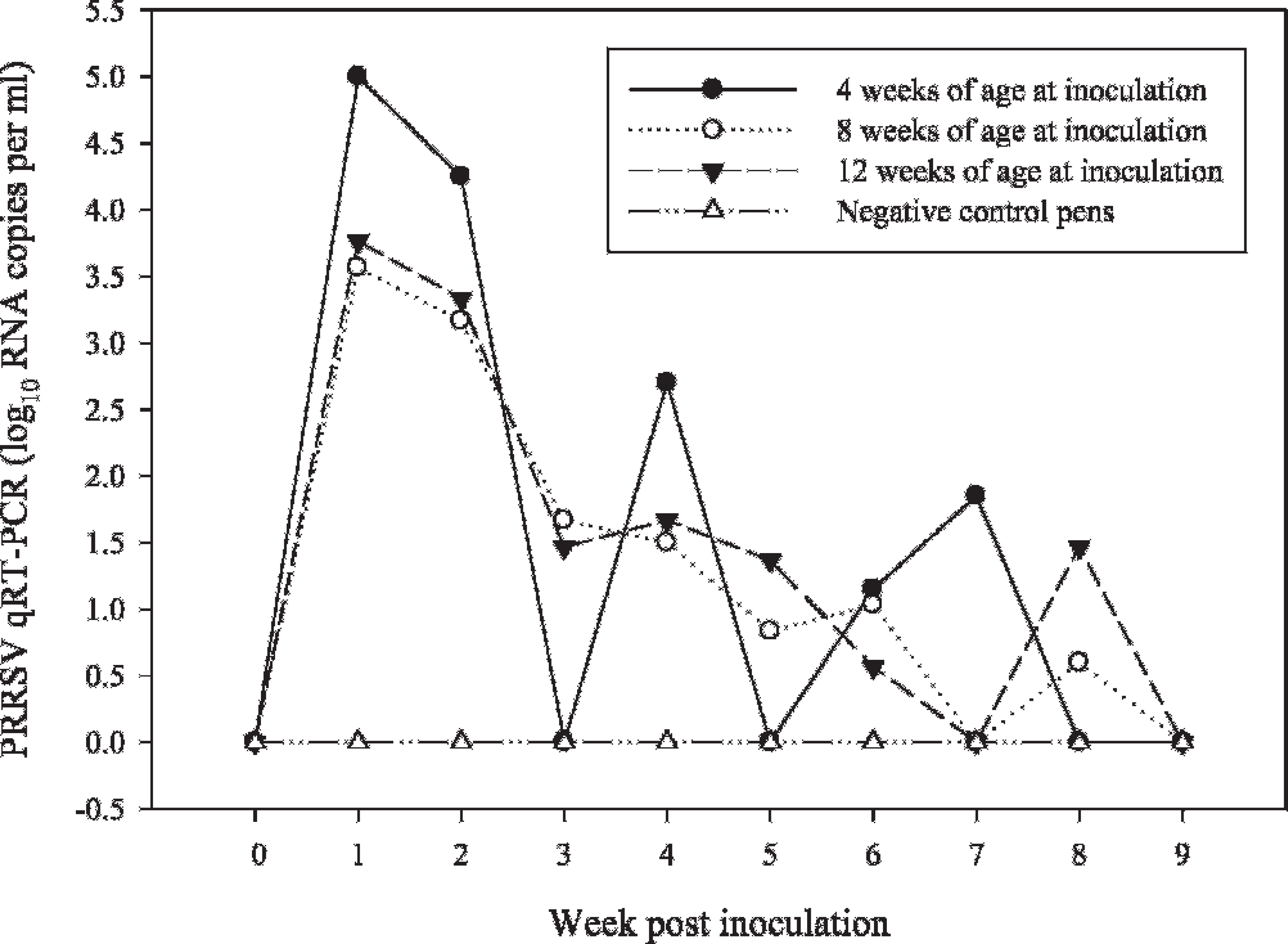
Laboratory B: Oral fluid
All samples collected prior to inoculation (
Serum samples were collected from all pigs twice weekly through 14 DPI, then weekly through 63 DPI. Analysis (MANOVA) of the real-time qRT-PCR data detected a difference (
Comparison showed that the levels of virus in serum and oral fluid samples followed a similar pattern, with oral fluid consistently containing a lower concentration of virus (Fig. 4). Both serum and oral fluid samples were real-time qRT-PCR positive from 3 DPI to 4 to 5 weeks postinoculation, with sporadic positives thereafter. Linear regression analysis estimated the correlation (r2) between virus concentration in serum and oral fluids at 0.56 (Fig. 5).
