Abstract
Mycoplasma bovis is an important cause of bovine respiratory disease (BRD) in newly received cattle at fattening operations. However, little information on its within-pen transmission dynamics during a BRD outbreak is available. Such information is nevertheless crucial to adapt control measures during M. bovis–associated BRD outbreaks. The objective of the current study was to determine whether single or multiple clones of M. bovis are present within a pen during a BRD outbreak that occurs early in the feeding period. Sixteen BRD outbreaks that naturally occurred in 12 pens of 8–12 bulls each (n = 112) newly received at 3 fattening operations were investigated. Two hundred and thirty-nine transtracheal aspirations (TTA) were performed during the outbreaks, and the M. bovis isolates obtained were characterized by pulsed-field gel electrophoresis (PFGE). Mycoplasma bovis isolates were recovered from TTA in 8 of the 16 BRD outbreaks that occurred. The within-pen prevalence of bulls positive for M. bovis during these outbreaks ranged from 8% to 100%. The PFGE analysis revealed that, even though bulls came from multiple origins, a single clone of M. bovis was present within a pen during BRD outbreaks with a high prevalence of M. bovis infection. The study therefore indicates that, even if M. bovis can recrudesce from carriers after stressful events such as transportation and commingling, the increased prevalence of M. bovis pulmonary infection observed during BRD outbreaks that are early occurring in the feeding period seems primarily due to the horizontal transmission of only 1 clone among cattle.
Bovine respiratory disease (BRD) is the most prevalent disease in cattle entering fattening operations. 3 Among the pathogens implicated in BRD, Mycoplasma bovis plays an important contributory role. 9 Indeed, M. bovis has been frequently identified within pulmonary lesions that occurred during the 2 months after arrival of cattle at fattening operations. 7 Furthermore, seroconversion to M. bovis during the first weeks after entrance is associated with an increased risk of being treated for BRD. 8 Early and efficient control of M. bovis infection could thus significantly decrease the overall consequences of BRD. 9
A good understanding of within-pen transmission dynamics of M. bovis during a M. bovis–associated BRD outbreak is needed to adapt control measures.5,9 However, to date, little information on the transmission dynamics of M. bovis in fattening operations is available in the veterinary literature.5,9
Strain typing can provide insights into the transmission dynamics of M. bovis. 5 Indeed, typing methods can determine relatedness among isolates that are part of the same chain of transmission and discriminate nonrelated isolates. 11 Among these methods, pulsed-field gel electrophoresis (PFGE) appears to be well adapted to monitor the transmission dynamics of M. bovis among cattle within a pen due to its high discriminatory power and reproducibility.1,5 The objective of the current study was, therefore, to characterize, by PFGE, the M. bovis isolates collected from the lower respiratory tract of bulls during BRD outbreaks that occur early in the feeding period to determine whether single or multiple clones of M. bovis are present within a pen.
One hundred and twelve bulls (initial body weight ± standard deviation [SD] = 346 ± 36 kg) were observed for 40 days following their arrival at 3 French fattening operations between November 2007 and March 2008 (Table 1). Bulls were purchased at auction markets and came from multiple sources (n = 43 different origins). After purchase, bulls were transported by truck to a central facility where they were sorted into groups of 8–12 bulls based on body weight. Bulls were then shipped to fattening operations by truckloads of 1–3 groups of bulls depending on the destination. After arrival, each group of bulls was housed in a pen separated from the other bulls fed in the fattening operation by fences that allowed nose-to-nose contact. None of the bulls received any vaccines or antibiotics at entry.
Descriptive statistics of 16 bovine respiratory disease (BRD) outbreaks observed in 12 pens of bulls during the first 40 days post-arrival at 3 fattening operations. The number of transtracheal aspiration (TTA) samples collected from the bulls during each BRD outbreak and the number of bulls positive for Mycoplasma bovis on TTA samples are also indicated.
