Abstract
Canine granulomatous hepatitis is an uncommon morphologic diagnosis that has been associated with a variety of diseases, including a number of systemic infectious etiologies. Formalin-fixed, paraffin-embedded (FFPE) tissues are typically the only source of liver tissue remaining for additional testing for the presence of infectious disease within granulomas. It is unclear if the more common infectious culprits of granulomatous hepatitis can be identified from such specimens. The aim of the current study was to retrospectively investigate archival FFPE liver tissue from dogs with granulomatous hepatitis for the presence of infectious agents. Semiquantitative analysis of copper accumulation in liver specimens was also performed. Medical records were examined for recorded evidence of systemic infectious disease diagnosis. Formalin-fixed, paraffin-embedded liver was prospectively evaluated for infectious agents via differential staining techniques (n = 13), eubacterial fluorescent in situ hybridization (n = 11), and Bartonella polymerase chain reaction assays (n = 15). An infectious cause of granulomatous hepatitis was not identified within liver tissue from any dog using these diagnostic methodologies. Six out of 25 (24%) dogs were diagnosed with concurrent systemic or localized bacterial infections at the time of presentation. Nine out of 17 (53%) dogs had excessive hepatic copper accumulation when evaluated by a semiquantitative histologic grading scheme or quantitative copper analysis. As definitive infectious causes of granulomatous hepatitis were not identified within archival liver biopsy samples, it was concluded that investigation of infectious etiologies within FFPE liver specimens using these diagnostic approaches may be of low yield.
Keywords
Introduction
Granulomatous hepatitis (GH) is a form of hepatic inflammation characterized by aggregations of activated macrophages, often with an epithelioid appearance, with infiltration of lymphocytes, plasma cells, and possibly fibroblasts within the liver. 19 This form of inflammation may occur as a result of a primary hepatic disorder or as a nonspecific response to a variety of chronic, extrahepatic disease processes. 19 Granulomas are identified in 2–15% of all liver biopsy specimens from human beings7,16,20 and in 1% of dogs with primary hepatitis. 17 Various intrahepatic infectious etiologies, including fungal disease, atypical bacteria, protozoa, and parasitic disease, have been reported as underlying causes for GH in people and dogs.3–6,8,9,13,14 While these agents cause GH when the liver becomes infected, it is unclear how often primary recognition of GH can be consequently attributed to one of these infectious agents. Because GH is an uncommon finding in dogs and is not recognized before routine histology has been performed, formalin-fixed, paraffin-embedded (FFPE) tissue is often the only resource remaining for additional testing of the liver for the presence of infectious disease following histopathologic diagnosis. Furthermore, opportunities for obtaining additional liver tissue for supplementary diagnostics are often limited due to owners’ concerns regarding finances or invasiveness of liver biopsy procedures. To the authors’ knowledge, studies have not been performed in dogs to date to determine whether the more common infectious causes of GH can be routinely identified within FFPE liver specimens.
The aims of the current study were to retrospectively investigate archival FFPE liver tissue from dogs with a histopathologic diagnosis of GH for the presence of select infectious agents, using differential staining techniques, eubacterial fluorescent in situ hybridization (FISH), and Bartonella polymerase chain reaction (PCR) assays. Medical records were examined for evidence of systemic infectious disease diagnosis and exposure to hepatotoxins associated with GH in dogs and human beings. Additionally, due to the association of copper storage disease in dogs with aggregates of macrophages, as well as infiltration with varying populations of lymphocytes, plasma cells, and neutrophils within the hepatic parenchyma,15,21,24,25,28 semiquantitative analysis of copper accumulation in the liver specimens was also performed.
Materials and methods
Case selection criteria
Dogs with a histopathological diagnosis of GH made between 2001 and 2010 were identified by searching histopathology reports of the pathology service at the North Carolina State University College of Veterinary Medicine (NCSU-CVM). Dogs were included in the study if the following 3 criteria were identified: GH lesions were reported, GH lesions were confirmed to be consistent with the diagnostic criteria described by the World Small Animal Veterinary Association (WSAVA) liver standardization group, 19 and a medical record was available for review. For the purpose of the study, cases of GH were included if there were 1 or more granulomas consisting of multifocal aggregations of activated macrophages, typically with peripheral lymphocytes and plasma cells and, on occasion, fibroblasts and collagen fibers. 19 Dogs with pyogranulomatous hepatitis were also included, as the lesions in these dogs included the presence of granulomas with infiltration of neutrophils. For dogs meeting the inclusion criteria, archived FFPE tissue was retrieved, when available, for PCR and FISH.
