Abstract
The preferred method to determine the prevalence of bovine spongiform encephalopathy (BSE) in a country is to use immunology-based rapid-tests. Though these tests are validated to detect C-type BSE disease–associated prion (PrPsc), test-specific properties may influence their ability to detect H- and/or L-type BSE PrPsc, where both are atypical from C-type PrPsc. Molecular characterization shows atypical BSE PrPsc to have a different sensitivity to proteinase activity and different affinities for certain prion-specific antibodies. It is important to understand how atypical BSE PrPsc may affect the performance of rapid-tests, which are typically dependant on the use of specific proteases and antibodies. The current study used experimentally generated C-, H-, and L-type BSE PrPsc to evaluate 3 tests used in various national BSE surveillance programs: an immunochromatographic assay, a standard sandwich enzyme-linked immunosorbent assay (stndELISA), and a PrPsc-conformation–specific ELISA (confELISA). Although BSE PrPsc type had some effects on rapid-test performance, analytical sensitivity for atypical BSE PrPsc on all 3 platforms was not significantly compromised. When testing for atypical BSE PrPsc, the 3 tests were able to meet the same requirements that the European Food Safety Authority set when evaluating the tests for C-type BSE PrPsc.
Keywords
A major concern in food and animal feed production is the potential contamination with transmissible spongiform encephalopathies (TSE), fatal neurological diseases caused by a rogue isoform of the prion protein (PrPsc).9,22 The outbreak of bovine spongiform encephalopathy (BSE) in Europe during the 1980s and 1990s remains the most significant event of a prion disease in agriculture. In the United Kingdom, hundreds of thousands of cattle were confirmed as BSE positive, and millions of preclinical animals are thought to have entered the human food chain. 19 Classical BSE can be transmitted from bovine to humans in the form of variant Creutzfeldt-Jakob disease via consumption of PrPsc-infected bovine tissues.4,13,19 Since this link was made, many countries have implemented active surveillance for BSE as a means of disease control and determining prevalence.11,19
Targeted surveillance programs for BSE in North America test animals displaying symptoms of BSE, such as ataxia, aggression, unexplained weight loss, and abnormal postures,3,20 qualifying them as a high-risk group. A sample of the brainstem is removed and tested for the presence of PrPsc. This can be done using immunology-based methods such as enzyme-linked immunosorbent assay (ELISA) or by Western blot analysis. Millions of bovines have been tested worldwide, and a small fraction of the BSE-positive animals have been identified as “atypical” compared to the more commonly observed classical (C-type) BSE. Atypical BSE cases are defined by an abnormal PrPsc isoform, and have been found in Europe, Asia, and North America.2,6,10,16,17 The existence of different types of BSE challenges surveillance programs, as it is imperative to successfully detect all types with comparable sensitivity on standard testing platforms. Of concern is whether atypical BSE types behave the same on diagnostic tests validated using C-type BSE. One study involving scrapie cases in France and Germany found that some TSE rapid-tests failed to detect 53 cases of atypical scrapie out of 276 known positive samples. 5
In TSE testing, proteinase K (PK) is often used to destroy the cellular isoform of the prion protein (PrPc), leaving behind the PK-resistant, disease-associated isoform, PrPsc.6,9,11,18 It is mainly the result of PK digestion that has lead to the identification and differentiation of 3 types of BSE PrPsc isoforms: C-type, H-type (“high”), and L-type (“low”). “High” and “Low” refer to apparent molecular weights of the PK-digested PrPsc proteins, as observed on Western blot.7,9,10,15,16,21 H- and L-type are “atypical” from the more common and widespread C-type BSE.
H- and L-type BSE PrPsc also have increased sensitivity to PK-related degradation under certain conditions, as compared to C-type BSE PrPsc.10,14 Because they can be more easily degraded by PK, weak positive H- and L-type BSE samples have the potential to register as false negatives, which is of concern in PK-based tests. If atypical BSE PrPsc possesses different PK sensitivity, and the monomeric PrPsc subunit of the fibril is cleaved differently by PK, 1 this could also suggest differences in structure between BSE PrPsc types, and thus the performance of diagnostic tests reliant on PrPsc conformation or fibril structure may be negatively affected as well.
