Abstract
Anaplasma phagocytophilum was detected in dogs from Brazil in the municipalities of Seropédica and Itaguaí, Rio de Janeiro state, by real-time polymerase chain reaction (PCR) using SYBR Green to detect the amplification. Of 253 samples, 18 (7.11%) were positive, with a threshold cycle (Ct) ranging from 31 to 35 cycles. The PCR product from a positive sample was cloned and sequenced. The sequence obtained demonstrated 100% identity with other A. phagocytophilum sequences published in the GenBank database. The analytical sensitivity of RT-PCR using SYBR Green system was able to detect 3 plasmid copies when defined numbers of plasmid copies containing 122 base pairs from the msp2 gene were used. The assay was considered specific when DNA from bacteria (Anaplasma platys, Anaplasma marginale, Ehrlichia canis, Neorickettsia risticii, Rickettsia rickettsii) closely related to A. phagocytophilum was placed in the reaction. These results demonstrate that the canine granulocytic anaplasmosis agent is present in regions in which dogs could be a source of infection for tick vectors. The current study reports the detection of A. phagocytophilum, a zoonotic agent responsible for Human granulocytic anaplasmosis, in Brazilian dogs.
Anaplasma phagocytophilum (formerly Ehrlichia equi, Ehrlichia phagocytophila, and human granulocytic agent) is an obligate intracytoplasmic bacterium that belongs to the family Anaplasmataceae, order Rickettsiales. 8 The bacterium infects granulocytes, predominately neutrophils, where it reproduces and forms small colonies called morulae. Dogs, horses, cats, ruminants, llamas, and human beings can be infected, 2 and it is transmitted by ticks of the Ixodes persulcatus complex, including Ixodes scapularis and Ixodes pacificus in the United States, Ixodes ricinus in Western Europe and I. persulcatus in Eastern Europe and Asia. 5
According to serological studies conducted in endemic areas, 13 dogs naturally infected with A. phagocytophilum generally remain healthy and become asymptomatic carriers. However, clinical manifestations such as lethargy, anorexia, depression, fever, and splenomegaly 14 are commonly described. Due to the close relationship between dogs and humans as well as their ticks, along with the difficulty of diagnosing the disease from clinical signs, the zoonotic potential of A. phagocytophilum should be considered, as dogs may be an important reservoir.
The diagnosis of A. phagocytophilum infections in dogs is commonly done by indirect immunofluorescence assays. However, possible co-infections and cross-reactivity among species within the same genogroup, especially Anaplasma platys, 18 should be considered. Therefore, the diagnostic strategy has recently focused on molecular methods. 16 Polymerase chain reaction (PCR) and real-time PCR have been developed to detect A. phagocytophilum DNA in peripheral blood, buffy coats, bone marrow, and spleen tissue samples. Some of these tests can amplify DNA from other rickettsiae species, but to determine whether the positive PCR result is actually due to A. phagocytophilum, the PCR product must be sequenced. Other techniques amplify only the A. phagocytophilum DNA and aim at the 16S ribosomal RNA (rRNA) gene or the major surface protein (msp2) gene. The methods used to detect msp2 are generally more specific to A. phagocytophilum than the methods based on 16S rRNA, in which other Anaplasma species or even other bacteria, such as Bartonella henselae, 9 can be detected. Use of the msp2 gene target improved specificity because it is not present in some more distantly related bacteria and has been shown to be an excellent target for PCR detection. 16 Other targets for PCR amplification include the genes msp4, groEL, rr, and anka of A. phagocytophilum. 16 According to the above-cited author 16 the detection limit of the PCR techniques has ranged from 0.25 to 25 infected cells.
The host diversity of A. phagocytophilum suggests the presence of different genotypes in the parasite, 4 a fact that can interfere in the molecular diagnosis of the agent. 6 Thus, primers previously assessed as being able to amplify different genotypes of A. phagocytophilum 6 were used in the current study. A real-time PCR assay based on SYBR Green was developed to evaluate the occurrence of A. phagocytophilum in dogs in the municipalities of Seropédica and Itaguaí, Rio de Janeiro state, Brazil.
A total of 253 dogs were randomly selected from the campus of Rio de Janeiro Federal Rural University and urban areas of Seropédica and Itaguaí (22° 57 ’133 “S and 43° 10’ 376” W). The study was carried out between the months of November 2009 and May 2010. An epidemiological questionnaire was applied to identify factors potentially associated with infection (age, sex, and breed).
