Abstract
A 6-year-old, 30-kg, female German Shepherd Dog, living in a leishmaniasis enzootic area, was presented with a severe rear limb motor disorder and a medical history of acute onset of fever. Routine hematology indicated neutrophilia. Spinal survey radiographs were consistent with osteomyelitis and discospondylitis. Because of the poor clinical prognosis and the painful nature of the lesions, the dog was euthanized at the owners' request. At necropsy, T13-L1 vertebrae had large areas of necrosis within the vertebral bodies. Histopathological findings were consistent with chronic, severe, fungal osteomyelitis and discospondylitis. Polymerase chain reaction identified Scedosporium apiospermum, a eutrophic filamentous fungus now recognized as an emerging agent of severe infections in immunosuppressed human patients.
Scedosporium apiospermum, 1 of 2 anamorphs of Pseudallescheria boydii, is a eutrophic filamentous fungus commonly isolated from soil, vegetation, polluted water, and animal feces in temperate climates. 6,16 It is now recognized as an emerging agent of severe fungal infections in immunosuppressed human patients. In humans, S. apiospermum has been classically implicated in subcutaneous infections and in asymptomatic pulmonary colonization, whereas systemic infections are more frequently observed in patients with an impaired immune system. 8 Few reports of scedosporiosis have been reported in animals, most of which are of traumatic origin. Scedosporium apiospermum infections in dogs have been associated with nasal infection, mycetoma, or keratomycosis. 4,15
A 6-year-old, 30-kg, female German Shepherd Dog was presented with a rear limb motor disorder. This dog lived both indoors and outdoors in a leishmaniasis enzootic area in the south of France. Deworming and vaccination had been conducted on a regular basis. Four weeks before presentation, the dog had an acute onset of fever, which resolved after a 10-day administration of enrofloxacin a (5 mg/kg, orally, once a day). However, the fever recurred after treatment was discontinued. Simultaneously, an insidious onset of a posterior motor disorder was observed, which evolved from paresia to paralysis 3 days before referral. The intravenous corticosteroid treatment by the referring veterinarian resulted in slight amelioration of this neurological disorder.
At presentation, body temperature was normal (38.4°C), mucosal surfaces appeared pink, and cardiac and respiratory examinations were unremarkable. There was a marked paravertebral muscular atrophy. The rear limbs were paretic with a severe proprioceptive deficit involving both rear limbs. Neurological examination showed increased patellar, cranial tibial, and femoral reflexes. The urinary bladder was full and could not be mechanically emptied. Palpation of the spine evoked severe pain at the thoracolumbar and lumbosacral junctions. The panniculus muscular reflex was absent posterior to the thoracolumbar junction. Hip manipulation was difficult, and creaking sounds were heard.
Routine hematology indicated leukocytosis (16,050 cells/μl; reference [ref.] interval: 5,000–15,000 cells/μl) with mature neutrophilia (11,765 cells/μl; ref. interval: 3,000–10,500 cells/μl). The other hematologic variables were within reference intervals. Serum total alkaline phosphatase activity, alanine aminotransferase activity, urea concentration, and creatine concentration were also within reference intervals. Biochemical abnormalities included hyperproteinemia (91 g/l; ref. interval: 52–82 g/l) with an albumin/globulin ratio <0.7 (ref. interval: 0.92–1.30). Serum protein electrophoresis revealed hyperproteinemia with normoalbuminemia (34.8 g/l; ref. interval: 22–35 g/l), normoalphaglobulinemia (alpha1, 2.6 g/l, ref. interval: 5–8 g/l; alpha2, 7.1 g/l, ref. interval: 5–10 g/l), hyperbetaglobulinemia (beta1, 6.2 g/l, ref. interval: 5–11 g/l; beta2, 13.0 g/l; ref. interval: 3–7 g/l), and hypergammaglobulinemia (27.3 g/l; ref. interval: 5–18 g/l). The serum protein electrophoretic pattern indicated a polyclonal gammopathy, consistent with a general antibody response associated with infection. Urinalysis was performed, and abnormalities were not found. Serological testing for Leishmania infantum (in-house immunochromatographic test on membrane) b was positive. Cytological examination of bone marrow smears revealed amastigotes and phagocytized erythroblasts within macrophages, megakaryocytic dyspla-sia, and neutrophilic hyperplasia. These results were consistent with active leishmaniasis.