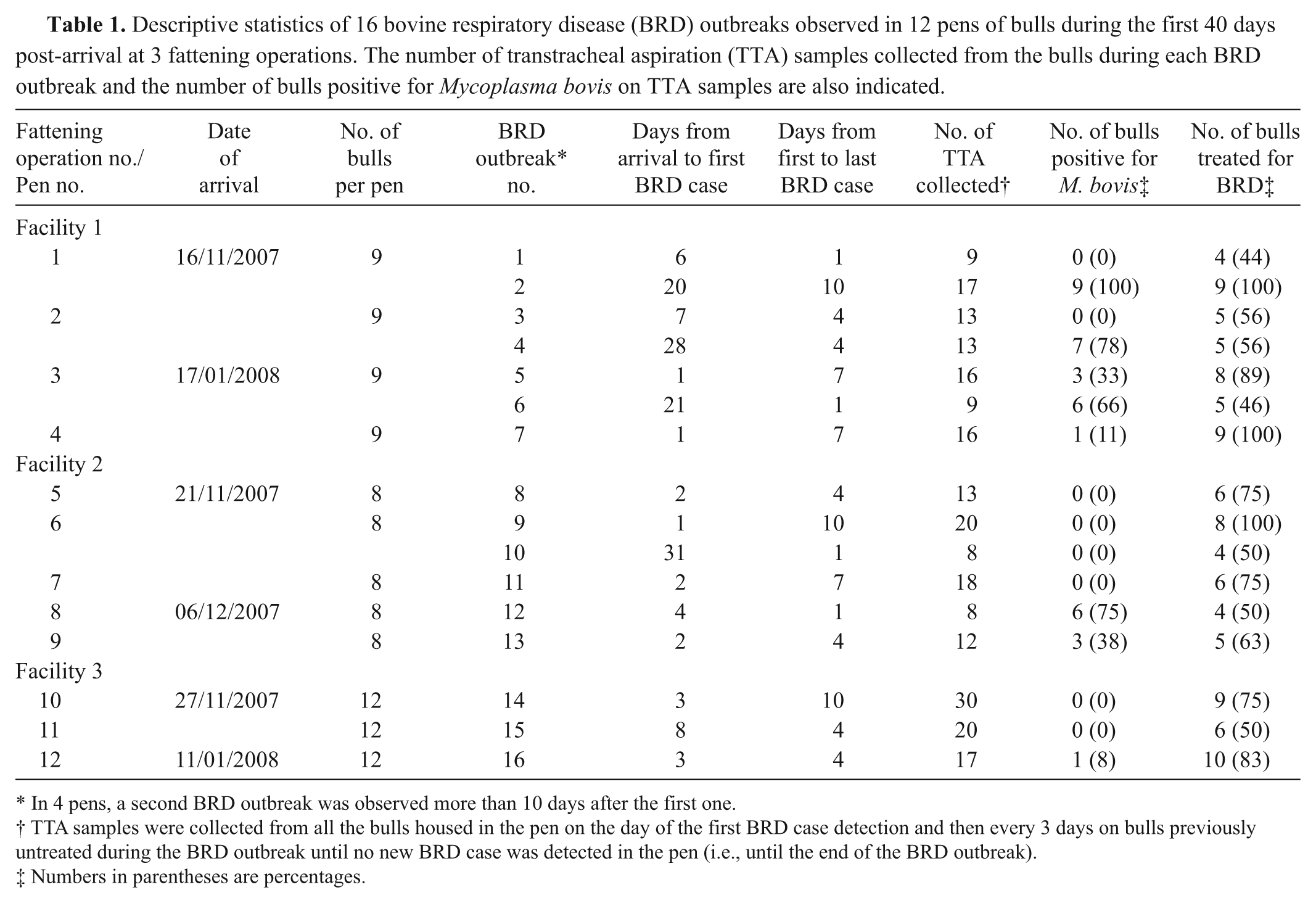
In 4 pens, a second BRD outbreak was observed more than 10 days after the first one.
TTA samples were collected from all the bulls housed in the pen on the day of the first BRD case detection and then every 3 days on bulls previously untreated during the BRD outbreak until no new BRD case was detected in the pen (i.e., until the end of the BRD outbreak).
Numbers in parentheses are percentages.
