Abstract
The purpose of the present study was to evaluate the diagnostic performance of a commercial serum antibody enzyme-linked immunosorbent assay (ELISA) modified to detect anti–
Introduction
“Oral fluid,” the liquid collected by placing an absorptive device in the mouth,
2
is a combination of serum transudate and saliva. Serum transudate enters the mouth from capillaries in the oral mucosa, crevicular gap, and gingival tissues.5,8 In human beings, the ease of collecting oral fluid samples has facilitated the implementation of this approach for large epidemiological studies. For example, in England, oral fluid samples were collected from 11,698 children at home by their parents and mailed to the laboratory for antibody testing.
3
Oral fluid specimens are used in human medicine and forensics for the diagnosis or detection of a variety of infectious agents,
23
hormones,
19
and drugs.
7
In particular, the recognition that both
Although oral fluid testing has not been widely applied to livestock health and wellness management, veterinary literature on the presence of antibodies, pathogens, and acute phase proteins in oral fluids from animals reflects the findings in human beings.
30
In swine, infectious agents, cortisol, acute phase proteins, and progesterone have all been detected in oral fluid specimens under experimental and field conditions.12,24,27–29 Increasingly, oral fluid samples are used for the surveillance of
Materials and methods
Experimental design
Oral fluid samples (
Oral fluid specimens
All procedures for pigs experimentally inoculated with PRRSV or vaccinated with licensed modified live virus (MLV) PRRS vaccine were approved by the Institutional Animal Care and Use Committees (IACUC) at Iowa State University (IACUC no. 10-10-7033-S) and Kansas State University (IACUC no. 4567). In all cases, oral fluid specimens were collected by allowing access to a short length of cotton rope suspended in the pen. After 20–30 min of pig interaction (biting) with the rope, oral fluid was extracted by passing the rope through a hand-operated wringer. b These procedures are fully described elsewhere (National Pork Board: 2011, Oral fluid collection in pigs booklet [NPB-03623]. Available at: http://www.porkstore.pork.org/producer/default.aspx?p=viewitem&item=NPB-03623&subno=&showpage=4&subcat=12. Accessed December 20, 2011).
In brief, 5-week-old pigs (
Oral fluid samples (
Expected PRRSV antibody–negative oral fluid field samples (
Protocol for PRRSV oral fluid IgG ELISA
The procedure for the commercial indirect ELISA a was modified to detect anti-PRRSV antibody in porcine oral fluid specimens. 14 The modified assay required 2 days to complete. In brief, on day 1, oral fluid samples were diluted 1:2 in dilution plates (150 µl oral fluid sample and 150 µl diluent) using the diluent provided with the test kit. Diluted oral fluid was then transferred to the 96-well PRRSV antigen-coated plates (250 µl per well per sample). Negative and positive kit controls were diluted 1:30 using the kit diluent (10 µl kit control and 290 µl diluent), and 100 µl was transferred to the assigned wells on the plates (i.e., A1 and B1 for negative controls; C1 and D1 for positive controls). Thereafter, plates were incubated for 16 hr at 4°C in a refrigerated incubator.
At the end of the 16-hr incubation period, the coated plates were washed 3 times with 400 µl of 1× kit wash solution. To detect bound antibody, reagents were brought to room temperature and then 100 µl of a solution containing appropriately diluted horseradish peroxidase (HRP)–conjugated anti-swine IgGFC secondary antibody f was added to each well and incubated for 30 min at 22°C. Thereafter, plates were washed 3 times with wash solution, then 100 µl of tetramethylbenzidine enzyme substrate solution was added to each well, and the plates were incubated at 22°C for 15 min. After 15 min, 100 µl of kit stop solution was added to each well. The plates were read at 650 nm using an ELISA plate reader g controlled by commercial software. h The reactions were measured as optical density (OD) and then converted to sample-to- positive ratios (S/P) using the following formula:
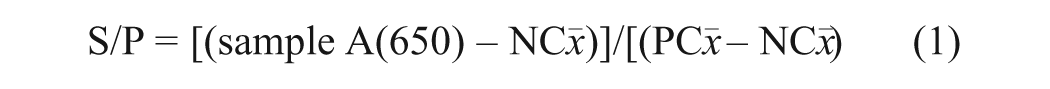
where
Preparation of anti-swine IgGFC secondary antibody
The appropriate dilution of HRP-conjugated anti-swine IgGFC antibody f was calculated for each ELISA kit lot to standardize the strength of the reaction and ensure the repeatability of results. This was achieved by determining the dilution of anti-swine IgGFC that matched the positive control OD value listed in the manufacturer’s Certificate of Analysis for the ELISA kit lot. Certificates of Analysis for specific kit lots can currently be obtained using the manufacturers’ Certificate of Analysis Request Form (http://www.idexx.com/view/xhtml/en_us/livestock-poultry/certificates-of-analysis.jsf).
