Abstract
The precision of a
Keywords
Introduction
Oral fluid samples are increasingly used for the surveillance of pathogens in commercial swine operations in North America. For example, the specimen type “swine oral fluid” was entered into the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL; Ames, Iowa) information management system in February 2010. During the remainder of 2010, 10,329 oral fluid specimens were received for testing. This number increased to 32,544 in 2011. In the first 3 months of 2012, the ISU VDL received 13,226 oral fluid samples. Thus, if the current level of submissions is maintained, the laboratory will receive >52,000 oral fluid samples in 2012. The majority of oral fluid samples received at the ISU VDL are tested by polymerase chain reaction (PCR)-based assays for
In 2012, the first antibody assay for swine oral fluids became available. 5 Although oral fluid antibody assays are already widely available in human diagnostic medicine for a variety of pathogens, 6 this is the first application in swine diagnostic medicine. Therefore, the objective of the current study was to evaluate the repeatability and reproducibility of the PRRSV oral fluid enzyme-linked immunosorbent assay (ELISA) a among 12 laboratories that offer diagnostic services to swine producers and veterinarians.
Materials and methods
Experimental design
Oral fluid samples (
Oral fluid specimens
Oral fluid specimens tested (
Oral fluid samples of known PRRSV antibody status originated from pigs inoculated with a type 2 PRRSV modified-live virus vaccine b and collected on 0, 10, 15, 20, 28, 35, 41, 49, 56, 75, and 91 days post vaccination (DPV). The samples were used to create 13 samples that were run on every plate. Specifically, the day 0 sample was used as in-house negative control; the day 75 sample was used to create high (1:5), medium (1:10), and low (1:20) positive controls using the PRRSV ELISA kit a sample diluent. The remaining 9 samples (i.e., DPV 10, 15, 20, 28, 35, 41, 49, 56, and 91) were split into 3 aliquots and then placed within the sample set such that all 9 were run on every plate.
Oral fluid ELISA procedures
Oral fluid samples were tested on a commercial PRRSV ELISA using a protocol designed to detect PRRSV antibody in oral fluid.3,5 In phase 1, all 12 laboratories used the same kit lot (40959-W721). The 11 laboratories that participated in phase 2 used 5 different kit lots: 5 used lot 40959-EG288, 3 used lot 40959-EG233, and 1 laboratory each used lots 40949-W721, 40949-W791, and 40949-X861. Phase 1 and phase 2 were completed before the data were analyzed.
To perform the test, oral fluid samples were diluted 1:2 in dilution plates using the diluent provided with the test kit. Two hundred fifty microliters of diluted oral fluid was then transferred to the 96-well PRRSV antigen-coated plates. Negative and positive kit controls were diluted 1:30 using the kit diluent and 100 µl transferred to the assigned wells on the plates (i.e., A1 and B1 for negative controls; C1 and D1 for positive controls). Thereafter, plates were incubated for 16 hr at 4°C. Three laboratories used refrigerated incubators; all other laboratories used standard refrigerators. At the end of the 16-hr incubation period, the plates were washed 3 times with 400 μl of 1× kit wash solution. To detect bound antibody, reagents were brought to room temperature and then 100 μl of a solution containing appropriately diluted horseradish peroxidase–conjugated anti-swine immunoglobulin G (IgGFC) secondary antibody c was added to each well and incubated for 30 min at 22°C. Thereafter, plates were washed 3 times with wash solution, 100 µl of tetramethylbenzidine enzyme substrate solution was added to each well, and the plates incubated at 22°C for 15 min. After 15 min, 100 µl of kit stop solution was added to each well. The plates were read at 650 nm and measured as optical density (OD).