Blood samples were collected by cephalic venipuncture and placed in sterile tubes containing ethylenediaminetetraacetic acid (EDTA). Subsequently, the tubes were centrifuged at 2,500 × g for 5 min, and the buffy coat was separated. Whole DNA from the 70-µl buffy coat samples was isolated using a commercial extraction kit, a according to the manufacture’s recommendations. The A. phagocytophilum genomic DNA, obtained from immunofluorescent slides b was used as positive control. A blood sample from an A. phagocytophilum–negative dog and water were used as negative controls. Carried out in triplicates, a real-time PCR assay based on the msp2 gene was performed using the primers 903f (5’-AGTTTGACTGGAACACACCTGATC-3’) and 1024r (5’-CTCGTAACCAATCTCAAGCTCAAC-3’). 6 The final reaction volume was 12 µl and contained 6 µl of Power SYBR Green PCR master mix c (2×), 0.9 µM of each primer, and 3 µl (approximately 90 ng) of total DNA. The optimized thermal cycler program was 95°C for 10 min, followed by 40 cycles of 95°C for 15 sec and annealing extension at 60°C for 1 min. After amplification, a dissociation curve was obtained to verify the specificity of amplification. Samples were considered positive with threshold cycle (Ct) levels less than 35 cycles and with amplified product at the same dissociation temperature as the positive control.
To check contamination of reagents and enzymes, a tube containing all PCR components, except the DNA sample, was included in each reaction. The DNA sample was replaced by an equivalent amount of ultrapure water. Filter tips were used in all stages of the experiment, and the procedures for DNA extraction, cloning, and PCR reactions were performed in separate rooms. The PCR was performed in a closed system.
Only one PCR product of A. phagocytophilum was purified, cloned, and sequenced. 17 Cloning was performed in plasmid, d according to the manufacturer’s recommendations. Plasmid DNA from positive clones was isolated by using a commercial kit e and submitted for sequencing. f Multiple sequence alignment was performed using the Basic Local Alignment Search Tool (BLAST) to evaluate the identity of the msp2 gene of A. phagocytophilum.
The analytical sensitivity of the assay was determined by evaluation of serial decimal dilutions of the amplicon cloned into plasmid pGEM-T. 1 The real-time PCR sensitivity was evaluated with and without 1 µl of buffy coat DNA from an unaffected dog. Plasmid DNA concentration and purity were verified using a spectrophotometer. g A curve of the plasmid copy number versus Ct values was plotted to determine the analytical sensitivity of the real-time PCR based on SYBR Green detection. The number of copies ranged from 3 to 3 × 106, with 6 separate dilution series performed for each point on the curve. A melt curve was also added at the end of this test to verify the specificity and ensure the primers’ efficiency.
The specificity of the assay was evaluated using DNA from related organisms and other pathogens transmitted by ticks, such as A. platys, Anaplasma marginale, Ehrlichia canis, Neorickettsia risticii, Rickettsia rickettsii, Babesia canis vogeli, Hepatozoon canis, and Theileria equi. The real-time PCR results were associated with sex, age, and breed using the chi-square test at 5% significance.
Eighteen (7.11%) of the 253 samples tested by real-time PCR had specific amplification for the msp2 gene. The average Ct value observed in the positive samples was 33.98 ± 0.5 cycles, ranging from 31 to 35 cycles. The melting temperature (Tm) of the amplified products showed had an average value of 77.97 ± 0.72°C, similar to the positive control (Fig. 1).