In brief, 4 dilutions (1:1,000; 1:1,500; 1:2,000; 1:2,500) of anti-swine IgGFC antibody
f
were prepared using the anti-pig IgG:HRPO conjugate provided with the ELISA
a
kit as diluent and then stirred for 48 hr at 4°C. To test the reactivity of the 4 dilutions of anti-swine IgGFC, kit negative and positive controls were diluted 1:30 (10 µl kit control and 290 µl kit sample diluent), and then 100 µl of diluted negative control was transferred into 48 wells (i.e., one half of the plate) and 100 µl of diluted positive control in each of the remaining 48 wells. Thereafter, the protocol for the PRRSV oral fluid IgG ELISA was conducted, as described above. Negative control OD values were used to detect nonspecific reactions. Positive control OD values were used to determine the correct dilution of secondary antibody in 2 steps: 1) linear regression (equation 2) was used to determine the equation of the line that described the relationship between positive control OD response and secondary antibody dilution (1:1,000; 1:1,500; 1:2,000; 1:2,500) and 2) the appropriate dilution of conjugated antibody dilution was calculated by substituting the mean positive control OD from the Certificate of Analysis for

where
IgM and IgA protocols
The commercial indirect ELISA was also modified to detect PRRSV-specific IgM (or IgA) antibody responses. To detect bound antibody, HRP-conjugated anti-pig IgM antibody i or anti-pig IgA antibody j was diluted 1:2,000 in conjugate diluent provided by the kit manufacturer. The reference standard oral fluid sample collected on day 0 was used as the negative control. The S/P values for IgM and IgA ELISAs were calculated as described in equation 1 using reference standard samples collected on DPV 10 and 35 used as positive controls for the IgM and IgA ELISAs, respectively.
For the PRRSV oral fluid IgM and IgA ELISA results to be valid, the mean of the kit positive control OD minus the mean of the kit negative control OD had to be ≥0.150 and the kit negative control mean OD ≤0.150. These validity checks were identical to those required by the manufacturer for the PRRSV serum ELISA.
Evaluation of IgG ELISA performance
Receiver operator characteristic (ROC) analyses were performed using MedCalc 9.2.1.0.
k
Diagnostic sensitivity, specificity, and 95% confidence intervals were estimated for a range of S/P values (≥0.20 to ≥0.40) using test results from samples of defined status. Positive samples for estimating diagnostic sensitivity included oral fluid specimens from experimentally inoculated pigs (
Results
Reference standards
Anti-PRRSV antibody isotype (IgM, IgA, IgG) kinetics in reference standard samples are given in Figure 1 for oral fluid samples collected on DPV 0, 10, 15, 20, 28, 35, 41, 49, 56, 75, and 91. On DPV 10, S/P ratios for IgM, IgA, and IgG were 1.00, 0.31, and 1.22, respectively. On DPV 15, S/P ratios peaked at 1.87, 1.53, and 4.29 for IgM, IgA, and IgG. Immunoglobulin M declined rapidly (i.e., the S/P ratio was 0.10 at DPV 28). For DPV 20–41, IgA S/P ratios ranged from a low of 0.59 (DPV 41) to a high of 1.05 (DPV 28). For DPV 49–91, IgA S/P ratios ranged from a low of 0.33 (DPV 75) to a high of 0.53 (DPV 91). Immunoglobulin G S/P ratios declined relatively slowly and steadily after DPV 15, reaching a low of 2.65 on DPV 75.