In the current study, standardized, homogeneous macerates from cattle experimentally inoculated with Canadian C-, H-, and L-type BSE cases (1 animal of each type) were tested with 3 commercial rapid-tests validated for BSE testing in Canada: an immunochromatographic assay (ICA), a a standard sandwich ELISA (stndELISA), b and a PrPsc-conformation–dependent sandwich ELISA (confELISA). c Both the ICA and stndELISA are reliant on PK degradation of PrPc, to select for PrPsc. In contrast, the confELISA uses a proprietary ligand to capture PrPsc by specific conformational recognition of PrPsc aggregates. Given the anomalistic nature of atypical BSE PrPsc, the present study aims to provide insight into its behavior on different rapid-tests, and how it may differ from C-type BSE PrPsc.
For a BSE rapid-test to comply with European Union requirements set for C-type BSE, the test should possess a sensitivity within a 2 log(10) range of the most sensitive BSE rapid-test available. The 2 log(10) range requirement for detection was set by the European Food Safety Authority (EFSA) in question no. EFSA-Q-2007-053, in accordance with EC no. 999/2001 regulations on monitoring BSE. The current study evaluated whether the 3 rapid-tests were within a 2 log(10) range of one another, in terms of detecting atypical BSE.
All work, including the inoculation of bovines, handling of infectious and noninfectious tissues and homogenates, and performance of rapid-tests was done in a biosafety level-3 facility. Bovines had been intracranially (IC) inoculated with BSE as previously described, 11 as per Canadian Council on Animal Care guidelines. Once clinical disease developed, animals were euthanized, and brain tissues from the frontal medulla, parietal medulla, thalamus, and colliculus were sampled from each animal. The BSE type was verified by Western blot (results not shown). The tissues were pooled into individual C-, H-, and L-type macerates, and used to prepare 50% (w/v) homogenates by weighing 0.5 g of macerate into every 1 ml of double-distilled H2O. Homogenates were then used as spiking material for each C-, H-, and L-type BSE dilution series for each test. All homogenization was done using a hand-held homogenizing tool with disposable mechanical shear tips. d
A BSE-negative macerate was prepared from brainstem tissues from 17 confirmed BSE-negative bovines. The macerate was used to prepare BSE-negative homogenates, which were then used as the negative diluent for the C-, H-, and L-type BSE serial dilutions. Although the PrPsc concentration changed throughout the serial dilutions, the total %w/v tissue composition of homogenate remained constant. Dilution sets for each rapid-test were made to comply with each individual rapid-test requirement in terms of %w/v tissue composition in the test homogenates, as well as in the correct buffer conditions, as done previously. 11
All tests were performed as per the package insert; results were read using the equipment and software recommended by each manufacturer. For the confELISA, the manufacturer provides an option of 3 protocols: “Standard,” “Short,” and “Ultra-Short.” In the current study, the “Short” protocol was followed. Due to the difficulty in quantifying true PrPsc concentration in any given sample, the units “mg−/mg+” were used in describing PrPsc quantity in any given sample (tissue-ratio). The tissue-ratio assigns a numerical value to the proportions of each sample comprised of normal tissue versus the proportion of tissue containing disease-associated PrPsc. This unit also coincides with a previous sensitivity analysis with C-type BSE. 11
Data from each of the 3 tests were fit with a saturative, one-site, total binding function, y = (Bmax × x)/(Kd + x) + (NS × x) + Background, e where Bmax is the maximum specific binding capacity of the test, Kd is the antibody–antigen dissociation constant, NS is the slope of any nonspecific binding, and Background is the baseline optical density in the test. e Because the current study evaluated BSE homogenates, likely differing in initial PrPsc concentration, it was difficult to attribute any differences in detection levels between BSE types to either test’s affinity for a specific BSE type, sample PrPsc concentration, or both. PrPsc distribution among different brain areas is a concern, as distribution is dependent on both incubation time and BSE type.7,12 Using a homogeneous macerate of different brain regions may dilute more positive regions with regions that are less positive.
