Abstract
An effective live animal diagnostic test is needed to assist in the control of chronic wasting disease (CWD), which has spread through captive and wild herds of white-tailed deer (Odocoileus virginianus) in Canada and the United States. In the present study, the diagnostic accuracy of rectal mucosa biopsy sample testing was determined in white-tailed deer from 4 CWD-infected captive herds. Specifically, the current study compared the immunohistochemical detection of disease-associated prion protein in postmortem rectal mucosa biopsy samples to the CWD status of each deer as determined by immunodiagnostic evaluations of the brainstem at the obex, the medial retropharyngeal lymph node, and the palatine tonsil. The effects of age, sex, genotype, and disease progression were also evaluated. Diagnostic sensitivity on rectal biopsy samples for CWD in white-tailed deer ranged from 63% to 100%; the pooled estimate of sensitivity was 68% with 95% confidence limits (95% CLs) of 49% and 82%. However, diagnostic sensitivity was dependent on genotype at prion protein gene (PRNP) codon 96 and on disease progression as assessed by obex grade. Diagnostic sensitivity was 76% (95% CLs: 49%, 91%) for 96GG deer but only 42% (95% CLs: 13%, 79%) for 96GS deer. Furthermore, diagnostic sensitivity was only 36% for deer in the earliest stage of disease (obex grade 0) but was 100% for deer in the last 2 stages of preclinical disease (obex grades 3 and 4). The overall diagnostic specificity was 99.8%. Selective use of antemortem rectal biopsy sample testing would provide valuable information during disease investigations of CWD-suspect deer herds.
Introduction
Chronic wasting disease (CWD) is a transmissible spongiform encephalopathy of cervid species with naturally occurring infections identified in Canada and the United States in white-tailed deer (Odocoileus virginianus), mule deer (Odocoileus hemionus), Rocky Mountain elk (Cervus elaphus nelsoni), and moose (Alces alces shirasi).1,18,19 Like other transmissible spongiform encephalopathies, CWD has a long incubation period lasting months to years, eventually leading to the accumulation of abnormal protease resistant prion protein (PrPCWD) in the brain, resulting in neurologic disease and death. 18
Chronic wasting disease was first identified in Colorado and has since been found in both domestic and wild cervids in expanding regions of North America.18,19 Chronic wasting disease has been identified in the provinces of Saskatchewan and Alberta in Canada and in 19 states across the United States (http://www.nwhc.usgs.gov/disease_information/chronic_wasting_disease/). Control of the disease has been hampered by the lack of a highly sensitive and specific antemortem diagnostic test method. 13 Current methods in Canada and the United States utilize brain stem, medial retropharyngeal lymph node, and palatine tonsil samples collected postmortem, which are then tested by immunohistochemistry (IHC), enzyme-linked immunosorbent assay (ELISA), and/or Western blot (WB) to detect PrPCWD. In white-tailed deer, PrPCWD accumulates within multiple lymphoid tissues such as lymph nodes, tonsil, and gut-associated lymphoid tissue during the subclinical phase of disease, prior to the accumulation of PrPCWD within the brain.7,8 Protease-resistant prion protein in white-tailed deer has a similar tissue distribution to scrapie within sheep and goats, and this wide tissue distribution of abnormal prion protein has been exploited by the utilization of antemortem rectal biopsy testing for the control and eradication of scrapie in Canada since 2006 and in the United States since 2008.3–5 Scrapie diagnostic testing on rectal biopsy samples by the U.S. Department of Agriculture (USDA) is used in specific types of epidemiologic investigations and has been found to be a safe and effective method in live animals. 3 In white-tailed deer, biopsy samples from palatine tonsil and rectal mucosa have both shown promise as possible antemortem methods to detect CWD.7,17,20 The rectal biopsy procedure has the advantages of being a simpler and more rapid procedure that can often be performed without sedation. 7
The purpose of the current study was to determine the CWD diagnostic accuracy of immunohistochemical detection of PrPCWD in rectal biopsy samples of white-tailed deer from known positive herds. Through a cooperative effort between the Canadian Food Inspection Agency (CFIA) and the USDA, rectal biopsy and other tissue samples were collected from white-tailed deer in 4 herds, each herd depopulated because of a previous diagnosis of CWD. All tissue samples were collected postmortem. Detection of PrPCWD in rectal biopsy samples was compared to the CWD infection status of each animal as determined by the official test methods of each country utilizing samples of the brainstem, retropharyngeal lymph node, and tonsil. All rectal biopsy samples were tested by IHC at a single laboratory. In addition, the effect of age, sex, genotype at prion protein gene (PRNP) codon 96, and stage of disease progression on the diagnostic accuracy of rectal mucosa biopsy testing was examined.
Materials and methods
Study populations
The white-tailed deer captive herds in the present study included 3 herds in Canada (Canada herds 1–3) and 1 herd in the United States (U.S. herd 1). For the purposes of the investigation, the study populations consisted of the deer euthanized during depopulation of each farm. At the time of depopulation, clinical signs of CWD were not evident in any deer from these herds. Canada herd 1 was a 65-hectare breeding operation established in northeastern Saskatchewan during the spring of 2005. Chronic wasting disease was first diagnosed in 2 deer that were found dead in September 2007 and tested as part of a mandatory CWD surveillance program. There were 122 adult deer and fawns tested for CWD in the facility at the time of depopulation in December 2007. Canada herd 2 was a 57-hectare breeding and hunting operation located in northwestern Saskatchewan. Chronic wasting disease was first diagnosed in 3 does that were slaughtered and then tested as part of a mandatory CWD surveillance program in January 2008. There were 385 deer (excluding deer < 12 months of age, which are not routinely tested for CWD as part of CFIA depopulation protocols) depopulated and tested for CWD at the facility in May 2008. Canada herd 3 was a 16-hectare breeding operation in northeastern Saskatchewan. Epidemiologic investigations had determined that this herd was the herd of origin of 2, previously identified, CWD-infected deer. The herd was confirmed to be infected in January 2011, and 48 deer (excluding deer < 12 months of age) were depopulated and tested for CWD in February 2011. U.S. herd 1 was a 72-hectare hunting operation in Wisconsin in which CWD was first diagnosed on the farm in an adult doe in the fall of 2008 as part of a voluntary CWD surveillance program. In the winter of 2008, at the time of the study and herd depopulation, the herd was composed of 66 adult deer and fawns.
Sample collection
After euthanasia, the sex and age of each deer was determined. The age of the deer was determined either by herd records or was estimated by visual inspection of the teeth. Brainstem at the obex, medial retropharyngeal lymph node, and/or tonsil and rectal mucosa biopsy samples were collected for CWD testing. More specifically, an approximately round, 2 cm in diameter portion of rectal mucosa immediately cranial to the anal mucocutaneous junction was sampled and then placed mucosal side down, within a sponge-lined histology cassette.