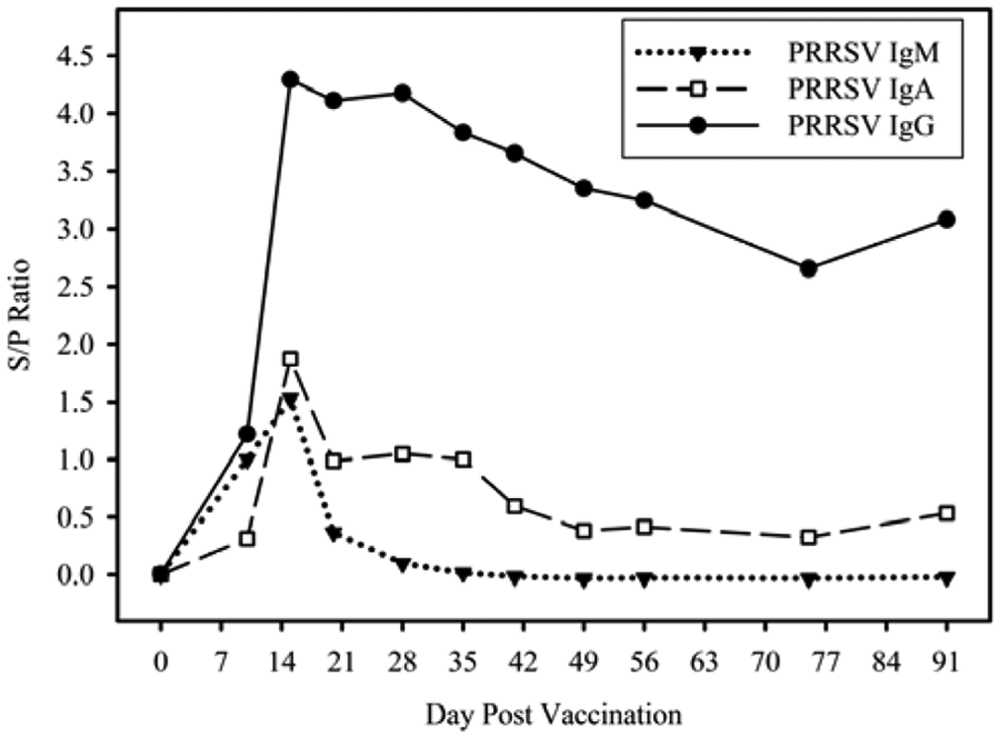
Kinetics of anti–
Experimental samples
Anti-PRRSV antibody isotype (IgM, IgA, IgG) kinetics in experimental samples are shown in Figure 2 for oral fluid samples collected on DPI 0, 5, 7, 9, 11, 14, 17, and 21. Mean IgM, IgA, and IgG S/P ratios on DPI 7 were 1.14 (95% confidence interval [CI]: 1.04, 1.24), 0.09 (95% CI: 0.06, 0.12), and 0.59 (95% CI: 0.50, 0.68), respectively. The mean IgM S/P ratio peaked at DPI 9 (1.88; 95% CI: 1.78, 1.97) and declined rapidly to a mean S/P of 0.10 (95% CI: 0.09, 0.13) on DPI 21. Mean IgA and IgG S/P ratios peaked at DPI 11 (1.41; 95% CI: 1.29, 1.53) and DPI 14 (3.69; 95% CI: 3.52, 3.85), respectively. Mean IgA and IgG S/P ratios remained relatively constant until the end of study at DPI 21.
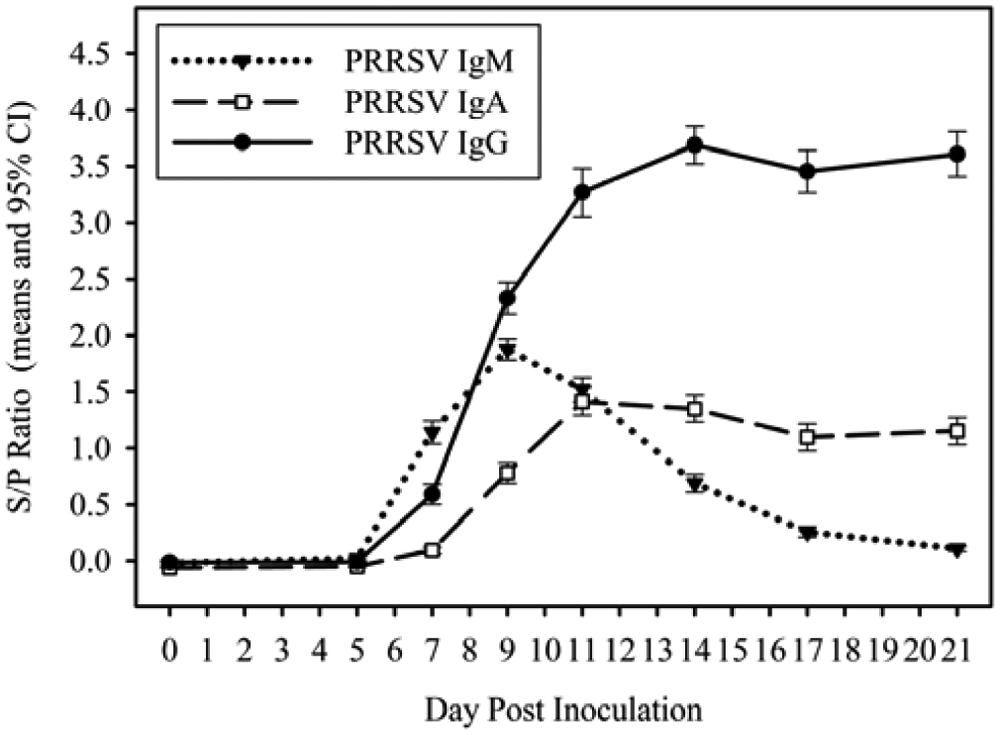
Kinetics of anti–
Field samples
Among the 60 pens on 10 sites from which oral fluid samples were collected over time, 39 pens had 1 or more PRRSV qRT-PCR–positive oral fluid samples over the course of the 18-week observation period. Expected PRRSV antibody–positive oral fluid field samples were defined as all samples collected from a pen after its first PRRSV qRT-PCR–positive sample. Based on this definition, 241 (40%) of 600 oral fluid samples were defined as expected antibody-positive field specimens. Specifically, these samples were from samples collected 2–18 weeks following the first qRT-PCR–positive result (Table 1).