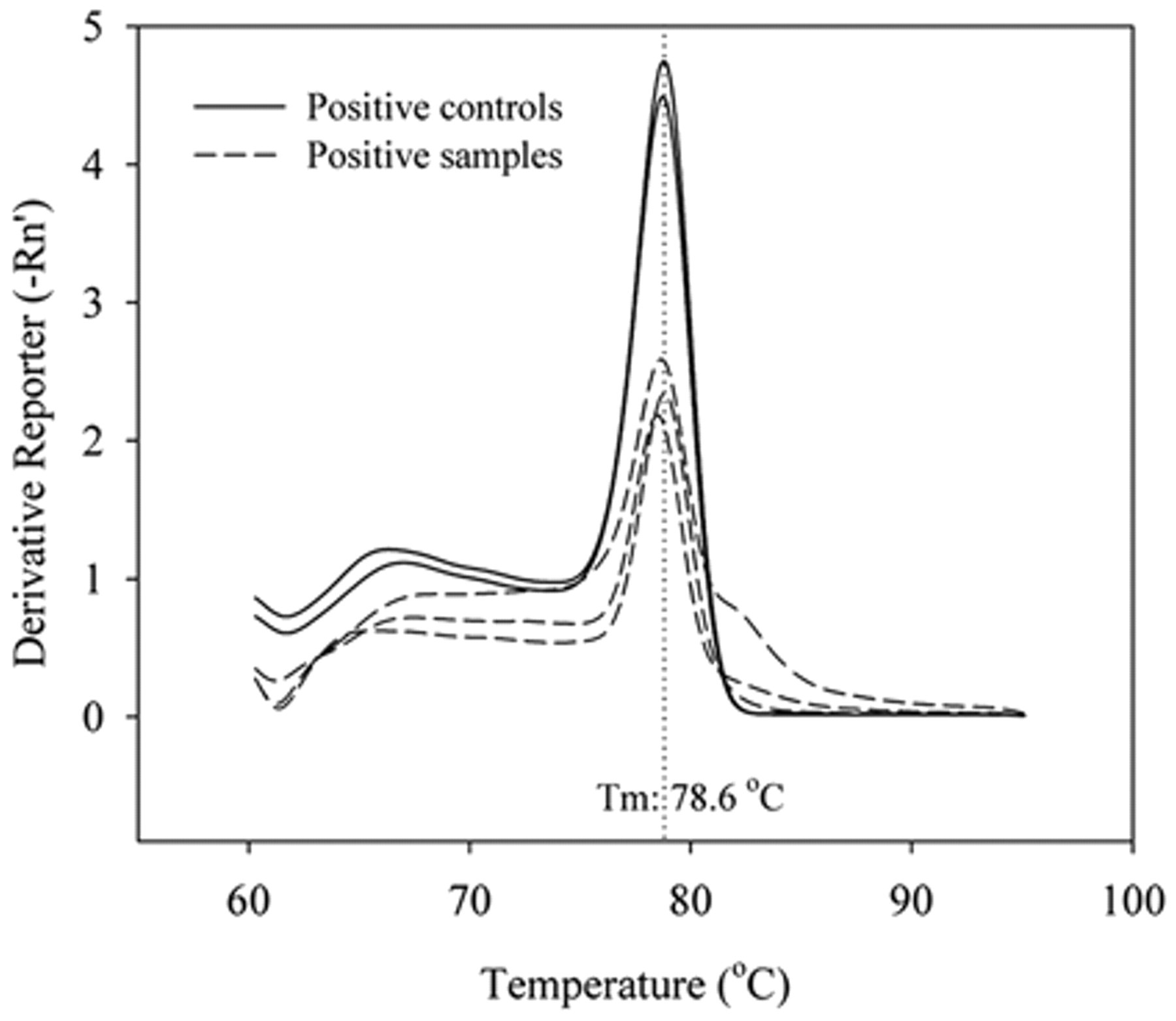
Positive control melt curve of the 122 base pair (bp) fragment from msp2 gene and melt curve of positive dogs’ samples, showing the specificity of real-time polymerase chain reaction for amplification of Anaplasma phagocytophilum using the SYBR Green system. Tm = melting temperature.
One positive sample was cloned, sequenced, and deposited in the GenBank database under the accession number HQ670750. The BLAST analysis revealed 100% identity [(122/122 base pair (bp)] for the msp2 gene with strains of A. phagocytophilum isolated from I. persulcatus ticks collected in the vegetation on wooded areas in Japan (accession no. FJ600612.1); from the blood of 3 dogs that were clinically ill with granulocytic anaplasmosis in Sweden and from 2 sheep experimentally infected with a cryopreserved isolate of A. phagocytophilum from the blood of a Norwegian flock with tick-borne fever (accession nos. DQ519566 and DQ519565). The sample sequenced in the current study also showed 100% identity with other isolates of A. phagocytophilum obtained from the blood of horses and human beings in the United States (accession nos. AY763480 and P000235).