To circumvent these issues, detection limits for each BSE type was treated as sample specific. Therefore, how each BSE-positive macerate performed on the confELISA, ICA, and stndELISA was compared relative only to itself. For this analysis, data points within the linear and proportional segments of each binding curve were taken 8 and plotted on a log10(x)–log10(y) graph, and fit with a nonlinear regression y = 10(slope×log(x)+yintercept), e in order to view the curve slopes, and calculate where the curve intersects each test’s cut-off value. Data points were weighted by 1/y2 to minimize data point relative distance to the fitted curve. Curve slopes for C-, H-, and L-type BSE were compared using a sum-of-squares F-test (P = 0.05) e to evaluate their identity to one another.
The H- and L-type BSE curves diluted out further than the C-type BSE curves; however, this does not mean that the tests are more sensitive for atypical BSE than C-type BSE. It should be emphasized that detection limits are sample specific, and the C-type BSE tissues used in the current study were less infected than tissues used in previous study, 11 resulting in lower detection thresholds in terms of mg−/mg+. This lower PrPsc concentration was also confirmed by IHC and Western blot analysis (results not shown), and is likely due to differences between individual animals, which were involved in different experiments. It is important to characterize the BSE PrPsc dilution curves by their slopes, because this better characterizes the dynamics of each test platform. Detection limitations are shown in Table 1, along with the range of analytical sensitivity among the rapid-tests for each BSE type. All BSE types remain within a 2 log(10) range of detection across test platforms.
Detection limits for each sample of bovine spongiform encephalopathy type on each rapid-test in the current study, defined by where each detection curve intersects each test’s cut-off value.*

H-type = high; C- type = classical; L-type = low. “High” and “Low” refer to apparent molecular weights of the PK-digested PrPsc proteins, as observed on Western blot.7,9,10,15,16,21 H- and L-type are “atypical” from the more common and widespread C-type bovine spongiform encephalopathy. ICA = immunochromatographic assay; stndELISA = standard sandwich enzyme-linked immunosorbent assay; confELISA = PrPsc-conformation–specific sandwich ELISA. Range of sensitivity (type vs. test) is the difference in log(10) between the most and least sensitive values (tests) for the respective BSE type. European Food Safety Authority regulations require a range no greater than a 2 log(10) difference between any one test and the most sensitive test available.
Of the 3 rapid-tests evaluated, the confELISA was the most sensitive for the 3 BSE types, which coincides with previous findings for C-type BSE. 11 The confELISA appeared to perform similarly with all 3 BSE types (Fig. 1). When plotted on the log(10) axes, C-, H-, and L-type BSEs had comparable slopes (Fig. 1B, Table 2). This suggests that C-, H-, and L-type BSE behave virtually the same on this test platform, and that the 3 BSE types would share a similar threshold for detection, provided initial PrPsc abundance is consistent among sample types. Because the confELISA platform is selective for PrPsc fibrils and does not use PK, it is also likely the best platform to quantify total PrPsc in a sample.