During the study period, feedlot staff observed the bulls twice daily for the detection of the following signs: depression, decreased rumen fill compared with pen mates, nasal or ocular discharge, cough, and increased respiratory rate. As soon as a bull displaying at least 1 of the above signs was detected in a pen under study, each bull housed in the pen was restrained in a chute by a veterinarian including the in-contact apparently healthy bulls, in order to perform a close physical examination and a transtracheal aspiration (TTA). Bulls with a rectal temperature ≥39.7°C and at least 1 other sign of respiratory tract disease received treatment a against BRD. Physical examinations and TTA samples were then repeated every 3 days on bulls previously untreated during the BRD outbreak until no new BRD case was detected in the pen (i.e., until the end of the BRD outbreak).
Transtracheal aspirations were performed as previously described. 6 Briefly, an area of skin covering the ventral aspect of the middle third of the trachea was shaved and disinfected. An intravenous catheter, b 75 cm in length with an external diameter of 2 mm, was used for the aspiration. The trochar part of the catheter was passed between 2 tracheal rings, and the flexible part of the catheter was introduced for approximately 40 cm in the direction of the lung. A 50-ml volume of physiologically balanced saline (i.e., NaCl 0.9%) was pushed into the catheter via a syringe. The fluid was then immediately retrieved by gentle suction using the syringe. On average, 5–10 ml of fluid was recovered and immediately placed into sterile plain tubes. After sampling, TTA samples were transported in a container at a temperature of 4°C and processed within 6 hr.
Mycoplasma bovis was isolated from TTA samples as previously described.1,2 Briefly, TTA samples were cultured at 37°C in 5% CO2 for 3–5 days on a PPLO (pleuropneumonia-like organism) medium (broth) modified as previously described. 10 After positive growth, the presence of M. bovis was assessed by dot immunobinding on membrane filtration, using polyclonal hyperimmune rabbit antisera prepared against M. bovis PG45. 10 The positive fresh cultures were then plated and incubated for 5–6 days for purification of M. bovis isolates by colony blotting. 2 Afterwards, 1 M. bovis colony per plate was subcultured and stored at −80°C until PFGE analysis. For PFGE analysis, Mycoplasma DNA was prepared in agarose plugs with overnight lysis at 55°C as previously described. 1 DNA plugs were then digested overnight with 30 units of SmaI at 24°C. This enzyme was chosen for its high discriminatory power. 1 The electrophoresis was undertaken in 1% agarose gel using a PFGE system c according to the program previously described. 1
To study the genetic diversity of M. bovis within a TTA sample, 5 TTA samples from a subset of positive bulls were cultured and 3–5 M. bovis colonies per sample were typed by PFGE. All PFGE-related analyses were performed using commercial software. d Isolate relatedness was assessed as previously described 11 (i.e., isolates with PFGE patterns differing by more than 4 bands were assigned to different main types [or clones]). Each main type was arbitrarily identified by a letter (A–E).
Sixteen BRD outbreaks were observed in the 12 pens under study during the 40 days following the arrival of bulls at the fattening operations (Table 1). In 8 pens, only 1 BRD outbreak was observed throughout the study period, whereas in the 4 remaining pens, 2 BRD outbreaks occurred more than 10 days apart from each other (mean ± SD = 15 days ± 4). Depending on the duration of the BRD outbreak and the number of bulls diagnosed and treated for BRD, from 8 to 30 TTA samples were collected (Table 1). In total, 239 TTA were performed during the study period.
Mycoplasma bovis isolates were recovered from TTA samples during 8 of the 16 BRD outbreaks (Table 1). During these outbreaks, the prevalence of bulls positive at least once for M. bovis on TTA samples ranged from 8% to 100% (median: 54%). In total, 35 bulls were positive for M. bovis: 25 bulls were positive once, 9 bulls were positive twice, and 1 bull was positive 4 times. It is interesting to note that, in 2 pens (pen nos. 1 and 2), M. bovis isolates were recovered during the second BRD outbreaks that occurred in these pens but not during the initial outbreaks 14–17 days earlier (Table 1).