Detection of
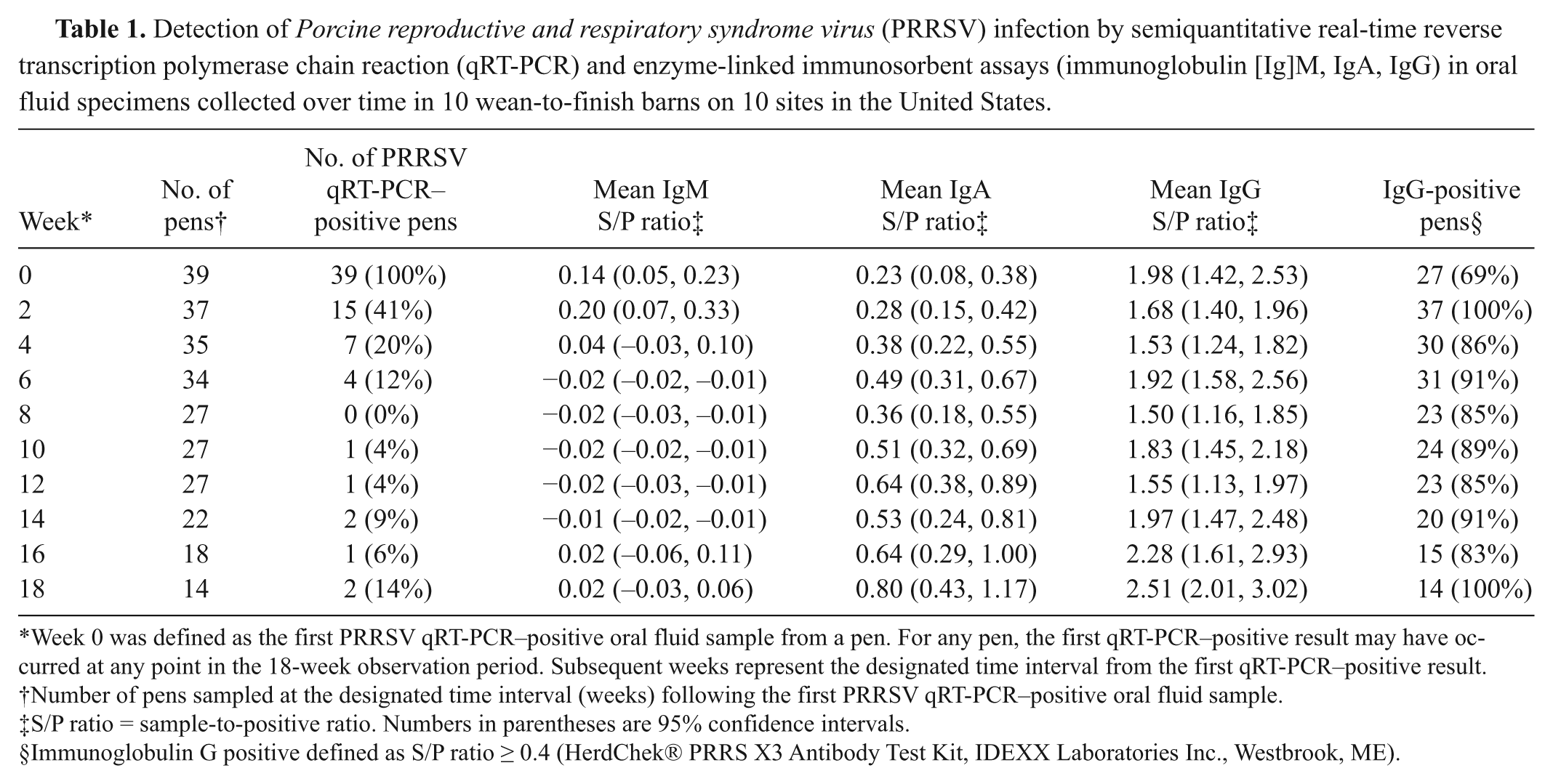
Week 0 was defined as the first PRRSV qRT-PCR–positive oral fluid sample from a pen. For any pen, the first qRT-PCR–positive result may have occurred at any point in the 18-week observation period. Subsequent weeks represent the designated time interval from the first qRT-PCR–positive result.
