Abstract
Semen from an apparently healthy 4-year-old American Quarter Horse was submitted to the National Veterinary Services Laboratories for Equine arteritis virus isolation. Visual inspection of the semen sample upon arrival noted it was unusually yellow in color. The semen sample was inoculated onto cell monolayers, and cytopathic effect was observed 5 days postinoculation. The resultant isolate tested negative for Equine arteritis virus, and was subsequently identified as Equine rhinitis A virus. Equine rhinitis A virus has been isolated from horse urine, but has not been described in stallion semen. The present study documents the isolation of Equine rhinitis A virus from stallion semen that was likely contaminated with urine at the time of collection.
Semen from a 4-year-old American Quarter Horse stallion was submitted to the National Veterinary Services Laboratories (NVSL) for Equine arteritis virus (EAV) isolation as prescribed for international movement of EAV-seropositive stallions. 7 Per the attending veterinarian, the animal appeared healthy with no signs of clinical disease. The refrigerated semen sample was received at the NVSL on the day following the collection. Testing of the semen for EAV was conducted using NVSL standard operating procedures, which closely follow the procedure outlined by the World Organization for Animal Health (OIE). 5 A portion of the sample was examined to confirm the presence of sufficient spermatozoa, and the sample was also visually inspected for color and gross particulate contamination. It was noted that the sample received was unusually yellow in color in comparison to normal equine semen samples, which are milky white.
On the day it was received at the laboratory, the sample was prepared for virus isolation and inoculated onto cell monolayers. Specifically, the sample was placed in an ice bath and sonicated (three 15-sec bursts at maximum power) to disrupt the spermatozoa, and serial dilutions (1:10 and 1:100) were prepared and inoculated into duplicate flasks of rabbit kidney (RK)-13 cells. a Serial dilutions were employed to circumvent the toxic effect of semen on cell culture. Inoculated flasks were incubated for 1 hr at 37°C followed by addition of overlay medium (Eagle minimum essential medium, pH 6.9, containing 2% calf serum b and 0.75% carboxymethyl cellulose c ). The monolayers were held at 37°C and examined for the presence of cytopathic effect (CPE) regularly. On day 5 postinoculation, CPE was observed in the flasks inoculated with the 1:10 sample dilution. The flasks were frozen at –70°C, thawed, and subpassaged onto RK-13 cell monolayers. Cytopathic effect was observed on the second passage flask at 1 day postinoculation, and the flask was frozen at –70°C on day 2 when CPE was observed on >90% of the cell sheet. The cell culture isolate was tested by a reverse transcription polymerase chain reaction (RT-PCR) targeting the open reading frame 1b gene of EAV 2 ; results were negative. The isolate was then examined by electron microscopy, and particles with the morphologic appearance of picornaviruses were observed (Fig. 1).
