Abstract
A 12-year-old female Argentinean Warmblood mare was evaluated because of respiratory noise. The horse resided in Calgary, Alberta, Canada, but had been imported from Argentina 28 months prior to presentation. Endoscopy of the upper respiratory tract revealed a single polypoid mass on the left arytenoid. The mass was surgically excised and was diagnosed histologically as rhinosporidiosis. Polymerase chain reaction and DNA sequencing were used to confirm the etiological agent. Four weeks postoperatively, endoscopy was repeated, revealing recurrence of the original lesion with multiple additional polypoid masses on the larynx and in the oropharynx. Resolution of the disease had not been attained at the time of publication. The current report outlines a case of rhinosporidiosis in an unusual anatomical and geographic location. The infection most likely originated in Argentina, with a prolonged subclinical phase. Due to increased travel of human beings and animals, there is potential for the introduction of exotic diseases into nonendemic areas.
A 12-year-old Argentinean Warmblood mare, imported from Buenos Aires, Argentina, 28 months previously, was examined for respiratory noise at the Moore Equine Veterinary Centre (Balzac, Alberta, Canada) in November 2009. Since importation, the mare had no additional travel history. Endoscopy of the nasopharynx revealed a 6-mm-diameter nodular mass on the corniculate process of the left arytenoid. The nasal cavities were unremarkable, and no nasal discharge was present. Surgical excision, with follow-up histopathology, was planned. The patient was presented for surgery 1 week later, at which time a significant increase in mass size was appreciated. The now 2.5 cm × 2.0 cm × 1.5 cm irregular to nodular, glistening, polypoid lesion was surgically excised from the left arytenoid, fixed in 10% neutral buffered formalin, and submitted to Prairie Diagnostic Services (Saskatoon, Saskatchewan, Canada) for histological evaluation (Fig. 1). Immediately following excision of the nodule, cryotherapy of the surgical site was performed.

Argentinean Warmblood mare. Gross section of the excised nodular polypoid arytenoid mass. Bar = 0.5 cm.
The formalin-fixed tissue was embedded in paraffin and sectioned at 4 µm. Sections were stained with hematoxylin and eosin, periodic acid–Schiff (PAS), and Gomori methenamine silver (GMS) and then examined using light microscopy. The mass consisted of submucosa expanded by numerous endosporulating sporangia of variable size (60–500 µm) and stage of maturation (Fig. 2). As previously described, juvenile sporangia typically had a central nucleus with a prominent nucleolus, surrounded by basophilic granular material and a PAS-positive, GMS-negative unilamellar wall.5,10 The nucleus was no longer visible in the intermediate sporangia, where immature endospores accumulated. The wall of the intermediate sporangia was bilamellar, the inner layer being GMS positive and the outer layer PAS positive.5,10 The mature sporangia retained the bilamellar wall, and endospore maturation progressed from peripheral immature endospores to centrally located mature endospores. 10 Within the collagenous stroma, dissecting between the sporangia, was a marked mononuclear infiltrate with plasma cells, lymphocytes, and fewer macrophages. Few of the sporangia were ruptured and associated with a focal marked neutrophilic inflammation. The surmounting mucosa was markedly hyperplastic with dramatic downgrowths into the submucosa and occasionally was noted to contain mature sporangia discharging endospores into the external environment. Variably sized islands of glandular epithelium were present within deep tissue, and cartilage formed the deep surgical margin. Sporangia were noted within 1 mm of the lateral surgical margins. Based on the characteristic histological appearance, laryngeal rhinosporidiosis was diagnosed. Although considered unlikely, due to the similarity of mature stages, coccidioidomycosis was a potential differential diagnosis.