Routine histopathology and special stains
Tissue sections were prepared from each archival liver tissue specimen, stained with hematoxylin and eosin, and reviewed by a board-certified anatomic pathologist (JM Cullen). Upon confirmation of a diagnosis of GH in accordance with WSAVA guidelines, lesions were subjectively scored as mild, moderate, or marked based upon the number of granulomas noted per approximately 20 liver lobules. Specifically, a grade of mild (group 1) was assigned to specimens containing <2 granulomas per lobule, moderate (group 2) if > 2 but <50% of the lobules were affected, and marked (group 3) if >50% of the lobules were affected. Sections were also stained with periodic acid–Schiff, Gomori methenamine silver, Fite, Giemsa, and Gram stains, each of which were examined for the presence of fungal and bacterial organisms. Sections were additionally stained with rhodanine for evaluation of copper accumulation. Copper content and distribution were graded from 0 to 5 using a previously published scoring system. 26 Positive and negative controls were used for all stains.
Fluorescent in-situ hybridization
Formalin-fixed, paraffin-embedded liver specimens were sectioned at 4 µm and mounted on poly-L-lysine–coated slides. Tissue sections were deparaffinized in xylene, rehydrated in a graded series of ethanol, and air dried prior to hybridization with FISH probes at a working concentration of 5 ng/µl. 11 The universal eubacterial probe Eub338, labeled at the 3’ end with FAM (fluorophore 6-carboxyfluorescein), was used to identify typical eubacteria. 11 A sense Eub probe (non-Eub338), labeled at the 5’ end with cyanine 3 was simultaneously applied to enable discrimination of nonspecific hybridization and autofluorescence. Positive control slides included FFPE sections of gastrointestinal mucosa containing surface-adherent bacteria. Hybridization of Eub338 and non-Eub338 probes to each specimen was performed using reagents and conditions as previously described. 11
PCR identification of Bartonella spp
In order to avoid PCR contamination, a sterile scalpel blade was used to cut slices from archival FFPE hepatic tissue, DNA extractions and amplification were performed in separate areas of the laboratory, and blank paraffin control blocks were used between sample blocks to minimize DNA carryover. 27 DNA was subsequently extracted using a commercial DNA kit, a following the manufacturer’s instructions, for the tissue extraction protocol. Identification of Bartonella species in extracted DNA was performed by PCR targeting the 16S-23S ribosomal RNA intergenic transcribed spacer (ITS) region. Amplification was performed in a 25-µl final volume reaction containing 12.5 µl of Tak-Ex Premix, b 0.25 µl of 30 µM of each forward and reverse primer, c 7 µl of molecular-grade water, and 5 µl of DNA from each sample tested. DNA templates for PCR-negative controls were prepared using blood samples of control dogs. Templates for PCR-positive controls were prepared using blood samples of patients with confirmed Bartonella henselae. In order to assess the quality of DNA obtained from each sample and identify potential inhibitory substances, a PCR reaction amplifying canine glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was performed as previously described. 1 Conventional PCR was performed d under the following conditions: a single hot-start cycle at 95°C for 2 min followed by 55 cycles of denaturing at 94°C for 15 sec, annealing at 66°C for 15 sec, and extension at 72°C for 18 sec. Amplification was completed by an additional cycle at 72°C for 1 min, and products were analyzed by 2% agarose gel electrophoresis with detection using ethidium bromide under ultraviolet light. Amplification products, if obtained, were sequenced to establish the Bartonella species and strain.
Review of medical records
Patient medical records were retrospectively reviewed. Signalment, history of drug or hepatotoxin exposure, clinical signs, physical examination findings, clinicopathologic test results, radiographic or ultrasonographic findings, and concurrent diagnoses were extracted and summarized.