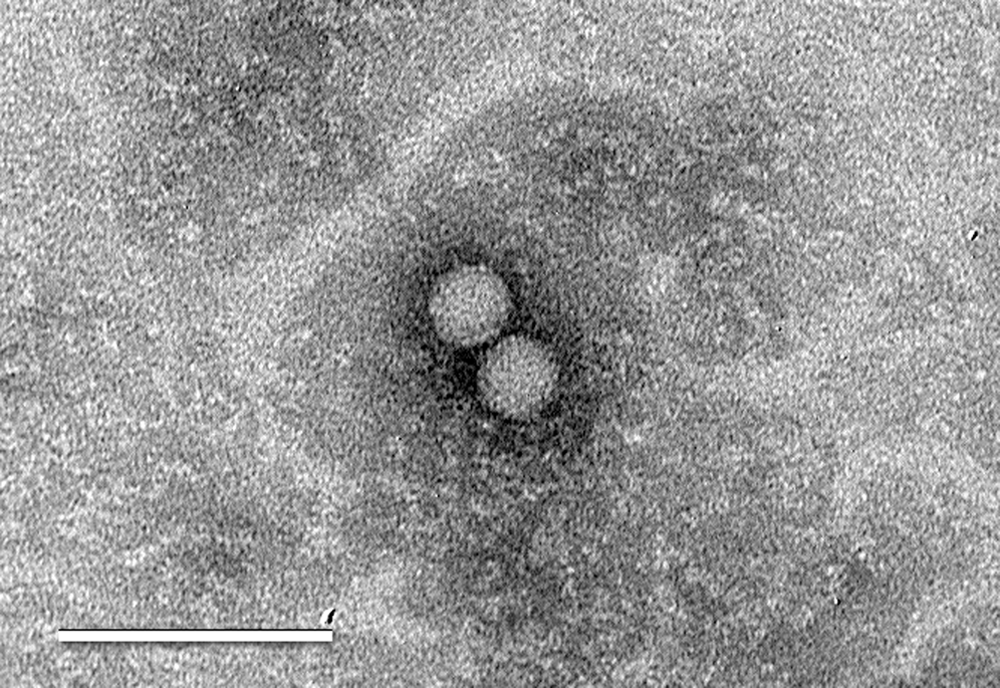
Electron micrograph of picornavirus-like particles observed in the rabbit kidney (RK)-13 cell culture isolate. Bar = 100 nm.
Viruses of the Picornaviridae family known to commonly infect horses are Equine rhinitis A virus (ERAV), a member of the Aphthovirus genus, and Equine rhinitis B virus (ERBV), a member of the Erbovirus genus. Both ERAV and ERBV may cause an acute febrile respiratory disease, but often horses infected with these viruses remain clinically normal. The high prevalence of serum neutralizing antibodies to ERAV and ERBV in U.S. horses, as well as horses from other regions of the world indicates that infection with equine rhinitis viruses is common (McCollum WH, Timoney PJ: 1992, Studies on the seroprevalence and frequency of equine rhinovirus-I and -II infection in normal horse urine. In: Proceedings of the sixth international conference on Equine Infectious Diseases, pp. 83–87. R&W Publications, Suffolk UK).1,3 Isolation of these viruses is often accomplished by inoculation of nasopharyngeal swab material onto susceptible cell cultures. 4 However, ERAV may also be isolated from the blood or tissues of infected horses, usually prior to the development of neutralizing antibodies. Equine rhinitis A virus–infected horses may also develop a prolonged carrier state when virus can readily be isolated from the urine (McCollum WH, Timoney PJ: 1992).
Equine rhinitis A virus and ERBV identification in isolates and in field samples is accomplished at the NVSL by RT-PCR. For isolates, outer primers are used in a single-step RT-PCR; a nested assay is employed for samples containing a lower level of virus, such as diagnostic specimens. Resultant amplicons from PCR testing are purified, d sequenced using the same PCR primers, e and aligned f with known viral sequences located in the GenBank library to confirm the presence and identity of virus.
Equine rhinitis A virus primers were designed g from the 5’-nontranslated and L protein regions of the equine rhinitis virus genome (GenBank accession no. DQ268580). The ERAV outer primers amplifying a 883-bp segment were ERA396-5′-GAAGGCGTTCGCAATAAGAC-3′ and ERA1278-5′-TTCCATCGTACCGACATCC-3′. The ERAV nested primers amplifying a 523-bp segment were ERA665-5′-AAGCCCAGCGTTAATAGC-3′ and ERA1186-5′-GTTTGGTCACTCACCCAATC-3′. Primer ERA1186 contained a single base modification from the GenBank sequence. The ERBV primers were designed from the 5′-nontranslated region of ERBV (GenBank accession no. X96871). Outer primers amplifying a 673-bp segment were ERB81-5′-GCTTGGCTCTCAGAAAGAATGG-3′ and ERB753-5′-GGTACCTGTAGGACATCCTTAG-3′; nested primers amplifying a 425-bp segment were ERB195-5′-TGTTGGTCGTTTGCCAATCG-3′ and ERB619-5′-GTGA GAGGTGCTGTAAACACTG-3′. The sensitivities of the RT-PCR assays were compared to cell culture by determining endpoint dilutions of NVSL reference virus stocks of ERAV and ERBV. h Both the ERAV and ERBV RT-PCR assays were found to be 100-fold more sensitive than cell culture in both the single-step and nested procedures (data not shown). However, for samples containing a lower level of virus, the additional nested reaction significantly improved visual detection and provided sufficient PCR product for sequencing (Fig. 2). The ERAV and ERBV primers were also tested on 3 NVSL repository isolates of ERAV and 10 NVSL repository isolates of ERBV where expected amplification occurred only with the homologous primers. The repository isolates had been previously identified by virus neutralization testing, and the identities were confirmed by sequencing during the current study.