Testing of rectal biopsy samples
Testing of the formalin-fixed rectal mucosa biopsy samples from all 4 herds was performed at the USDA National Veterinary Services Laboratories (Ames, Iowa) following a previously described procedure. 3 Briefly, after routine tissue processing, the biopsy samples were embedded in paraffin (with the mucosal surface facing, and parallel to, the front surface of the block). A 5-μm section was obtained at a depth of approximately 250 μm and a second section at a depth of approximately 350 μm from the surface of the mucosa. Immunohistochemical testing was performed following the USDA CWD test protocol with an automated immunostainer a using the mouse monoclonal 99/97.6.1 anti–prion protein antibody b as the primary antibody. 14 Positive control tissues, consisting of scrapie-positive brain and lymph node, were included with each staining run. Tissues were considered positive if there was immunohistochemical staining of the appropriate character and location in the tissue (Fig. 1).3,18 Tissues were considered negative if immunohistochemical staining of the appropriate character and location in the tissue was not detected in the tissue. Any lymphoid tissue samples, including rectal biopsy samples that contained less than 6 lymph follicles in the entire sample, were considered inadequate to make a negative test result determination. No minimum number of lymphoid follicles was required to make a positive determination. Pathologists interpreting the rectal biopsy samples were blinded to test results from other tissues.
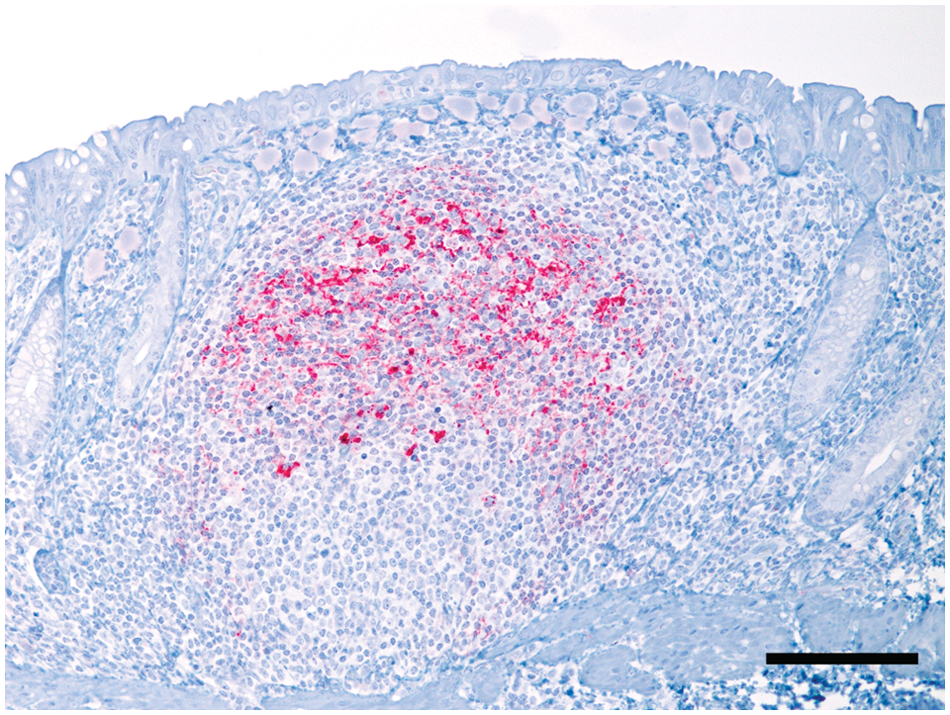
Rectal mucosa; white-tailed deer (Odocoileus virginianus). A prominent lymphoid follicle in the lamina propria contains abundant immunohistochemical staining for protease resistant prion protein (PrPCWD). Streptavidin–alkaline phosphatase immunohistochemistry using an anti-prion antibody and hematoxylin counterstain. Bar = 100 µm.
Testing of obex, lymph node, and tonsil samples
Obex, lymph node, and palatine tonsil samples from U.S. herd 1 were tested by the immunohistochemical procedure as described previously for the rectal biopsy samples. Testing of the obex, lymph node, and palatine tonsil samples from Canada herds 1–3 was carried out by IHC, ELISA, and/or WB and included positive and negative controls on each assay. All 3 methods were applied to obex, lymph node, and palatine tonsil samples from Canada herd 1. At the time of testing samples, from Canada herds 2 and 3, the ELISA method was being used to first screen all 3 tissues, after which a concordant IHC or WB result was used to confirm all positive samples. The methods have been previously described for IHC using a mouse monoclonal 99/97.6.1 anti–prion protein antibody c as the primary antibody with an automated immunostainer.a,2,14 Immunodetection by ELISA d and WB e was performed on unfixed tissue samples using commercial test kits according to the manufacturer’s instructions and as previously described.2,6 The ELISA absorbance cut-off value for PrPCWD detection was calculated by adding a fixed value of 0.09 to the mean optical density of the negative controls. 6 The WB protein bands were evaluated using a digital imaging system and analysis software. f
Grading of obex samples as an estimate of disease progression
Chronic wasting disease progression was estimated using IHC in each deer that was positive on brain tissue by each country’s initial screening test (ELISA testing for Canada herds 1–3) and IHC testing for U.S. herd 1. Obex grading was performed similarly to previously described methods. 12 Obex grades were assigned based on the following criteria: grade 0, no IHC PrPCWD staining present in the obex or ELISA negative on fresh obex samples; grade 1, minimal IHC PrPCWD staining primarily restricted to the ventral area of the dorsal motor nucleus of the vagus (DMNV); grade 2, moderate IHC PrPCWD staining in the DMNV and the nucleus of the solitary tract; grade 3, heavy IHC PrPCWD staining present in the DMNV with lighter staining that extended to other adjacent nuclei (including the nucleus of the solitary tract) and white matter; grade 4, IHC PrPCWD staining present in the DMNV, adjacent nuclei, and white matter.
Determination of codon 96 polymorphism
Two methods were used for the determination of the codon 96 polymorphism. For Canada herds 1 and 2 and U.S. herd 1, DNA was extracted from archived fixed or frozen tissue. The open reading frame of the PRNP gene was amplified and sequenced as described using primers specific for the cervid functional gene and excluding amplification of the cervid processed pseudogene.
10
For Canada herd 3, genomic DNA samples were isolated and purified from the ethylenediamine tetra-acetic acid–anticoagulated blood or homogenized brain tissue using an automated DNA extractor. Real-time polymerase chain reaction (PCR) assays were performed using primers (5’-AGCCACATGGTGGTGGAG-3’; 5’-TG GTTTACTGGGCTTGTTCC-3’) and allelic-specific probes (FAM-TCAA
Statistical methods
Data sets were recorded in spreadsheets i and imported into SAS j for data handling, summary and analyses, and graph production. Within each herd, one-way table analysis (the Frequency procedure, SAS) was used to compute the exact binomial and 95% confidence intervals of intraherd prevalence of CWD. Stepwise selection and exact conditional logistic analyses (the Logistic procedure, SAS) were used to determine the effects of deer factors (age, sex, and PRNP codon 96) on the probability of a positive CWD infection status. The model used reference cell coding and lenient P values for model entry (0.5) and elimination (0.1). Exact odds ratios with 95% upper and lower confidence limits (95% CLs) conditioned by all significant explanatory factors were subsequently determined. Association between disease progression (estimated by obex grade) and PRNP genotype at codon 96 was determined by requesting Somers D statistics (the Frequency procedure, SAS) for all study herd deer with a positive CWD infection status and complete data set.