All laboratories provided results as both OD and sample-to-positive control (S/P) ratios. The S/P ratio was calculated as:
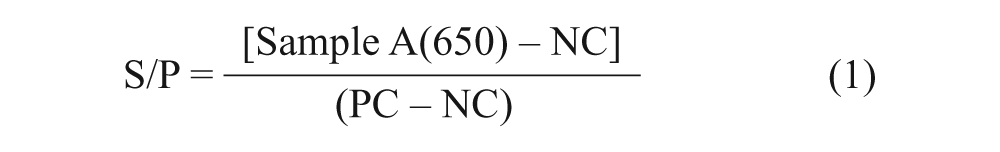
where NC and PC represented the mean OD of the 2 negative control wells and 2 positive control wells, respectively. The S/P ratios ≥ 0.40 were considered positive for PRRSV antibody. 5
Statistical analysis
The consistency of the PRRSV oral fluid ELISA was quantified by estimating reliability coefficients (ρ) for assay repeatability (within laboratory) and assay reproducibility (between laboratories) using the results from oral fluid samples of known (
For calculation of repeatability reliability coefficients, the model and associated parameters were based on

where τ
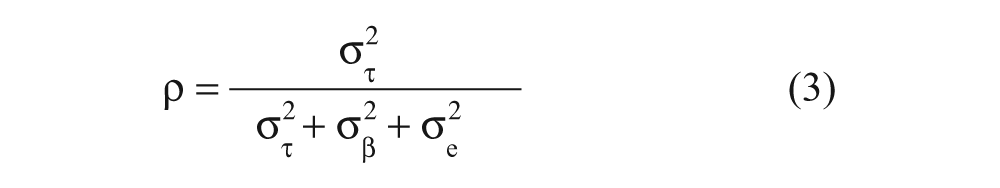
where
Reproducibility reliability coefficients were estimated for 3 conditions (case 1: samples of unknown status, case 2: samples of known status, and case 3: all oral fluid samples) using equation (4):
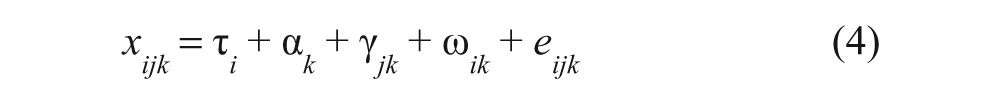
