Abstract
Canid herpesvirus 1 (CaHV-1) is a well-known cause of fatal hepatic and renal necrosis in neonatal puppies. In adult dogs infected with CaHV-1, papulovesicular genital lesions may be observed. CaHV-1 infection during pregnancy can lead to embryonic resorption, abortion, and stillbirth. In high-density dog populations, CaHV-1 can also contribute to kennel cough. Furthermore, recent literature has clearly documented that CaHV-1 can induce ocular disease in immature and adult dogs. The current study describes a case of fatal CaHV-1 infection in a 9-year-old spayed female Bichon Frise dog. Following a history of vomiting and diarrhea, the dog deteriorated and subsequently died. The main lesions were multifocal areas of necrosis with intranuclear inclusion bodies in the liver, adrenal gland, and small intestine, similar to the lesions observed in CaHV-1-infected puppies. Infection with CaHV-1 was confirmed on samples of liver by polymerase chain reaction, immunohistochemistry, and in situ hybridization. There was no indication of immunosuppression in this dog. Based on the results presented herein, CaHV-1 should be included in the list of differential diagnoses of hepatic necrosis in adult dogs.
Canid herpesvirus 1 (CaHV-1) is classified under the genus Varicellovirus within the subfamily Alphaherpesvirinae.1,8 In young puppies, 1–4 weeks of age, the virus typically induces a fatal multisystemic necrohemorrhagic disease. Infection is thought to occur primarily as the puppies pass through the birth canal. Puppies infected in utero may be resorbed, aborted, or stillborn, depending on the time of gestation. 4 Several factors, such as body temperature regulation and immune status, are considered to be important in the development of resistance to systemic viral infection in young puppies. 3 Systemic infection has rarely been reported in older immunosuppressed animals, but adult immunocompetent dogs usually exhibit mild, self-limiting respiratory, genital, or ocular signs. 4 The current report describes a case of fatal systemic CaHV-1 infection in an adult immunocompetent dog.
A 9-year-old spayed female Bichon Frise dog was presented to the referring veterinarian with a 2-day history of vomiting, diarrhea, and decreased appetite. Physical examination revealed pale yellow mucous membranes with multiple petechiae. The dog was weak and lethargic and had a rectal temperature of 39°C. A Bordetella bronchiseptica vaccine was administered on the initial day of presentation, and the dog was placed on low-residue food. On the following day, the dog returned with a history of vomiting clear material the previous night, and an injection of maropitant citrate a was administered. Blood work revealed increased serum levels of alanine aminotransferase (236 U/l, reference range: 5–107 U/l) and aspartate aminotransferase (236 U/l, reference range: 5–55 U/l). There was also mild hypokalemia (3.5 mEq/l, reference range: 4.0–5.6 mEq/l). The complete blood count analysis showed no abnormalities. The dog presented again 3 days later, with coughing as the primary complaint. Physical examination revealed bilateral oculonasal discharge, and the rectal temperature was 38.2°C. Lateral abdominal radiographs showed a slightly enlarged liver. By the following day, the rectal temperature had decreased to 36.4°C. At this time, the dog was given a guarded to poor prognosis. It died on the way to the Michigan State University Veterinary Teaching Hospital (East Lansing, Michigan).