The diagnostic sensitivity of real-time qRT-PCR for serum and oral fluid samples corresponded to the percentage of inoculated animals (or pens) that tested positive (Table 1). For oral fluid samples, the mean diagnostic sensitivity for the first 4 weeks postinoculation was 88% for laboratory A and 81% for laboratory B. By comparison, the mean diagnostic sensitivity of laboratory A on individual pig serum samples for the same 4-week period was 89%. Neither laboratory reported positive real-time qRT-PCR results for serum or oral fluid samples from negative controls (i.e., diagnostic specificity was 100%).
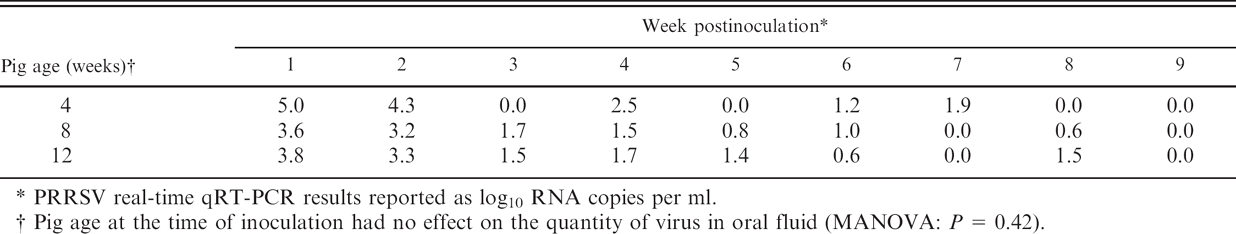
Laboratory A: Oral fluid
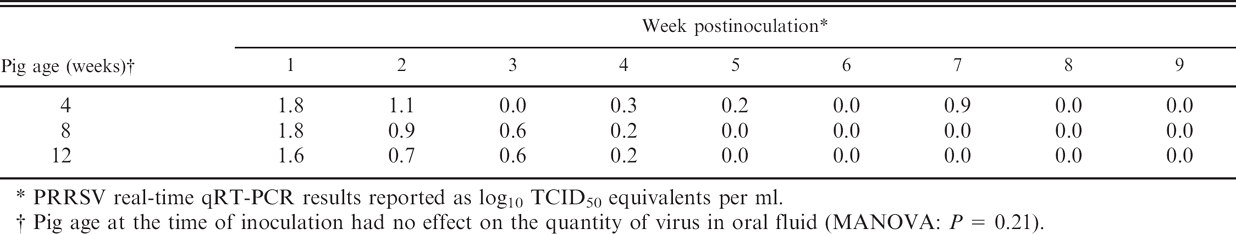
PRRSV real-time qRT-PCR results reported as log10 TCID50 equivalents per ml.
Pig age at the time of inoculation had no effect on the quantity of virus in oral fluid (MANOVA:
Laboratory B: Oral fluid
PRRSV real-time qRT-PCR results reported as log10 RNA copies per ml.
Pig age at the time of inoculation had no effect on the quantity of virus in oral fluid (MANOVA:
Porcine reproductive and respiratory syndrome virus enzyme-linked immunosorbent assay
Statistical analysis of the serum antibody response found a significant difference in ELISA S/P values by pig age at the time of inoculation (MANOVA:
In contrast, a comparison of oral fluid ELISA results found no significant difference in least square means between PRRSV-inoculated and negative-control pens (MANOVA:
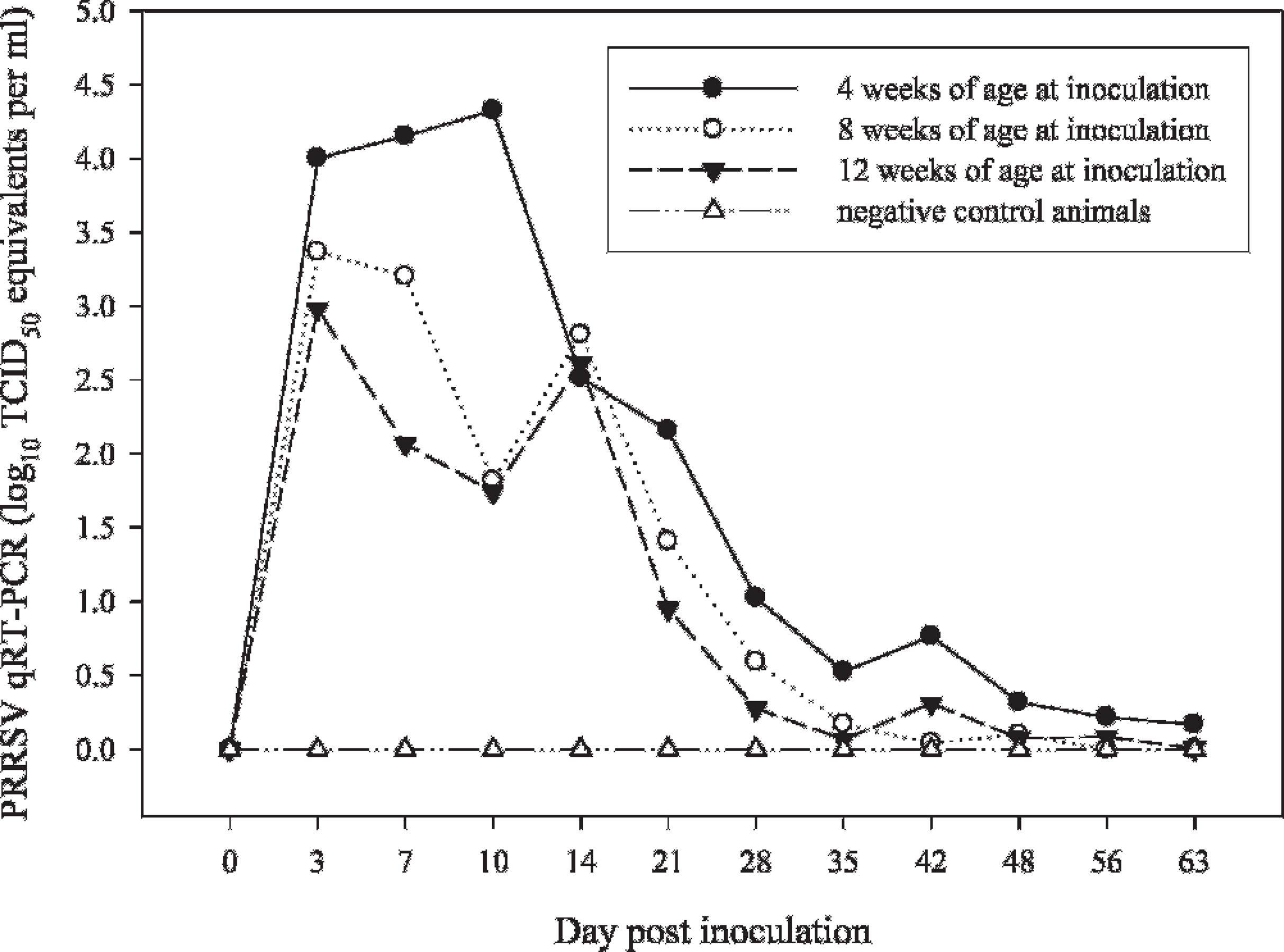
Serum
Indirect fluorescent antibody test
Anti-PRRSV IgG was detected by IFAT in 20 of 154 oral fluid samples from inoculated pens, with positives sporadically distributed across the postinoculation observation period. No anti-PRRSV IgA was detected in oral fluid samples with the protocol described. Samples from negative control pens were negative for both IgG and IgA.
Discussion
In humans, viral infections in which the agent is present in oral fluids include
Serum
PRRSV real-time qRT-PCR results reported as log10 TCID50 equivalents per ml.
Within columns, means with different superscripts were significantly different by Tukey Honestly Significantly Different test.