Forty-seven M. bovis isolates were recovered from TTA samples. Of these 47 isolates, 3 were not cultivable after storage and thus, were not analyzed by PFGE (Table 2). A PFGE pattern was obtained for all the 44 remaining isolates (i.e., typeability = 100%) and 5 PFGE main types (or clones) comprising between 2 (A) to 24 (D) isolates were observed after SmaI digestion (Fig. 1). The study of the genetic diversity of M. bovis within a TTA sample revealed no diversity. Indeed, identical PFGE profiles were obtained from the 3–5 colonies typed per TTA sample.
Pulsed-field gel electrophoresis types (A–E) of Mycoplasma bovis isolates recovered from transtracheal aspiration samples performed during bovine respiratory disease (BRD) outbreaks.
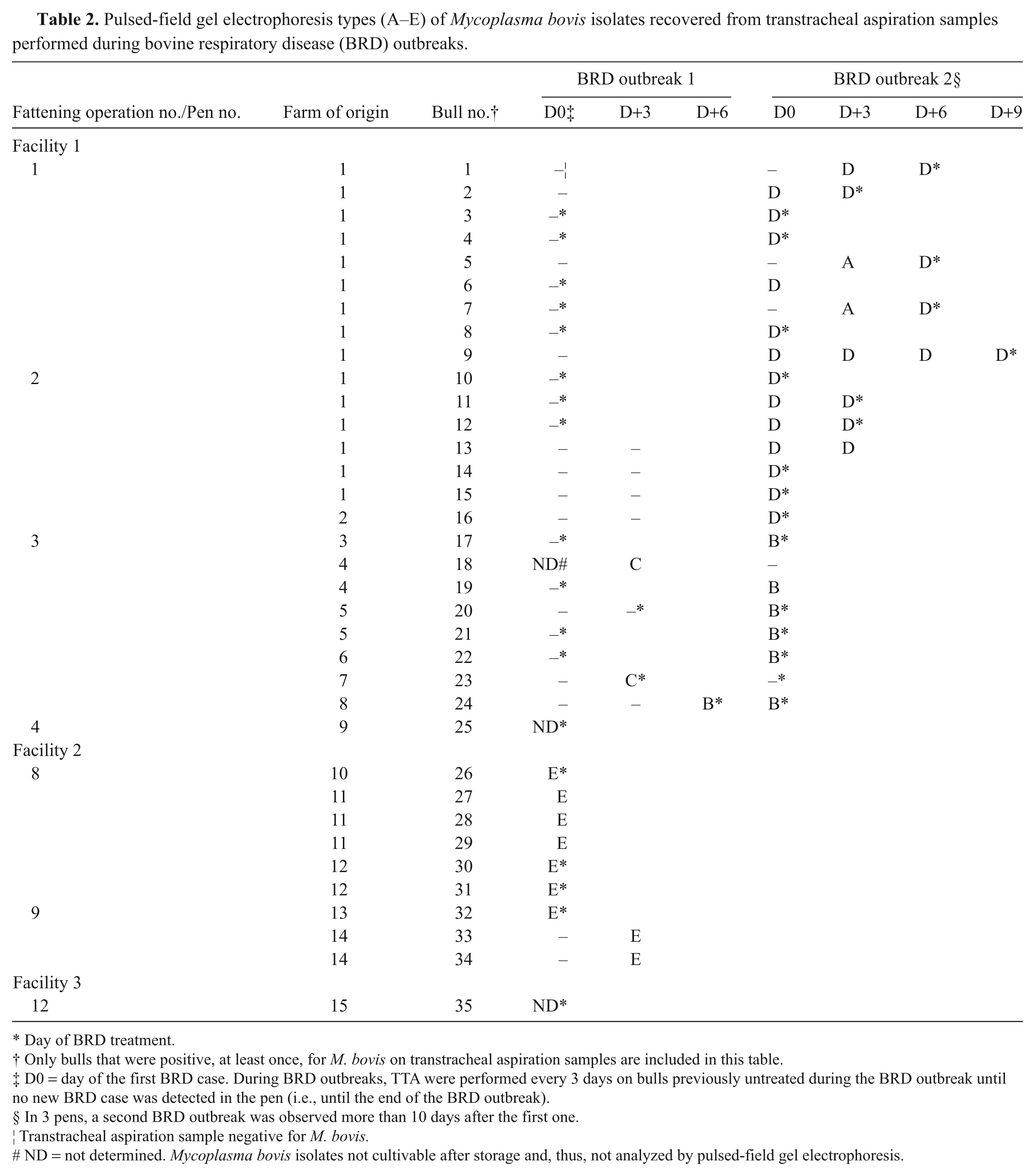
Day of BRD treatment.
