Abstract
Since 1987, when bovine spongiform encephalopathy (BSE) emerged as a novel disease in cattle, enormous efforts were undertaken to monitor and control the disease in ruminants worldwide. The driving force was its high economic impact, which resulted from trade restrictions and the loss of consumer confidence in beef products, the latter because BSE turned out to be a fatal zoonosis, causing variant Creutzfeldt—Jakob disease in human beings. The ban on meat and bone meal in livestock feed and the removal of specified risk materials from the food chain were the main measures to successfully prevent infection in cattle and to protect human beings from BSE exposure. However, although BSE is now under control, previously unknown, so-called atypical transmissible spongiform encephalopathies (TSEs) in cattle and small ruminants have been identified by enhanced disease surveillance. This report briefly reviews and summarizes the current level of knowledge on the spectrum of TSEs in cattle and small ruminants and addresses the question of the extent to which such atypical TSEs have an effect on disease surveillance and control strategies.
Keywords
Introduction
Transmissible spongiform encephalopathies (TSEs), also termed prion diseases, comprise scrapie in sheep and goats, chronic wasting disease (CWD) in North American cervids, and bovine spongiform encephalopathy (BSE) in bovids. Human TSEs include Creutzfeldt—Jakob disease (CJD), Gerstmann—Sträussler—Scheinker disease (GSS), fatal familial insomnia (FFI), and kuru (Table 1). All of these are transmissible, neurodegenerative disorders that lead inevitably to death after a long incubation period from months to decades. Morphologically, they are characterized by spongiform changes and astrocytic gliosis in the central nervous system (CNS). 37,185,191 Neuronal loss is observed in most human TSEs, but absent or only subtle in scrapie or BSE. 103 In addition, a pathologic conformer (PrPd[disease associated], alternatively referred to as PrPSc[scrapie]) of the host-encoded physiologic prion protein (PrPc[cellular]) accumulates in brains, and in some TSEs, in lymphoid tissues and other organs as well. 131 The mature prion protein spans approximately 230 amino acids and is partially glycosylated at 2 sites. In contrast to PrPc, a fraction of PrPd forms detergent-insoluble aggregates and reveals resistance to enzymatic proteolysis. In vitro, proteinase K (PK) completely degrades PrPc and the N-terminus of PrPd, whereas a PrPd core fragment resists this treatment and is designated PrPres[PK resistant]. The PrPd is regarded as the major, if not the only, component of the causative agent, designated prion. 139 The conversion of PrPc to PrPd is triggered either by the uptake of PrPd from an external source or results from spontaneous PrPd formation. Specific mutations of the PrP gene are considered to facilitate this conversion. According to the prion hypothesis, PrPd can impose its conformation on PrPc molecules, resulting in an autocatalytic amplification. 140 Therefore, prion diseases could be either of spontaneous (sporadic) origin, genetically determined (familial), or acquired (infectious).
Classical scrapie is a contagious disease and has been recognized as a clinical entity for more than 250 years. It is endemic in many countries and can cause important losses in affected small ruminant populations; however, it is not considered a zoonosis. The classical type of BSE (C-BSE) emerged as a novel disease in the United Kingdom in 1986. Comprehensive epidemiologic investigations identified ruminant-derived meat and bone meal (MBM), which was widely used as a supplement in concentrate feed, as the vehicle that recycled the agent in the cattle population. 188 The C-BSE epidemic spread to other countries by export of infected animals and contaminated MBM. Since then, it has been reported from most European countries, Japan, Israel, Canada, and the United States, where affected animals were again processed to MBM, which eventually was fed back to domestic cattle. Enormous efforts were undertaken to monitor and control the disease, especially when its major impact on public health became evident in that it was transmissible to human beings by ingestion of infectious tissues, causing a hitherto unknown new variant of CJD (vCJD). 35,95,189 The ban on MBM in livestock feed and the removal of specified risk materials from the food chain were the main measures to successfully prevent infection in cattle and to protect human beings from BSE exposure.
Transmissible spongiform encephalopathies in animals and human beings. *
BSE = bovine spongiform encephalopathy (C- = classical type, H- = high type, L- = low type); BASE = bovine amyloidotic spongiform encephalopathy; CWD = chronic wasting disease; FSE = feline spongiform encephalopathy; TME = transmissible mink encephalopathy; CJD = Creutzfeldt—Jakob disease; vCJD = variant CJD; GSS = Gerstmann—Sträussler—Scheinker disease; FFI = fatal familial insomnia.
For a long time, the surveillance for TSEs in livestock relied solely on the identification and reporting of clinically suspicious animals with subsequent neuropathologic postmortem confirmation of the disease (passive surveillance). In 1999, Switzerland was the first country that applied screening tests to non—BSE-suspect, healthy slaughtered, and perished cattle, which enabled infected cattle to be detected at a preclinical or unrecognized early clinical stage. 60 Such active surveillance has subsequently been adopted worldwide and has led to a significant increase in not only the number of cases but also often the identification of index cases in countries that had previously claimed to be free from BSE. 25 In Europe, active TSE surveillance has also been extended to small ruminants (2002) and cervids (2006). These species were presumably exposed to BSE-contaminated feed concentrates and have subsequently been proven to be susceptible to BSE upon experimental challenge. 54,75 Unlike cattle, they were considered to have the potential to transmit BSE under natural conditions (i.e., to represent a contagious disease) because this occurs in both scrapie and CWD, respectively. Although natural transmission of BSE has been demonstrated within an experimentally infected sheep flock, 11 this situation still remains to be addressed in goats and deer species. The main concern of natural transmission is that BSE could become established in these populations and would present challenges to traditional disease control approaches.
With the enhancement of surveillance in cattle and small ruminants, previously unnoticed, so-called atypical types of BSE and scrapie were identified. These differed in their phenotypic presentation from what had been observed in these diseases previously. The objective of the present review is to briefly compile the current level of knowledge on the spectrum of TSEs in cattle and small ruminants and to address the question of the extent to which such atypical TSEs have an effect on current disease surveillance and control strategies.
Diagnostic methods for TSE in ruminants
According to the guidelines of the World Organization of Animal Health (OIE), European Union (EU), and national regulations, the diagnosis of TSE in ruminants involves screening tests, confirmatory tests, and discriminatory tests. Although screening tests are applied in active surveillance programs, confirmatory tests are used for a final diagnosis in clinically suspicious animals that were identified in the passive surveillance stream and as secondary tests to confirm or reject screening test—positive samples. As standard screening and confirmatory tests do not allow for reliable differentiation between classical and atypical scrapie on the one hand and BSE in small ruminants on the other, specific discriminatory test formats have been developed to identify any BSE among TSE-positive small ruminants.
Histopathologic lesions and immunochemical characteristics of the prion protein
(PrPd) deposits in the central nervous system of classical and
atypical scrapie—affected small ruminants. In classical scrapie, lesions and
PrPd deposits are most prominent in the nuclei of the obex region in
the medulla oblongata:
Screening tests
The implementation of active surveillance programs for BSE and scrapie requires test formats for a rapid diagnosis (within a few hours) and a high sample throughput. In the past 10 years, a range of Western blot (WB) and enzyme-linked immunosorbent assay (ELISA) screening tests has been evaluated for this purpose by the European Commission and by national authorities. 65,68,70 Depending on the diagnostic performances, the tests were approved specifically for cattle or for small ruminants or for both. Most of them involve a PK digestion step before immunochemical PrPres detection to discriminate TSE-positive from TSE-negative animals.
Active surveillance is routinely performed on unfixed medulla oblongata samples that are removed postmortem from the skull of slaughtered and perished animals via the foramen magnum. In the laboratories, a subsample of the obex region is then processed further. Autolysis does not seem to directly affect the test performances, 47,127,149,176 but a critical point is identifying and correctly sampling the target structures in cases of advanced tissue disintegration.