In equation (4),
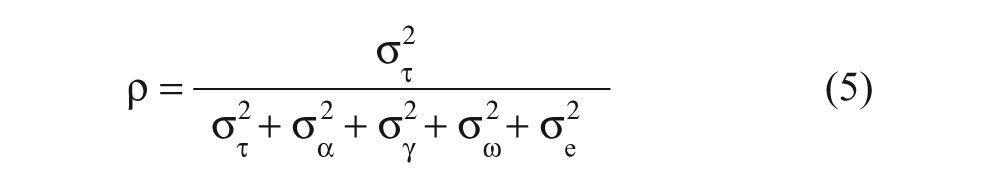
where the variance
Variance estimates for equations (2) and (4) were calculated from random effect models solved using commercial statistical software. d The S/P ratios within linear random effects models were analyzed using the GLIMMIX procedure. Oral fluid samples, laboratory, kit lot number, and interaction between oral fluid and laboratory were treated as random effects.
Results
A total of 3,156 PRRSV oral fluid ELISA results were produced by the 12 laboratories that participated in phase 1 (Fig. 1). Among these results, 1,581 (50.1%) were positive at a S/P cutoff of ≥0.40. In phase 2 (Fig. 2), the 11 participating laboratories produced 2,885 results with 1,438 positives. As illustrated in Figures 1 and 2, the dispersion of the results increased as the magnitude of the S/P response increased. The number of samples tested, mean ELISA S/P ratios, and S/P standard deviation are shown in Table 1 by laboratory. Among the samples of unknown PRRSV antibody status (