The diagnostic accuracy (sensitivity and specificity) for determining CWD infection status of white-tailed deer by rectal biopsy testing was determined per herd using the Frequency procedure (SAS) as implemented in SAS Usage Note 24170 (http://support.sas.com/kb/24/170.html). The Surveyselect procedure (SAS) was used to randomly select 1 diagnostic result per deer in Canada herds 2 and 3 in which 2 diagnostic rectal mucosa biopsy samples had been collected but assigned discordant outcomes. Random selections were made within herd strata defined by disease status, age group, and codon 96 in order to avoid inadvertent introduction of selection bias. A meta-analytic bivariate approach was used to compute pooled estimates of diagnostic sensitivity and specificity. 11 The generalized linear random-effects model (random subject effect, herd ID; the GLIMMIX procedure, SAS) was based on code provided previously. 9 The meta-analytic pooled estimates were first computed using only the herds of the current study (n = 4) and then inclusive of data from 2 additional herds (n = 6 total) from a similarly conducted U.S. study. 7 Unless otherwise stated, all meta-analyses converged with satisfaction of the convergence criterion.
Results
Case herds summary statistics
Deer herd populations, PRNP genotype at codon 96, sex, age, and CWD infection status are summarized in Table 1. Canada herd 1 was composed of 122 deer, which were depopulated in December 2007, 38 (31%) of which were determined to be CWD positive by testing of obex, lymph node, and/or palatine tonsil samples. Eleven (9%) of these deer had rectal biopsy samples with an insufficient number of lymphoid follicles and were excluded from the rectal biopsy statistical analyses. Canada herd 2 contained 385 deer when the facility was depopulated in May 2008, 79 (21%) of which were determined to be CWD positive based on testing of obex, lymph node, or palatine tonsil. Forty-seven (12%) of these deer had rectal biopsy samples with an insufficient number of lymphoid follicles and were excluded from the rectal biopsy statistical analyses. Deer from the breeding facility ranged from 1 to 12 years of age, but for 210 (55%) of these deer, age was recorded only as “M” for mature (4–8 years old) and were thus excluded from further analyses that included age as a continuous covariate. Thirty-two (67%) of the 48 deer in Canada herd 3 at the time of depopulation were determined to be CWD positive. Twenty-one (44%) of these deer had rectal biopsy samples with an insufficient number of lymphoid follicles and were excluded from the rectal biopsy statistical analyses. U.S. herd 1 contained 66 deer at the time of depopulation of which only a single deer (approximately 2%) was determined to be CWD positive. Eleven (17%) of these deer had rectal biopsy samples with an insufficient number of lymphoid follicles and were excluded from the rectal biopsy statistical analyses.
White-tailed deer (Odocoileus virginianus) herd characteristics for chronic wasting disease (CWD) prevalence with 95% confidence limits (95% CLs), PRNP genotype at codon 96, sex, and age.
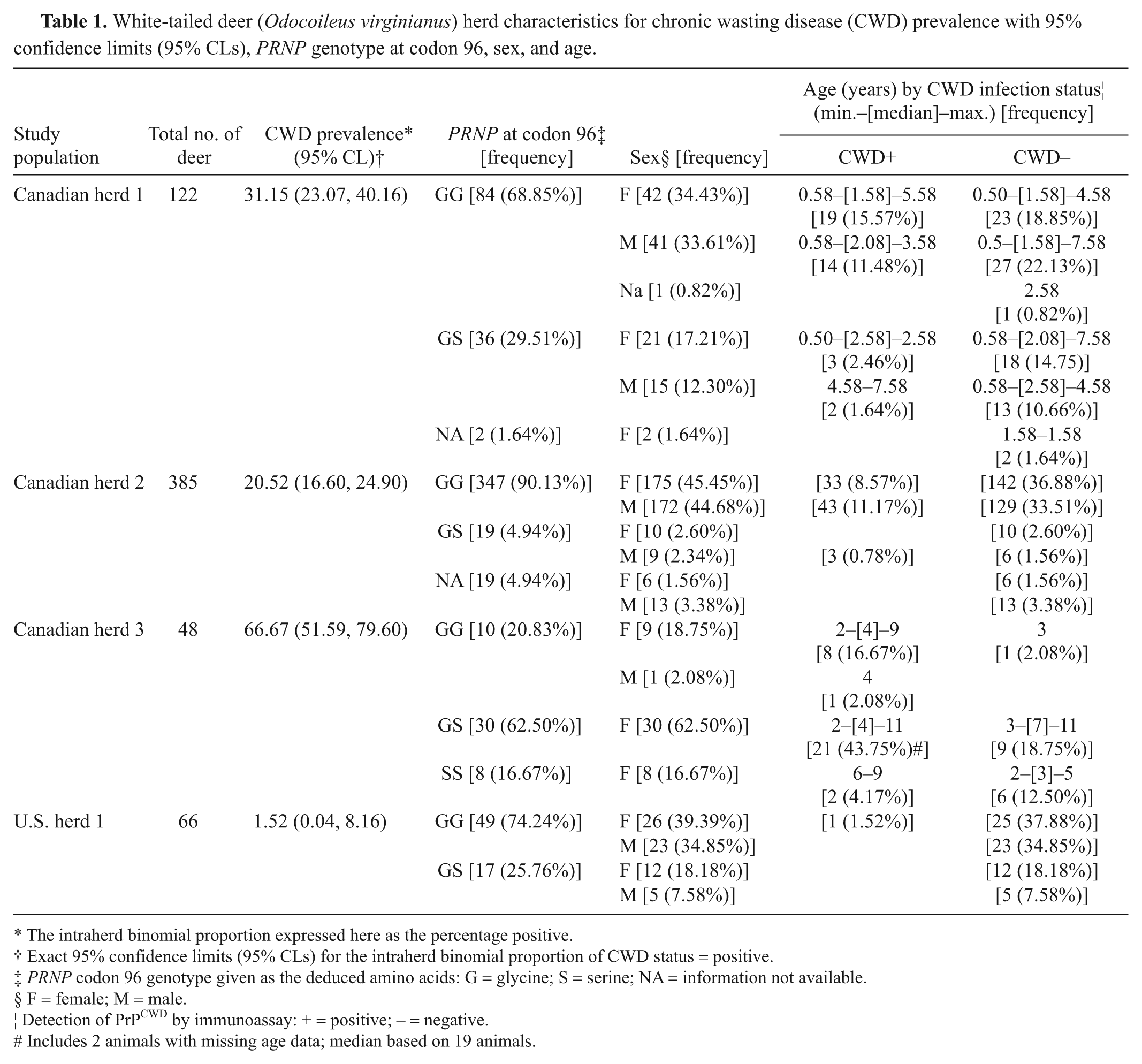
The intraherd binomial proportion expressed here as the percentage positive.
Exact 95% confidence limits (95% CLs) for the intraherd binomial proportion of CWD status = positive.