Only bulls that were positive, at least once, for M. bovis on transtracheal aspiration samples are included in this table.
D0 = day of the first BRD case. During BRD outbreaks, TTA were performed every 3 days on bulls previously untreated during the BRD outbreak until no new BRD case was detected in the pen (i.e., until the end of the BRD outbreak).
In 3 pens, a second BRD outbreak was observed more than 10 days after the first one.
Transtracheal aspiration sample negative for M. bovis.
ND = not determined. Mycoplasma bovis isolates not cultivable after storage and, thus, not analyzed by pulsed-field gel electrophoresis.
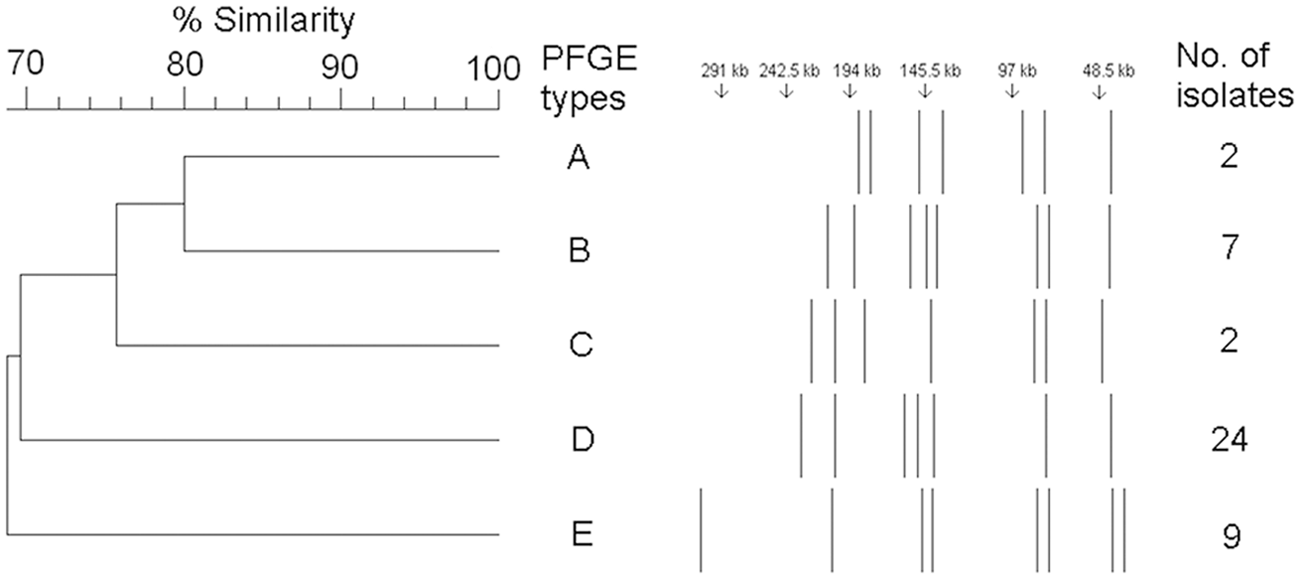
Dendrogram and schematic representation of pulsed-field gel electrophoresis (PFGE) fragments of 44 Mycoplasma bovis isolates with SmaI digestion. The dendrogram was created by unweighted pair group method with arithmetic mean cluster analysis using Dice coefficients of similarity with 1.0% optimization and 2.0% position tolerance settings. Each PFGE type was arbitrarily identified by a letter (A–E).