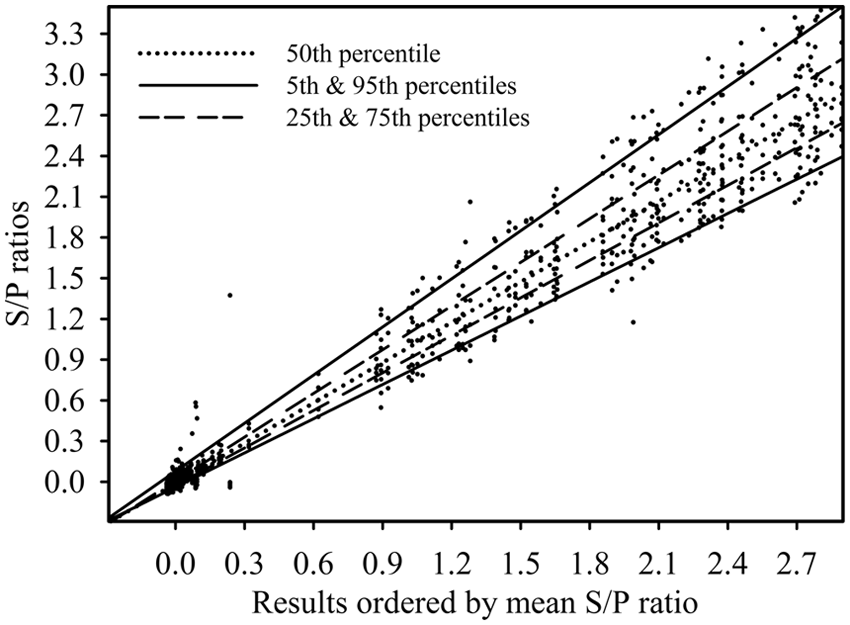
Phase 1. Results from 224 samples tested in each of 12 laboratories. All laboratories using the same
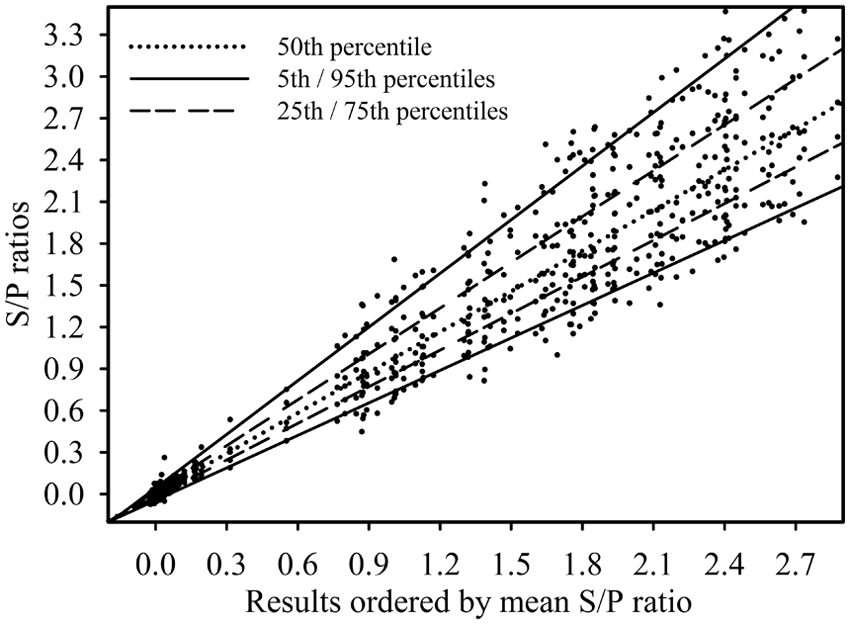
Phase 2. Results from 224 samples tested in each of 11 laboratories on a total of 5
Summary of results used to estimate test variance components in the
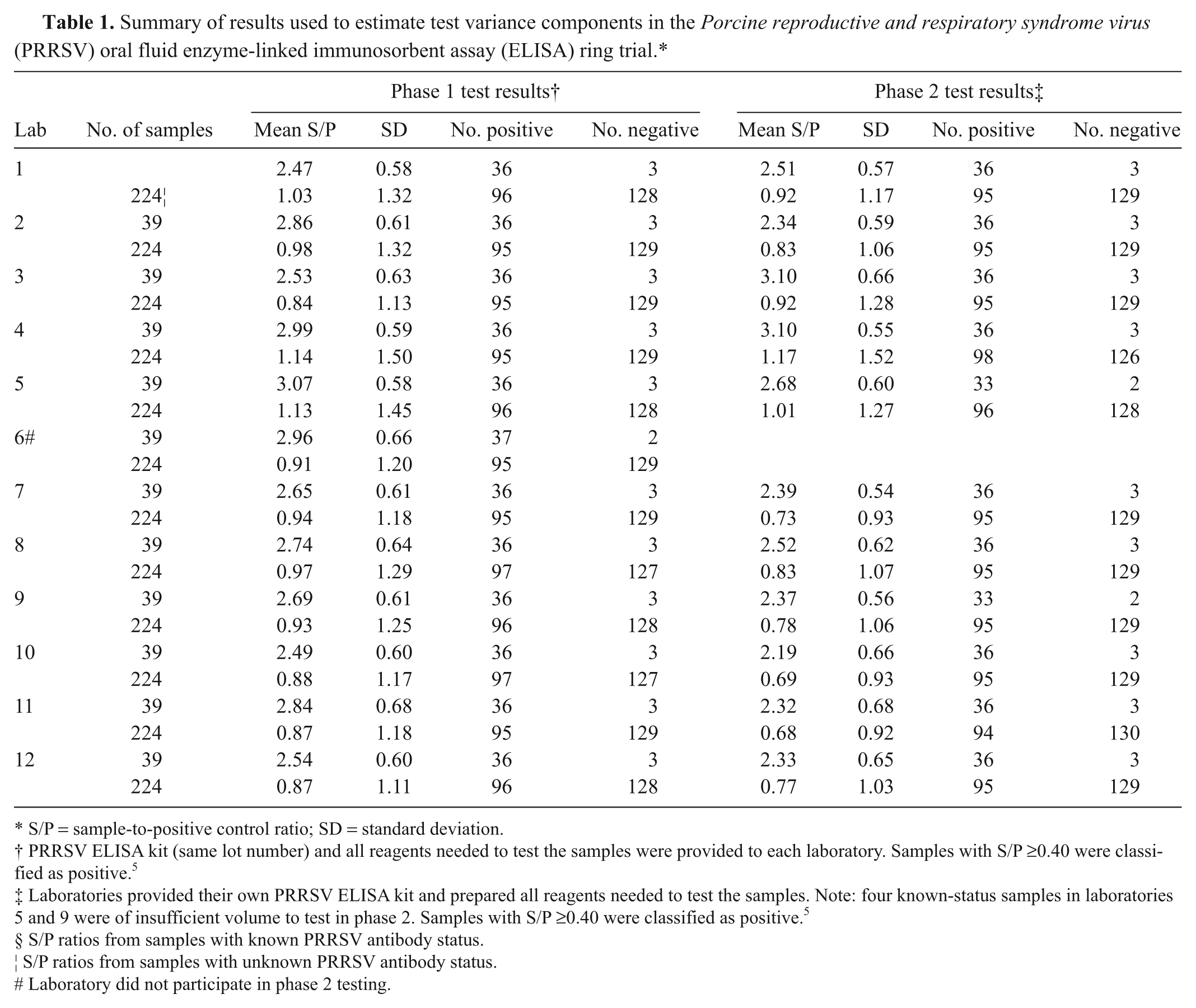
S/P = sample-to-positive control ratio; SD = standard deviation.
PRRSV ELISA kit (same lot number) and all reagents needed to test the samples were provided to each laboratory. Samples with S/P ≥0.40 were classified as positive. 5
Laboratories provided their own PRRSV ELISA kit and prepared all reagents needed to test the samples. Note: four known-status samples in laboratories 5 and 9 were of insufficient volume to test in phase 2. Samples with S/P ≥0.40 were classified as positive. 5
S/P ratios from samples with known PRRSV antibody status.
S/P ratios from samples with unknown PRRSV antibody status.
Laboratory did not participate in phase 2 testing.