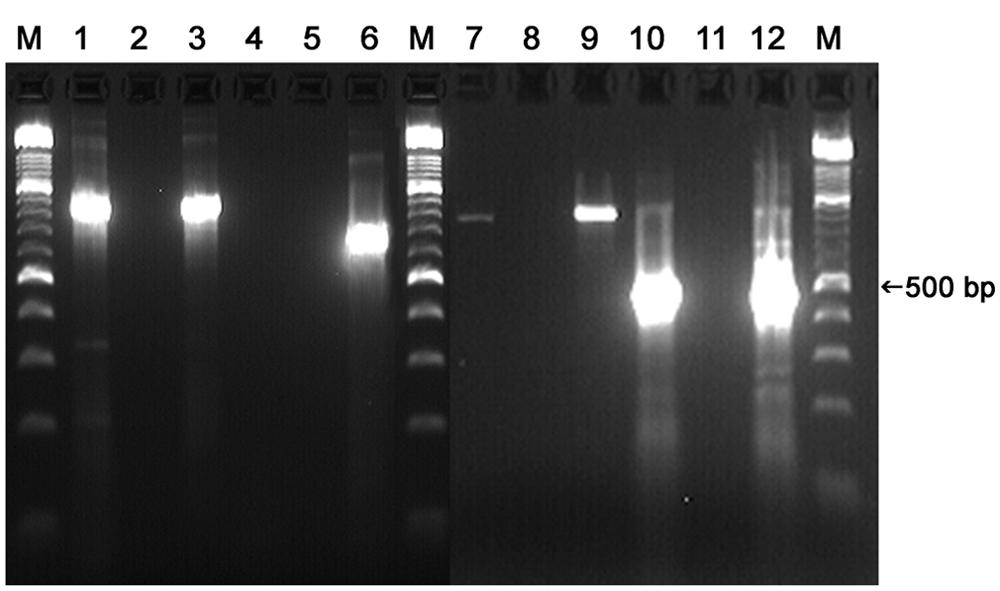
Reverse transcription polymerase chain reaction (RT-PCR) testing of the virus isolate and the original semen sample with Equine rhinitis A virus (ERAV) and Equine rhinitis B virus (ERBV) primers. Single-step RT-PCR testing for ERAV (lanes 1–3) and ERBV (lanes 4–6); semen isolate in lanes 1 and 4, no template control in lanes 2 and 5, positive ERAV control in lane 3, positive ERBV control in lane 6. ERAV RT-PCR testing of original semen sample in lanes 7–9 (single-step) and 10–12 (nested) with semen sample in lanes 7 and 10, no template control in lanes 8 and 11, and ERAV control in lanes 9 and 12. Lane M: molecular weight marker.