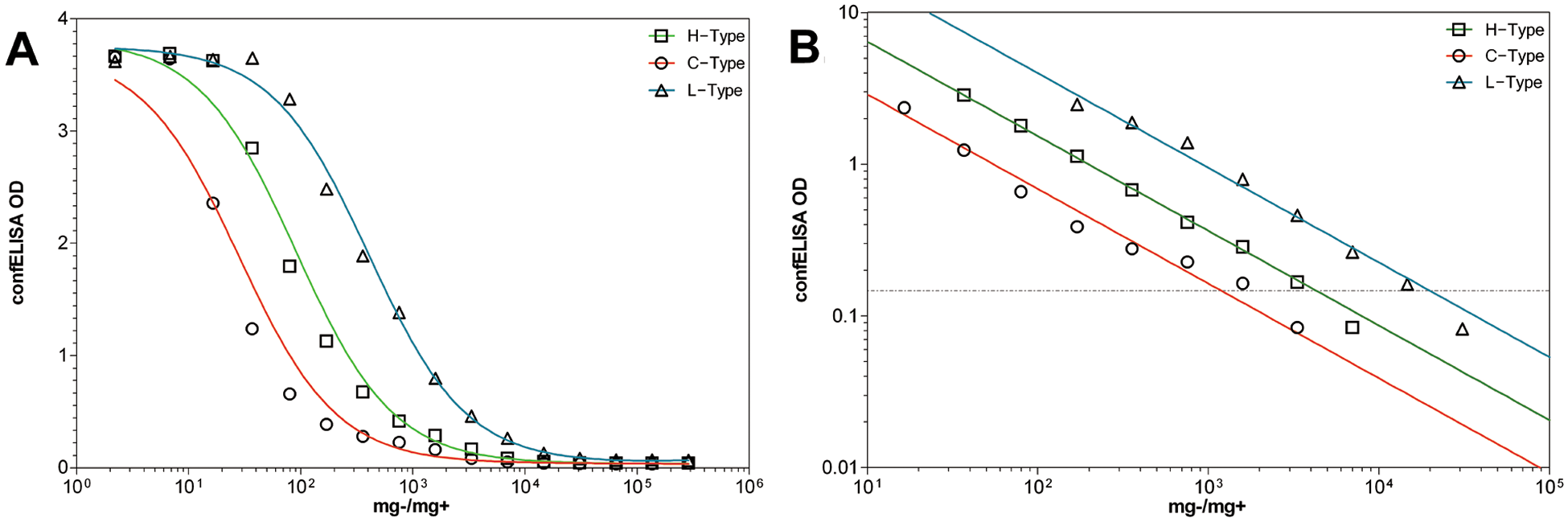
The appearance of experimentally generated Canadian H-, C-, and L-type bovine spongiform encephalopathy, in serial dilution, on the PrPsc-conformation–specific sandwich enzyme-linked immunosorbent assay (confELISA).
The goodness-of-fit values (R2) for the fitted one-site total binding curves, as well as the curves’ slope values (n = 3).*
H-type = high; C- type = classical; L-type = low. “High” and “Low” refer to apparent molecular weights of the PK-digested PrPsc proteins, as observed on Western blot.7,9,10,15,16,21 H- and L-type are “atypical” from the more common and widespread C-type bovine spongiform encephalopathy. ICA = immunochromatographic assay; stndELISA = standard sandwich enzyme-linked immunosorbent assay; confELISA = PrPsc-conformation–specific sandwich ELISA. Numbers in parentheses are R2.
In terms of the ICA, the F-test preferred a model where the 3 BSE types share a similar slope (Fig. 2, Table 2). That being mentioned, the range of detection between the ICA and the confELISA regarding L-type BSE was the widest between the tests, but did not exceeded 2 log(10) (Table 1).