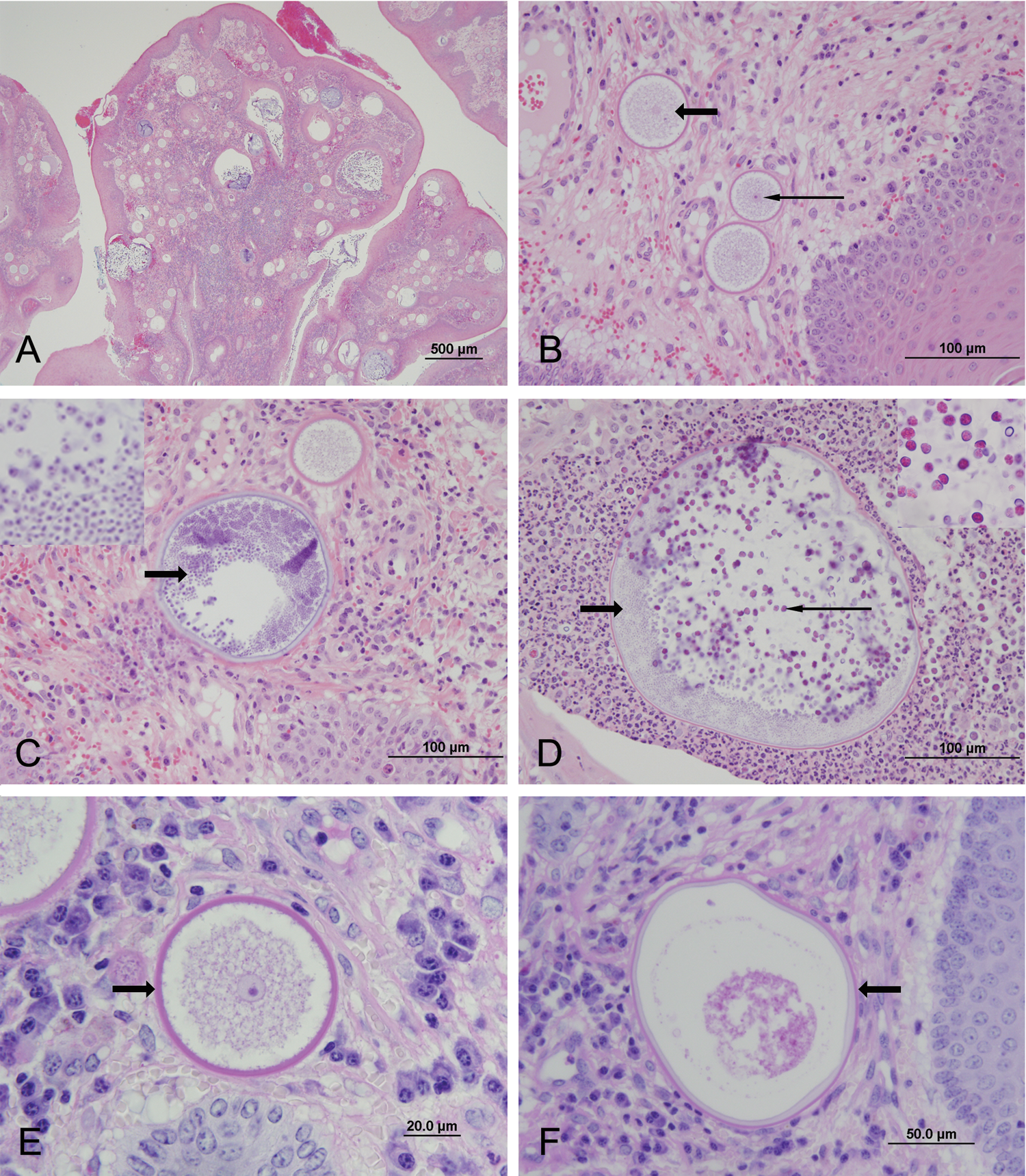
Argentinean Warmblood mare.
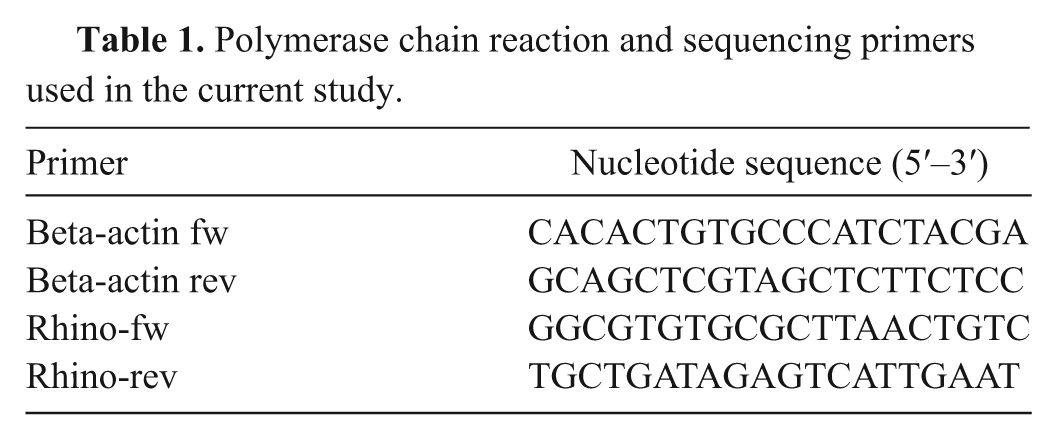
To definitively confirm the causative agent, polymerase chain reaction (PCR) was performed. DNA was extracted from patient formalin-fixed tissue, as well as formalin-fixed normal equine larynx (negative control), by deparaffinization using xylene and by digestion using a lysis buffer containing 100 mM NaCl, 500 mM Tris, and 10% sodium dodecyl sulfate. Freeze–thaw techniques were used to break apart the fungal wall and expose the DNA. Proteinase K (5 µl of 10 mg/ml stock solution) was added, the samples were heated overnight at 56°C, and DNA was extracted with phenol–chloroform. Pellets were resuspended in 100 µl of Tris-EDTA buffer. To confirm that amplifiable DNA was present in both the patient and control samples, PCR amplification for the housekeeping beta-actin gene (Table 1) was performed, producing bands of expected size (247 bp) in each sample. Polymerase chain reaction amplification using
Polymerase chain reaction and sequencing primers used in the current study.