Spinal survey radiographs disclosed narrowing of the vertebral canal at the lumbosacral junction (cauda equina syndrome) as well as moderate to marked bilateral hip arthrosis. In addition, a large area of osteolysis and osteoproliferation involved the T13 and L1 vertebrae (Fig. 1). These vertebral lesions were consistent with osteomyelitis and discospondylitis; however, a bone tumor could not be excluded. Because of the poor clinical prognosis and the painful nature of the osseous lesions, the dog was euthanized at the owners' request.
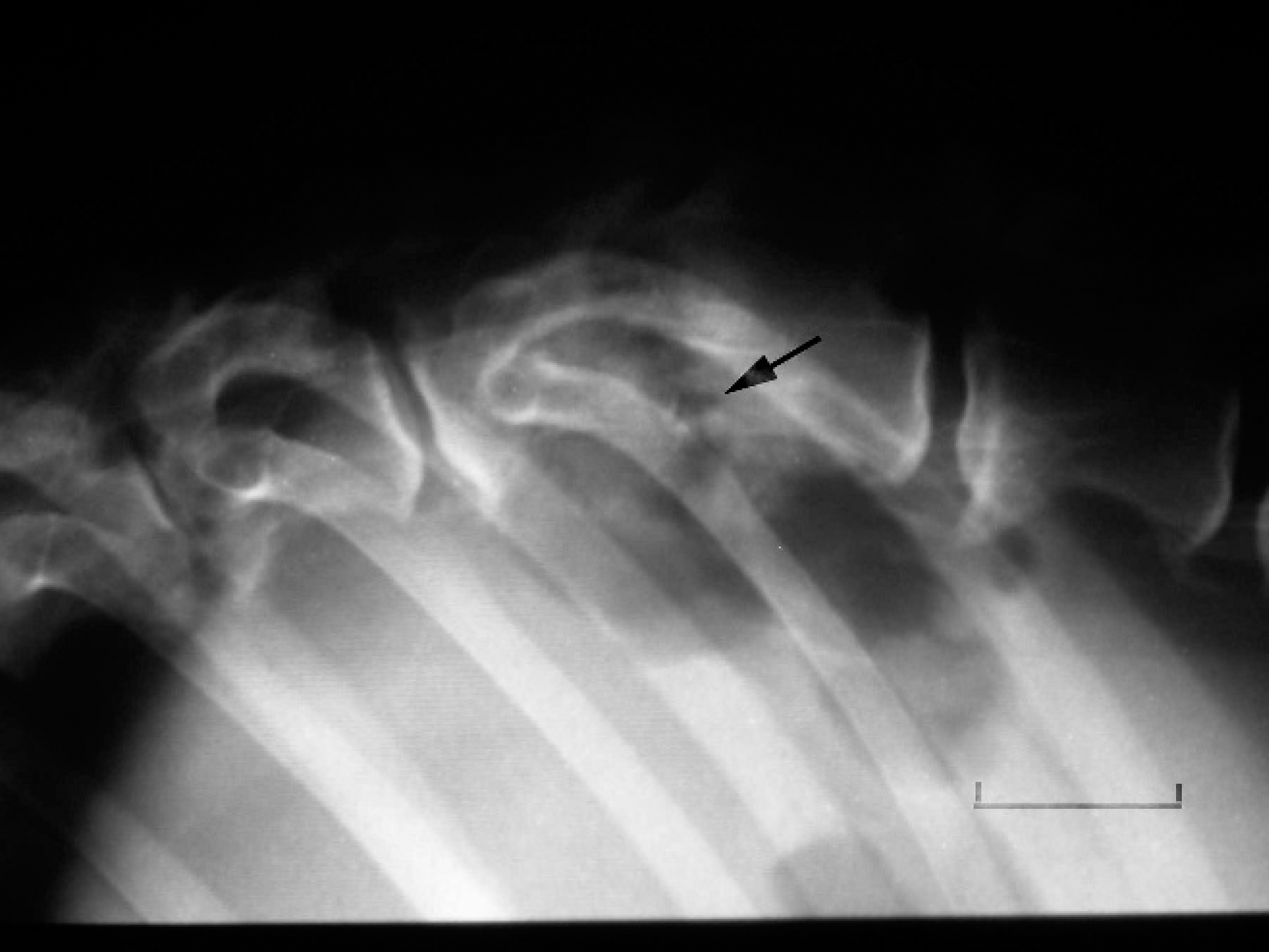
Lateral spinal survey radiograph. Notice the areas of osteolysis and osteoproliferation involving the T13 and L1 vertebrae (arrow). The radiographic changes are consistent with discospondylitis and vertebral osteomyelitis. Bar = 3 cm.