Confirmatory tests
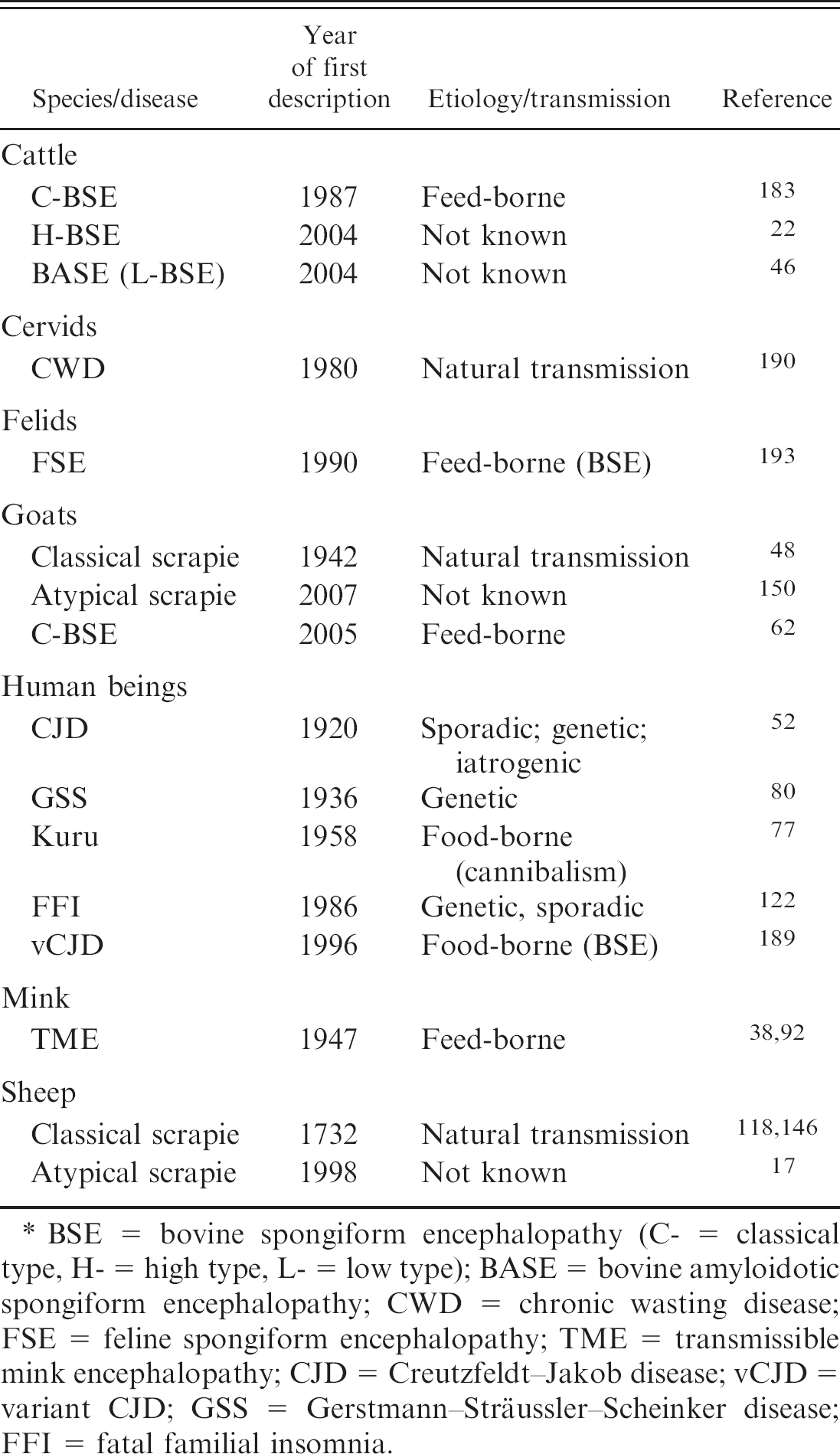
Histopathology. Historically, laboratory confirmation of clinical TSEs was routinely accomplished by histologic examination of the brain for disease-specific lesions. In affected animals, characteristic spongiform changes and neuronal vacuolation are most apparent in the medulla oblongata at the level of the obex. For scrapie, the dorsal motor nucleus of the vagus nerve (DMNV; Fig. 1A), 191 and for BSE, the solitary tract nucleus, the spinal tract nucleus of the trigeminal nerve, and the DMNV 180 are consistently affected and, therefore, the most suitable diagnostic target sites. However, histologic examination may be hampered by tissue autolysis.
With growing knowledge of the involvement of PrPd in the disease pathogenesis, the availability of appropriate antibodies, and the increasing efficiency of the detection methods, immunochemical procedures for PrPd detection in situ by immunohistochemistry (IHC) and in vitro by WB are increasingly being applied. Currently, histopathology is no longer the method of choice for TSE confirmation. However, it is important to be aware of TSE characteristic lesions to identify any cases in routine histopathologic examination of cattle brains submitted for reasons other than as a TSE suspect.
Immunohistochemistry. The IHC techniques allow for specific detection of PrPd deposits in formalin-fixed, paraffin-embedded tissues. This can only be achieved by stringent denaturing treatments of the tissue sections before antigen detection (e.g., by formic acid treatment and hydrated autoclaving, which suppress PrPc labeling and enhance specific labeling of PrPd). Similar to histopathologic lesions, PrPd deposits are most prominent in the above-mentioned obex nuclei for both classical scrapie (Fig. 1B) and BSE. The recognition of the presence of characteristic immunolabeling patterns in these target areas leads to confirmation of the disease. Immunohistochemistry has become the standard confirmatory test because its interpretation is less affected by tissue autolysis and because there is evidence of a higher level of diagnostic sensitivity compared with histologic examination alone. 145,148,192
Western blot. The detection of PrPres by WB requires unfixed (fresh or frozen) tissue samples. The tissues are homogenized and then subjected to limited proteolysis with PK. In WB analysis, PrPres is then detected immunochemically by prion protein—specific antibodies. The typical WB signature of PrPres in classical scrapie and BSE, as for most TSEs, is a 3-band pattern in the range of 18–30 kD of molecular mass, which represents unglycosylated, monoglycosylated, and diglycosylated PrPres moieties, respectively (Fig. 1G). The so-called OIE-confirmatory WB technique uses relatively high amounts of starting material (4 g) and PrPres concentration steps by lengthy ultracentrifugation before PK treatment. 164 Very recently, the OIE has approved the use of more convenient WB techniques for BSE confirmation in combination with screening tests. 192
Discriminatory tests
Although screening tests for small ruminant TSEs were also evaluated to detect BSE cases using brain material from experimentally BSE-infected sheep, 66 the tests do not differentiate between scrapie and BSE. Therefore, TSE-confirmed small ruminants are subjected to specified discriminatory testing. The molecular basis for these techniques is that, in BSE, a more C-terminal cleavage of PrPd occurs when treated with PK in vitro 96,163 and intracellularly by endogenous proteases in vivo. 107 Hence, BSE can be discriminated from most scrapie isolates using monoclonal antibodies that bind to the N-terminus of PrPres in either WB or IHC. However, because some scrapie isolates (termed CH1641-like) show features in WB analysis similar to BSE 10,96 and can only be differentiated from BSE by IHC, 104 such classification is preliminary, and samples in which BSE cannot be excluded still require confirmation in bioassays (see below). The discrimination of different types of BSE in cattle currently relies mainly on the PrPres signature in WB, which is described in detail below.
PrPd detection in the lymphoreticular system
In many classical scrapie cases and in CWD, PrPd is detectable in the lymphoreticular system (LRS) at a preclinical stage and before its accumulation in the CNS. 173 Thus, postmortem testing of LRS, in addition to nervous tissues, may increase the diagnostic sensitivity and assist in active surveillance and disease eradication on the herd level. It can also be used as an antemortem test on biopsies of tonsils, third eyelids, or rectoanal mucosa—associated lymphoid tissue, all of which contain lymphoid tissues and are, in principle, accessible in live animals. 81,82,135,147 In the EU, rapid tests for small ruminant TSEs have also been specifically validated on LRS tissues 68 but are, at present, not approved for that purpose. The main disadvantage of using biopsies in rapid tests is the accuracy of sampling and the uncertainty of sufficient lymphoid follicles being comprised in the specimen. In contrast, IHC techniques allow the identification of these structures by microscopic examination. Immunohistochemical protocols have also been established to discriminate BSE from classical scrapie in the LRS. 105,169
Disease phenotypes and prion pathogens
The phenotype of a TSE is defined by the clinical, neuropathologic, and biochemical characteristics displayed by an affected host. It results from the interaction of the host with the agent. Currently, the following main criteria are established to describe the phenotype of a TSE:
Clinical signs Incubation period Neuroanatomical distribution and intensity of lesions (lesion profiles) Neuroanatomical PrPd distribution and type of deposition Biochemical characteristics of PrPd
Transmissibility and host range Infectivity distribution in affected hosts
These criteria are also used in biological strain—typing experiments, which compare attack rates, incubation times, and lesion profiles of different TSE agents and isolates them upon transmission to panels of conventional, inbred mice lines. 32 Because of apparent species-barrier effects, not all TSEs transmit to such wild-type rodent models. The transmission may also be inefficient, requiring several passages to adapt the agent to the host to stabilize its phenotypic features. Transgenic mouse models overcome such limitations; these are mice that (over-) express heterologous PrPc on a murine, PrPc-ablated, genetic background. 89 The TSE isolates that exhibit different disease phenotypes in the same experimental model are then considered to represent distinct TSE agents, also referred to as prion strains. However, because the nature of the pathogen is still under debate, it remains difficult to develop a mechanistic understanding of how prion-strain characteristics are encoded. According to the protein-only hypothesis, which claims that the infectious agent consists only of PrPd without the involvement of any nucleic acids, strain characteristics are proposed to be enciphered in the higher-order structure of PrPd isoforms. 140
In recent years, strain-typing strategies have increasingly included biochemical criteria: the degree of resistance of PrPd to PK treatment, the site of PK cleavage, PrPres glycoform ratios, and conformational stability of PrPd. Novel techniques, such as infrared spectroscopy, 167 luminescent-conjugated polymers, 155 or organotypic slice culture and cell culture assays, 72,124 as well as in vitro PrPd amplification systems, 144 have become available and are promising tools that will help shed light on the enigma of prionstrain diversity in the near future.