Using a set number of insert-vector copies, the analytical sensitivity of real-time PCR using SYBR Green system was evaluated. The detection limit of the technique was 3 plasmid copies, each containing an msp2 gene from A. phagocytophilum. The correlation coefficient of the seven dilutions tested in the standard curve was 99%, with Ct ranging from 15.15 ± 0.17 cycles in the first dilution (3 × 106 plasmid copies) to 33.91 ± 1.07 cycles in the last dilution (3 plasmid copies; Fig. 2). The sensitivity remained the same with the addition of buffy coat DNA from an unaffected dog. The curves had the same slope during the exponential phase of amplification, demonstrating the reaction’s efficiency even a points with few targets (Fig. 3). When real-time PCR specificity was evaluated in relation to other closely related bacteria, only A. phagocytophilum DNA produced a positive fluorescent signal.
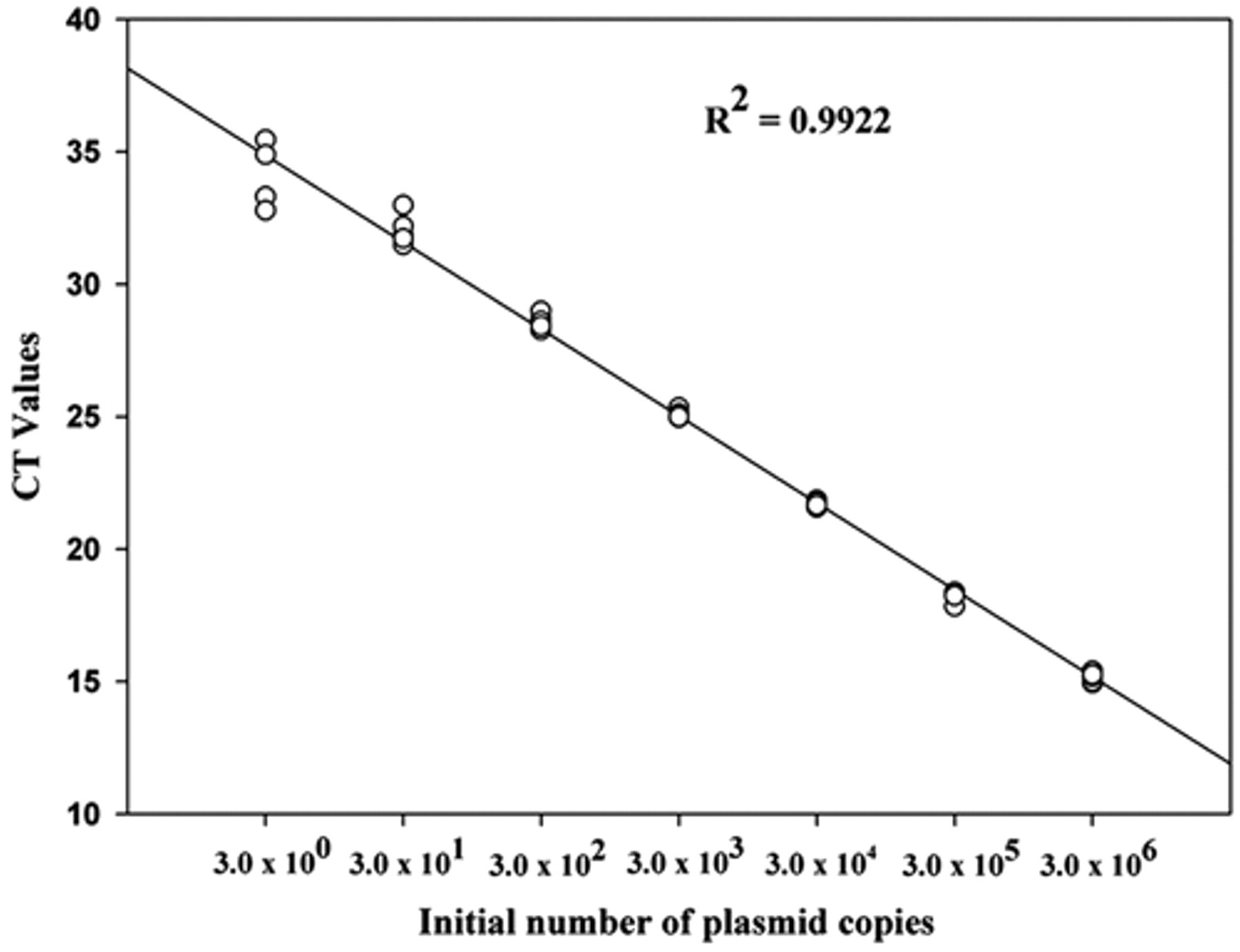
Standard curve plotted from serial decimal dilutions of plasmid DNA, containing the Anaplasma phagocytophilum msp2 gene. The threshold cycle (Ct) value obtained by real-time polymerase chain reaction using SYBR Green system was plotted as a function of the initial number of plasmid copies.