Number of pens sampled at the designated time interval (weeks) following the first PRRSV qRT-PCR–positive oral fluid sample.
S/P ratio = sample-to-positive ratio. Numbers in parentheses are 95% confidence intervals.
Immunoglobulin G positive defined as S/P ratio ≥ 0.4 (HerdChek® PRRS X3 Antibody Test Kit, IDEXX Laboratories Inc., Westbrook, ME).
Mean oral fluid IgM, IgA, and IgG S/P ratios at the time of the first detection of PRRSV by qRT-PCR were 0.14 (95% CI: 0.05, 0.23), 0.23 (95% CI: 0.08, 0.38), and 1.98 (95% CI: 1.42, 2.53), respectively (Table 1). Peak IgM S/P ratios 0.20 (95% CI: 0.07, 0.33) were observed 2 weeks after the first detection of PRRSV by qRT-PCR and declined thereafter. Immunoglobulin A and IgG S/P ratios were relatively steady but tended to increase over the course of the observation period, reaching a peak in samples collected 18 weeks after the first qRT-PCR–positive sample (Fig. 3).
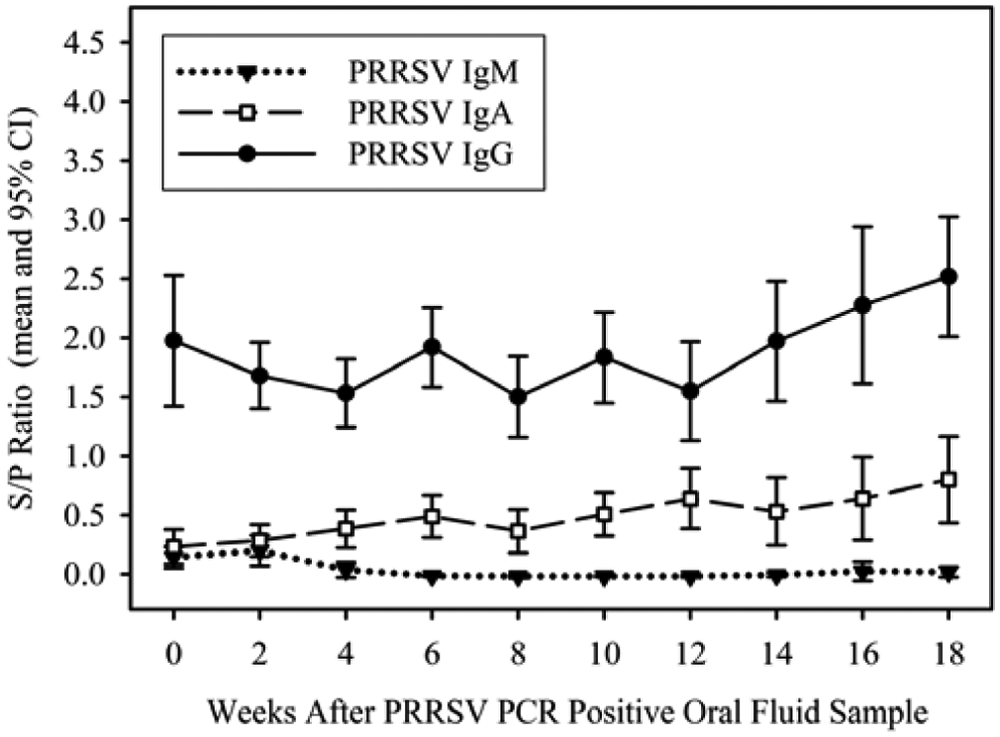
Kinetics of anti–
IgG indirect ELISA diagnostic performance
Estimates of IgG indirect ELISA performance were based on samples of defined status. Positive samples (
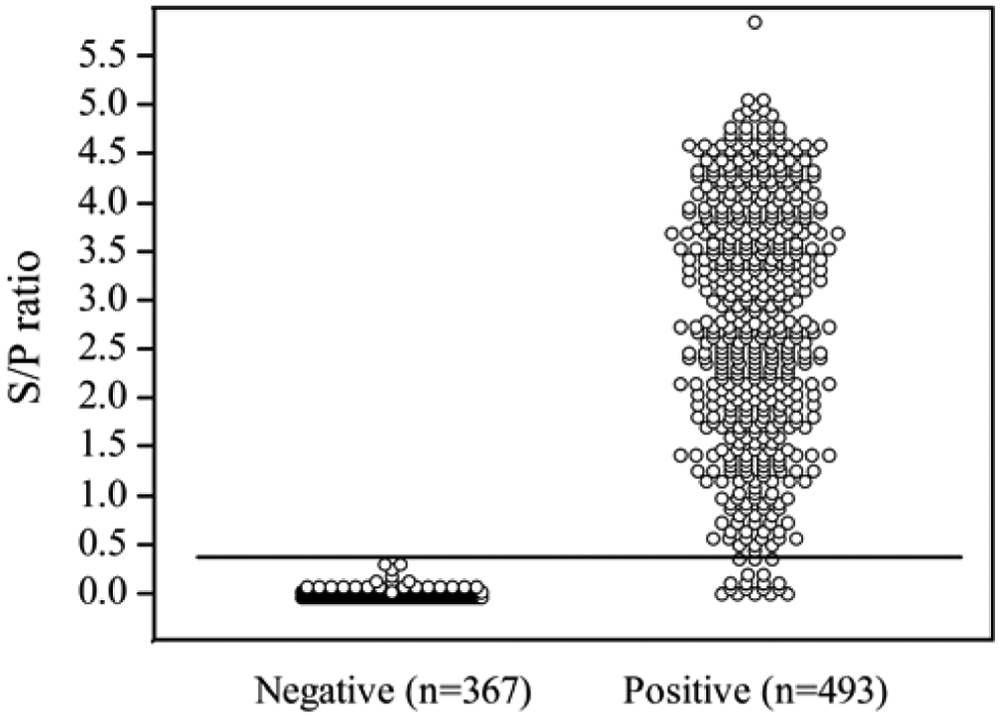
Distribution of
Diagnostic performance of an enzyme-linked immunosorbent assay performed using a protocol modified to detect anti–
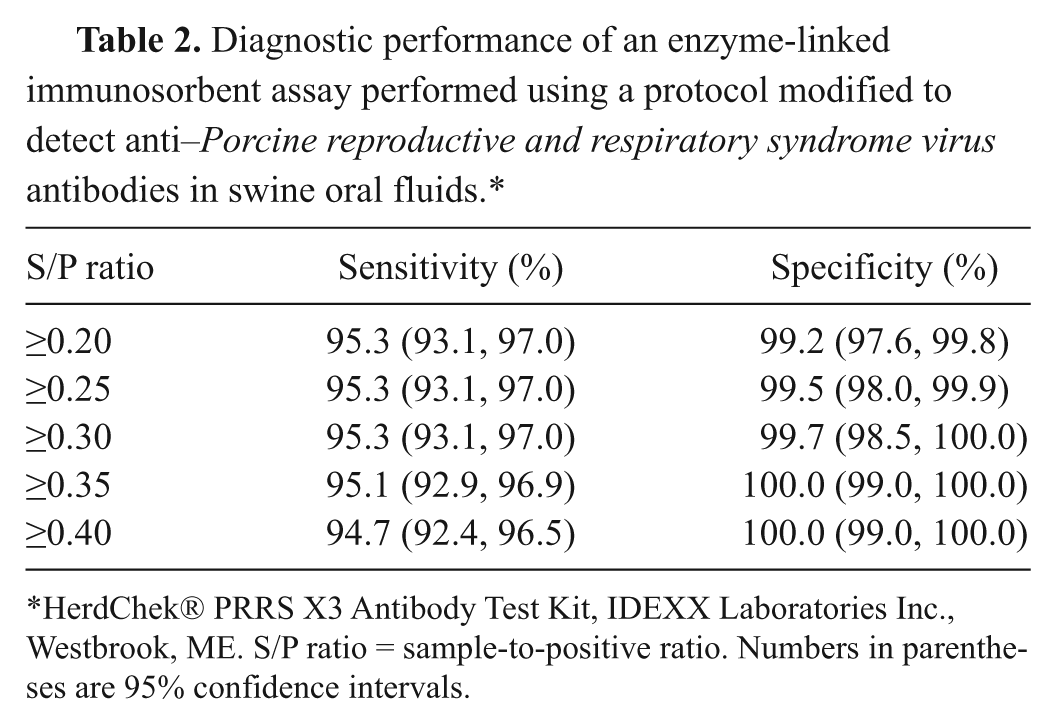
HerdChek® PRRS X3 Antibody Test Kit, IDEXX Laboratories Inc., Westbrook, ME. S/P ratio = sample-to-positive ratio. Numbers in parentheses are 95% confidence intervals.