Results
Routine histopathology and special stains
Twenty-five dogs were identified as meeting the inclusion criteria, of which 15 dogs had archival liver tissue available for evaluation via differential staining techniques, FISH, and/or Bartonella PCR. Fourteen specimens were obtained antemortem via needle (n = 6) or surgical (n = 8) biopsy, whereas 1 sample was obtained at necropsy. All specimens included in the current study were adequate for determining a histopathological diagnosis of GH and contained at least 2 lesions. Two dogs had focal periportal, centrilobular, or midzonal discrete granulomas and mild inflammatory infiltration within liver tissue and were categorized as group 1 (Fig. 1A). Ten dogs had multifocal discrete granulomas and were categorized as group 2 (Fig. 1B). Three dogs had locally extensive granulomatous inflammation and were categorized as group 3 (Fig. 1C). Sufficient archival liver tissue was available from 13 out of 15 dogs for evaluation by means of special stains. Fungi, acid-fast bacteria, Gram-negative bacteria, and Gram-positive bacteria were not identified in liver specimens from any of these dogs. Rhodanine staining was performed on liver specimens from 12 out of 15 dogs. Five dogs had histologic evidence of increased copper accumulation, of which 3 were designated with a grade of >2, which is considered abnormal and implicated copper as a potential etiologic factor. 22 In these 3 samples, copper was distributed predominantly in zone 3 (centrilobular). The remaining 2 samples had copper accumulation predominantly within clustered macrophages throughout the sample and along the septae (Fig. 2). Insufficient archival tissue was available to retrospectively quantify tissue copper concentrations.
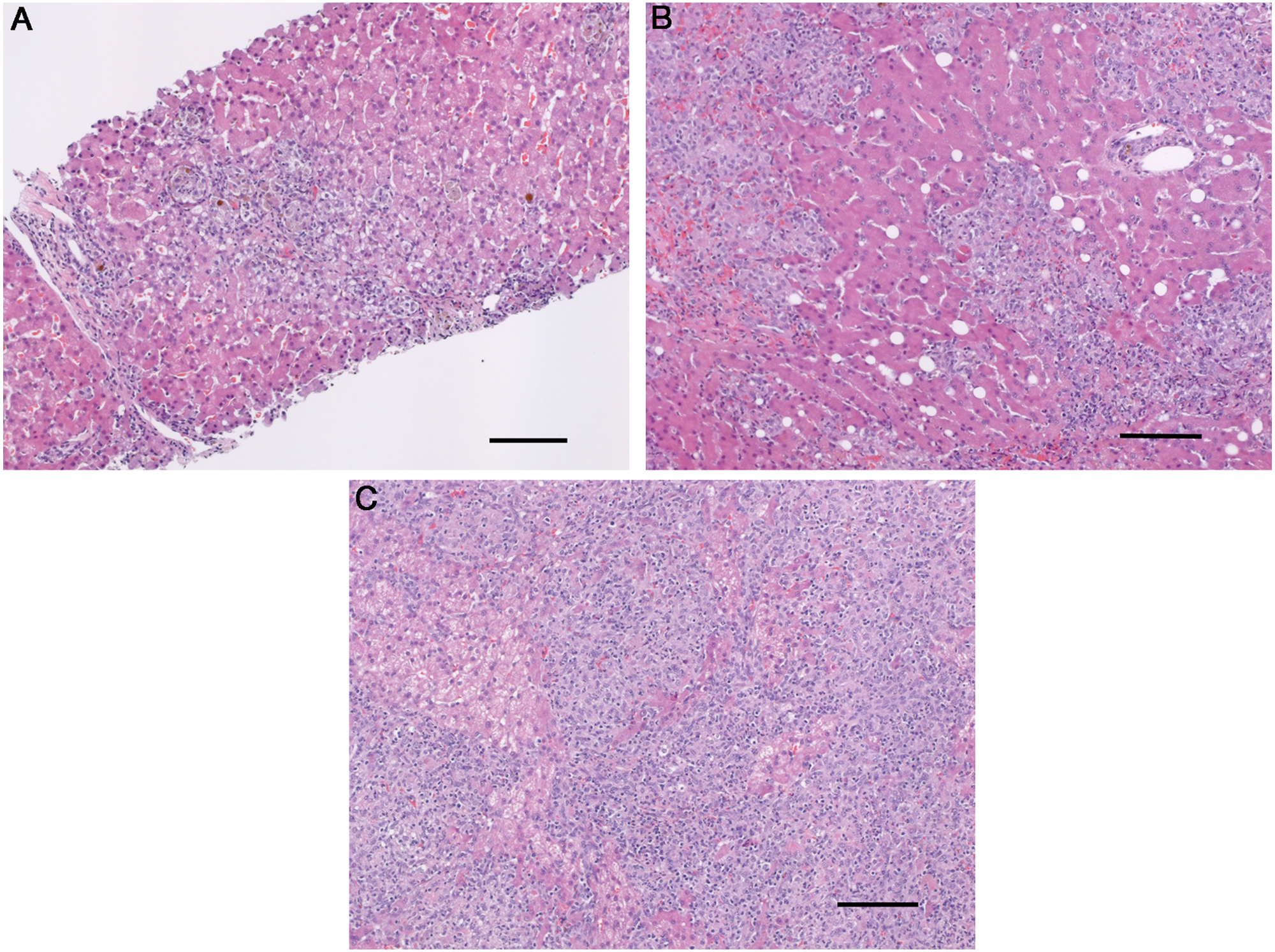
Liver.
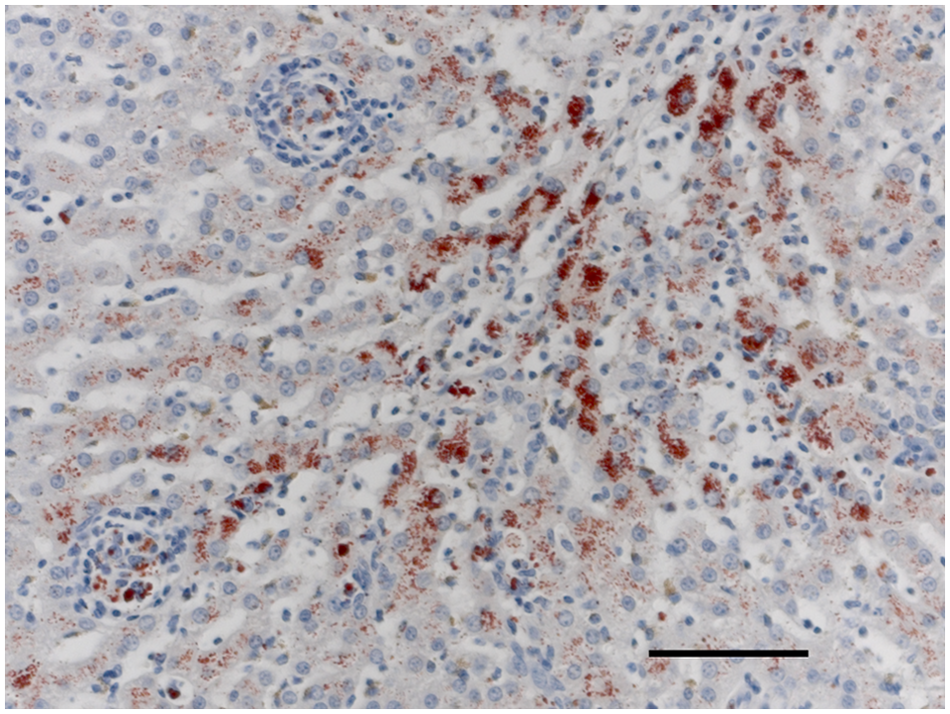
Liver. Copper accumulation predominantly within clustered macrophages. Rhodanine. Bar = 75 μm.
Fluorescent in-situ hybridization
Eleven of the 15 FFPE archival liver specimens were evaluated by FISH. Bacteria were not identified within granulomas, bile ducts, Kupffer cells, or hepatic parenchyma. Positive hybridization of Eub338 was observed for each positive control sample.