PRNP codon 96 genotype given as the deduced amino acids: G = glycine; S = serine; NA = information not available.
F = female; M = male.
Detection of PrPCWD by immunoassay: + = positive; – = negative.
Includes 2 animals with missing age data; median based on 19 animals.
Effects of age, sex, and genotype at PRNP codon 96 on likelihood of a positive CWD infection status
The potential influence of age, sex, and genotype at PRNP codon 96 on the likelihood of a positive CWD infection status at the time of depopulation was investigated. Having only 1 positive case, U.S. herd 1was excluded from this analysis. A significant dependence on age or sex at the time of depopulation was not detected for deer in any of the Canadian herds. A positive CWD infection status was significantly dependent on codon 96 for deer from Canada herds 1 and 3 but this factor did not reach statistical significance for deer from Canada herd 2 (Table 2). From the exact conditional logistic analysis, odds ratios relative to 96GG deer were significant for both 96GS deer and 96SS deer such that 96GS deer were overall approximately 3 times less likely, and 96SS deer approximately 19 times less likely, than 96GG deer to be CWD positive at the time of depopulation.
Effect of genotype at PRNP codon 96 on the likelihood of a positive chronic wasting disease infection status in Canada herds of white-tailed deer.*
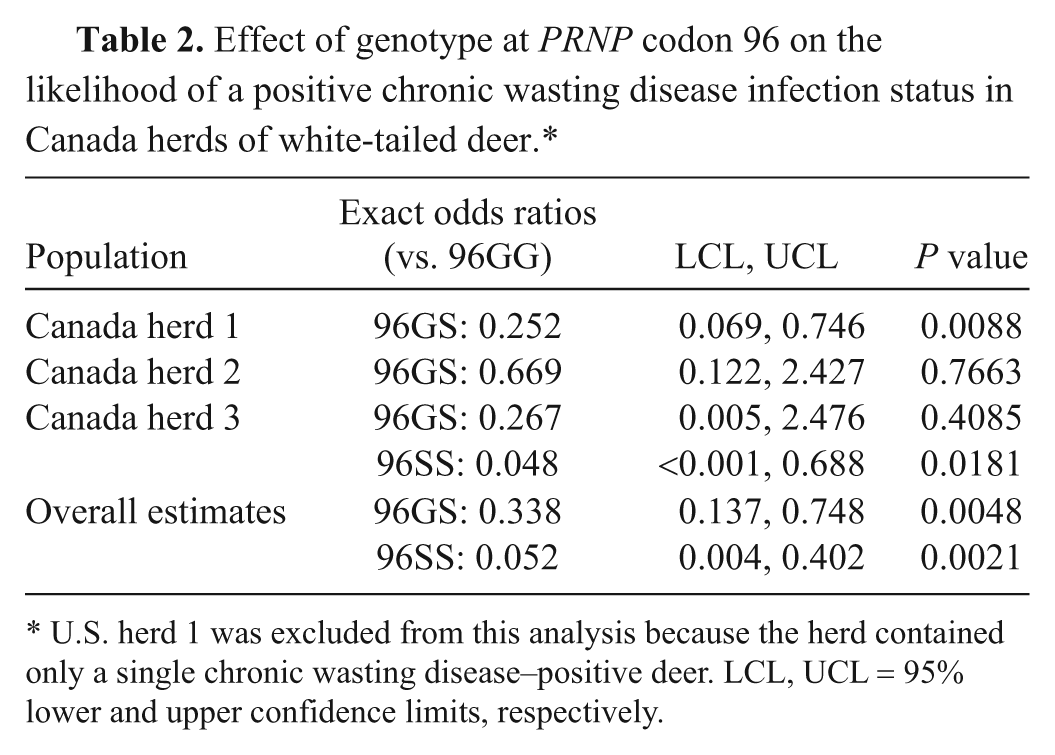
U.S. herd 1 was excluded from this analysis because the herd contained only a single chronic wasting disease–positive deer. LCL, UCL = 95% lower and upper confidence limits, respectively.
Diagnostic accuracy of postmortem rectal biopsy testing
The diagnostic sensitivity of postmortem rectal biopsy testing for determination of CWD infection status is summarized in Figure 2 as a forest plot accompanied by summary data for each herd. The diagnostic sensitivity for individual herd investigations varied from 63% to 100%. The pooled estimate of diagnostic sensitivity (Fig. 2) for the herds of this study (n = 4 herds) was 68% with 95% CLs of 49% and 82%.
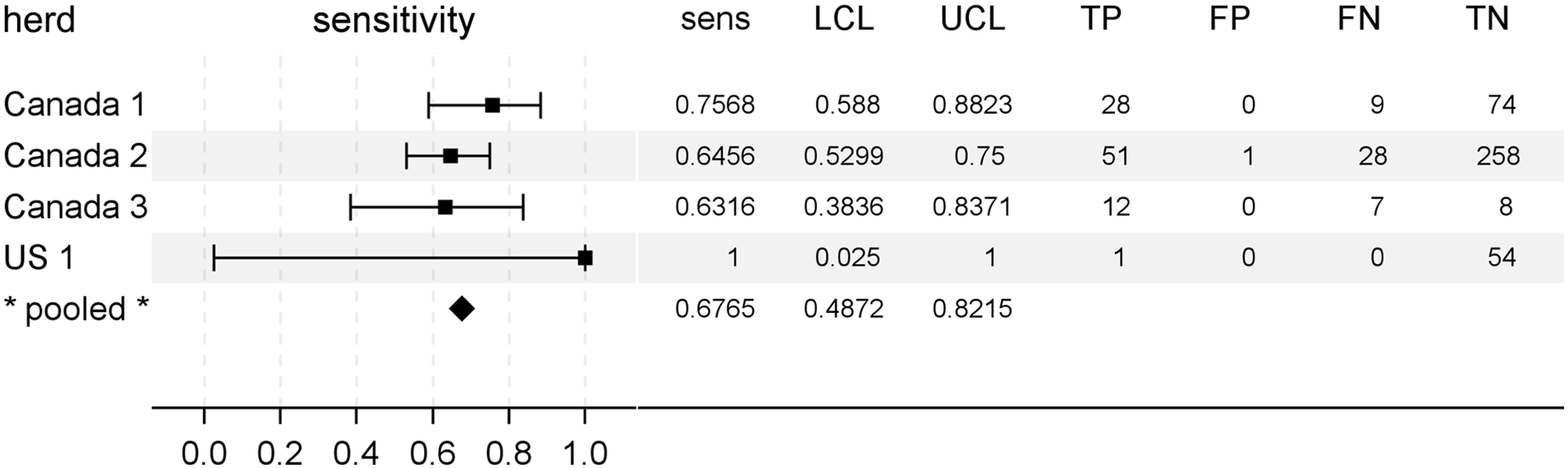
Rectal biopsy sample diagnostic sensitivity for chronic wasting disease in preclinical North American white-tailed deer (Odocoileus virginianus). A forest plot is shown to aid comparison of test sensitivities between herds (squares) and the pooled estimate (diamond). To the right of each plot is given the exact measures of sensitivity (sens), lower and upper 95% confidence limits (LCL, UCL), and the frequencies of rectal biopsy test results categorized relative to gold standard testing. TP = true-positive; FP = false-positive; FN = false-negative; TN = true-negative.