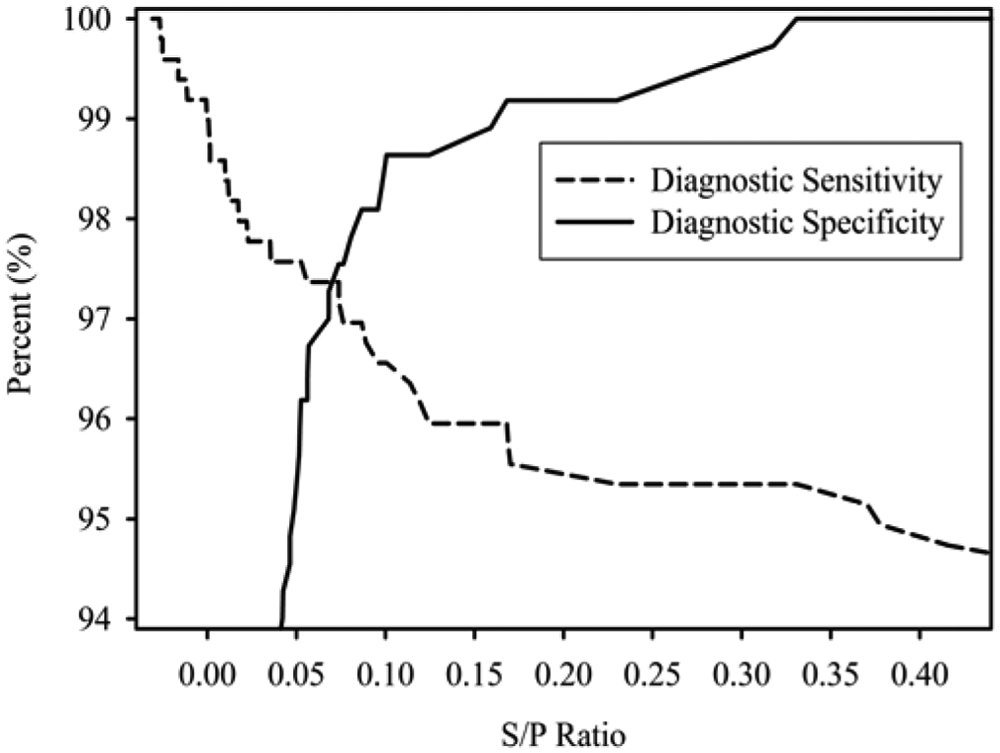
Estimates of
Discussion
In the current study, the detection of anti-PRRSV antibodies in oral fluid specimens was evaluated using a commercial PRRSV indirect ELISA a performed using a procedure adapted to the oral fluid matrix. The validity of this approach was supported by prior evidence that pigs, like human beings and other species, produce detectable levels of specific antibodies in oral fluid in response to infection with PRRSV, as well as other pathogens. 30 Essentially, the ELISA procedure was modified to calibrate the reactivity of the assay to the lower concentration of antibody present in oral fluid relative to serum. Specific modifications included larger sample volume, less dilution of the sample, higher secondary antibody concentration, and longer sample incubation. 14 A key step to creating a reproducible test was determining the kit lot– specific dilution of secondary antibody. This was done by titrating the secondary antibody against a 1:30 dilution of positive and negative controls and then calculating the dilution of secondary antibody that matched the OD values for the positive and negative controls in the manufacturer’s Certificate of Analysis. This step provided repeatable results and resulted in a cutoff in the oral fluid IgG ELISA that matched the cutoff recommended by the manufacturer for the serum ELISA (S/P ≥ 0.40).
Anti-PRRSV IgM, IgA, and IgG responses were evaluated in pen-based oral fluid samples using isotype-specific ELISAs. Immunoglobulin M, IgA, and IgG were readily detected in populations in which the infection was synchronized among all individuals (i.e., reference standards and experimental samples). However, this was not the case in field samples. The reason for the difference in responses in synchronized versus field samples is unknown, but it may be hypothesized that asynchrony in the spread of PRRSV infection in the field resulted in animals at different stages of the antibody response. Consequently, IgM and IgA from pigs in the early stages of infection would have been diluted by oral fluid from animals not secreting those antibody isotypes. In addition, IgG, both because of its higher concentration and its higher affinity, may have outcompeted IgM and IgA for the antigenic sites available in the ELISA plate.18,22
In contrast to IgM and IgA, IgG was readily detected at diagnostically useful levels in reference standards, experimental samples, and field samples for an extended period. That is, IgG was consistently detected in reference standards through the termination of sample collection at DPV 91 and in experimental samples and field samples throughout their respective observation periods (i.e., 21 and 126 days).