At necropsy, the T13 and L1 vertebrae had large foci of necrosis within the vertebral bodies (Fig. 2). The outlines of both vertebrae were uneven, and there was destruction of the intervertebral disk. The spinal cord appeared normal. Except for the paravertebral muscular atrophy, necropsy findings were otherwise unremarkable.
Histological examination of the T13 and L1 vertebrae showed severe diffuse pyogranulomatous osteomyelitis and discospondylitis with marked osteolysis and periosteal proliferation of reactive bone. Inflammatory infiltrates consisted mainly of epithelioid macrophages admixed with a few multinucleate Langhans-type giant cells and a moderate number of neutrophils. Numerous foci of necrosis were also present. On hematoxylin and eosin stain, many round to oval structures with a birefringent capsule were present that measured 10–14 μm in diameter. Thin, 8–10-μm-diameter, septate, branching fungal hyphae were observed and were best visualized in periodic acid-Schiff (PAS) stained tissue sections (Figs. 3, 4). The capsule of the round structures were PAS negative. The histopathological findings were consistent with chronic, severe, pyogranulomatous, fungal osteomyelitis and discospondylitis. As the fungus could not be speciated by conventional light microscopy, a polymerase chain reaction (PCR) analysis was performed on formalin-fixed, paraffin-embedded samples. DNA was extracted using a commercial extraction kit. c Portions of the internal transcribed spacer (ITS) and 5.8S ribosomal RNA (rRNA) genes were amplified using primers ITS1 and ITS4, purified, and sequenced. 18 The sequencing reaction was performed in a 10-μl volume containing 50 ng of sample DNA, 4 pmol of primers, and 4 μl of BigDye Mix. d The positive control mixture was similar but used 50 ng of target DNA that had been extracted from Aspergillus fumigatus (strain CBS 144.89). The negative control specimen was prepared by adding 2 μl of distilled water (instead of DNA extract) to the