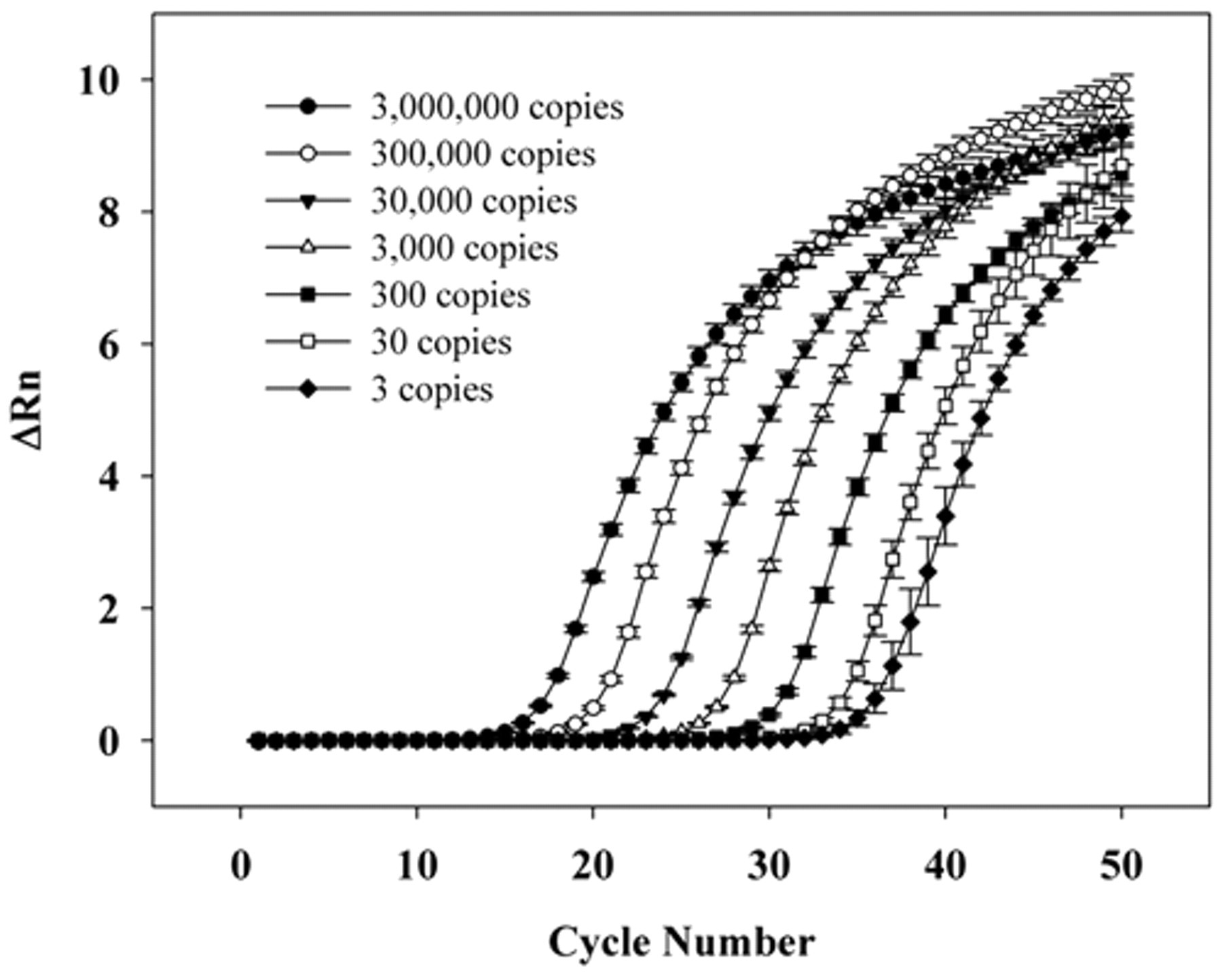
Real-time polymerase chain reaction analytical sensitivity, showing the amplification curves of plasmid DNA, containing the Anaplasma phagocytophilum msp2 gene, in serial dilutions (3–3,000,000 copies).