Regarding diagnostic specificity of postmortem rectal biopsy sample testing for determination of CWD infection status in white-tailed deer, only 1 sample was scored as positive in an animal in which PrPCWD was not detected (in this context referred to as a “false-positive”; Fig. 2). Thus, estimates of diagnostic specificity under these conditions ranged from 99.6% in Canada herd 2 to 100% in all other study herds. The pooled estimate of specificity was 99.8% (n = 4 herds, 95% CLs: 84.1%, 100.0%). Given the near uniform accuracy of postmortem rectal biopsy testing with regard to diagnostic specificity, further investigations were limited to testing for the effects of deer factors on rectal biopsy sample diagnostic sensitivity.
Effects of age, sex, genotype at PRNP codon 96, and stage of disease progression on diagnostic sensitivity
Considering the significant effects of PRNP codon 96 polymorphism on the temporal patterns of PrPCWD detection in rectal biopsy samples of deer and, in the present study, on the likelihood of a positive CWD infection status, the effect of genotype at codon 96 on rectal biopsy sample diagnostic sensitivity was examined. 20 For individual herds, exact estimates of diagnostic sensitivities ranged from 67% to 100% in 96GG deer and from 0% to 60% in 96GS deer (Fig. 3). Only 8 96SS deer were identified in the current study, 2 of which were determined to be CWD positive (Table 1). PrPCWD was not detected in the rectal biopsy samples from either of the 2 CWD-positive 96SS deer. The pooled estimates of rectal biopsy sample diagnostic sensitivity by genotype at codon 96 were 76% (95% CLs: 49%, 91%) for 96GG deer and 42% (95% CLs: 13%, 79%) for 96GS deer; a pooled estimate for 96SS deer was not made given the number of cases.
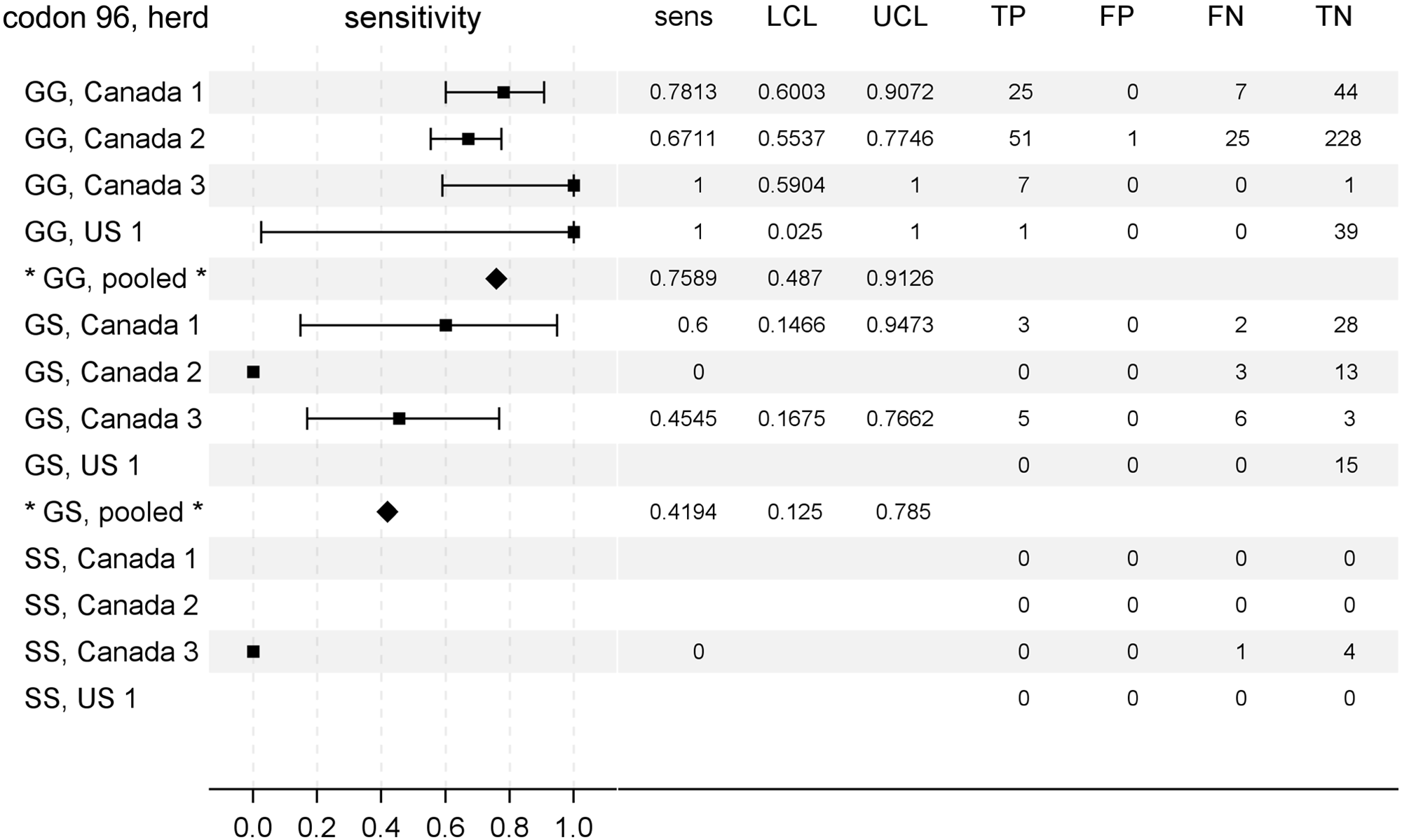
Rectal biopsy sample diagnostic sensitivity for chronic wasting disease in preclinical North American white-tailed deer (Odocoileus virginianus) characterized by PRNP codon 96 genotype. A forest plot is shown to aid comparison of test sensitivities between herds (squares) and the pooled estimates (diamonds). To the right of each plot is given the exact measures of sensitivity (sens), lower and upper 95% confidence limits (LCL, UCL), and the frequencies of rectal biopsy test results categorized relative to gold standard testing. TP = true-positive; FP = false-positive; FN = false-negative; TN = true-negative.
All deer in the present study had unknown sources and timing of exposure and were deemed to be nonclinical at the time of depopulation. Thus, brainstem accumulation of PrPCWD at the level of the obex (obex grade, where 0 = not detected and 4 = maximum accumulation) was used as a surrogate indicator of disease progression in CWD-positive deer. As seen in Figure 4, there was a clear association of early disease progression (low obex grades) with lower estimates of rectal biopsy sample diagnostic sensitivity. Given smaller numbers of CWD-positive deer within each obex grade subgroup, the measures of sensitivity were somewhat variable between herds. Nevertheless, the modeled effect of obex grade on rectal biopsy sample diagnostic sensitivity was significant in the meta-analysis of the study herds (P = 0.0230). The pooled estimates of sensitivity were 36% for obex grade 0 deer, 83% for obex grade 1 deer, 93% for obex grade 2 deer, and 100% for deer with obex grades 3 or 4.