At necropsy, the dog weighed 2.17 kg. Significant gross lesions included diffuse red and tan mottling of the liver (Fig. 1). In addition, multifocal petechiae were present on the gingival mucous membranes and the serosa and mucosa of the entire gastrointestinal tract and the urinary bladder. Beginning at the distal third of the jejunum and extending throughout the remainder of the small intestine, the mucosa was diffusely reddened. The cecum and colon contained moderate amounts of mucoid, dark red to black feces. Other findings included icteric sclerae and oral mucous membranes. Representative sections of brain, heart, lung, liver, spleen, adrenal gland, urinary bladder, kidney, stomach, small intestine, and colon were fixed in 10% neutral buffered formalin, sectioned at 5-µm thickness, stained with hematoxylin and eosin, and examined histologically. The liver had multifocal to coalescing areas of hemorrhage and coagulative necrosis. Hepatocytes adjacent to the areas of necrosis often contained eosinophilic, intranuclear inclusions that displaced the chromatin to the periphery of the nucleus (Fig. 2). Necrotic foci and intranuclear inclusions were also seen within the adrenal glands and jejunal crypt epithelium. Sections of lung revealed moderate intra-alveolar edema and fibrin exudation, with mild alveolar histiocytosis, multifocal alveolar hemorrhage, and multifocal necrosis of the alveolar septa. Multifocal lymphoplasmacytic interstitial nephritis was also observed. A systemic herpesviral infection was suspected on the basis of gross and microscopic lesions.

Liver; dog. Multifocal hepatic necrosis and hemorrhage.
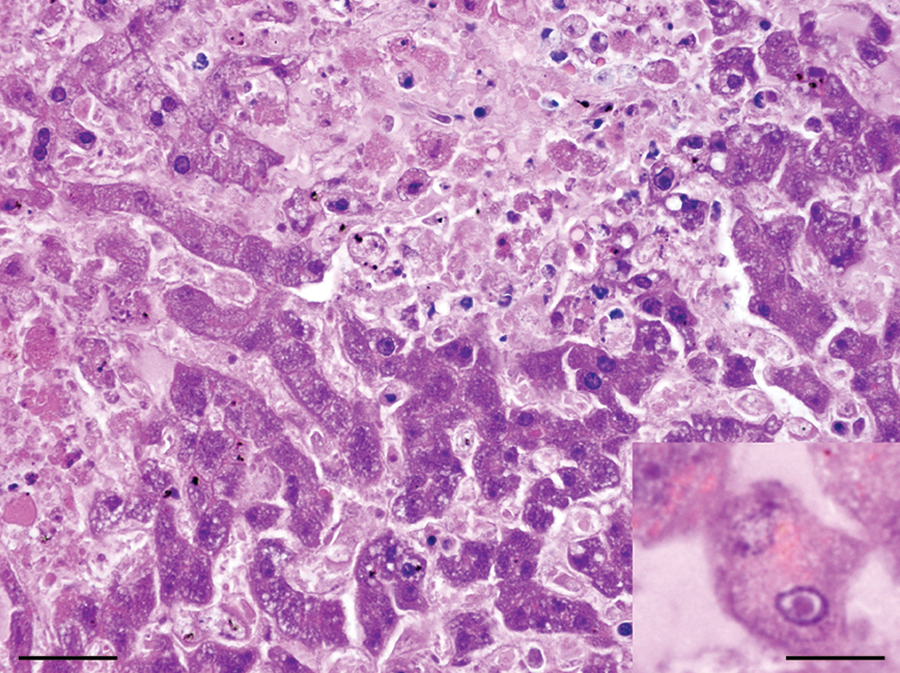
Liver; dog. The normal hepatic cord architecture is replaced by areas of necrosis and nuclear debris. Hepatocytes adjacent to the areas of necrosis contain eosinophilic, intranuclear inclusions that displace the chromatin to the periphery of the nucleus (inset). Hematoxylin and eosin. Bar = 50 µm; inset: bar = 150 µm.