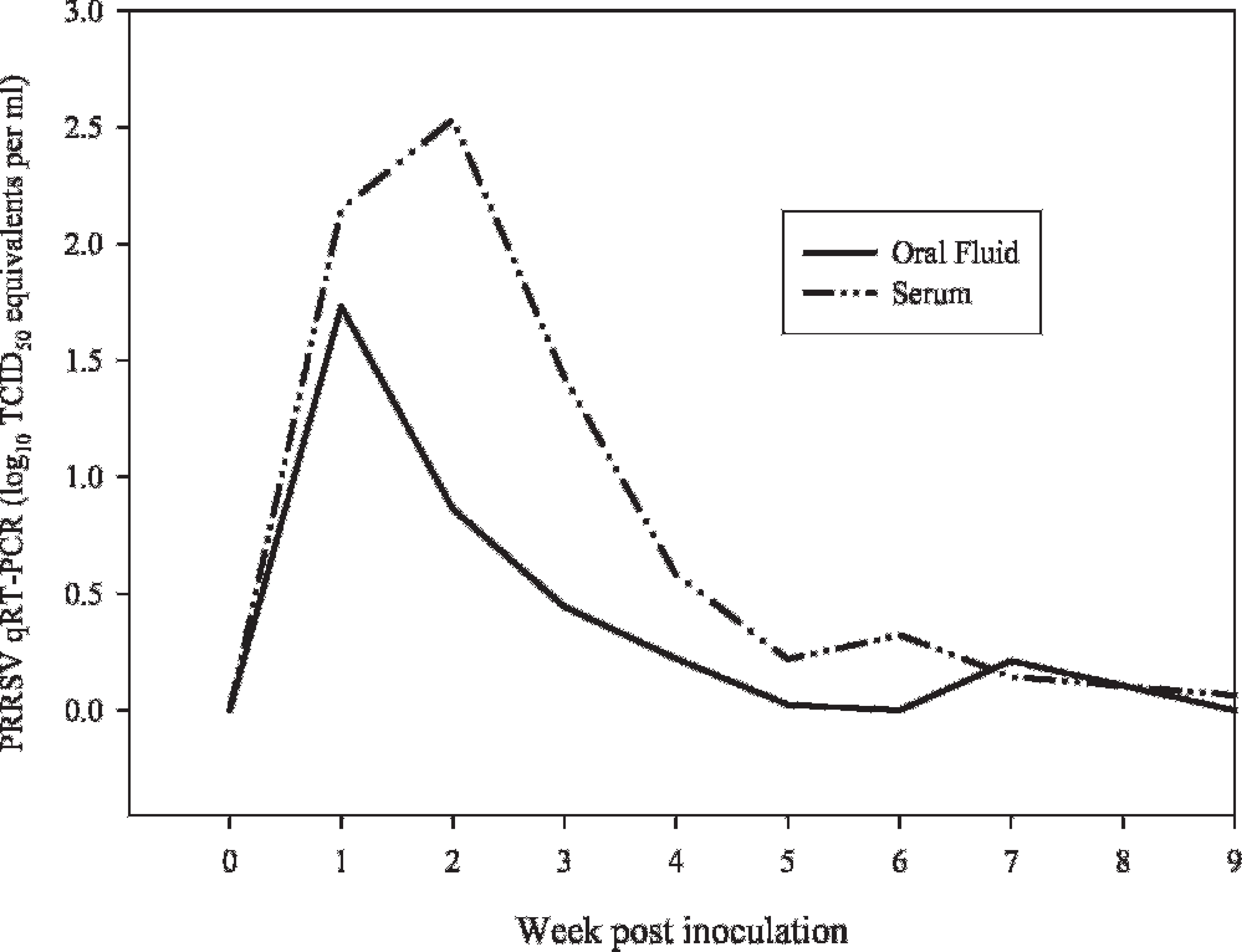
Serum and oral fluid
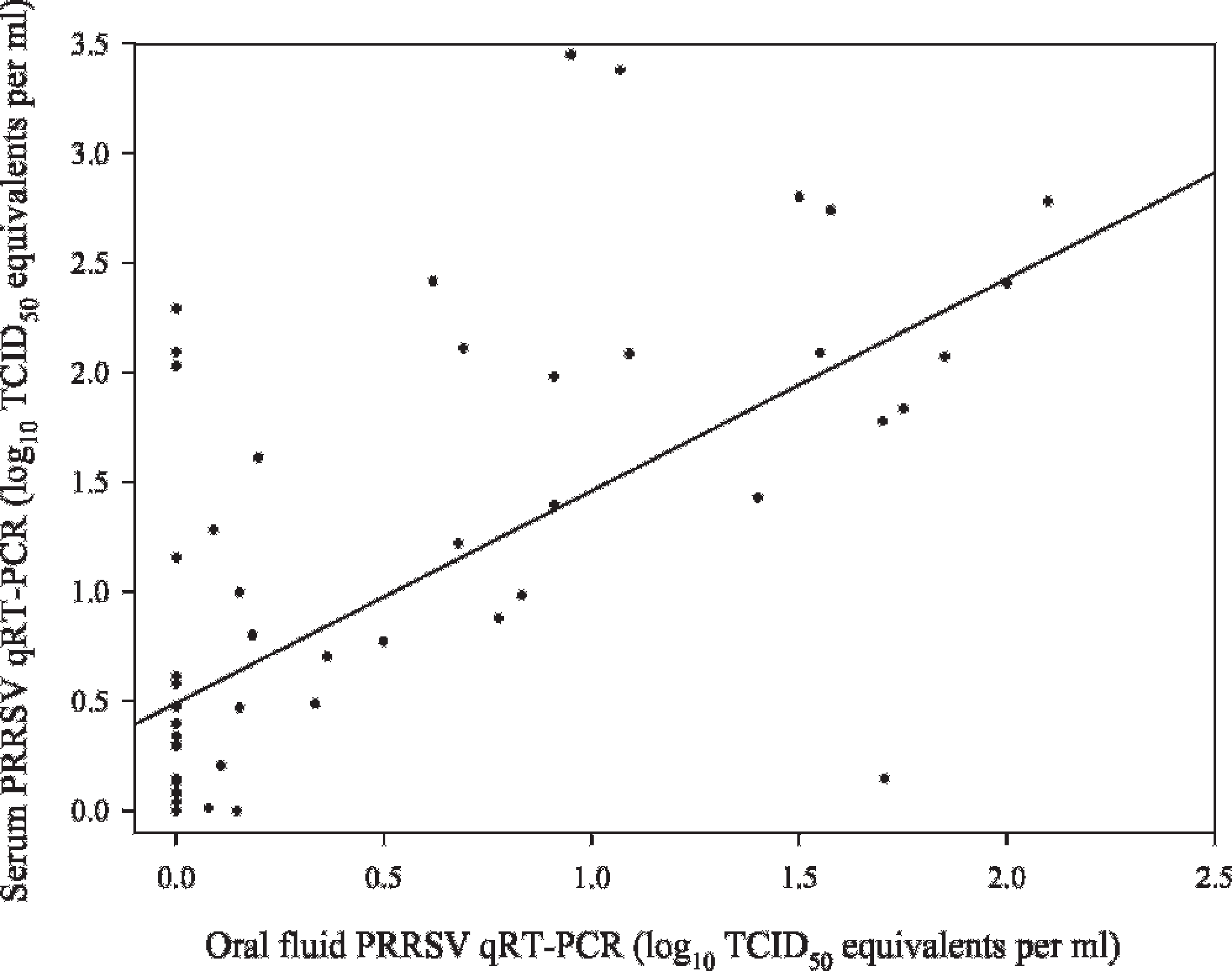
Correlation between serum and oral fluid
In animals, the presence of pathogens in oral fluid has generally been described in the context of transmission (e.g.,
The use of oral fluids in veterinary diagnostic medicine has been minimal, for the most part limited to the diagnosis of
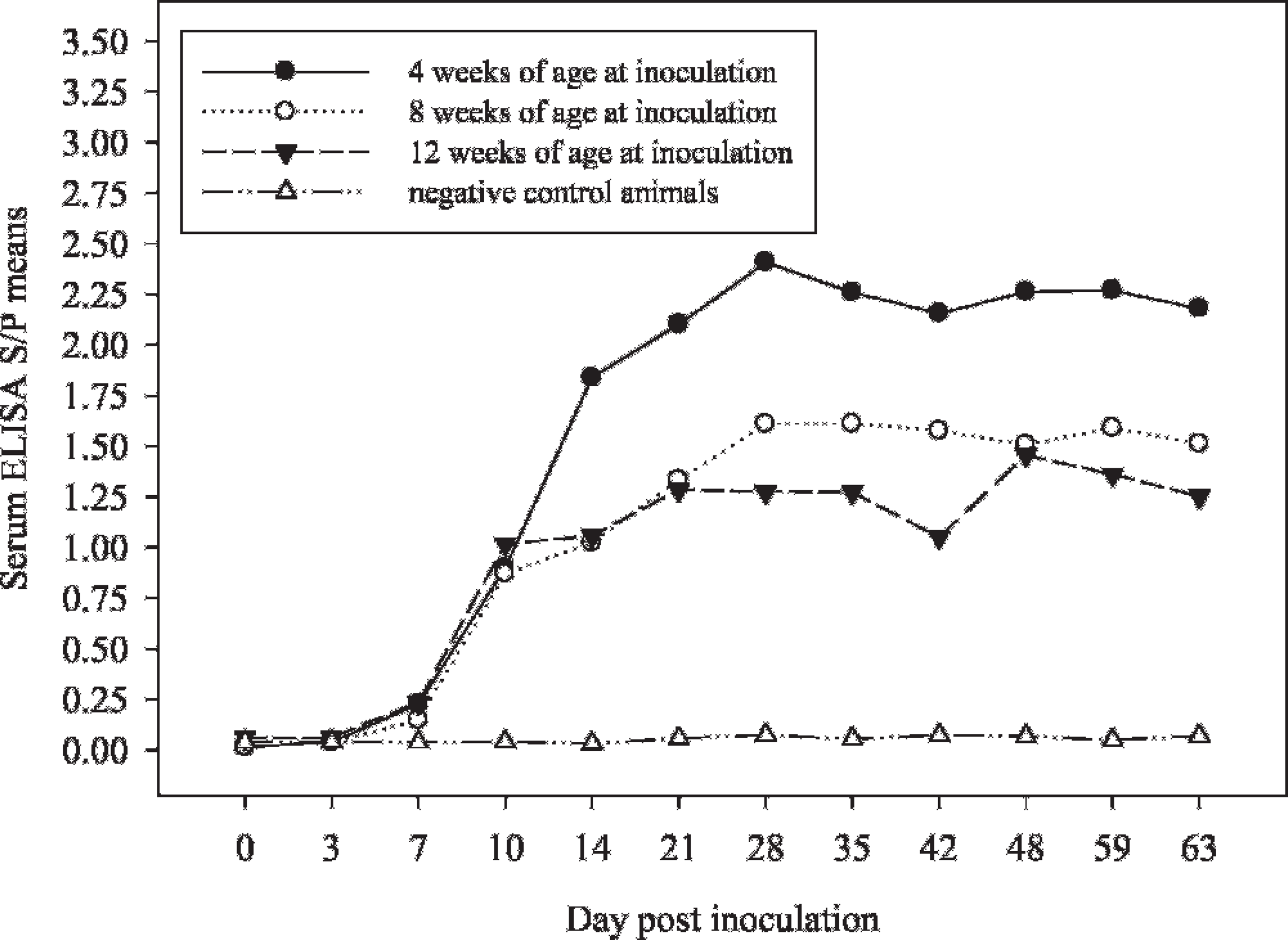
Serum
Serum
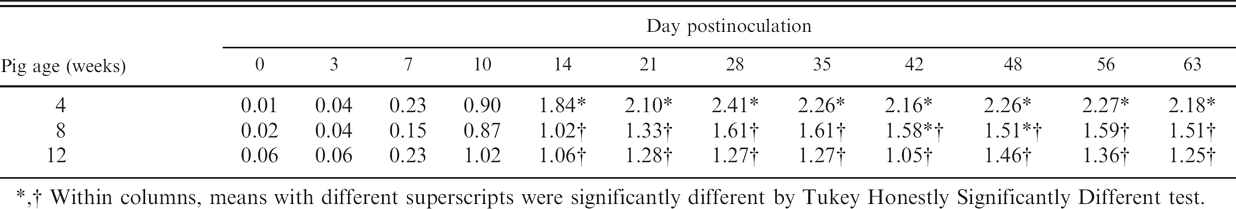
Within columns, means with different superscripts were significantly different by Tukey Honestly Significantly Different test.
Within columns, means with different superscripts were significantly different by Tukey Honestly Significantly Different test.
The research reported here represents a further investigation of the application of oral fluids to veterinary diagnostic medicine. In this experiment, the collection of oral fluid samples from pens of pigs was determined to be easy and efficient. Normal pig behavior was conducive to sample collection, that is, pigs naturally investigate and chew on new objects within the pen (i.e., rope).
Acknowledgements
The study was supported in part by Pork Checkoff funds distributed through the National Pork Board, PO Box 9114, Des Moines, IA 50306. The authors thank the faculty and staff of the Iowa State University Veterinary Diagnostic Laboratory for advice and technical support.
Footnotes
a.
Becton Dickinson, Franklin Lakes, NJ.
b.
Fisher Scientific, Logan, UT.
c.
Ace Hardware Corporation, Colorado Springs, CO.
d.
QIAGEN, Inc., Valencia, CA.
e.
Applied Biosystems, Foster City, CA.
f.
Integrated DNA Technologies, Coralville, IA.
g.
GenBank, Bethesda, MD.
h.
Bethyl Laboratories, Montgomery, TX.
i.
IDEXX Laboratories, Inc., Westport, ME.
j.
SAS Institute, Cary, NC.