Classical scrapie, the archetype of prion diseasesé
Classical scrapie (la tremblante [French], Traberkrankheit [German]) occurs naturally in sheep, goats, and mouflons. The infection spreads from ewes to offspring after parturition and spreads horizontally within and between flocks. Affected animals usually reach the clinical stage after an incubation period of 2–4 years. Behavioral changes, hyperesthesia, ataxia, and pruritus, the latter being manifested by intense itching, alopecia, and dermal abrasion, characterize the clinical picture in classical scrapie. Collapse and death sometimes occur rapidly within days of onset but, more often, after several weeks of progressing disease. 171
Different strains of the classical scrapie agent naturally occur in sheep and presumably also in goats. These strains were mainly characterized in the United Kingdom by biological-strain typing in mice, and approximately 20 strains could be discriminated based on differences in incubation times and lesion profiles. 33,58,59 However, the significance of that diversity for disease pathogenesis and control remains to be established.
The PrPd deposits are identified in the CNS first, in the DMNV but also in the intermediolateral column in the spinal cord, before they accumulate in other gray matter structures of the brain. 173,174 A broad spectrum of different morphologic PrPd deposition types has been observed in situ by IHC, and their neuroanatomical distribution and intensity can be scored to generate PrPd profiles that also serve for phenotype definition. 84 Western blot analysis of PrPres in nervous and lymphoid tissues reveals the 3-band pattern most commonly observed in TSEs. For most classical scrapie field isolates, the proportion of monoglycosylated and diglycosylated PrPres are approximately equal, 1,87,161 and epitope-mapping studies indicate that PK cleavage of PrPd occurs near amino acid position 85–90. The molecular mass of the unglycosylated PrPres is approximately 19–20 kD (Fig. 1G). 94,96 Some natural scrapie isolates reported from the United Kingdom and France revealed a molecular signature in WB different from the usual one and having a lower molecular mass of the unglycosylated moiety, similar to BSE. The isolates resembled the experimentally sheep-passaged scrapie isolate CH1641 and were thus termed CH1641-like. 119,163
The susceptibility of sheep to classical scrapie is highly associated with specific polymorphisms of the ovine PrP gene at codons 136, 154, and 171. Although PrP alleles encoding V136R154Q171 (PrPVRQ; International Union of Pure and Applied Chemistry single letter amino acid code) or PrPARQ are associated with disease, PrPARR alleles are considered to confer a high, although not complete, degree of resistance. 14,90 On this basis, some countries implemented breeding plans toward scrapie eradication aiming at increasing the proportion of scrapie-resistant animals in the population. 3 By contrast, PrP polymorphisms observed in goats differ from those in sheep, and a genetic basis for resistance to classical scrapie has not yet been established, although case-control studies in different countries identified some candidate genotypes for this purpose. 172
Atypical scrapie, a newly identified TSE in small ruminants
In 1998, sheep with unusual clinical, neuropathologic, and biochemical characteristics, when compared with the classical type of scrapie, were reported from Norway. Accordingly, this condition was termed Nor98. 17 By active TSE surveillance, similar cases (also referred to as atypical or discordant cases) have been identified since 2002, first in sheep from France and Germany and then in a series of other countries. 39,42,63,71,78,120,126,130,132,133,137 In some of those countries, the numbers of such cases even exceeded those of classical scrapie, whereas in others, such as Portugal, Sweden, Poland, and New Zealand, atypical cases were found without classical scrapie ever having been observed previously (Ministry of Agriculture and Forestry of New Zealand: 2009, Atypical scrapie/Nor 98 detection in single sheep from the national flock. Available at http://www.maf.govt.nz/mafnet/press/2009/281009-atypical-scrapie-detection.htm. Accessed on June 22, 2010). 78,133 Atypical and Nor98 isolates also showed discrepant results in screening tests that had been approved for small ruminant TSE surveillance at that time. Tests that used more stringent PK digestion conditions failed to detect such cases, whereas others with milder conditions clearly scored positive. 39
Defining the disease phenotype of atypical and Nor98 scrapie cases in the natural host is puzzling because, in active surveillance, it is difficult to collect reliable data on the clinical signs and brain tissues, apart from the caudal brainstem. Moreover, interpretation of the histopathologic lesions and PrPd deposition patterns is often compromised by advanced tissue autolysis. These animals were older than the average of classical scrapie—affected sheep, and the salient clinical sign, whenever reported, was ataxia, whereas pruritus and wool-loss were not observed. Immunochemical analysis by WB showed a PrPres banding pattern clearly distinct from that in classical scrapie, with a strongly labeled lower band of approximately 7 kD (Fig. 1G). Intriguingly, the obex was free from histopathologic lesions, and PrPd deposits were weak in the spinal tract nucleus of the trigeminal nerve and absent in the DMNV. In the Norwegian cases, for which whole brains were available, lesion and PrPd deposits were much more pronounced in the cerebellar cortex and the more rostral structures of the brain.
The definite proof that atypical and Nor98 scrapie cases do indeed involve a transmissible agent was provided when the disease was transmitted by intracerebral (i.c.) inoculation to mice that were transgenic for ovine PrP. 117 Transmissibility was also confirmed into sheep. 158 The neuropathologic and biochemical signature of Norwegian Nor98 and French atypical cases in the mice were indistinguishable from each other, but clearly different from classical scrapie. Hence, it was concluded that both were caused by the same newly identified TSE agent. Therefore, from this point onward, the term atypical scrapie will be used throughout, although this should not imply similarity between all such cases with respect to the phenotypic criteria listed above. Recently, more comprehensive data from clinically affected field cases and experimentally i.c.-infected sheep were reported. 114,128,130,158 Indeed, there is a degree of conformity to the PrPres WB signature, and it appears to be conserved among isolates when compared with the Norwegian cases. Independent studies from France, Germany, and Sweden came to the conclusion that the complex WB pattern in atypical scrapie is likely the result of overlapping signals from at least 2 different, truncated, glycosylated PrPres fragments. 5,88,110 However, depending on the antibodies applied and because of differences in PK digestion and electrophoresis conditions, the WB pattern in atypical scrapie cases, especially of the bands with a higher molecular mass, varied considerably among different laboratories, which makes a comparison of isolates difficult. There is also evidence of a certain degree of diversity in the neuroanatomic distribution of PrPd and histopathologic lesions among individual sheep, as well as between sheep and goats (Fig. 1C-F) when whole brains are examined. 16,130,143,150,186 Whether such differences are related to host factors (e.g., the PrP genotype and route of transmission) or to different strains of the agent is currently under investigation. By reviewing the available data, preliminary diagnostic criteria to discriminate atypical scrapie from small ruminant BSE and classical scrapie were defined in an opinion of the European Food Safety Authority, Biohazard Panel (Table 2). 67
Classical BSE in cattle and small ruminants
Classical BSE in cattle
Because the precise definition of the clinical aspects is a prerequisite toward recognizing and reporting diseases by means of passive surveillance, the clinical phenotype of C-BSE has been investigated in depth in the wake of the epidemic in the United Kingdom and also in Switzerland in the early 1990s. 27–29,183,187 Given the long incubation period of several years, the disease occurred in adult cattle with an average age of 5–6 years. Besides hypersensitivity to external stimuli and abnormal behavior (anxiety, restlessness, and sometimes aggression), most affected animals presented changes in locomotion. However, the signs varied markedly, and some were rather nonspecific, like poor general condition, chronic weight loss, and reduced milk production.
The baseline for the histopathologic phenotype of BSE in cattle has been defined by analyzing the neuroanatomic distribution and severity of vacuolar and spongiform lesions in brains of several hundreds of British BSE cases over a period of several years. The lesion profile was conserved in cattle with clinical BSE, indicating that the disease phenotype did not change during the course of the epidemic. 157,178 Outside Great Britain, lesion profiles covering the entire brain were described in BSE cases from France and Switzerland. 56,73 These cases were consistent with the British findings and supported the notion that the disease phenotype was similar, irrespective of its geographical occurrence.