Direct fluorescent antibody testing using a commercially available antibody against canine herpesvirus b was positive on cryostat sections of liver (data not shown). Polymerase chain reaction targeting a portion of the thymidine kinase gene of CaHV-1 15 was also positive. Sequencing of the purified PCR product showed that the amplified sequence was 100% homologous to other thymidine kinase sequences of CaHV-1 deposited in GenBank. Canid herpesvirus 1 antigens were detected in sections of liver immunolabeled with an automated immunostainer c using a commercial antibody specific for canine herpesvirus b and an alkaline phosphatase red detection system d (Fig. 3). Additionally, in situ hybridization for CaHV-1 was performed on serial sections of formalin-fixed, paraffin-embedded liver using an automated slide-processing system. e A digoxigenin-labeled oligonucleotide probe (DIG-5′-GATATTGTGCAAGGTATATACTCAGTACAAGACCGGAAGCTGC-3′) was designed on the basis of a highly conserved region of the thymidine kinase gene sequence of CaHV-1 (nucleotide positions 330–372, GenBank accession no. D83054). Positive intranuclear signal was detected in the areas of coagulative necrosis, confirming the presence of canine herpesviral DNA within the lesion (Fig. 4). Tissue sections of a young puppy naturally infected with CaHV-1 were used as a positive control. Duplicate attempts to isolate the virus in Madin–Darby canine kidney cells and an additional attempt in A72 cells were made with fresh, filtered liver homogenates but were unsuccessful. Polymerase chain reaction for Canine adenovirus serotype 1 was negative. 5 Canine parvovirus and Canine distemper virus were not detected by immunohistochemistry on sections of small intestine.10,14 Aerobic culture of the liver was negative.
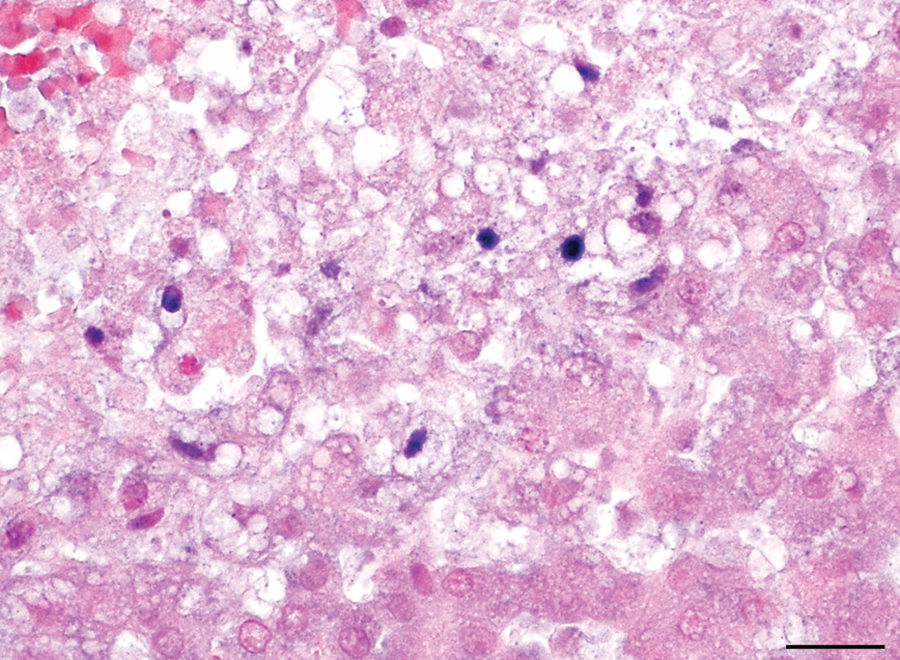
Liver; dog. Canid herpesvirus 1 proteins are detected at the periphery of the areas of hepatic necrosis. Immunohistochemistry. Bar = 50 µm.
