Abstract
A 15-year-old Belgian gelding was referred for fever, depression, and respiratory distress. Lung biopsy revealed interstitial fibrosis consistent with chronic interstitial pneumonia. Equid herpesvirus 5 (EHV-5) DNA was detected by polymerase chain reaction (PCR) in bronchoalveolar lavage and biopsy specimens. A presumptive diagnosis of equine multinodular pulmonary fibrosis (EMPF) was made, and the horse was administered a systemic treatment with corticosteroids and antiviral drugs. Despite initial clinical improvement, 4 weeks later, the condition of the horse rapidly deteriorated, and the animal was euthanized. Postmortem examination confirmed the presumptive diagnosis of EMPF. The EHV-5 DNA load in different tissues was estimated using a quantitative real-time PCR. Lung had a remarkable viral load, higher than in other organs, especially within the pulmonary fibrotic nodules, and a linkage between high viral burden and the most severely affected tissues was observed. The results suggest that the quantitative real-time PCR is a useful tool to quantify the EHV-5 load in different organs and to understand the relationship between EHV-5 and EMPF. The bronchoalveolar lavage was determined to be a good clinical sample to estimate the EHV-5 load in lung.
Equine multinodular pulmonary fibrosis (EMPF) is a fibrotic interstitial lung disease of horses, described for the first time in 2007 and associated with Equid herpesvirus 5 (EHV-5; family Herpesviridae, subfamily Gammaherpesvirinae, genus Percavirus 2 ) infection in horses older than 4 years. 6,10,14–16 EHV-5 is considered ubiquitous in horse populations, but probably distributed with different prevalence based on the age of the host. 4,8 To date, all reported cases of EMPF have been associated with EHV-5 infection. However, the role of EHV-5 in the development of EMPF still remains to be determined. The current report describes the clinical, pathomorphological, and biomolecular findings in a 15-year-old Belgian gelding presenting respiratory signs due to EMPF, in which a quantitative analysis of EHV-5 DNA loads from bronchoalveolar lavage (BAL) fluid, lungs, and other tissues demonstrated a differential tissue-related virus burden, directly proportional to the severity of the lesions.
A 15-year-old Belgian gelding was referred to the Veterinary Teaching Hospital of Perugia University for a 3-week history of fever, respiratory distress, and depression. Prior to referral, the horse was empirically treated with antibiotics, nonsteroidal anti-inflammatory drugs, and dexamethasone. At admission, on physical examination, the horse was quiet and slightly lethargic, and had hyperemic and jaundiced mucous membranes, with a rectal temperature of 38.1°C. Evaluation of the respiratory system revealed an abdominal breathing pattern with reduced thoracic expansion, increased inspiratory and expiratory efforts, as well as diffuse inspiratory crackles, and no evidence of nasal discharge. Hematological abnormalities included mild leukocytosis (9.9 × 103 cells/µl, reference [ref.] range: 5–10 × 103/µl, with 50% hypersegmented neutrophils, 35% lymphocytes, 7% band neutrophils, 5% monocytes, 3% eosinophils), and mild anemia (6.83 × 106 red blood cells/µl, ref. range: 6–12 × 106 red blood cells/µl; packed cell volume 30.8%, ref. range: 33–42%; hemoglobin 10.2 g/dl, ref. range: 11–13 g/dl). The serum biochemical profile revealed total protein of 7.6 g/dl (ref. range: 6–7.5 g/dl), hyperbilirubinemia (total bilirubin 3.68 mg/dl, ref. range: 0.5–2.8 mg/dl; unconjugated bilirubin 2.41 mg/dl, ref. range 0.3–1 mg/dl), and a mild increase in muscle enzyme activities (lactate dehydrogenase 279 U/l, ref. range: to 400 U/l; alkaline phosphatase 335 U/l, ref. range: 86–285 U/l; creatine kinase 38 U/l, ref. range: to 55 U/l; aspartate aminotransferase 217 U/l, ref. range: to 240 U/l), and mild hypoxemia on blood gas analysis (PaO2 83 mmHg, ref. range: 89–115 mmHg; PaCO2 36 mmHg, ref. range: 37–49 mmHg; saturation 96%, ref. range: 95–100%).