mix. The positive PCR amplification obtained with DNA from a single paraffin-embedded tissue block is shown in Figure 5. The unique PCR product was analyzed on a genetic analyzer. e The amplicon sequence was directly compared to other fungal sequences with BLASTn (nucleotide database using a nucleotide query; www.ncbi.nlm.nih.gov/blast/Blast.cgi). The sequence of the amplicon showed 100% similarity with that of Pseudallescheria boydii (teleomorph of S. apiospermum) reference strain CBS 101723 (GenBank accession no. AY878947). Thus, S. apiospermum was the etiology of the vertebral osteomyelitis and discospondylitis in this dog.

The T13 and L1 vertebrae, dissected at necropsy, contain large foci of necrosis within the vertebral bodies (arrows). The contour of both vertebrae are uneven, and there is destruction of the intervertebral disk (arrowhead). The spinal cord appears normal.
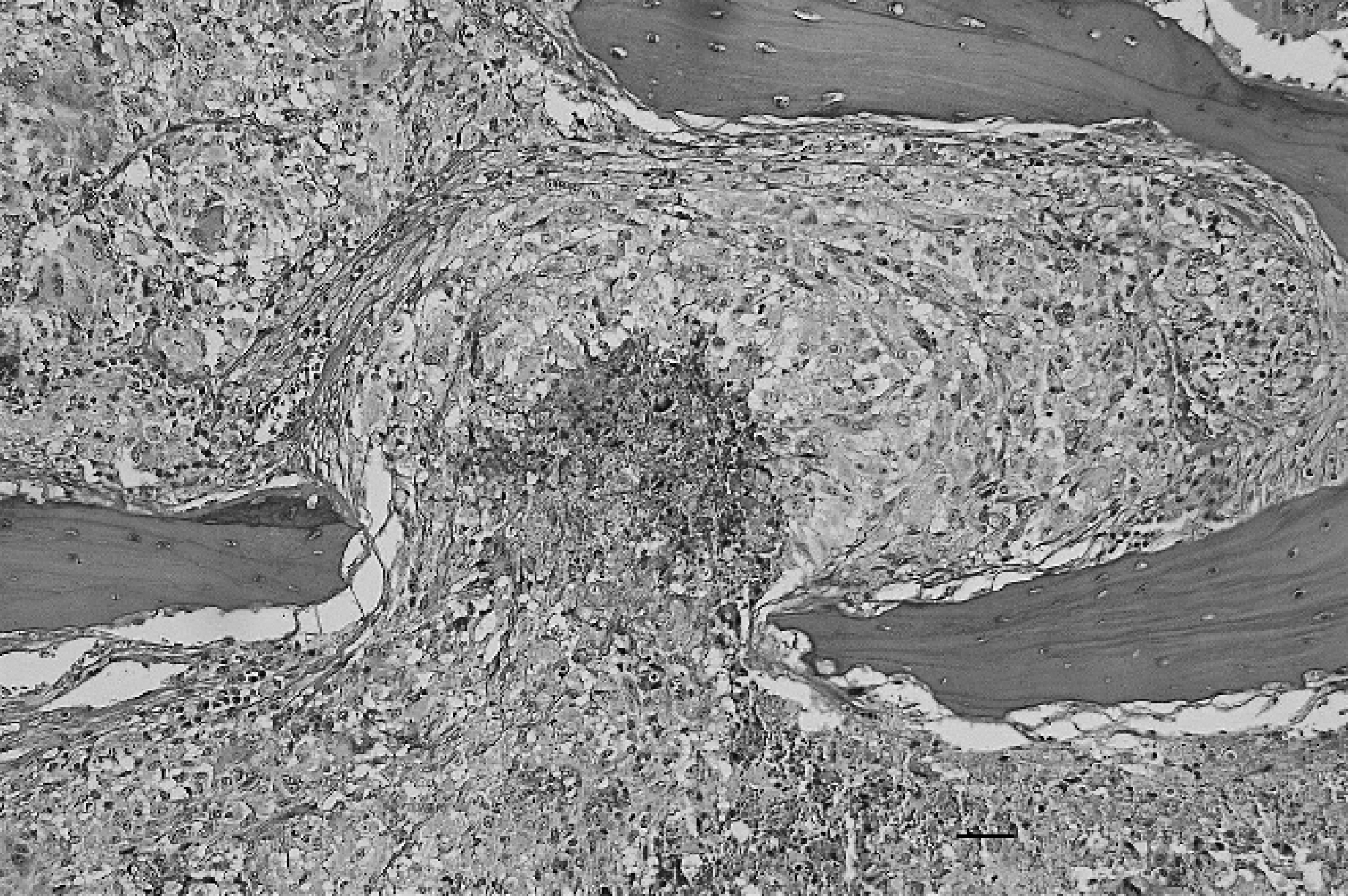
Pyogranulomatous inflammation of the bone marrow with a focus of central necrosis. Note the severe osteolysis of the bone trabeculae. Hematoxylin and eosin. Bar = 40 μm.
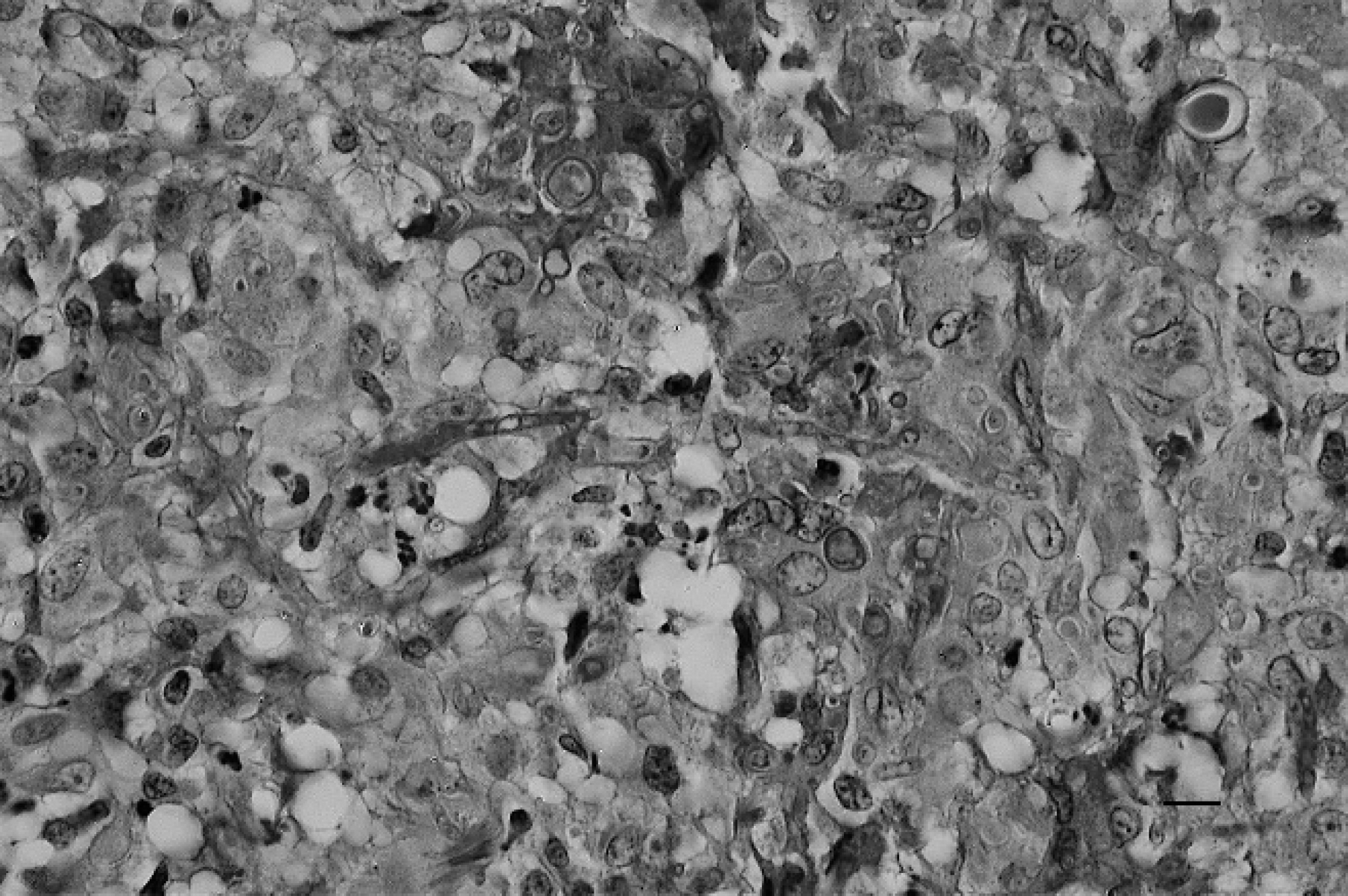
Pyogranulomatous infiltrate with thin, septate, branching hyphae and large encapsulated spores. Periodic acid–Schiff stain. Bar = 10 μm.

Ethidium bromide-stained, agarose, polymerase chain reaction (PCR) gel. Lane 1: positive amplification of Scedosporium apiospermum DNA from paraffin-embedded sample; lane 2: PCR positive control (DNA from Aspergillus fumigatus strain CBS 144–89); lane 3: PCR negative control (distilled water substituted for DNA template); lane 4: 1-kb molecular marker.
Scedosporium apiospermum is a eutrophic filamentous fungus that is common in temperate climates. It can be isolated from soil or potted plants and is able to grow in a poorly aerated environment. Its growth is promoted by manure-enriched or polluted environments. 6,16 Systemic diseases caused by S. apiospermum have been referred to as allescheriasis, graphitosis, monosporiosis, petriellidiosis, pseudallescheria, pseudallescheriasis, or scedosporiosis. 10 This organism is now recognized as a potent and emerging pathogen of severe infection in immunosuppressed human patients.