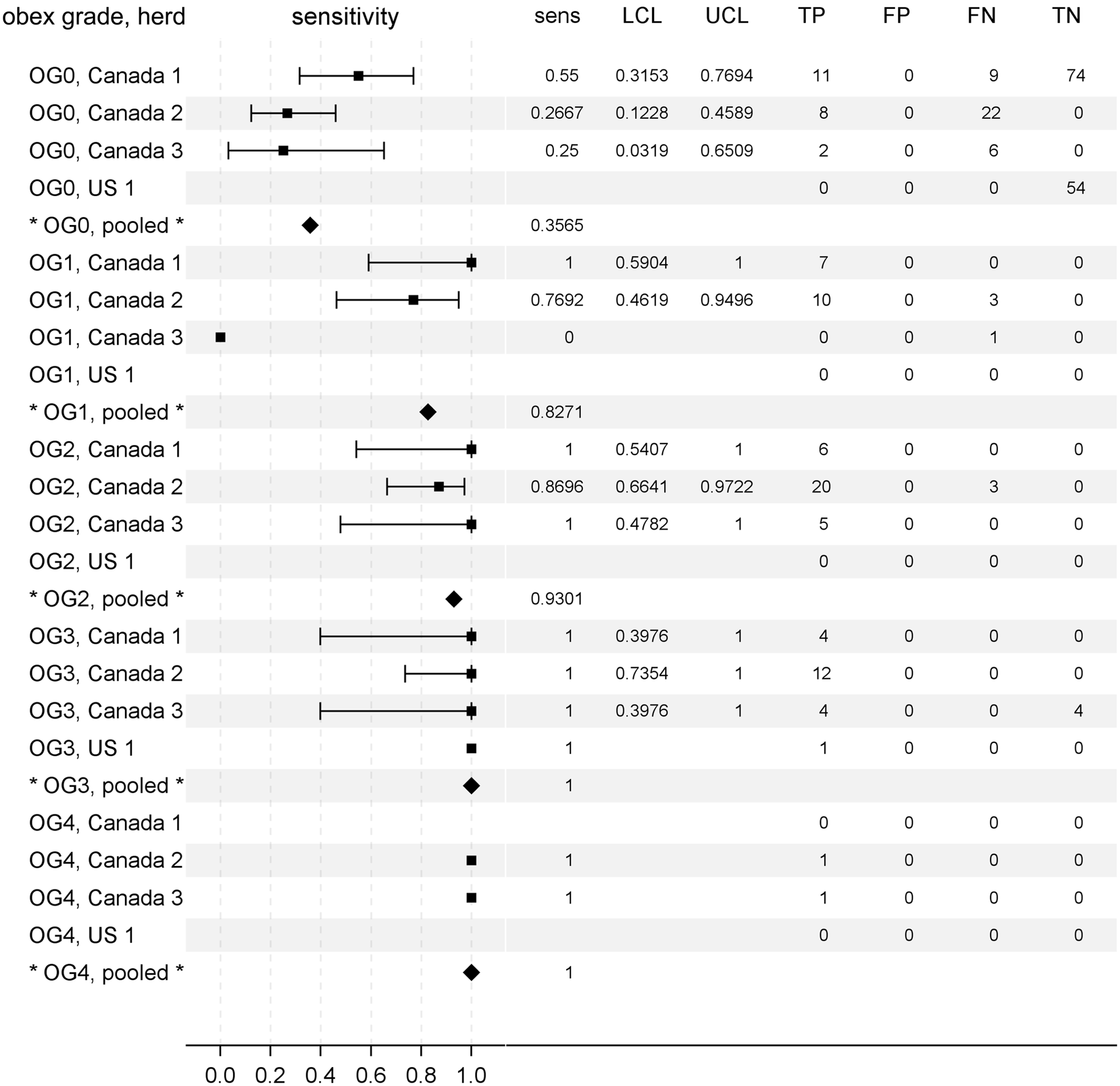
Rectal biopsy sample diagnostic sensitivity for chronic wasting disease (CWD) in preclinical North American white-tailed deer (Odocoileus virginianus) characterized by brainstem accumulation of protease resistant prion protein (PrPCWD) at the level of the obex (obex grade). Obex grade (OG) ranged from 0 (PrPCWD not detected) to 4 (maximum accumulation). A forest plot is shown to aid comparison of test sensitivities between herds (squares) and the pooled estimates (diamonds). To the right of each plot is given the exact measures of sensitivity (sens), lower and upper 95% confidence limits (LCL, UCL), and the frequencies of rectal biopsy test results categorized relative to gold standard testing. TP = true-positive; FP = false-positive; FN = false-negative; TN = true-negative).
To test the hypothesis that reduced rectal biopsy sample diagnostic sensitivity in white-tailed deer with G96S polymorphism might be associated with slower disease progression, the association of obex grade with genotype at codon 96 in deer with a positive CWD infection status at the time of depopulation was measured. As shown in Figure 5, the proportion of deer in later stages of disease progression (obex grades > 0) decreased with G96S polymorphism, though this association did not reach statistical significance (Somers D = −0.2066, 95% CLs: −0.4188, 0.0057; P = 0.0645). When similarly stratified by stage of disease progression, G96S polymorphism also appears to be negatively associated with the proportion of positive lymph follicles per rectal biopsy sample of CWD-positive white-tailed deer (Fig. 6).
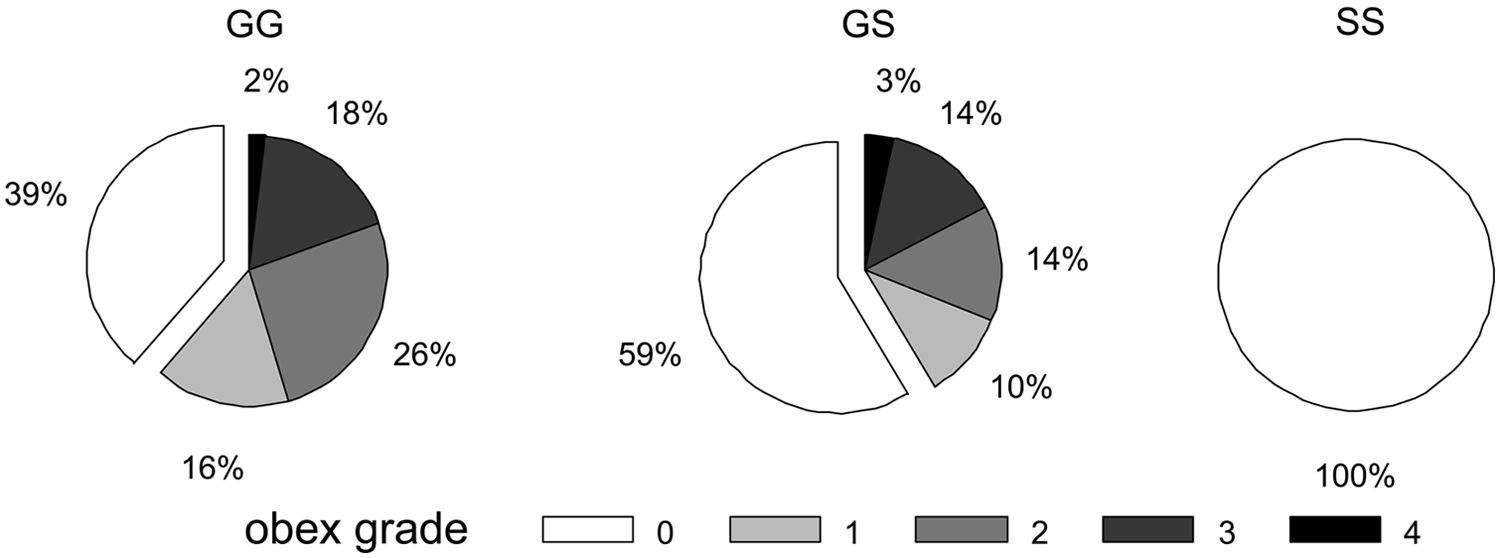
The proportional brainstem accumulation of protease resistant prion protein (PrPCWD) at the level of the obex (obex grade) in North American white-tailed deer (Odocoileus virginianus) with preclinical chronic wasting disease (CWD) grouped by genotype at PRNP codon 96. Note the decreased proportion of deer with G96S polymorphism that have grade 1 or greater accumulation of PrPCWD. Data represent the pooled records of deer for which obex grades (scale: 0, PrPCWD not detected; 4, maximum accumulation) and genotypes were available (Canada herds 1, 2, and 3 representing 119 GG, 29 GS, and 2 SS deer).