Criteria for the classification of classical scrapie, atypical scrapie, and bovine spongiform encephalopathy (BSE) in small ruminants (adapted from an EFSA BIOHAZ-Panel opinion). 67
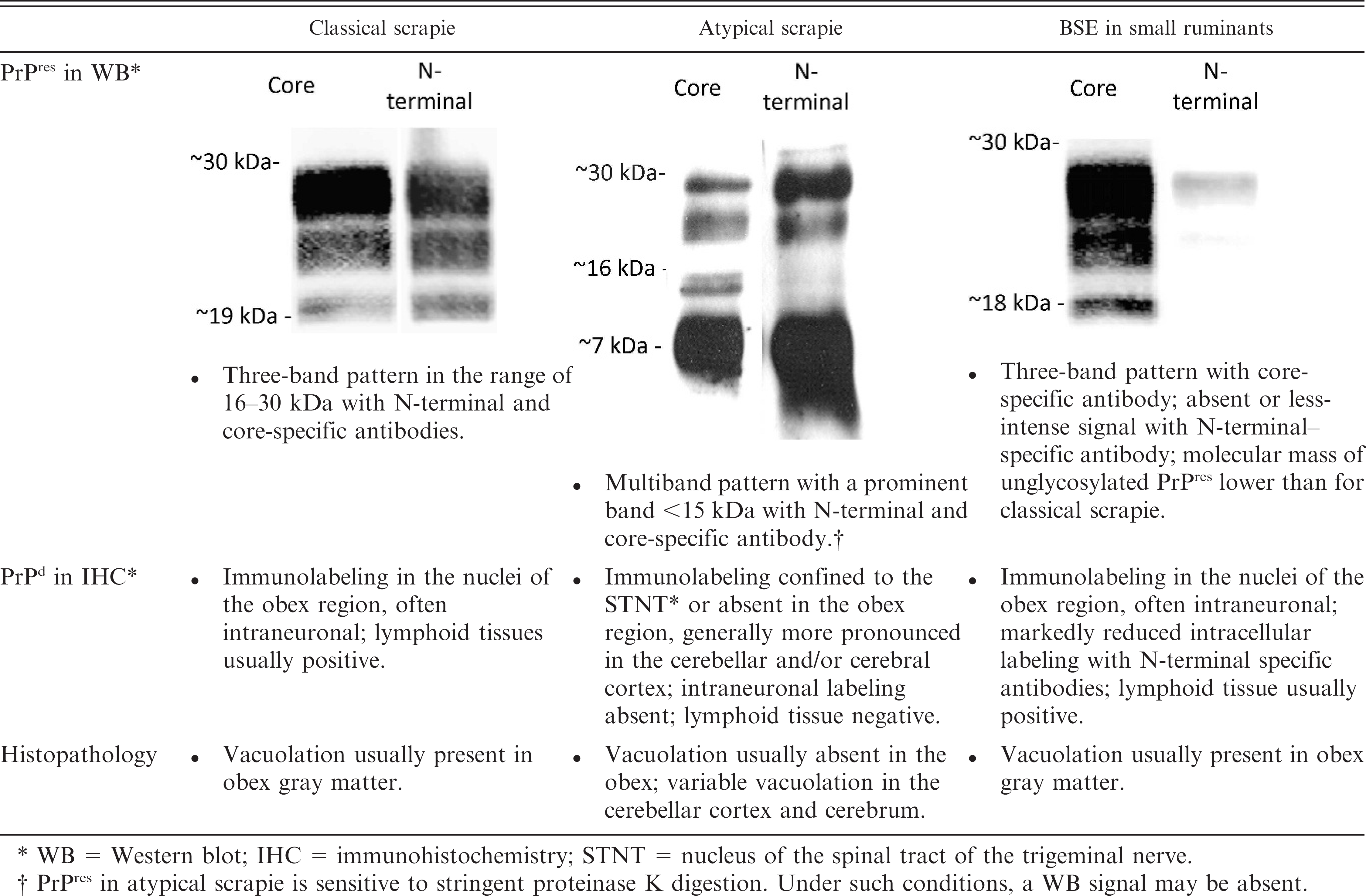
WB = Western blot; IHC = immunohistochemistry; STNT = nucleus of the spinal tract of the trigeminal nerve.
PrPres in atypical scrapie is sensitive to stringent proteinase K digestion. Under such conditions, a WB signal may be absent.
With the availability of in situ PrPd detection methods, it has been shown, although only in a limited number of cattle with clinical BSE, that the PrPd distribution throughout the neuraxis corresponded with the distribution pattern of the vacuolar and spongiform changes. 56,86 More comprehensive case numbers were analyzed systematically at the level of the obex. The target nuclei defined for the histopathologic diagnosis consistently showed distinctive immunolabeling patterns in IHC. 44,45,134 These nuclei were also the structures where PrPd was detectable in the absence of lesions in preclinical BSE cases that had been first identified through herdculling schemes. 145,148,160
The typical BSE WB profile has a 3-band pattern with a clearly more prominent diglycosylated PrPres band, as compared with the monoglycosylated band (Fig. 2A, C-type), 95 and was, therefore, distinct from those observed in most natural, classical scrapie cases. However, as the profound analysis of the molecular PrPd characteristics has not been part of routine WB confirmatory tests in BSE-affected cattle until recently, the extent of variation in these characteristics among affected animals and among brain regions within the same individual has been poorly addressed. More precise investigations now underpin the understanding that molecular PrPres phenotype in classical BSE cases indeed appears well conserved 7,40,101 and independent of the brain region under investigation. 166
Biological strain typing by transmission of individual samples from BSE-affected cattle to inbred mouse lines has been accomplished for 12 cases from the United Kingdom 31,53 and for 2 cases from Switzerland (M. Bruce, unpublished data) and revealed a closely similar disease phenotype with respect to incubation times and lesions profiles.
Based on these findings, there has been no evidence in favor of a significant diversity in the phenotypic representation of BSE in cattle, and scientists believed that the disease was caused by a single prion strain. On the other hand, it must be emphasized that, for most BSE cases, only fragmentary information was available. It cannot not be excluded that a proportion of them differed to some degree from the commonly observed phenotype and remained unrecognized as such.
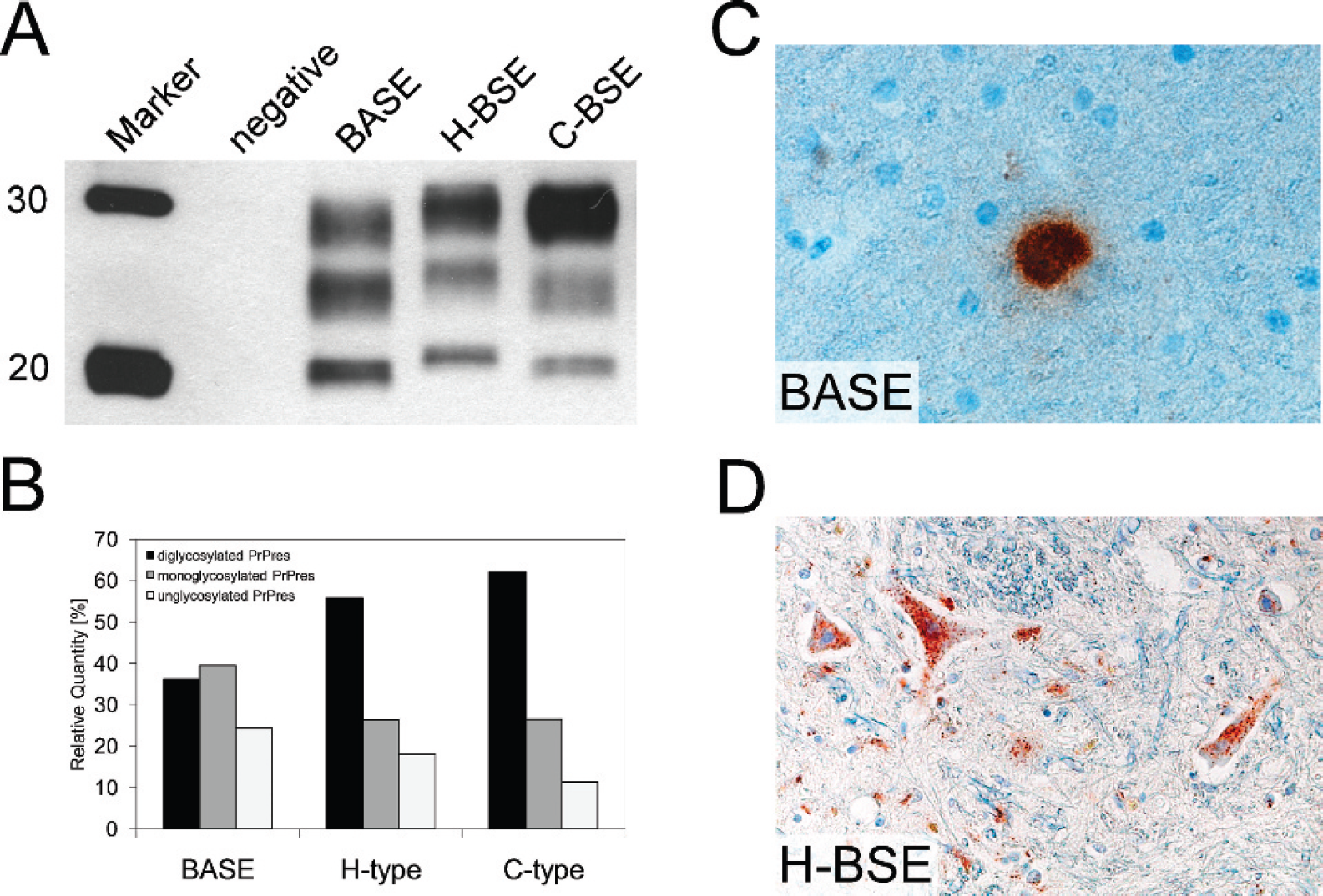
A, discrimination of different types of bovine spongiform encephalopathy (BSE).