In the present study, no significant association was found (p > 0.05) with sex, breed, and age of dogs with positive results from real-time PCR detection. Regarding sex, 9 male and 9 female dogs were positive by PCR. Infection with A. phagocytophilum was more frequent (55.55%, n = 10) in dogs between 5 and 10 years old, followed by dogs between 2 and 5 years old (16.66%, n = 3), 6 months and 2 years old (11.11%, n = 2), and 1 dog over 10 years old (5.55%, n = 1). Among the positive dogs, 66.6% (n = 12) had no defined breed, while there were 2 Labrador Retrievers, 1 Poodle, 1 Rottweiler, 1 Doberman Pinscher, and 1 Border Collie.
The current study reports on the detection of A. phagocytophilum infection in Brazilian dogs, which suggests that the potentially zoonotic agent responsible for human granulocytes anaplasmosis currently exists in Brazil. Although the presence of A. phagocytophilum immunoglobulin G antibodies had been previously reported in Brazilian dogs, 12 the molecular diagnosis of this rickettsia has not been achieved yet, in spite of previous attempts using the nested PCR technique, 3 which could be attributed to the low sensitivity of the molecular methods used.
The development of techniques for accurate detection of DNA fragments in real time was a significant advance in PCR technology. Real-time PCR has several advantages over conventional PCR, including speed, simplicity, sensitivity, reproducibility, quantitative capability, and low risk of contamination. 11
The greater analytical sensitivity of the msp2 assay is likely due to the use of SYBR Green PCR master mix, which is more efficient amplification chemistry. The SYBR Green assays can increase detection limits as much as 10–100-fold over ethidium bromide–stained agarose gel, 7 which is traditionally used for the detection of PCR amplification. The primers used in the current study presented high sensitivity to different strains A. phagocytophilum, including strains from the Upper Midwestern United States, Europe, China, and Guatemala. 6 As such primers may be very useful in investigations of anaplasmosis in other regions, where the organism is likely to occur, they were used in the present study. The above-cited study 6 evaluated 97 dogs using real-time PCR as diagnostic method, targeting 16S rRNA, and none of the samples was considered positive, while when the mps2 gene was used as a target, 7 dogs were positive for A. phagocytophilum. One likely reason for the excellent analytical sensitivity of the msp2 assay is that the primers can amplify each of the multiple copies on the msp2 gene that are present in the A. phagocytophilum genome. 16
The real-time PCR using SYBR Green system developed in the current study had high sensitivity and reproducibility to diagnose A. phagocytophilum infection in dogs (Fig. 2). The results indicated that rickettsia parasitemia in dogs was very low, which may explain the relatively high values of Ct observed. The detected low rickettsia parasitemia in dogs would explain why other studies conducted in Brazil have failed to detect A. phagocytophilum infection in dogs. 3
The current study was conducted between the months of November and May, when there is increased activity of the adult stage of Amblyomma cajennense. 15 Although Ixodes amarali have been detected parasitizing skunks of the genus Didelphis in Seropédica, 10 there are no parasitism reports in dogs by Ixodes species to date. These data suggest the possibility that A. cajennense may be involved in the transmission of A. phagocytophilum in the region studied. In Berlin, A. phagocytophilum infection in dogs was higher between the months of April and September. The disease’s seasonal distribution most likely coincides with the activity peak of nymphal and adult tick species of Ixodes, suggesting that adults could be the main vectors. 14 The results are similar to those reported by a present study regarding the seasonality of A. cajennense.
While not statistically significant, the finding that frequency of infection increases with age is consistent with previous studies. 9,14 The detection of A. phagocytophilum in Brazilian dogs is the starting point for further studies of cell culture isolation, molecular characterization of strains, and identification of potential vectors in Brazil. The objective of further research will focus on additional genotypes that may be circulating in Brazil. Such studies would improve epidemiological surveillance aimed at the prevention of new A. phagocytophilum infections in Brazil and other South American countries.
Footnotes
a.
DNeasy Tissue Kit, Qiagen Inc., Valencia, CA.
b.
Fuller Laboratories, Fullerton, CA.
c.
Power SYBR® Green PCR Master Mix, Applied Biosystems, Foster City, CA.
d.
pGEM-T® Easy Vector System, Promega Corp., Madison, WI.
e.
Plasmid Miniprep PureYield™ System, Invitrogen Corp., Carlsbad, CA.
f.
ABI Prism®310 Genetic Analyser, Applied Biosystems, Foster City, CA.
g.
NanoDrop 2000c, Thermo Scientific, Wilmington, DE.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
The Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the finantial support.