Summary of
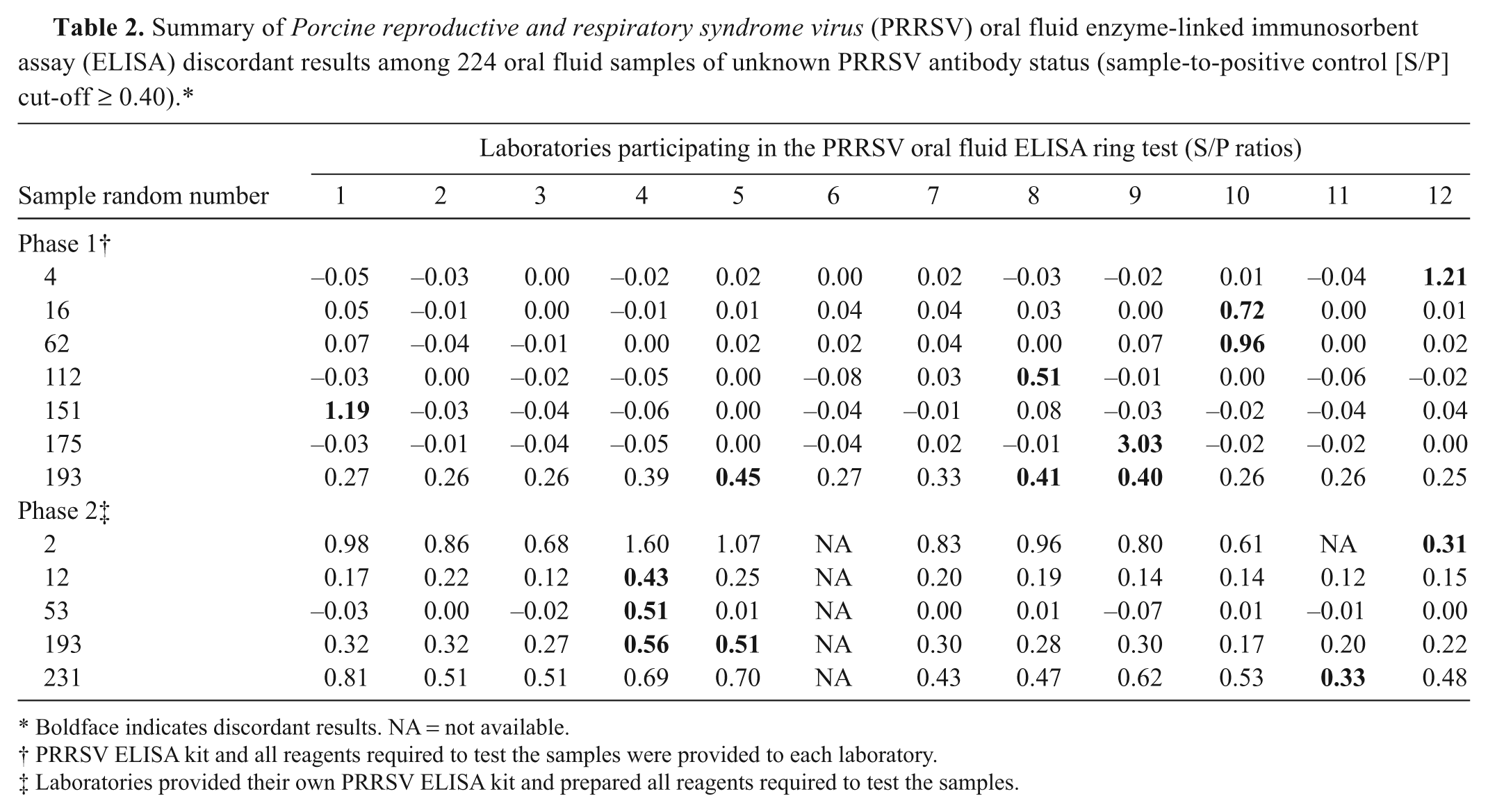
Boldface indicates discordant results. NA = not available.
PRRSV ELISA kit and all reagents required to test the samples were provided to each laboratory.
Laboratories provided their own PRRSV ELISA kit and prepared all reagents required to test the samples.
Repeatability (within laboratory) reliability coefficients calculated from linear random effects models are shown in Table 3. The within laboratory agreement ranged from 0.942 to 0.997 in phase 1 and from 0.953 to 0.998 in phase 2. Laboratory 6 reported 1 false-positive result in phase 1, which resulted in a within laboratory reliability coefficient of 0.724 for that laboratory. No statistically significant difference in repeatability was detected for any laboratory when comparing phase 1 versus phase 2 reliability coefficients.