Reports of scedosporiosis in animals are infrequent. To date, S. apiospermum infections in dogs have been associated with nasal infection, mycetoma, or keratomycosis. 1,2,4,15 Only 2 cases of Scedosporium inflatum osteomyelitis have been described in veterinary medicine, 1 in an adult beagle dog involving the humerus 14 and another in a horse with arthritis and osteomyelitis of the metacarpophalangeal joint. 17
In humans, the clinical presentation of scedosporiosis is largely dependent on the fungal strain, portal of entry, and immune status of the patient. 3,10 Three clinical syndromes can be distinguished: 1) localized disease after trauma, such as subcutaneous mycetoma, arthritis, osteomyelitis, endophthalmitis, or onychomycosis; 2) large asymptomatic or symptomatic colonization of cavities, mainly pulmonary cases, but also sinusitis or otitis; and 3) invasive systemic diseases such as pneumonia, endocarditis, or central nervous system disorders. 10 Classically, S. apiospermum has been implicated in subcutaneous infections and asymptomatic pulmonary colonization. Systemic infections are more frequently observed in patients with an impaired immune system or in victims of near drowning 10 ; however, infections also have been documented in immunocompetent patients. 8 Vertebral osteomyelitis associated with S. apiospermum is infrequent in humans, with less than 10 cases reported in the medical literature. 10 Progressive spinal pain is the most common presentation in human patients. Systemic fungal infections in humans are usually related to direct extension of pulmonary infections, hematogenous spread, or direct inoculation of the organism. In most of these reports, dissemination of mycotic infection often involves the central nervous system.