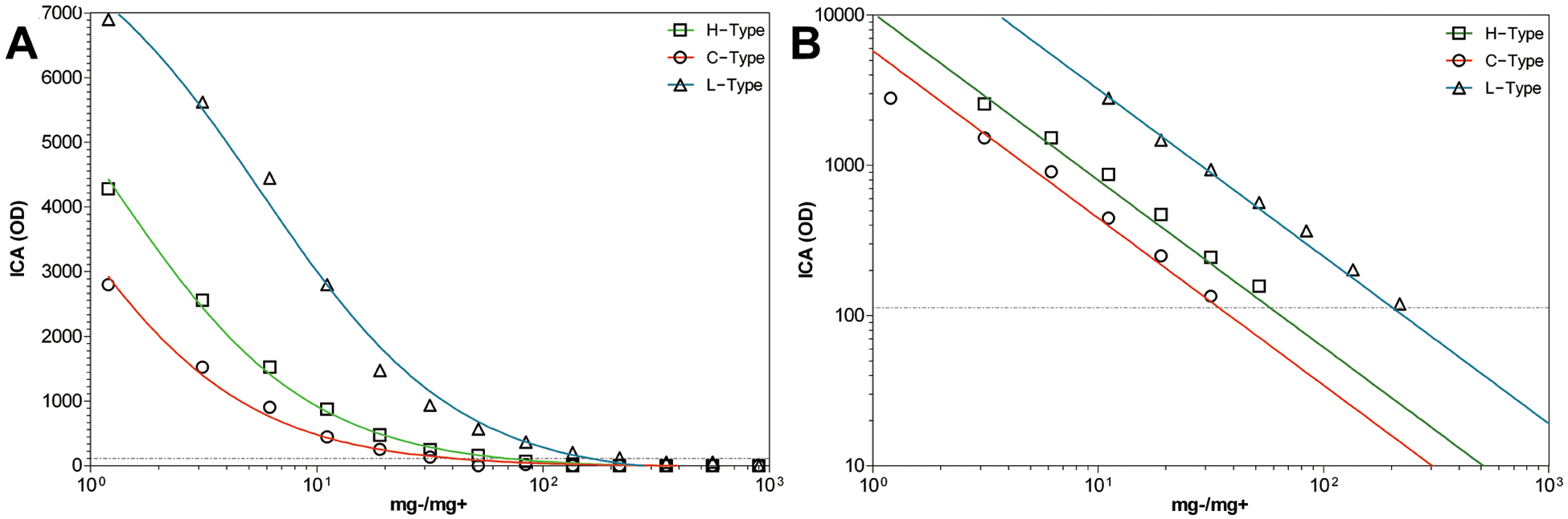
The appearance of experimentally generated Canadian H-, C-, and L-type bovine spongiform encephalopathy, in serial dilution, on the immunochromatographic assay (ICA).
The performance of the stndELISA was definitely affected by BSE type. Interestingly, H-type BSE, which was weaker than the L-type BSE sample set on the ICA and the confELISA, matched the L-type BSE as the strongest sample set in the stndELISA (Fig. 3). Detection of H-type BSE appeared to be enhanced on the stndELISA. However, it also generated the steepest binding curve slope (Table 2), suggesting that H-type BSE may be more sensitive to PK digestion conditions in the stndELISA than C- or L-type BSE. Nonetheless, H-type BSE performed better on the stndELISA than on the ICA, also using PK, in terms of analytical sensitivity. One possible explanation for this is that 1 of the 2 stndELISA antibodies targeted an epitope more toward the N-terminus of the PrPsc monomer, thereby compensating for the increased PK sensitivity with a higher antibody affinity. H-type PrPsc maintains 16 amino-acid residues toward the amino-terminus of the peptide after PK cleavage, which is in contrast to C- and L-type BSE.1,10,14
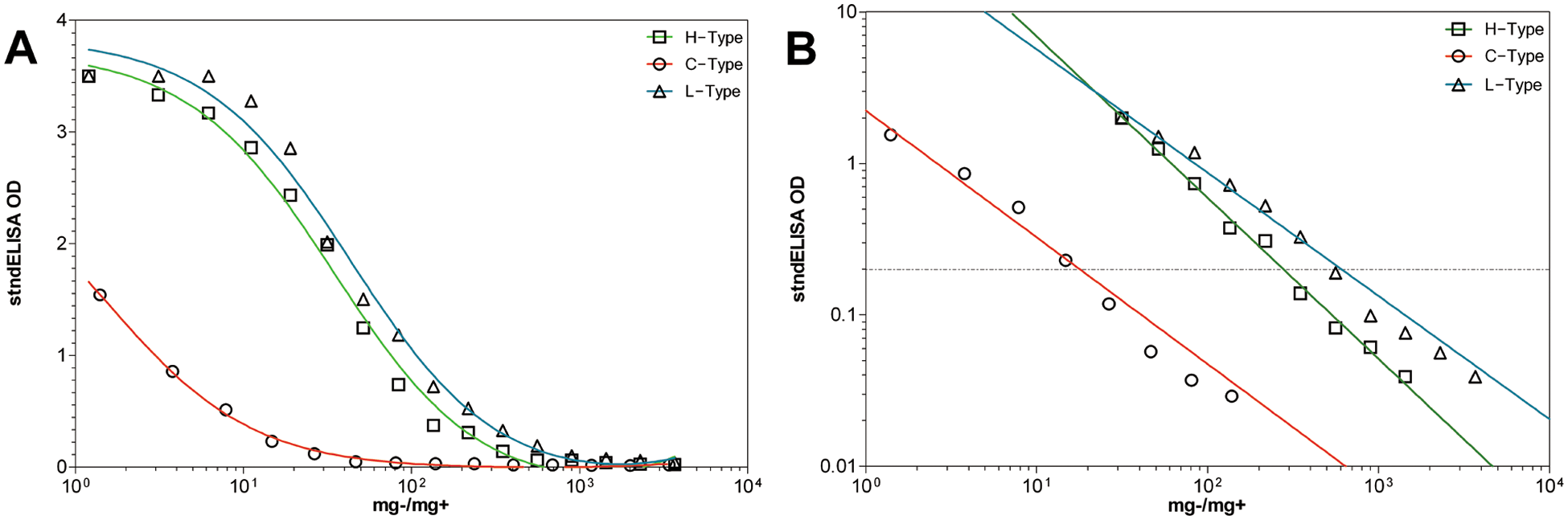
The appearance of experimentally generated Canadian H-, C-, and L-type bovine spongiform encephalopathy, in serial dilution, on the standard sandwich enzyme-linked immunosorbent assay (stndELISA).
In conclusion, the present study found that BSE PrPsc type did have relatively mild effects on test reactivity, but nothing that falls beyond the 2 log(10) rapid-test range indicated for C-type BSE by the EFSA. The current study also reinforces that these tests maintained a 2 log(10) range of sensitivity for C-type BSE (Table 1). H-type