Biochemical typing of PrPres by Western blot reveals a slightly
higher molecular mass of PrPres in high-type BSE (H-BSE) as compared
with classical BSE (C-BSE) and bovine amyloidotic spongiform encephalopathy
(BASE). A BSE-negative sample served as control. Molecular masses of a protein
marker are indicated on the left in kDa.
Classical BSE in small ruminants
The phenotype of C-BSE in small ruminants has been investigated in sheep and goats after experimental oral and parenteral challenge. In these studies, the clinical and histopathologic features were essentially indistinguishable from those in classical scrapie. 75,76,97,113 However, discrimination is possible by specific WB, 163,168 ELISA, 159 and IHC 83,106,107,169 procedures (Table 2) and by biological strain typing. 31,33
The scenario of BSE entering the sheep or goat population was the driving force for monitoring for TSEs in small ruminants. This became a realistic threat when a natural case was recognized in a French goat in 2003. 62 Thereafter, the EU increased TSE surveillance numbers in sheep and especially in goats, implementing a multistage testing strategy to identify any additional cases. In some countries, retrospective investigations on historic small-ruminant TSE cases have been undertaken. 34,93,177 By these means, another goat was identified in Scotland that showed immunohistochemical features indistinguishable from BSE. 106 This case is being investigated further. Despite these efforts, these 2 goats have remained the only cases confirmed so far worldwide, and no cases have been identified in sheep.
In many countries, the justification to control TSEs in small ruminants was to reduce the risk of BSE in sheep and goats. This was also the motivation for the implementation of genetic-based control strategies in sheep under the assumption that the genetic basis for classical scrapie resistance applied also for BSE because sheep with the PrPARR/ARR genotype appeared resistant to infection and showed no apparent peripheral distribution of infectivity or PrPd. 12,13,108 In different studies, it has been shown that PrPARR/ARR sheep are susceptible to BSE infection, 2,15,99 although discrepancies among studies may be, in part, dependent on the conditions of experimental challenge, and none of the studies accurately mimic natural exposure. Genetic breeding may, therefore, not be equally effective in the control of BSE in sheep as it is supposed to be in classical scrapie, and this rationale may in time require reevaluation.
Atypical BSE in cattle
When active surveillance for BSE in cattle was implemented throughout the EU in 2001 and later also in countries outside of Europe, WB procedures were increasingly used to further characterize suspect samples that were detected by the screening tests applied first. Almost simultaneously, BSE cases with aberrant electrophoretic PrPres profiles were identified in France, Italy, and Japan.
The French cases showed unglycosylated PrPres in the caudal brainstem and was of conspicuously higher molecular mass than classical BSE is. Antibodybinding studies revealed that this was due to more N-terminal PK cleavage of PrPd, compared with that in classical BSE, 22 similar to the situation in classical scrapie. Such isolates were later designated H-BSE, where H refers to the higher molecular mass of the PrPres. By contrast, in the Italian cases, the molecular mass of PrPres was slightly lower than classical BSE is and, more obvious, the monoglycosylated PrPres band was at least equally intense as the diglycosylated one. In these animals, whole brains were sampled, and the tissues collected were also suitable for IHC analysis. The morphologic PrPd deposition differed from those previously observed in BSE cases. Relatively few deposits were found in the obex region but many more occurred in the more rostral structure of the brain, namely, in the thalamus and the olfactory bulb. The deposits predominated in the form of amyloidlike plaques, which were also reported for TSEs in human beings, such as GSS, sCJD, kuru, and vCJD, but were not previously reported in cattle (Fig. 2C). 46 The Italian investigators designated this newly identified disease phenotype bovine amyloidotic spongiform encephalopathy (BASE).
In Japan, an unusually young animal (23 months) scored suspiciously in a BSE screening ELISA, and the sample also gave a positive result in a sensitive WB technique, with a banding pattern distinct from classical BSE control samples. 194 However, very little material was available, and other confirmatory methods could not be applied, with attempts to transmit the disease to bovine PrP transgenic mice failing. 195 Hence, the significance of the findings remains difficult to assess.
At the present time, the classification as C-BSE, H-BSE, and BASE relies mainly on the biochemical properties of PrPres. A systematic molecular typing strategy has recently been proposed and validated. 101 Besides the comparison of the electrophoretic profile, this approach also includes the analysis of reactivity patterns with different PrP-specific monoclonal antibodies in WB. In this experimental setup, H-type BSE reveals 1) an unglycosylated PrPres of a molecular mass of about 1.5 kD higher, compared with that in C-BSE (Fig. 2A); 2) reactivity with N-terminal antibodies that bind to the amino acids 101–107 of the bovine prion protein; and 3) an additional band migrating at about 10–12 kD and a complex banding pattern in the range of 18–30 kD with antibodies that bind to the PrP C-terminus. The latter, it has been proposed, 21 results from overlapping signals with an additional glycosylated C-terminal fragment of PrPres, which is missing in C-BSE and BASE. Differences in the molecular mass of PrPres between BASE and C-BSE are a less-robust criterion, and the identification of BASE relies primarily on the comparison of band intensities from the different PrPres glycoforms (Fig. 2B).
Reported cases of bovine amyloidotic spongiform encephalopathy (BASE) and high-type bovine spongiform encephalopathy (H-BSE) worldwide. 162
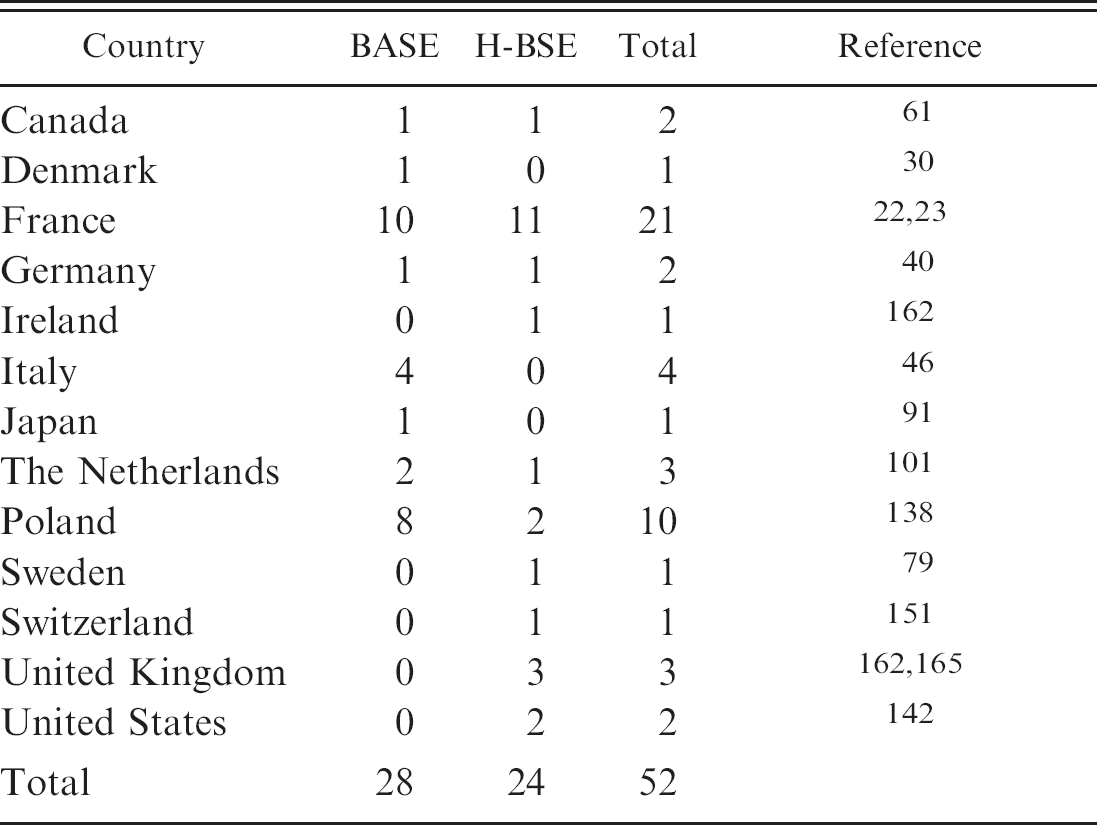
Since these first reports, some 50 cases similar to BASE (also referred to as L-BSE) or H-BSE have been recorded in a series of countries, either newly identified or by analyzing BSE cases retrospectively while focusing on their specific features (Table 3). All were linked by their falling within the age range of 8–20 years and were, therefore, older in comparison to the average age of 5–6 years in C-BSE. Except for a 19-year-old zebu bull (Bos indicus), which was identified as a clinical suspect in a zoo in Switzerland and later classified as having H-BSE, 151,166 these atypical cases have been detected exclusively by active surveillance. Similar to atypical scrapie, this limited the availability of reliable information on the clinical signs and prevented comprehensive examination of neuronal and extraneuronal pathology. Of all H-BSE cases, a complete brain was available only for the zebu, and despite distinct biochemical PrPres features, the histopathologic lesions and the morphologic PrPd depositions in the brain were similar to classical BSE (Fig. 2D).