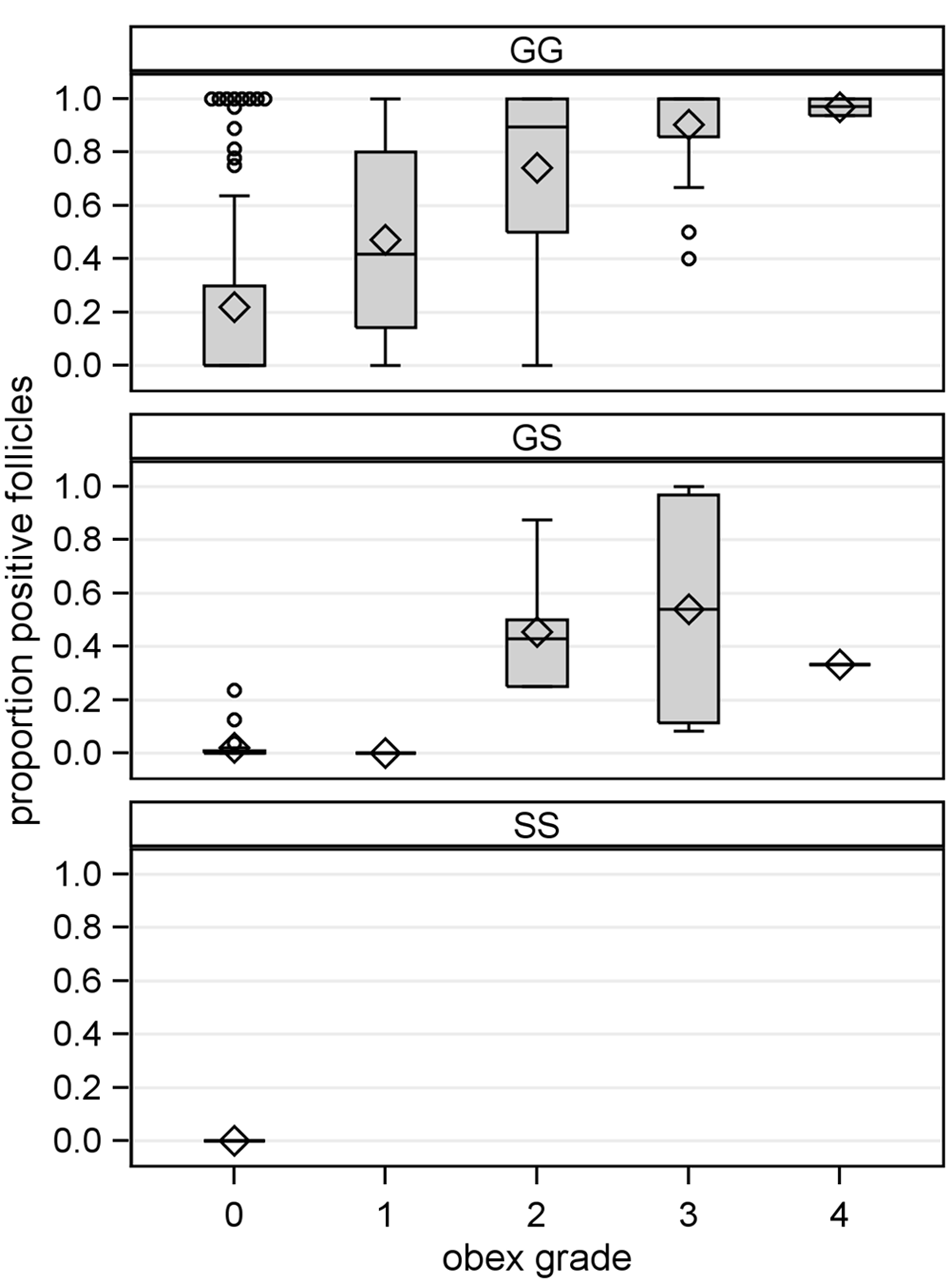
The proportion of protease resistant prion protein (PrPCWD)-positive rectal mucosal lymphoid follicles in North American white-tailed deer (Odocoileus virginianus) with preclinical chronic wasting disease (CWD) and grouped by genotype at PRNP codon 96 and disease progression. Disease progression estimated by brainstem accumulation of PrPCWD at the level of the obex (obex grade: 0, PrPCWD not detected; 4, maximum accumulation). Shown are box plots depicting the interquartile range (gray box with limits of 25th and 75th percentiles), mean (diamond) and median (gray box center line), and data falling within (stems) and outside (open circles) the 1.5 interquartile range. Data represent deer for which the proportion of positive rectal mucosal lymphoid follicles could be determined and for which obex grades and genotypes were available (Canada herds 1, 2, and 3 representing 196 GG, 35 GS, and 2 SS deer).
Discussion
A safe and effective live animal diagnostic test is needed to assist in the control of CWD, which currently relies on testing of samples collected only after the death of the deer. Identification of CWD-infected herds is further hindered because the disease has a long subclinical period. 18 The current study included over 600 clinically normal white-tailed deer, in 4 genetically and geographically diverse herds, and showed that rectal biopsy sample testing was an effective method in detecting subclinical CWD infection. The diagnostic sensitivity of rectal biopsy testing for determining CWD infection status ranged from 63% to 100% in the 4 white-tailed deer herds in the study. The aim of the meta-analytic modeling in the present study was to improve the estimate of diagnostic sensitivity accuracy in a manner more broadly applicable to future application.9,11 Thus, the pooled estimate of diagnostic test sensitivity of rectal biopsy testing was 68% when based on the 4 herds of the current study, which was refined to 74% (95% CLs: 60% and 85%) after inclusion of data from 2 additional herds from a similarly conducted study (data not shown). 7 Conversely, false-negative results should be expected from rectal biopsy samples in generally one-fourth of all CWD-infected deer. Thus, a negative test result based on a single rectal biopsy sample should not be interpreted as a deer being free of CWD infection.