PCR identification of Bartonella spp
Bartonella species DNA was not amplified from any of the 15 FFPE archival liver tissue samples from dogs with GH, including a specimen from a dog with documented B. henselae bacteremia. Bartonella henselae DNA was successfully amplified from each positive control specimen.
Review of medical records
The median age of the 25 dogs was 6 years (range: 2–17 years). Ten dogs were spayed females, 2 dogs were intact females, 11 dogs were castrated males, and 2 were intact males. Fifteen different breeds were represented, including mixed breed (5), Labrador Retriever (4), American Staffordshire Terrier (3), and Rottweiler, West Highland White Terrier, Vizsla, Pomeranian, Chesapeake Bay Retriever, Saint Bernard, corgi, Cairn Terrier, springer spaniel, Beagle, Wheaten terrier, German Shorthaired Pointer, Munsterlander (1 each). Six dogs were euthanized, and the liver was sampled at necropsy. Surgical (13/19) or percutaneous needle (6) biopsy techniques were used to sample the liver in the remaining dogs. The primary indications for liver biopsy were identified as clinical signs referable to hepatic disease, including an increase in liver enzyme activities (22/23 dogs) or abnormal ultrasonographic appearance of the liver (18/22 dogs). Commonly reported presenting complaints were anorexia or inappetence (15), lethargy (12), weight loss (12), vomiting (11), fever (11), polyuria and polydipsia (7), lymphomegaly (4), and edema (3). None of the dogs had an identified exposure to a known hepatotoxin.
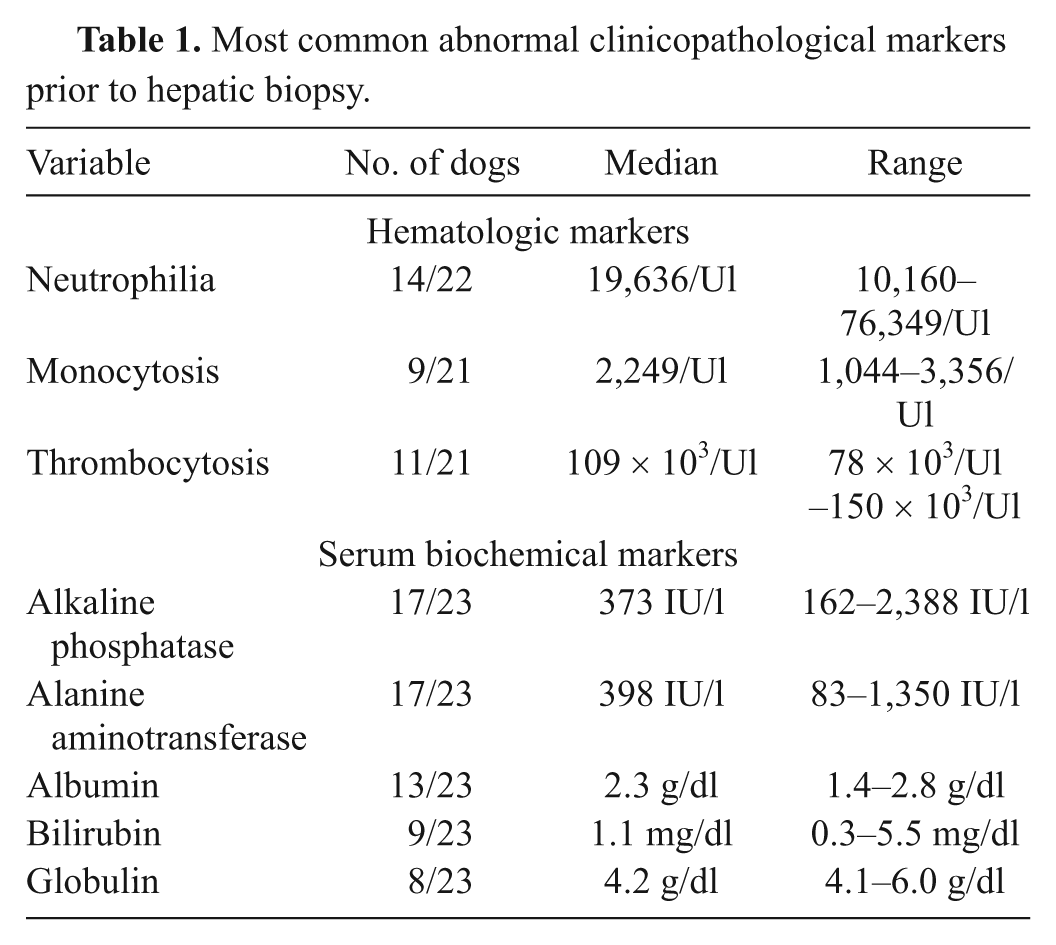
The most common hematologic abnormalities in dogs with GH included neutrophilia, monocytosis, and thrombocytopenia (Table 1). Increased alkaline phosphatase or alanine aminotransferase activities were noted in all but 1 dog for which serum biochemical testing was performed. Hypoalbuminemia, hyperbilirubinemia, and hyperglobulinemia were also commonly identified (Table 1).