A single PFGE main type (i.e., clone) was recovered during 4 of the 6 BRD outbreaks where at least 2 bulls were positive for M. bovis (Table 2). During the 2 remaining BRD outbreaks, which occurred in pens.1 and 3, 2 different PFGE main types were recovered from positive bulls. However, it should be noted that, in pen no. 1, even though 2 PFGE main types (A and D) were present at the beginning of the BRD outbreak, only 1 main type (D) was recovered from all the infected bulls at the end of the outbreak. Similarly, in pen no. 3, even though 2 PFGE main types (C and B) were recovered from 3 positive bulls during the first BRD outbreak which occurred in this pen, only 1 of these 2 PFGE main types (B) was recovered from the 5 newly infected bulls observed during the second BRD outbreak, which occurred 10 days later.
Identical PFGE main types were shared only between groups of bulls that arrived on the same date in the fattening operations (i.e., same truckload). Indeed, no PFGE main type was shared between groups that arrived at different dates at the same fattening operation or between groups fattened in different operations (Table 2).
The current study provides new insights on the transmission dynamics of M. bovis in bulls newly received at a fattening operation. The PFGE analysis revealed that, even though bulls came from multiple origins, a single “dominant” clone of M. bovis was present within a pen during BRD outbreaks with high prevalence of M. bovis infection. This finding is in agreement with a previous study 1 that also described the clonal spread of only 1 “epidemic” clone of M. bovis per BRD outbreak in veal calves. Such a result indicates that, even if M. bovis can recrudesce from carriers after stressful events 5 such as transportation and commingling, the increased prevalence of M. bovis pulmonary infection observed during BRD outbreaks that are early occurring in the feeding period seems primarily due to the horizontal transmission of only 1 clone among cattle.
It is interesting to note that, in the present study, some M. bovis clones seemed more contagious than others. Indeed, in pen no. 3, 2 different clones were present in the lower respiratory tract of bulls during the first days after entrance and only 1 of these clones spread a few days later to all negative in-contact bulls housed in the pen. Similarly, in pen no. 1, 2 clones were present at the beginning of the BRD outbreak and only 1 clone infected all the bulls at the end of the outbreak. This apparent difference in the ability of M. bovis clones to spread may explain why only 1 clone per pen was recovered during the M. bovis–associated BRD outbreaks. Further research is needed to investigate the factors that could explain this apparent difference of contagiousness between M. bovis clones observed in the present study.
The source of the M. bovis clones that spread among pen mates seems to be the newly acquired bulls (i.e., presence of M. bovis–infected bulls at entrance). The M. bovis isolates recovered from groups of bulls that arrived at different dates at the same fattening operation had PFGE patterns that differed by more than 4 bands indicating unrelated isolates using previously described criteria. 11 This finding is in agreement with previously published results 4 highlighting that frequent additions of new cattle to a herd increased the likelihood of identification of different strains of M. bovis in the population.
The presence of older bulls housed in neighboring pens at the fattening operations seems also to have provided a source of infection for the newly acquired cattle in the present study. Once established, infection of the respiratory tract by M. bovis is often persistent, 1 and chronically infected cattle can be a source of infection for in-contact susceptible animals. 5 Mycoplasma bovis–chronically infected cattle in neighboring pens were most likely the source of the M. bovis clone that spread in pens 1 and 2. In these 2 pens, no M. bovis was recovered from the lower respiratory tract of bulls during the first BRD outbreak that occurred few days after entrance, but the bacterium was recovered from 78% to 100% of bulls during the second BRD outbreak that occurred later during the fattening period. Therefore, it seems that M. bovis can be transmitted either to susceptible cattle by newly acquired cattle or older cattle housed in neighboring pens. In conclusion, the present study shows that a clonal transmission of M. bovis within pens of cattle could occur early during the fattening period, with the source of the M. bovis strain spreading among cattle being either the newly acquired cattle or the older cattle housed in neighboring pens.
Footnotes
a.
Resflor, MSD Animal Health, Angers, France.
b.
Centracath, Vygon SA, Ecouen, France.
c.
CHEF-DRIII system, Bio-Rad Laboratories, Hercules, CA.
d.
BioNumerics version 5.1, Applied Maths Inc., Austin, TX.
Declaration of conflicting interests
The author(s) declared no potential conflict of interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was partly funded by the French ministry of Agriculture under project MOZAE and partly funded by MSD Animal Health.