Rectal biopsy diagnostic testing of deer should be done concurrently with PRNP genotype testing in order to make a more accurate assessment of test results. In an experimental inoculation study of CWD in white-tail deer, PrPCWD was detected at earlier time periods postinoculation in tonsil and rectal mucosa in codon 96GG deer as compared to 96GS and 96SS deer. 20 As demonstrated in the present study, knowledge of the genotype is important because rectal biopsy sample test sensitivity was 76% for 96GG deer, but dropped to 42% for 96GS deer. Additionally and not surprisingly, the rectal biopsy test sensitivity was the greatest (100%) in deer with advanced disease as evidenced by abundant PrPCWD in the brain and the lowest (36%) in deer in which PrPCWD was not detected in brain tissue and only detected in lymph tissues; thus in agreement with a previous study. 7 Higher rates of false-negative rectal biopsy results are expected in white-tailed deer in earlier stages of CWD disease progression and in deer having PRNP G96S polymorphism.
The diagnostic specificity of rectal biopsy sample testing for determining CWD infection status was 99.8% and not 100% for the 4 herds of the current study due to a single deer in Canada herd 2 classified as a false positive. The rectal biopsy from the 2-year-old, codon 96GG deer had a single positive lymphoid follicle out of 143 total follicles, and the positive staining was repeatable on serial tissue sections. Possible explanations for these conflicting test results include ineffective subsampling of the obex and lymph node samples, which may have had limited focal PrPCWD deposition and led to false-negative obex and lymph node results. An abnormal PrPCWD tissue distribution pattern due to individual animal variation is another possibility. Other studies have identified deer in which the PrPCWD tissue distribution did not follow the expected pattern of appearing in select lymph tissues before appearing in the brainstem (e.g., a white-tailed deer in which PrPCWD was found only within brain tissue and not within lymph node samples). 8
The effect of genotype polymorphism at codon 96 was also examined because it has been shown to correlate with the CWD infection status of deer.8,10 Chronic wasting disease infection status based on brain, lymph node, and palatine tonsil testing was negatively associated with polymorphisms of codon 96, such that deer with G96S polymorphism were less likely than GG deer to be test-positive at the time of depopulation. Though this association with the heterozygous genotype (96GS) reached statistical significance only in Canada herd 1, the estimated odds ratios for this genotype in Canada herds 2 and 3 were of similar magnitude and direction. However, the effect of serine at codon 96 does not prevent CWD infection, as evidenced in the respective 70% and 25% infection rates in 96GS and 96SS deer of Canada herd 3. Disease progression, as measured by obex grading, also appeared to be slower in 96GS and 96SS deer as compared to 96GG herd mates, a conclusion consistent with previous findings.7,8,20 There may be other unidentified polymorphisms of the prion gene that have additional influence on the pathogenesis of CWD.
Although the samples in the current study were collected postmortem, rectal biopsy testing for CWD and scrapie on live animals has been shown to be safe for the animal and relatively simple to perform by the surgeon.3,15 When performing the rectal biopsy, care must be taken to identify the proper biopsy site in order to obtain a sample that contains lymphoid follicles. Lymphoid follicles are most abundant within an approximately 1 cm–wide band of mucosa and submucosa immediately cranial to the stratified squamous epithelium of the rectoanal junction; samples collected proximal or distal to that location have fewer or no lymphoid follicles, which results in nondiagnostic samples. 7 Fewer lymph follicles are also found within rectal biopsy samples collected from older animals.3,16 In the present study, the percentage of samples with an insufficient number of follicles occurred at an unexpectedly high rate (44%) in Canada herd 3, while the other 3 herds had rates of 9–17%, which is similar to a previous study. 7 Because the procedure causes minimal discomfort to the animal, the procedure is often performed using only restraint in proper animal handling facilities and topical anesthesia.3,15 In contrast, the only other commonly used live animal CWD test used in research is the palatine tonsil biopsy. 20 The palatine tonsil biopsy is a more complex procedure that carries potentially greater risk due to the necessity of sedation or general anesthesia, but has shown a greater sensitivity at detecting PrPCWD in the early stages of disease.7,20
The current study provides additional evidence that rectal biopsy samples can be used successfully to detect subclinical CWD infections in white-tailed deer. Polymorphisms of codon 96 and stage of disease progression have major effects on the expected level of diagnostic sensitivity, so any interpretation of biopsy sample results in which PrPCWD is not detected should consider these 2 factors. Genotype testing can easily identify the codon 96 polymorphism of each deer. However, estimating the level of disease progression is much more difficult and often impossible, but could potentially be estimated based on the predicted date of exposure. One potential use for this test may be for epidemiologic investigations of CWD-suspect deer herds. Selective use of this test to investigate herds suspected of having subclinical CWD, in conjunction with traditional postmortem testing, could provide valuable information to animal health officials as well as reduce the costs associated with postmortem testing.
Footnotes
Acknowledgements
The authors wish to recognize the valuable contributions of the laboratory technicians at the TSE Unit at the Canadian Food Inspection Agency (CFIA)–Ottawa Laboratory Fallowfield: Patricia Shaffer, Steven Foster, and Dariush Ghazi; USDA, National Veterinary Services Laboratories: Jennifer Lamoreux, Sharon Lund, Julie Lease, and AnnaStacia Penrod; and the USDA, Animal Disease Research Unit: Desiree Lesiak. The authors thank statistician Bruce Mackey (USDA, Agricultural Research Service, Albany, CA) for advice and critical review of the statistical methods used in this study. Mention of trade names or commercial products or enterprises in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the USDA or CFIA.
a.
Ventana Medical Systems Inc., Tucson, AZ.
b.
Anti-Prion 99, Ventana Medical Systems Inc., Tucson, AZ.
c.
Anti-prion F99/97.6.1, VMRD Inc., Pullman, WA.
d.
TeSeE ELISA, Bio-Rad Laboratories, Hercules, CA.
e.
TeSeE Western Blot, Bio-Rad Laboratories, Hercules, CA.
f.
Chemi Doc XRS imager and Quantity One, version 4.6 software, Bio-Rad Laboratories, Hercules, CA.
g.
Applied Biosystems, Carlsbad, CA.
h.
iQ5 Multicolor Real Time PCR System, Bio-Rad Laboratories, Hercules, CA.
i.
Excel, Microsoft Corp., Redmond, WA.
j.
Software version 9.2, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The U.S. Department of Agriculture holds the patent on anti-prion antibody F99/97.6.1, and K. O’Rourke is the inventor. Other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported through funding from the Canadian Food Inspection Agency (CRTI-085), USDA Animal and Plant Health Inspection Service, and Agricultural Research Service (CRIS 5348-32000-030-00D).