Most common abnormal clinicopathological markers prior to hepatic biopsy.
Twenty-two dogs had abdominal ultrasound performed. An abnormal appearance and/or size of the liver were identified in 18 dogs. Fifteen dogs had abnormal hepatic echogenicity with the liver described as hypoechoic (8), hyperechoic (4), or heterogeneous (3), while microhepatica was reported in 6 dogs and hepatomegaly in 5 dogs. The liver was normal in size and echogenicity in 4 dogs, 3 of which had peritoneal effusion. Trace to minimal peritoneal effusion was noted in a total of 8 dogs.
Hepatic copper was evaluated in 9 out of 25 dogs at initial presentation via semiquantitative histologic grading scheme (8 dogs) and/or quantitative copper analysis (3 dogs). Six out of 9 (67%) dogs had evidence of hepatic copper accumulation.
Extrahepatic infectious diseases were identified at the time of diagnosis of GH in 6 out of 25 dogs. Diseases included vegetative endocarditis of the mitral valve (Staphylococcus intermedius; 1 dog), B. henselae bacteremia based on PCR assay of blood (1 dog), bacterial urinary tract infection by Escherichia coli (2 dogs) and S. intermedius (1 dog), and diskospondylitis with a concurrent Serratia marcescens epidural abscess (1 dog). Culture of liver and/or bile was performed in 11 dogs. Aerobic cultures of hepatic biopsy tissue from 6 dogs yielded growth of S. intermedius from 1 dog. Aerobic (6/6) or anaerobic (3/6) bile cultures from 6 dogs did not yield bacterial growth. Concurrent noninfectious diseases identified at the time of diagnosis included renal dysplasia, progressive epilepsy, cerebellar cortical degeneration, pyogranulomatous pneumonia, eosinophilic enteritis, inflammatory bowel disease, and acute myeloid leukemia (1 dog each).
Discussion
Granulomatous hepatitis is considered an uncommon diagnosis in dogs with liver disease. A variety of infectious etiologies have been reported in association with GH, including Bartonella spp., Histoplasma spp., Leishmania spp., and Mycobacteria spp.5,6,8,18 A recent retrospective case review in human beings detected an infectious agent within archival, paraffin-embedded liver tissue in 3.63% of granulomatous hepatic lesions utilizing PCR. 4 Using an approach restricted to diagnostic investigation of archival FFPE tissue from dogs with GH, the present study did not identify any infectious etiologies within the liver despite a focus on the anticipated common pathogens using differential staining techniques, FISH, and molecular testing for Bartonella spp. Previous studies have shown that PCR and FISH can be effective approaches to identifying DNA of infectious agents in FFPE tissue samples.4,11,12 However, due to limitations of these techniques in demonstrating all potential eubacteria, differential staining techniques were also performed.