The origin of atypical TSEs
In a series of BASE and H-BSE cases, the PrP coding gene has been sequenced to identify any genetic determinants for the disease, but, with the exception of 1 H-BSE case reported from the United States, differences from classical BSE cases were not found. 36,40,50 This particular U.S. animal carried a mutation, previously unknown in cattle, that encoded for lysine instead of glutamic acid at codon 211 (E211K). 141 A similar mutation at the homologous codon (E200K) in human beings is considered the most frequent cause of genetic CJD. 116 Hence, this animal potentially represents the first case of a genetic TSE in cattle, but a genetic etiology of H-BSE does not seem to be the rule. For all other H-BSE and BASE cases, no candidate genetic determinants have been identified.
Several scenarios have been put forward with respect to the etiology of atypical BSE in cattle and atypical scrapie in small ruminants. For many scientists, the most favored hypothesis is a spontaneous origin similar to sporadic prion diseases in human beings. Indeed, this is supported by the relative old age of the animals, their wide geographic distribution, the lack of epidemiologic links to other TSEs, and (at least shown for atypical scrapie) the lack of PrPd deposits in tissues other than the CNS. This implies that such cases were always present in small ruminant and cattle populations and remained unrecognized as such in the past. The diseases may also reflect a natural process of maturation or aging and, therefore, share some similarities to other protein-misfolding disorders in human beings. 49 Alternatively, if these TSEs transmit naturally or by oral uptake of MBM, they may also result from yet unidentified intraspecies or interspecies transmission events or from being coupled with host factors (e.g., the age at the time of exposure) that affect the phenotypic representation.
Disease surveillance and the implications of atypical TSEs
The efficiency of disease surveillance regimes depends on several factors. Although passive surveillance requires recognition of the clinical signs and a high disease awareness and motivation of farmers and veterinarians to notify cases, key points in active surveillance are the definition of the target population, the type of sample to be analyzed, and the performance of the screening tests applied. In the end, for both surveillance streams, powerful laboratory diagnostic procedures are indispensible for confirming or rejecting a TSE suspicion and also for discriminating atypical from classical forms of the diseases.
Passive surveillance
Only a few of the atypical TSE cases has been identified upon clinical suspicion, and passive surveillance appears inefficient in detecting such animals. Different factors may contribute to this situation. First, little is still known on the pathogenesis and clinical aspects of these TSEs. Many of the cases appeared asymptomatic at the time of diagnosis (by active surveillance), and it remains to be established at which stage in the time-course of the disease affected animals present clinical signs and whether truly subclinical infections occur. Assuming the clinical signs differ from those in the classical disease, there is a risk that such cases are overlooked or misinterpreted. As a result, they may escape from passive surveillance. Second, the prevalence of atypical TSE types, especially those of BSE, appears very low. In France and Germany, it has been estimated that for H-BSE and BASE, the prevalence is as low as 1 case per 3 million adult cattle and may, therefore, lie below the detection limit of passive surveillance. 22,40 In contrast, for atypical scrapie, the prevalence was estimated to be much higher (1–25 cases per 10,000 adult animals). 74 However, it must be stressed that the economic value of individual sheep and goats is low, and for this reason, single diseased animals, in which the disease appears unrelated to any health problem on a herd level, are often not subject to veterinary intervention. Finally, underreporting of clinical TSE-suspicious animals may result from an intentional lack of compliance. Farmers and veterinarians may be afraid of severe social and economic consequences, although it should be stated that this point does not apply specifically to the atypical types and may also depend on compensation policies.
Active surveillance
Active surveillance schemes largely overcome passive surveillance limitations. On the other hand, they are costly and require a logistical infrastructure and means of tracking laboratory results back to the farm of origin and the individual animal. The traceability is ensured by individual animal identification and animal movement databases in many countries for cattle, but not so for sheep and goats. In cattle, the targeting of fallen stock and emergency slaughtered animals proved to be more efficient than mass screening of regularly (healthy) slaughtered cattle in detecting C-BSE. 60 Whether this also holds true for atypical scrapie is still under debate. Although the disease prevalence was calculated to be higher in fallen stock compared with regularly slaughtered animals in some countries, it was just the opposite in others. 57,74 The situation is even less clear for H-BSE and BASE in cattle. Although many of these cases were reported in fallen stock or emergency slaughtered animals, 23,138 statistical analysis of the prevalence in different surveillance streams are still missing. Active TSE surveillance targets adult animals. This makes sense because, in younger animals, the disease is usually not yet detectable. With the exception of the Japanese case mentioned above, atypical TSEs were only observed in relatively old animals. Based on the current epidemiologic situation, a risk assessment, and cost—benefit analysis, 69 the age limit for active BSE surveillance in cattle was increased to 48 months in some EU member countries. This still includes the age group considered at risk for atypical BSE, and therefore, this modification is not expected to result in an underestimation of case numbers. In contrast, introducing an upper age limit for active TSE surveillance may severely affect the observed incidence of such atypical TSEs.
Sampling
For the diagnosis of C-BSE, classical scrapie, and small-ruminant BSE, the obex region of the medulla oblongata can be sampled conveniently postmortem via the foramen magnum without the need to open the skull. In atypical scrapie, it is clearly documented that the cerebellar cortex or even more rostral structures demonstrate the most intense PrPd deposits, rather than the obex. This situation should result in modifications of the sampling and testing strategies, if the purpose is to identify as many atypical scrapie cases as possible. However, such an approach requires removal of the whole brain and is expensive and difficult if large numbers of samples are to be analyzed. European Union regulations now require, as a minimum, additional sampling of parts of the cerebellum, which can also be accomplished via the foramen magnum by using appropriate sampling devices (European Commission TSE Reference Laboratory: 2010, TSE strain characterization in small ruminants. Available at http://www.defra.gov.uk/vla/science/docs/sci_tse_rl_handbookv4jan10.pdf. Accessed on June 22, 2010). Notwithstanding the aberrant PrPd distribution in atypical scrapie, but as a compromise between costs and benefit, some screening test are approved to be used only on obex samples because of their relative high analytical sensitivity. 60 In addition, for classical sheep and goat scrapie, it has repeatedly been shown that many preclinical cases accumulate PrPd only in the LRS and not in the brain. 26,85 Current active surveillance schemes for classical and atypical scrapie on obex samples, therefore, always result in an underestimation of their true prevalence.
Substantially less is known about the neuroanatomical distribution of lesions and PrPd in H-BSE and BASE. The H-BSE—positive zebu showed no differences in these criteria compared with C-BSE, but in the 2 Italian BASE index cases, the rostral structures of the brain were predominantly affected. More baseline data are definitely needed to map best-suited diagnostic target structures for H-BSE and BASE, respectively.