Most cases of S. apiospermum infection in dogs and horses are of traumatic origin. 7,12,14,15 In the dog of the present report, the portal of entry of S. apiospermum was unknown; the owner did not mention the presence of wounds or nasal discharge before presentation of the dog for illness. The relationship between scedosporiosis and leishmaniasis in this dog is also unclear. However, phagocytic capabilities of macrophages from Leishmania-infected animal models (mainly mouse and hamsters) are altered via disturbed cytokine production. 9 In northeastern Spain (an endemic area of leishmaniasis), a 2007 report demonstrated that chronic infection with leishmaniasis may predispose dogs to secondary vector-borne infections (Tabar L, Francino O, Altet L, et al.: 2007, PCR-based of coinfection with multiple vector-borne pathogens related to leishmania infantum infection in dogs from north eastern Spain [Abstract], p. 203. Seventeenth European Society of Veterinary Internal Medicine-Companion Animals Congress, September 13–15, Budapest, Hungary). Thus, chronic leishmaniasis may explain the unusual localization of S. apiospermum infection in this dog to the thoracolumbar vertebrae. Another explanation may be the predisposition of German Shepherd Dogs to develop fungal infection (especially disseminated aspergillosis) and discospondylitis. There also may be a relationship in this breed with defective immunoglobulin A production and altered mucosal immunity, which may allow initial penetration of the fungus. 5
Scedosporium infections are difficult to diagnose clinically, and histological lesions are similar to those due to Aspergillus sp. and other hyphomycetes. Routine special stains, such as PAS, do not discriminate S. apiospermum from other related fungi. Therefore, mycological culture is recommended for specific identification. When culture has not been performed or only formalin-fixed tissues are available for study, as in the current case, PCR analysis and genetic sequencing of the amplicons may be used to specifically identify the fungus from infected tissues. For example, a previous study distinguished S. apiospermum from A. fumigatus by an in situ hybridization technique using rDNA probes. 11
Scedosporiosis is often resistant to treatment with traditional antifungal drugs such as amphotericin B, miconazole, or itraconazole. When possible, surgical excision of infected tissue is the optimal form of treatment. In one human patient, successful treatment of a chronic, fungal, vertebral infection was achieved with a combination of surgery (abscess debridement, surgical drain placement, ostectomy, osseous autograft implantation, and vertebral stabilization), drug administration (intravenous and oral voriconazole administration for at least 1 year), and bed rest. 8,13 In dogs, systemic scedosporiosis is usually fatal; however, localized infection such as that involving the nasal cavities may be cured with appropriate treatment. One previous study described treatment of nasal fungal infection by endoscopic removal of the fungal mass, which resulted in clinical improvement within 2 weeks. 4 In another case report, a nasal granuloma was treated by oral administration of ketoconazole (10 mg/kg of body weight, twice a day). Medication decreased the nasal discharge after 1 month of treatment and was continued until the dog died in a car accident 7 months later. 2
To the authors' knowledge, this is the first description of chronic vertebral osteomyelitis and discospondylitis due to S. apiospermum infection in the dog. Moreover, S. apiospermum infection has never been reported in a dog from France. The present case report documents that S. apiospermum may cause vertebral osteomyelitis and discospondylitis in dogs. Furthermore, the clinical presentation and histological findings of S. apiospermum infection may be similar to those of A. fumigatus infection. Specific identification of S. apiospermum, A. fumigatus, and other hyphomycetes is best accomplished by techniques such as mycological culture, PCR analysis, or immunostaining.
Footnotes
a.
Baytril® Taste Tabs™ Antibacterial Tablets, Bayer AG, Leverkusen, Germany.
b.
Speed® LEISH, Virbac France SAS, Carros cedex, France.
c.
DNeasy® Tissue Kit, Qiagen SA, Courtaboeuf cedex, France.
d.
Applied Biosystems, Courtaboeuf cedex, France.
e.
ABI PRISM™, Applied Biosystems, Courtaboeuf cedex, France.