For RT-PCR testing, total RNA was extracted i from 250 µl of the diagnostic sample (cell culture isolate, semen, swab suspension, or tissue homogenate). The extracted RNA was suspended in 20–25 µl of RNase-free water. A single-step RT-PCR j following manufacturer’s recommendations was performed using 2.0 µl of the RNA in a 50-µl RT-PCR reaction containing a 0.6 µM concentration of the outer primers, and the following cycling conditions: 50°C for 30 min, 95°C for 15 min, followed by 35 cycles of 94°C for 45 sec, 58°C for 45 sec, and 72°C for 1 min; and a final extension of 72°C for 9 min. k For the nested reaction, 1.5 µl of the first stage amplification product was added to 48.5 µl of a PCR mix prepared per manufacturer recommendations l containing a 0.5 µM concentration of the nested primers. The nested reaction employed the following cycling conditions: 95°C for 15 min, followed by 30 cycles of 94°C for 45 sec, 58°C for 45 sec, and 72°C for 1 min; and a final extension of 72°C for 9 min. The RT-PCR products were analyzed by agar gel electrophoresis using a nucleic acid dye m for DNA visualization.
Testing of the semen cell culture isolate using the ERAV and ERBV outer primers resulted in amplification only with the ERAV primers (Fig. 2, lanes 1–6), and the resultant 831 base amplicon shared an 87–89% nucleotide identity to multiple strains of ERAV (GenBank accession nos. L43052, DQ268580, DQ272127, DQ272128, DQ272577, DQ272578). The sequence did not align significantly to any other pathogen. After identification of the semen sample isolate, the remaining 3 ERAV isolates in the NVSL repository (TN 2002, CA 2004, and MD 2004) were also subjected to RT-PCR, and the amplicons were sequenced using the same primers. The 3 isolates shared similar nucleotide identity to the same ERAV strains within the GenBank library (87–91%), indicating that the percent identity observed with the semen sample isolate was within normal range.
Subsequent to the identification of ERAV in the isolate, the original semen sample was tested by nested RT-PCR using the ERAV primers (Fig. 2, lanes 7–12). The resultant 504 base amplicon was identical to the ERAV isolate obtained from the same sample, confirming that the source of the ERAV isolate was the semen sample.
While ERAV-infected horses may shed virus in their urine well past the detection limit in other body fluids or tissues, ERAV has not been previously described in horse semen. In a previous study, over 2,500 urine samples were collected from apparently healthy horses at Kentucky racetracks in 1989 (McCollum WH, Timoney PJ: 1992). Equine rhinitis A virus was isolated from 17% of the urine samples, and ERBV was not isolated from any of the horse urine tested. It was also demonstrated at that time that ERAV may be recovered from horse urine nearly 6 months postinfection. One report has also speculated upon the possibility of venereal infection of ERAV from urine-contaminated horse semen. 6 Considering the observed high infection rate of horses to ERAV, the occurrence of ERAV in the urine of infected horses, the yellow color of the semen tested in the present report, and the absence of evidence that ERAV-infected stallions shed the virus in semen, it is likely the semen in this investigation was contaminated with ERAV-infected urine at the time of collection. Because this isolate was obtained from a horse awaiting export, it was not possible to collect urine or additional semen to confirm if the actual source of ERAV was urine or semen.
Footnotes
a.
CCL-37™, American Type Culture Collection, Manassas, VA.
b.
Bovine calf serum, HyClone Laboratories, Logan, UT.
c.
Carboxymethylcellulose sodium salt, medium viscosity, Sigma-Aldrich, St. Louis, MO.
d.
QIAquick® PCR Purification Kit, Qiagen Inc., Valencia, CA.
e.
3500xl Sequence Detection System, Applied Biosystems, Foster City, CA.
f.
Align Plus 5, Scientific and Educational Software, Cary, NC.
g.
Primer Designer 5, Scientific and Educational Software, Cary, NC.
h.
Equine rhinitis A virus, stock (NVSL 060-EDV), and equine rhinitis B virus, stock (NVSL 061-EDV); National Veterinary Services Laboratories, Ames, IA.
i.
Trizol LS, Invitrogen Corp., Carlsbad, CA.
j.
OneStep RT-PCR kit, Qiagen Inc., Valencia, CA.
k.
GeneAmp® PCR System 9700, Applied Biosystems, Foster City, CA.
l.
HotStarTaq® DNA polymerase, Qiagen Inc., Valencia, CA.
m.
GelRed™, Biotium Inc., Hayward, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