Diagnostic tests
Diagnostic procedures are required to be fit-for-purpose. The purpose of TSE surveillance is primarily to estimate the prevalence of infection, to facilitate risk analysis, and to assist in the demonstration of the efficiency of control policies. The BSE screening tests have been intensively validated in Switzerland 24,145 and the EU before their approval (European Commission: 1999, The evaluation of tests for the diagnosis of transmissible spongiform encephalopathy in bovines. Available at http://ec.europa.eu/food/food/biosafety/bse/bse12_en.pdf. Accessed on June 22, 2010; European Commission: 2002, The evaluation of five rapid tests for the diagnosis of transmissible spongiform encephalopathy in bovines [2nd study]. Available at http://ec.europa.eu/food/food/biosafety/bse/bse42_en.pdf. Accessed on June 22, 2010). These validation studies comprised large sets of defined positive and C-BSE—negative bovine samples. Although some tests, in principle, proved able to detect H-BSE and BASE as well, as shown simply by both having been identified by active surveillance, the tests were not formally validated for that purpose, and it currently remains unclear how fit they are.
That such a situation may be challenging was obvious when screening tests, originally developed for cattle BSE, were applied, without being evaluated for this purpose, in active small-ruminant TSE surveillance in Europe in 2002. First, there were challenges to the confirmation of diagnoses because confirmatory tests had not been optimized for the detection of atypical scrapie. Soon, it also became clear that the capacity of some screening tests to detect atypical scrapie was poor, 39 which was at least partially related to the finding that PrPres in atypical scrapie was found to be less resistant to PK treatment. The poorly performing tests used more stringent PK conditions than the other tests. Consequently, the atypical scrapie incidences varied noticeably between countries, depending on the screening tests being applied. 74 However, this changed with small-ruminant TSE tests that were specifically validated for this purpose. 68 However, for these studies, only a few samples of experimental ovine BSE and atypical scrapie cases were available. Even more important, the studies only comprised sheep samples, and their performance to detect TSEs in goats still remains to be determined.
Laboratory quality control
To provide a high standard to the performance of laboratories and tests, most countries implemented rigorous, postapproval control procedures that included quality management requirements, kit-batch release controls, on-site inspections, and interlaboratory comparison trials. These procedures are indispensible, because false-negative and false-positive test results are highly undesirable. By these means, TSE screening tests became exemplary for the validation and control of laboratory diagnostic procedures in the veterinary field. 129,152,153 However, because of the lack of reference materials, these measures do not specifically address the performance of identifying atypical TSE cases in most countries.
Transmissibility of different bovine spongiform encephalopathy (BSE) isolates to transgenic and wild-type (wt) mice by the intracerebral route. *
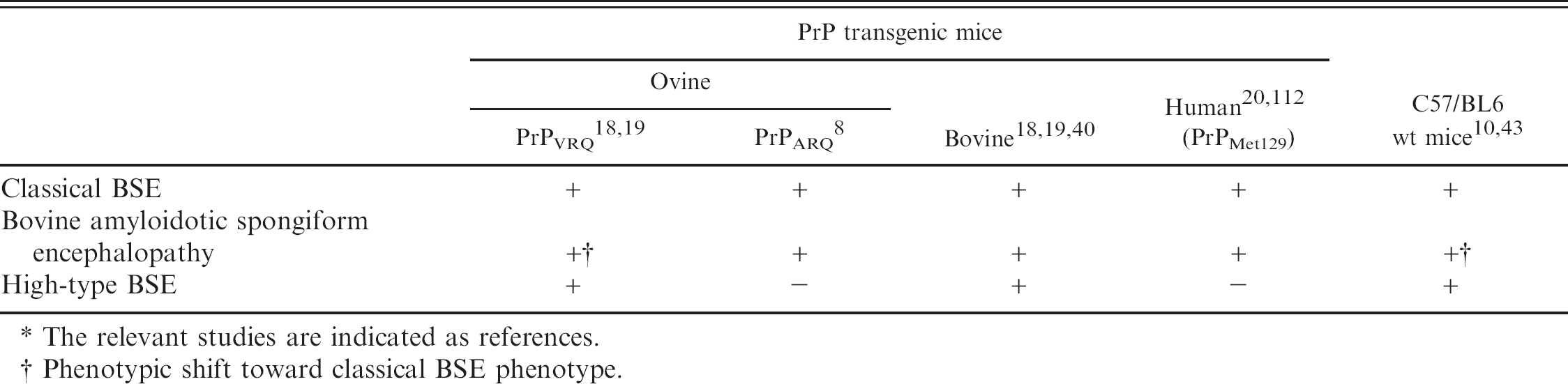
The relevant studies are indicated as references.
Phenotypic shift toward classical BSE phenotype.
Taken together, there are still many uncertainties, making it difficult to assess the efficiency of current surveillance regimes for atypical TSEs. Experimental transmission studies will help to elucidate their phenotypic features and deliver reference materials for the evaluation of test formats and laboratory procedures. It will then be a subject of risk and cost—benefit assessments to provide a decision baseline for the potential modification of surveillance policy.
Disease control and the implications of atypical TSEs
Control of BSE in cattle
The key to C-BSE disease control lies in preventing recycling of the agent to susceptible animals by excluding MBM from feedstuff. There is no evidence of horizontal transmission of C-BSE. Possible transmission to other species, via MBM, was a particular major concern, especially to pigs, sheep, and goats. Although pigs have been demonstrated to be susceptible to C-BSE by parenteral inoculation, they failed to develop disease when challenged orally with massive doses of C-BSE brain material. 55,125,181 Therefore, and in the apparent absence of any naturally occurring prion diseases in pigs, it is considered unlikely that C-BSE became established in pig populations. 102,109,111 As outlined above, the situation was very different in small ruminants, in terms of susceptibility, although the likelihood of exposure via feed was historically much lower because MBM was less frequently used than in pig feed.
Recently, BASE was experimentally transmitted to cattle by i.c. inoculation in 2 studies in Italy 121 and Japan. 100 In all animals, the BASE phenotype was conserved. Interestingly, the clinical picture in BASE described in the Italian study involved dullness, and the muscles were amyotrophic, neither of which has been observed in C-BSE. Similar experiments for H-BSE are ongoing in several countries, but the results are not yet available in the veterinary literature. Secondary cases of H-BSE and BASE have not been reported, and there is also no evidence of a temporal or spatial relation between atypical and C-BSE cases. Data from France and Poland show that the H-type and BASE incidence was constant over years and not related to that of C-BSE. 23,138 It is, therefore, tempting to postulate that H-BSE and BASE indeed represent sporadic TSEs. Yet, this will be difficult to prove and can only be substantiated by exclusion of other origins. In view of future disease-control strategies, it is important to address the potential of intraspecies and interspecies transmission experimentally. To this end, transmission studies to PrP transgenic mice and other species were initiated. The transmissibility of different BSE isolates to mice is illustrated in Table 4. Several lines of evidence indicate a relatively high potential of BASE to cross over to other species. The BASE readily transmitted to conventional C57/BL6 mice and to human-PrP, bovine-PrP, and ovine-PrP transgenic mice, as well as to a primate. 20,40,51,112 Interestingly, upon transmission and serial passage in ovine PrPVRQ transgenic mice and C57/BL6 mice, its phenotype shifted toward that observed in classical BSE. This supports the hypothesis that BASE was possibly the origin of the C-BSE epidemic and eventually changed its phenotypic representation upon subpassage in cattle or other hosts. 18,43
Transmission studies in ovine PrPARQ mice also demonstrated similarities between BASE and a bovine-passage of a transmissible mink encephalopathy (TME) isolate derived from an outbreak in the United States in 1985. Transmissible mink encephalopathy is a rare, noncontagious, feed-borne prion disease in farmed mink with an hitherto unknown origin. These findings gave rise to the hypothesis that BASE-contaminated feed was the source of the TME outbreaks. 8
For H-BSE, the situation is different. Disease transmission succeeded to wild-type C57/BL6 9 and ovine PrPVRQ transgenic mice with a phenotype distinct from BASE and C-BSE, but failed in human PrP 20 and ovine PrPARQ transgenic mice. 8 Based on these studies and the different phenotypic representation of C-BSE, BASE, and H-BSE in these mice, it was concluded that the 3 phenotypes involve distinct prion pathogens. Beyond this, the interpretation of data with regard to disease transmissibility and pathogenesis obtained in mouse models is always limited. For instance, intracerebral or intraperitoneal inoculation may not reflect species barrier issues as relevant to oral inoculation.