Thoracic radiography revealed a diffuse and severe interstitial pattern associated with air bronchograms and thickened peribronchial walls. Thoracic ultrasonography demonstrated a bilaterally generalized pleural roughening, several comet tails, and multiple, hyperechogenic small scattered areas (<0.5 cm in diameter), but no obvious nodular thickening. Transtracheal wash (TTW) and bronchoalveolar lavage (BAL #1) fluid were collected for microbiology and cytology. Cytological examination of both specimens revealed mild inflammation predominantly characterized by segmented neutrophils; furthermore, in BAL #1, rare large foamy macrophages were present, with numerous single to clustered, oval, 3–6 µm, deep blue yeast-like bodies.
Twenty-four hours after admission, the horse developed intermittent fever (with peaks at 39.8°C), worsening of respiratory signs, and moderate hypoxemia. Waiting for lab results, a support therapy was set up with gentamicin (6.6 mg/kg intravenously every 24 hr), cefquinome (1 mg/kg intramuscularly every 24 hr), flunixin meglumine (1.1 mg/kg intravenously every 24 hr), and theophylline (7 mg/kg orally every 12 hr).
Multiple transcutaneous lung biopsies were collected from the left and right lungs, under ultrasonographic guidance using a biopsy needle. The samples were collected from diffusely affected parenchyma. Histological examination of the biopsy specimens showed a moderate to severe diffuse interstitial fibrosis with mixed-cell infiltration, consistent with chronic interstitial pneumonia.
No bacterial or fungal growth was obtained from TTW, BAL #1, and lung biopsy specimens under aerobic and anaerobic conditions. A qualitative semi-nested polymerase chain reaction (PCR) analysis for Equid herpesvirus (EHV)-1, -2, -4, and -5 3,8,11,13 was performed on DNA of TTW and lung biopsy, and was positive for EHV-2 and EHV-5 on TTW and only for EHV-5 on lung biopsy. Viral isolation attempts from lung biopsy on rabbit kidney (RK)13 cells were negative after 5 passages.
Based on clinical, radiographic, and histological findings, a presumptive diagnosis of multinodular pulmonary fibrosis was made. The horse was treated with a tapering course of dexamethasone (0.2 mg/kg intramuscularly every 24 hr for 7 days then a daily 10% decrease in dose) to control inflammatory response, with acyclovir (30 mg/kg orally every 12 hr) to limit viral replication and fluconazole (14 mg/kg orally as loading dose followed by 5 mg/kg orally every 24 hr) to reduce the growth of the yeast. Antimicrobial therapy with cefquinome was continued to prevent a possible bacterial infection due to a possible immunosuppression caused by steroids. During the first 3 weeks of treatment, the horse showed clinical improvement, but transitory signs of iatrogenic Cushing-like syndrome appeared, manifested as polyuria and periorbital oedema.
At 4-weeks re-evaluation, thoracic radiography and ultrasonography showed a worsening of the interstitial pattern, with an increased number of comet tails and small, consolidated areas. BAL fluid collection was repeated (BAL #2), and the fluid was characterized cytologically by macrophages, hypersegmented neutrophils, and mast cells. Neither bacterial nor fungal growth was observed from BAL #2; PCR for EHV-5 nucleic acid was still positive.
A week later, the general condition of the horse worsened dramatically showing depression, high fever (39.3°C), nasal discharge, severe dyspnoea, and hypoxemia. Despite aggressive support therapy, the clinical picture did not improve, and the horse was euthanized due to poor prognosis.
At necropsy, both lungs were enlarged, did not collapse, and preserved rib imprints; the consistency was increased with multifocal to coalescing whitish to tan fibrous nodules (Fig. 1); bronchial lymph nodes were markedly enlarged with edematous cut surfaces. No gross abnormalities were detected in other organs. Samples of several tissues and specimens from 3 different sites of the lung (lung #1 from apical lobe, lung #2 from caudal lobe, and lung #3 from a fibrotic nodule) were collected for microbiological, biomolecular, and histological examinations (Table 1).