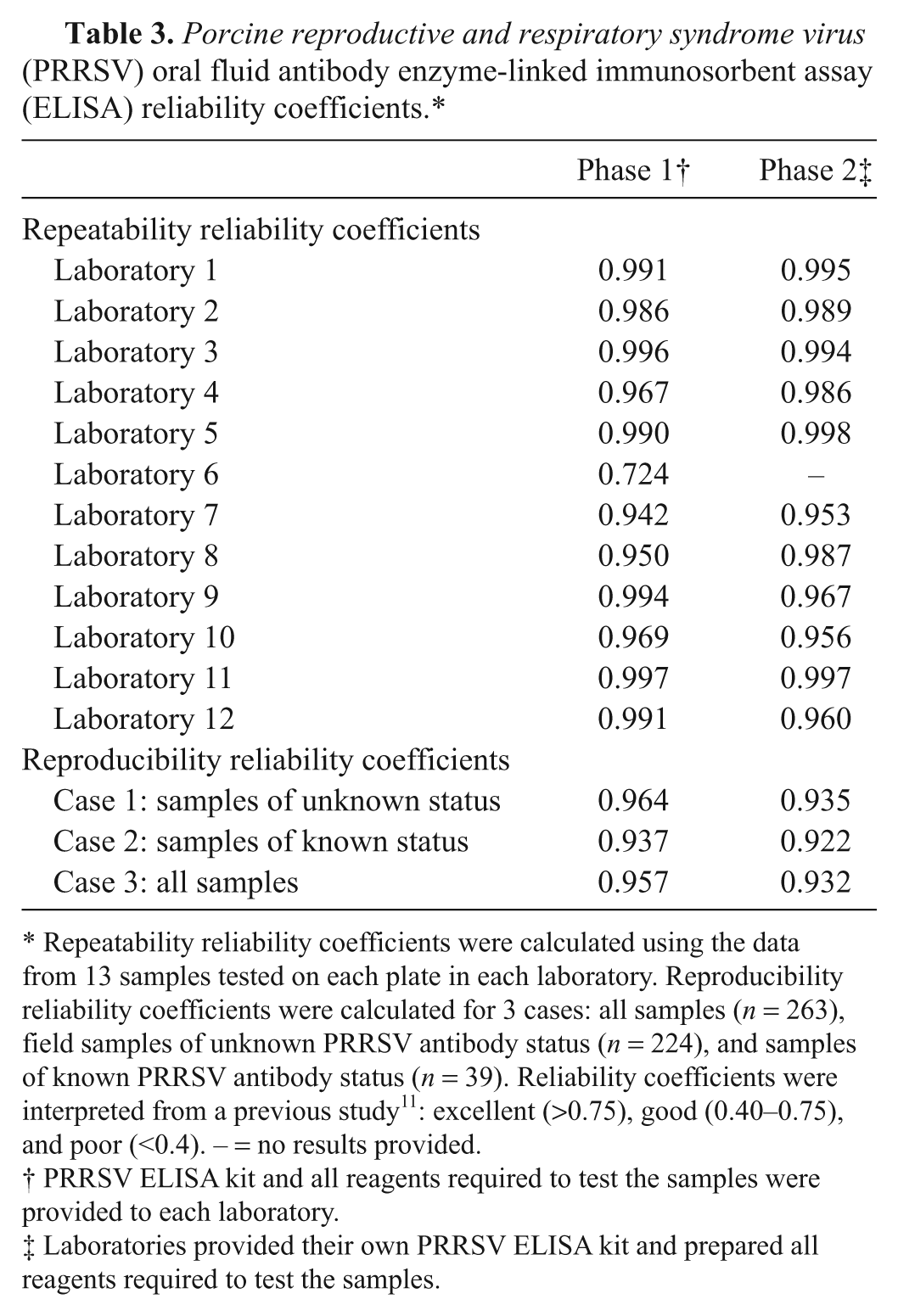
Repeatability reliability coefficients were calculated using the data from 13 samples tested on each plate in each laboratory. Reproducibility reliability coefficients were calculated for 3 cases: all samples (
PRRSV ELISA kit and all reagents required to test the samples were provided to each laboratory.
Laboratories provided their own PRRSV ELISA kit and prepared all reagents required to test the samples.
Reproducibility (between laboratories) reliability coefficients were calculated using results from 224 samples of unknown status and 39 samples of known status tested in all laboratories. As shown in Table 3, this allowed for the calculation of reproducibility reliability coefficients for 3 conditions: case 1—samples of unknown status (
Estimates of the variance components used to calculate reproducibility reliability coefficients for the

Variance components were estimated in a linear random effects model using the sample-to-positive control (S/P) ratio data from all oral fluid samples (
PRRSV ELISA kit and all reagents required to test the samples were provided to each laboratory.
Laboratories provided their own PRRSV ELISA kit and prepared all reagents required to test the samples.