Retrospective review of the medical records data identified a diagnosis of an extrahepatic bacterial infection in 24% (6/25) of dogs in the current study. These infections included E. coli and S. intermedius urinary tract infection, S. intermedius vegetative endocarditis, S. intermedius hepatic infection, S. marcescens epidural abscess, and B. henselae bacteremia. It is unclear if these infections played a role in the pathogenesis of GH, in part because sufficient archival liver tissue for the PCR and FISH analysis was only available from the dog with B. henselae infection. Polymerase chain reaction did not amplify B. henselae DNA from the liver of this particular dog. Although PCR is considered to be the most sensitive approach to diagnosis of tissue B. henselae infection, it is not positive in all cases. 2 Because of the retrospective nature of the medical records review aspect of the study, a uniform approach to systemic infectious disease diagnosis in these dogs cannot be assured and, therefore, the existence of undiagnosed infectious diseases cannot be ruled out.
It is recognized that excessive copper accumulation in dogs can cause prominent macrophage aggregates.15,19,24,25,28 In such cases, special stains for copper are required to avoid an inappropriate search for infectious agents. In the current study, 17 dogs with GH had semiquantitative or quantitative evaluation of intrahepatic copper performed. Nine of the dogs (53%) were identified as having intrahepatic copper accumulation. Hepatic granulomas in these dogs were predominantly clustered around necrotic hepatocytes or were found in association with copper accumulation in the centrilobular regions, which is suggestive of a primary copper-associated hepatitis. Biliary excretion is the predominant route of copper elimination; therefore, increased hepatic copper concentrations may also be the result of cholestasis secondary to chronic liver disease. Three of the 9 dogs with copper accumulation in the current study were Labrador Retrievers and 1 was a West Highland White Terrier, which are breeds predisposed to primary hepatic copper accumulation. This observation is of potential medical importance, as chelation therapy may be indicated as part of the treatment regimen. Based on this finding, hepatic copper quantification is advised in GH cases, and further investigation, including quantitative copper analysis within a larger group of dogs with GH, is warranted.
A variety of noninfectious diseases were diagnosed in the dogs reported herein including inflammatory bowel disease, renal dysplasia, acute myeloid leukemia, cerebellar cortical degeneration, eosinophilic enteritis, and pyogranulomatous pneumonia. Therefore, GH may represent a form of “reactive hepatopathy” in some dogs, and recognition of GH should prompt a diagnostic investigation that also includes noninfectious systemic diseases. None of the dogs were diagnosed with malignant histiocytic sarcoma; therefore, it is less likely that GH in the dogs reported herein represented evidence of a metastatic or an overt neoplastic process. While several drugs and hepatotoxins have been associated with GH in dogs and human beings, 10 a careful review of the medical records failed to disclose exposure to these agents. Given the retrospective nature of the medical record review, it is certainly possible that some important exposures were not recognized.
In conclusion, a diagnostic investigation of archival FFPE liver tissue for infectious etiologies using the approaches described in the present study is unlikely to be of high yield. While study of a larger number of dogs with GH may have disclosed different results, the incidence of GH in dogs is low, and to the authors’ knowledge, the present study is the largest yet compiled. It is likely that infectious agents are less often direct causes of GH than is presumed. This is supported by results of the only prior retrospective study of GH in dogs (n = 9) where the only definitive diagnoses identified were dirofilariasis, histoplasmosis, histiocytosis, lymphoma, and lymphangiectasia. 3 However, there are a number of reasons for possible false-negative test results in the present study and in the approaches described, including the deleterious effects of formalin and time on DNA integrity,23,29,30 insufficient sample size, and organism numbers in the sample that are below the limit of detection. Based on results of the present study, future studies to detect infectious agents in fresh or fresh frozen liver tissue from dogs with GH appear to be warranted. Additionally, because of the unexpectedly high number of dogs having GH associated with intrahepatic copper accumulation, liver copper quantification is recommended in dogs diagnosed with GH.
Footnotes
a.
QIAamp DNA tissue mini kit, Qiagen, Valencia, CA.
b.
Fisher Scientific Co., Pittsburgh, PA.
c.
Integrated DNA Technologies Inc., Coralville, IA.
d.
Mastercycler EP gradient, Eppendorf North America, Hauppauge, NY.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported a grant from the Firestone Canine Research Endowment, North Carolina State University, College of Veterinary Medicine, Raleigh, North Carolina.