Atypical BSE has not been detected in animals born after the implementation of the complete ban of MBM from feed in the respective countries. If this holds true in the future, it may mean that feed controls will prevent cases of atypical BSE and that they may not, therefore, be spontaneous TSEs. In the end, the crucial question of whether H-BSE and BASE transmit vertically, horizontally, or by the oral route (e.g., via MBM) still remains to be investigated. Until then, prudence is warranted, and the classification of atypical TSEs as spontaneous prion diseases would be premature. When pressure grows to lift the ban on the inclusion of MBM in farm animal feed in the future, such aspects clearly need consideration to assess the risk of reemergence of BSE in livestock.
Control of TSEs in small ruminants
In countries with a high incidence of classical scrapie, disease control is challenging and often frustrating. New Zealand and Australia are the only countries that successfully completed the elimination of classical scrapie by applying rigorous control measures after it had been imported into the domestic population. 123 Despite large efforts, some countries have been struggling with the disease for decades. 136 The main factors that contribute to this difficult situation are the long incubation time of several years and a tremendous resistance of the agent to degradation, resulting in its persistence in the farm environment. 136 Under such circumstances, animal movement restrictions and “stamping out” policies may not be sufficient to control or even eradicate the disease. Genotyping and selection for classical scrapie resistance may assist in reducing exposure of contact sheep, preventing reinfections in affected flocks, and reducing the overall susceptibility in the population. In the United Kingdom, for example, the National Scrapie Plan, implemented in 2001, led to a significant drop in the incidence of classical scrapie cases. 170 For atypical scrapie in sheep, the genotype preference appears to be almost the converse of that for classical scrapie. 16 A high proportion of atypical scrapie cases are of PrP genotypes, which confer the highest degree of resistance to classical scrapie (e.g., PrPARR/ARR). A side effect of selective breeding to control for classical scrapie may, therefore, be an increase in the proportion of atypical scrapie—susceptible sheep.
A fundamental question prompted by the detection of atypical scrapie was whether it could possibly have arisen as a result of the infection of small ruminants with BSE. This appears very unlikely because, when BSE was experimentally transmitted to sheep and goats, the neuropathologic and biochemical phenotype in such animals differed completely from that observed in the atypical cases. This is also supported by transmission experiments to ovine PrPARQ transgenic mice. 6
Unlike classical scrapie, to date, there has been no evidence that atypical scrapie is a contagious disease. Usually such cases occurred in single animals and only in some instances have secondary cases been identified mainly in large flocks, which do not, however, necessarily indicate natural disease transmission. 16 Moreover, PrPd in atypical scrapie has not yet been detected outside the CNS (for instance in the LRS). A study in the United Kingdom 156 identified 3 cases of atypical sheep scrapie in a classical scrapie—free research flock, which was kept under stringent containment and which had founder animals imported from New Zealand, a country considered to be free from classical scrapie. By process of exclusion, the authors found it likely that these 3 cases were not infected from an exterior source. 156 Taken together, and similar to what has been proposed for atypical BSEs in cattle (see above), this situation gave rise to the hypothesis that atypical scrapie is poorly transmissible, if at all, under natural conditions and that it may occur spontaneously. Retrospective studies in the United Kingdom also indicate the presence of atypical scrapie as early as 1987 in the population. 34,177 It may, therefore, have existed for a long time but remained unrecognized as such because of its relatively low prevalence.
Data on the potential of atypical scrapie to cross over to other species are still limited. A 2009 study, 64 showed that transmission to porcine PrP transgenic mice resulted in a shift of its phenotype, similar to cattle and ovine BSE isolates. The same effect was observed when cattle BSE was transmitted to pigs by i.c. inoculation. 115,154 Such situations may challenge the suitability of biological strain typing using single transgenic mouse models without a proper validation for this purpose. Even more important, it points to the possibility that TSE strains may change their phenotype upon transmission to other hosts, rendering them indistinguishable from other TSE sources. In this sense, it will be necessary to investigate the phenotype of atypical scrapie when transmitted to cattle and that of atypical BSE isolates when transmitted to sheep and goats, especially after oral challenge. It also highlights the continued need to refine and validate the tools used to define strains.
Public health measures and the implications of atypical TSEs
Classical BSE clearly is a public health concern, and since 1996, some 200 cases of vCJD have been reported, with 170 located in the United Kingdom. Currently available diagnostic tests only allow for the reliable detection of infected animals in the late phase of the incubation period, but BSE infectivity has been detected earlier in experimentally infected cattle. 179,182 Testing for BSE cannot, therefore, be considered ultimately effective in protecting consumers, but it may help to maintain consumer confidence. Certainly, complete BSE eradication would best solve this situation. At present, the most important measure to prevent human exposure to BSE is the removal of specified risk material from ruminant carcasses. These materials have been identified to contain C-BSE infectivity in transmission experiments and are designated to be removed from carcasses intended for the production of food for human beings. 4,41,179,184
Unlike the demonstrated link between C-BSE and vCJD, no association has been identified between atypical BSEs and TSEs in human beings; there is no epidemiologic link that may indicates a zoonotic situation. Still, the molecular signature in BASE was reported to be reminiscent of some sporadic CJD cases, and upon transmission to human PrP transgenic mice and primates, the BASE agent seemed to replicate more efficiently compared with classical BSE. 20,51,112
It is currently unknown whether tissues other than those defined for C-BSE carry infectivity in BASE or H-BSE. More data are needed from transmission studies or from in-depth analysis of tissues harvested from field cases, where whole carcasses have been available. Until then, atypical BSEs are assumed to present a risk similar to C-BSE and treated accordingly. If the agents show infectivity distributions different from C-BSE, especially in tissues that are not yet defined as specified risk materials, it may have implications on risk management and public health.
It must also be emphasized that the number of atypical BSE cases is small, and, therefore, the risk of exposure can expected to be less compared with that resulting from a much higher number of C-BSE cases during the C-BSE epidemic. Consequently, the demonstration of a zoonotic link may not prompt regulatory action because the likelihood of exposure is low.
The definition of specified risk materials in sheep and goats is much more difficult. This is because, in BSE-affected small ruminants, infectivity is more widely spread in the body 175 and has also been detected in blood. 98 However, only the CNS, the spleen, and the tonsils are considered high-risk materials and, therefore, defined as specified risk materials. These also include tissues in which atypical scrapie infectivity has been identified so far, namely the brain, but it still remains to be determined whether other tissues contain atypical scrapie infectivity. To date, atypical and classical scrapie are not regarded as zoonotic. Whether or not consumption of small-ruminant tissues is relevant for public health purposes depends on the presence of classical BSE in these species. If the classical BSE prevalence were to increase in small ruminants, the definition of specified risk material would have to be enlarged and would then probably include the whole animal.
Conclusions and outlook
The BSE crisis during the past 20 years has shaken the livestock industry to its core and caused enormous economic losses worldwide. Although the crude numbers of identified BSE cases in most countries is relatively low, the breadth of the epidemic in the United Kingdom, with more than 180,000 detected cattle, has raised fears of a global expansion to similar levels. The risk to human beings, which could result in an incurable neurologic disease, damaged consumer confidence in beef products and resulted in a considerable market crisis in that sector of the economy. Because of the nature of the disease, namely, the long incubation period and the unusual properties of the agent, it took almost 2 decades to finally control the disease, although the risk to consumers appears to have been contained while the epidemic declined. Although classical BSE now fades out in the cattle populations, novel, so-called atypical TSEs have been recognized in cattle and small ruminants. Significant effort has been directed toward characterizing these isolates, toward understanding whether or not they can be controlled by the measures established for C-BSE, and toward examining the extent to which they may represent a public health risk. At present, it still remains to be determined whether these isolates potentially transmit to other animals under natural conditions or by means of feeding MBM. Several lines of evidence lead to the hypothesis that such atypical variants occur sporadically at a constant level. This would indicate a potential to give rise to further epidemics and may represent a permanent risk for the public if some measures are not maintained indefinitely. If classical BSE is ever considered to be eradicated, requests to lift the ban on inclusion of MBM in ruminant feed and specified risk materials in food may need to be considered. In case atypical TSEs in ruminants indeed turn out to be spontaneous diseases, their eradication will be difficult. At this stage, the impact of atypical TSEs on public health and disease control certainly needs to be reassessed. However, with decreasing coverage of active surveillance, it will be difficult to identify such cases in the future. This dilemma points to the importance of maintaining a certain level of efficient TSE surveillance until there is clarity regarding risks from atypical TSEs.
Acknowledgements
The authors thank Miroslaw Polak (Pulawy, Poland) and Fernando Arnolfo, Christina Casalone, and Barbara Iulini (Turino, Italy) for BASE tissue samples. Moreover, the authors acknowledge Michael Garrood (Zürich, Switzerland) for editing and Danny Matthews (United Kingdom) for scrutinizing the manuscript. This work is funded by the Swiss Federal Veterinary Office.