Discussion
A recent analysis of the PRRSV oral fluid ELISA reported test sensitivity of 94.7% (95% confidence interval [CI]: 92.4, 96.5) and specificity of 100% (95% CI: 99.0, 100.0). 5 Further evaluation of this ELISA was motivated by the fact that the protocol differs significantly from the protocol for the PRRSV serum antibody ELISA. That is, in contrast to the serum antibody assay, the PRRSV oral fluid ELISA uses a sample volume of 250 μl (100 μl for serum), a 16-hr incubation period at 4°C (30 min at 22°C for serum), and requires the operator to purchase and correctly prepare the appropriate concentration of anti-swine IgGFC secondary antibody.3,5 To fully evaluate the precision of the assay, the study was conducted in a ring test involving various North American laboratories that were expected to offer this test to swine clientele on a routine basis.
Visual analysis of the cumulative results (Fig. 1) showed greater dispersion of S/P ratios in phase 2 (i.e., laboratories tested the samples on a total of 5 ELISA kit lots using reagents they had prepared), as compared with phase 1 (i.e., samples were tested on a single kit lot with pre-diluted reagents). However, as shown in Table 2, relatively few discordant qualitative results were seen either in phase 1 (7 discordant samples) or phase 2 (5 discordant samples). With one exception, discordant results were single events. The one exception was a sample (#193) that was reported positive in 3 out of the 12 laboratories in phase 1 and in 2 out of the 11 laboratories in phase 2. The frequency of discordant results for this sample can be explained by the fact that its mean S/P value lies close to the positive cutoff (S/P ≥ 0.40). Thus, small differences in quantitative measurements produced divergent categorical results.
Reliability coefficients were used as quantitative summary measures of agreement. By definition, the repeatability reliability coefficient is a measure of intra-laboratory assay agreement, and the reproducibility reliability coefficient is a measure of inter-laboratory agreement. 12 A previous study 11 has suggested that coefficients >0.75 indicate excellent reliability. The limitation of this approach is that reliability coefficients reflect both the assay and the distribution of the analyte in the samples tested. That is, given the same assay, reliability coefficient estimates will be affected as the concentration and/or prevalence of the analyte in the population of samples tested varies. 2
Repeatability reliability coefficients were calculated using 13 samples tested on each ELISA plate in every laboratory (Table 3). Coefficients ranged from 0.724 to 0.997 (phase 1) and from 0.953 to 0.998 (phase 2). No statistically significant differences were detected in phase 1 versus phase 2 repeatability reliability coefficients in the 11 laboratories participating in both phases. Based on the reliability coefficient criteria described previously, 11 the results were shown to be highly repeatable in the participating laboratories.
Reproducibility reliability coefficients were calculated for 3 cases (Table 3): samples of unknown PRRSV antibody status (case 1,
Cumulatively, the current study showed that a commercial serum antibody ELISA adapted to oral fluid samples was highly reproducible within and between laboratories responsible for swine health testing. The results support the routine use of this test in laboratories providing diagnostic service to pig producers. Thus, herd monitoring based on oral fluid antibody testing could be one part of a PRRSV control and/or elimination program. The successful ring trial of a commercial serum antibody ELISA to the oral fluid diagnostic suggests that antibody assays for other pathogens could also be modified to this approach.
Footnotes
a.
HerdChek PRRS X3 Ab Test (lot no. 40959-W721), IDEXX Laboratories Inc., Westbrook ME.
b.
Ingelvac PRRS MLV, Boehringer Ingelheim Vetmedica Inc., St. Joseph, MO.
c.
Pig IgG-Fc Antibody (catalog no. A100-104P), Bethyl Laboratories Inc., Montgomery, TX.
d.
SAS version 9.2, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research authorship, and/or publication of this article: Authors A. Ballagi, A. Rice, and S. Lizano are employed by IDEXX Laboratories Inc. The remaining authors declare no conflicting interests with respect to their authorship or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported in part by Pork Checkoff funds distributed through the National Pork Board (PO Box 9114, Des Moines, IA 50306) and the PRRS CAP, USDA NIFA award 2008-55620-1932. HerdChek PRRS 3X ELISA kits were provided by IDEXX Laboratories Inc., Westbrook, ME.