BSE displayed different performance characteristics on the stndELISA compared to those of C- and L-type BSE. C- and L-type BSE share comparable slopes, while H-type BSE has a steeper slope; however, when compared to the other PK-based test (i.e., the ICA), H-type BSE performed better on the stndELISA. This could be related to differences in antibody affinity in the various test kits.
It should be mentioned that the protocol of the stndELISA has been changed by the manufacturer to a new “Short Assay Protocol” which was used in this study. While the short assay protocol resulted in a reduced sensitivity for C-type BSE compared to previous results using the original protocol, 11 the stndELISA remains within the acceptable range of 2 log(10) range of the confELISA, being the most sensitive for C-type BSE.
Despite these differences, neither the atypical H- or L-type BSE seemed to be significantly compromised on any of the 3 tests in terms of test sensitivity range. The results indicate that the 3 rapid-tests evaluated are suitable for detecting atypical BSE within the allotted sensitivity range set out by the EFSA for C-type BSE.
Footnotes
a.
Bovine Spongiform Encephalopathy (BSE) Antigen Test Kit, Immunochromatographic Assay, Prionics-Check PrioSTRIP, V3.0; Prionics AG, Schlieren-Zurich, Switzerland.
b.
TeSeE SAP Purification/Detection Kit User’s Manual, Bio-Rad Laboratories, Marnes-La-Coquette, France.
c.
Bovine Spongiform Encephalopathy Antigen Test Kit, EIA, V9.0, IDEXX Laboratories, Westbrook, ME.
d.
OMNI International, Kennesaw, GA.
e.
GraphPad Prism v5.04, GraphPad Software, La Jolla, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Alberta Livestock and Meat Agency (ALMA; grant no. 2010R043R), and the Alberta Prion Research Institute (APRI; grant no. 201000005).