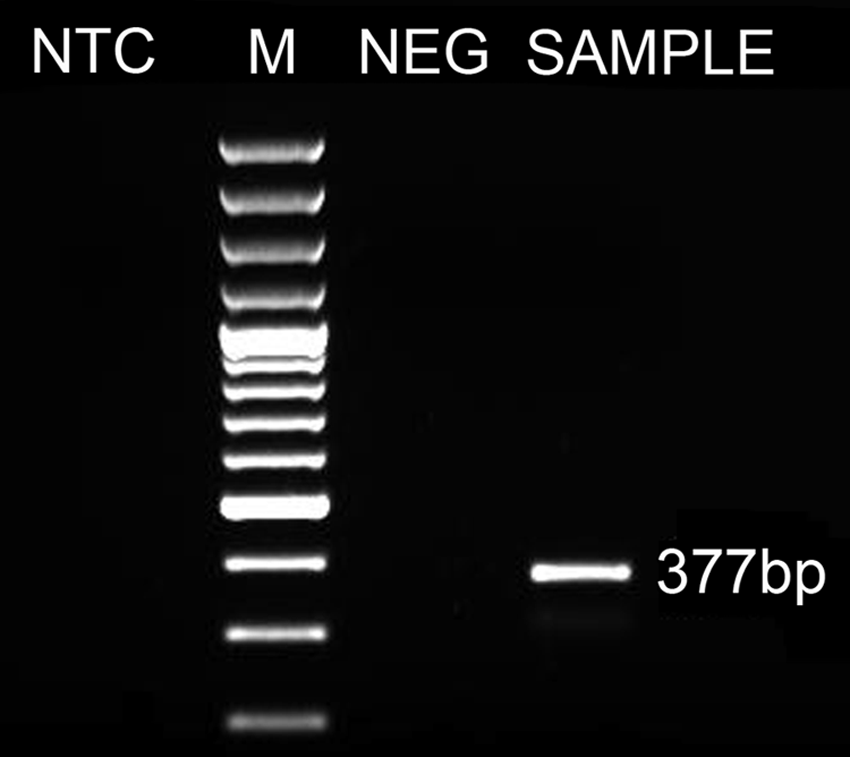
Agarose gel electrophoresis of
Amplified DNA from the
Four weeks postoperatively, endoscopy of the nasopharynx was repeated, revealing recurrence of the original lesion with multiple additional nodular masses on the arytenoid, epiglottis, aryepiglottic folds, and oropharynx. Histopathology confirmed the presence of
Although historically controversial, recent phylogenetic analysis has clarified the taxonomic classification of
The diagnosis in the current case report was confirmed through the characteristic histopathological appearance, PCR, and DNA sequence analysis. The patient had been imported from an endemic area and was presumably already infected at that time, as there was no known opportunity for exposure since. A similar scenario in the United Kingdom was recently reported, where 4 polo ponies imported from Argentina harbored the disease.11,12 The infection was not apparent at the time of importation in any of the above-mentioned horses and remained subclinical in 1 of the polo ponies for 10 months.
12
In the current case, the period between last known possible exposure and onset of clinical signs was 28 months, significantly exceeding previous reports.4,12,16 In addition, the minimal changes noted in the oropharyngeal lesions over the last 14 months of the case report further reinforce the ability of this organism to persist subclinically for long periods of time. With increased international travel of human beings and animals, there is increased potential for the introduction of exotic diseases in nonendemic countries, particularly when the subclinical phase is prolonged. Direct transmission has not been reported with
Spontaneous regression of lesions has been reported, 10 but this is exceedingly rare. Complete surgical excision, preferably with electrocautery, is the recommended therapeutic approach. 1 The reported rate of recurrence is variable in the literature. In the case reported herein, sporangia extended beyond the polyp stalk, to the surgical margins. Despite local cryotherapy at the time of surgery, recurrence was documented within 4 weeks. In addition to regrowth of the original lesion, multiple additional masses were identified. Dissemination and embedding of endospores from the original lesion, as part of the natural life cycle of the organism, or spillage of endospores during surgical excision, can account for the additional masses. Recurrence was also documented in the Belgian Warmblood report, 14 months after surgical excision, 14 suggesting that extended follow-up times may be necessary in some cases.
Although rhinosporidiosis has been recognized, but poorly understood, since 1900, the current case report describes a documented occurrence in western Canada and an uncommon anatomical location. The extended subclinical phase in this patient reinforces the potential for importation and the necessity to consider exotic diseases in cases where there is any travel history. Resolution of the disease had not been attained at the time of publication, and no further cases had been diagnosed, to the authors’ knowledge, in the area.
Footnotes
Acknowledgements
The authors thank Dr. J. Hill for her assistance and expertise with molecular diagnostics and DNA sequence analysis.
a.
Invitrogen Canada Inc., Burlington, Ontario, Canada.
b.
EZ-10 Spin Column PCR Purification kit, Bio Basic Inc., Markham, Ontario, Canada.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