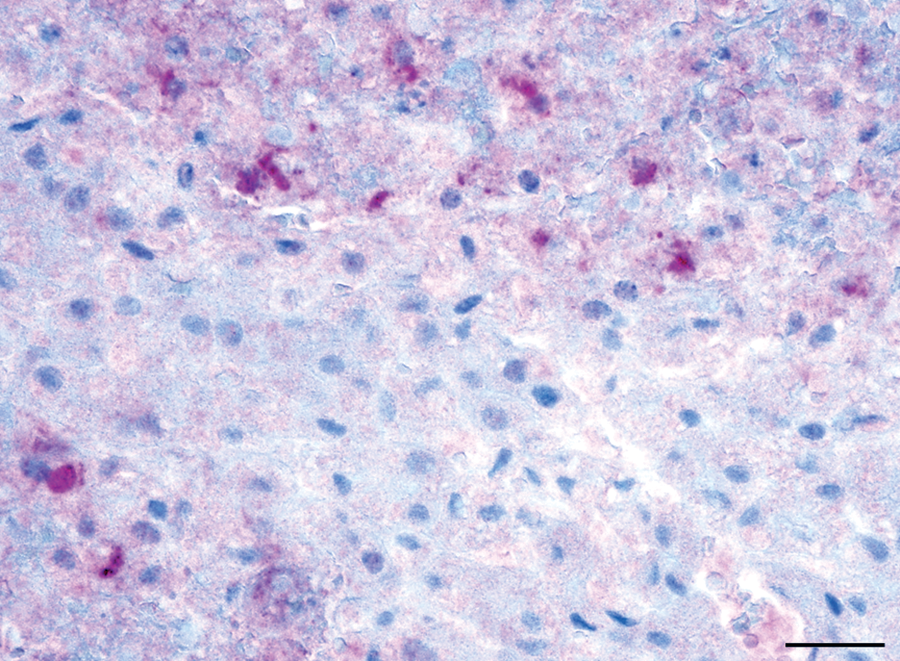
Liver; dog. Canid herpesvirus 1 nucleic acid is present at the periphery of the areas of hepatic necrosis. In situ hybridization. Bar = 50 µm.
Immunocompetent adult dogs infected with CaHV-1 usually exhibit mild, self-limiting respiratory, genital, or ocular signs. 4 Systemic disease due to CaHV-1 infection was reported in 1 adult dog, where the dog was considered to be immunocompromised secondary to chemotherapy for lymphoma. 9 In the current case, however, the dog’s medical history did not indicate the administration of glucocorticoids or any other type of immunosuppression; the complete blood cell count findings were insignificant; and there was no evidence of concurrent disease or lymphocyte depletion in lymphoid tissues at necropsy.
A hallmark of herpesvirus infections is that the acute phase of infection is followed by lifelong latency. As is typical for alphaherpesviruses, the primary latency sites of CaHV-1 are neurons within sensory nerve ganglia, such as the trigeminal, lumbosacral, and vestibular nerve ganglia.1,2,4,11-13 In addition, latent viral DNA has been detected in lymphoid tissues. 11 The virus can be reactivated when an animal becomes stressed or pregnant or receives systemic immunosuppressive doses of steroid therapy.1,4,7 In contrast, topical corticosteroid eye drops are unlikely to cause reactivation of latent CaHV-1 that result in eye lesions. 6 Differentiating between an acute viral infection and the reactivation of a latent viral infection would be difficult. It has been reported that the humoral immune response against CaHV-1 is rather short-lived. Consequently, viral reactivation in a seronegative dog cannot be excluded. 7
The fatal outcome of the CaHV-1 infection in the adult dog in the present study is unusual. Acute infection with a novel CaHV-1 strain with high virulence could explain this clinical outcome. Unfortunately, the biological properties of this virus could not be investigated, as attempts to isolate it were negative. Searching for genetic evidence of enhanced virulence determinants would require extensive sequencing of DNA from both this strain and a reference strain and was not feasible for this case. Regardless, CaHV-1 infection is a potential differential diagnosis in cases of acute fatal liver disease in adult immunocompetent dogs.
Footnotes
a.
Cerenia®, Pfizer Animal Health, New York, NY.
b.
Canine Herpesvirus FITC FA Conjugate, VMRD Inc., Pullman, WA.
c.
BenchMark, Ventana Medical Systems Inc., Tucson, AZ.
d.
Enhanced V-Red Detection kit, Ventana Medical Systems Inc., Tucson, AZ.
e.
Discovery XT, Ventana Medical Systems Inc., Tucson, AZ.
The author(s) declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication received the support of the Diagnostic Center for Population and Animal Health, Lansing, MI.