Previous reports on anti-PRRSV serum antibody kinetics using an indirect fluorescent antibody test reported that IgM was first detected 5–7 DPI13,20,26 and IgG was detected 9–11 DPI.20,34 Thus, the kinetics of serum IgM and IgG responses reported in the literature and the findings of the current study for oral fluid were similar. In contrast, IgA was first detected by immunoperoxidase monolayer assay in serum at 14 DPI, reached its maximum concentration at 25 DPI, and was undetectable after 35 DPI. 17 The reported detection of IgA in serum was later and shorter compared with the response observed in oral fluid. Overall, the observations for oral fluid are in agreement with general understanding of antibody kinetics in serum and on mucosal surfaces. Likewise, these data are compatible with previous reports that IgA is locally produced by serum-derived plasma cells in salivary glands and duct-associated lymphoid tissue, whereas IgM and IgG are primarily derived from serum, with some contribution from local production. 30
The use of pen-based oral fluid specimens, an unusual sample for an antibody ELISA, requires comment because an understanding of the epidemiological and statistical properties of the sample is central to the interpretation and analysis of the assay results. In sampling terminology, an oral fluid sample is best described as a grab sample from a sample unit (pen), where “grab sample” is defined as a sample collected “at a specific point in time and at a specific location” 16 and “sample unit” is the smallest division of the population and the level at which sample size calculations and random sampling procedures are performed.9,32,33 Examples of sample units include “an animal, a group of animals, i.e., a pen, a herd . . . . ” 33 Oral fluid ELISA results are amenable to diagnostic interpretation because the pathogen-specific epidemic curve and immune responses of the individual pigs within the pen are highly correlated (dependent). The link with specific time and place provides for the statistical analysis of the results, including analysis of geospatial relationships between pens in the context of the larger population.
A pen-based sample should not be confused with a pooled sample. More universally known as a “composite sample,”
10
a pooled sample is created by combining 2 or more sample unit specimens into 1 aliquot for testing, thereby losing sample unit identity in the process.
5
Pooled samples are typically used to screen populations at a lower cost than testing individual samples using a 2-step process termed the
Cumulatively, the present study showed that a commercial serum antibody ELISA a adapted to oral fluid specimens can be used to detect anti-PRRSV antibody. Testing for PRRSV antibody using pen-based oral fluids could provide an efficient, cost-effective approach for monitoring maternal antibody, vaccination compliance, and herd immune parameters in commercial herds and surveillance of PRRSV infections in elimination programs. The successful adaptation of a commercial serum antibody ELISA to the oral fluid matrix suggests that antibody assays for other pathogens could also be modified to this purpose.
Footnotes
a.
HerdChek® PRRS 3X ELISA, IDEXX Laboratories Inc., Westbrook, ME.
b.
BL-38, Dyna-Jet Products, Overland Park, KS.
c.
Ingelvac® PRRS MLV, Boehringer Ingelheim Vetmedica Inc., St. Joseph, MO.
d.
Becton, Dickinson and Company, Bedford, MA.
e.
Applied Biosystems, Foster City, CA.
f.
Pig IgG-Fc Antibody (catalog no. A100-104P), Bethyl Laboratories Inc., Montgomery, TX.
g.
EL800 micro plate reader, BioTek Instruments Inc., Winooski, VT.
h.
GEN5™, BioTek Instruments Inc., Winooski, VT.
i.
Pig IgM Antibody (catalog no. A100-100P), Bethyl Laboratories Inc., Montgomery, TX.
j.
Pig IgA Antibody (catalog no. A100-102P), Bethyl Laboratories Inc., Montgomery, TX.
k.
MedCalc Software, Mariakerke, Belgium.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors A. Ballagi and A. Rice are employed by IDEXX Laboratories Inc. The remaining authors declare no conflicting interests with respect to their authorship or the publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by Pork Checkoff funds distributed through the National Pork Board (PO Box 9114, Des Moines, IA 50306) and the PRRS CAP, USDA NIFA award 2008-55620-1932. HerdChek® PRRS 3X ELISA kits were provided by IDEXX Laboratories Inc., Westbrook, ME.