Results of qualitative polymerase chain reaction (PCR), quantitative real-time PCR, and histological lesions in samples of a horse with equine multinodular pulmonary fibrosis.*
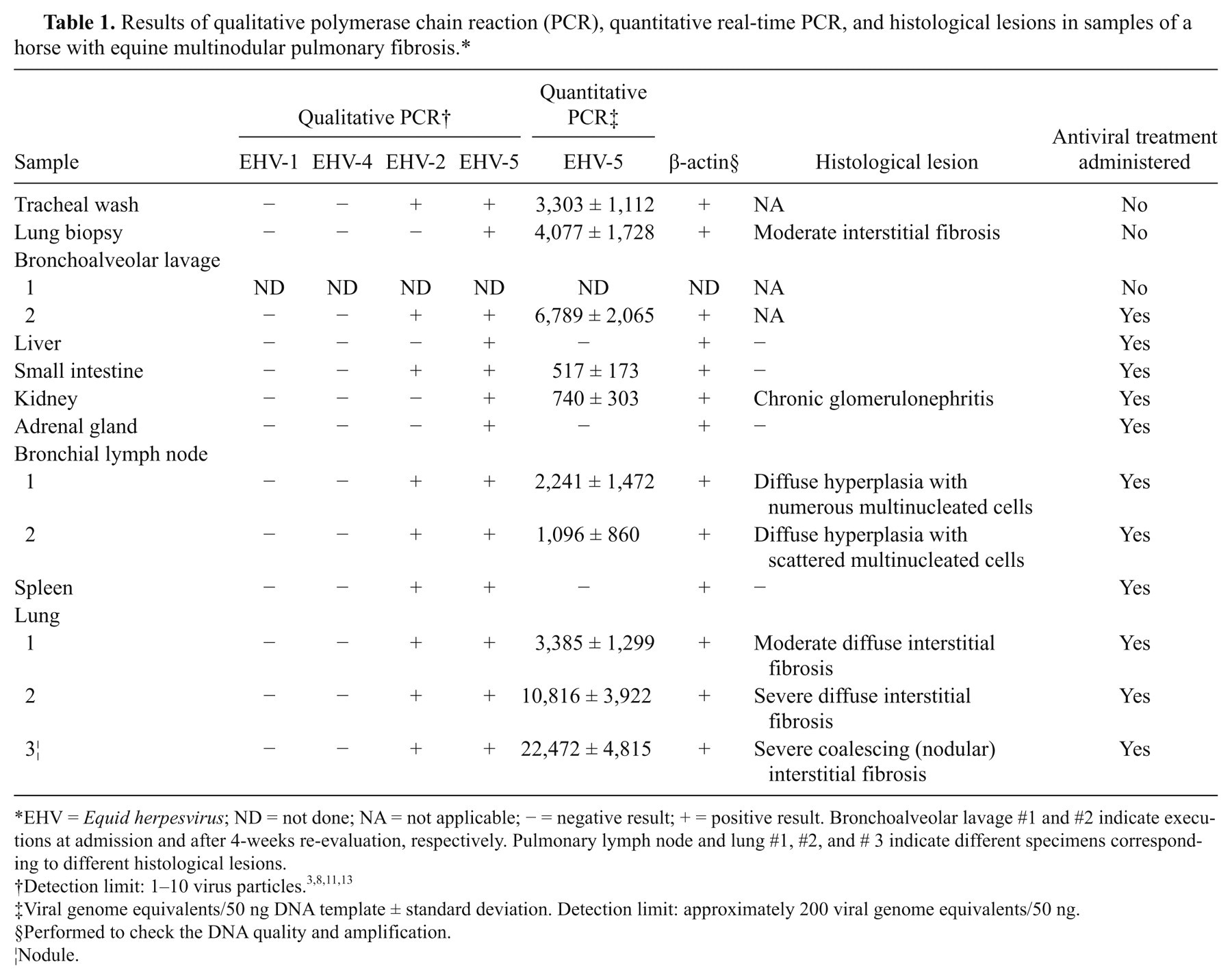
EHV = Equid herpesvirus; ND = not done; NA = not applicable; − = negative result; + = positive result. Bronchoalveolar lavage #1 and #2 indicate executions at admission and after 4-weeks re-evaluation, respectively. Pulmonary lymph node and lung #1, #2, and # 3 indicate different specimens corresponding to different histological lesions.
Viral genome equivalents/50 ng DNA template ± standard deviation. Detection limit: approximately 200 viral genome equivalents/50 ng.
Performed to check the DNA quality and amplification.
Nodule.
Histologically, 80% of lung parenchyma was effaced by a diffuse interstitial fibrosis, most prominent in subpleural spaces, that multifocally coalesced into ill-defined fibrous nodules (Fig. 2); alveolar spaces were lined by hyperplastic type II pneumocytes, and filled by fibrous tissue, large macrophages exhibiting erythrophagocytosis and hemosiderosis, and rare multinucleated cells, but no evidence of intranuclear inclusions was seen. Lymph nodes were diffusely hyperplastic with multiple multinucleated giant cells and marked subcapsular histiocytosis. Gross and histological features were consistent with the multifocal coalescing form of EMPF. 15
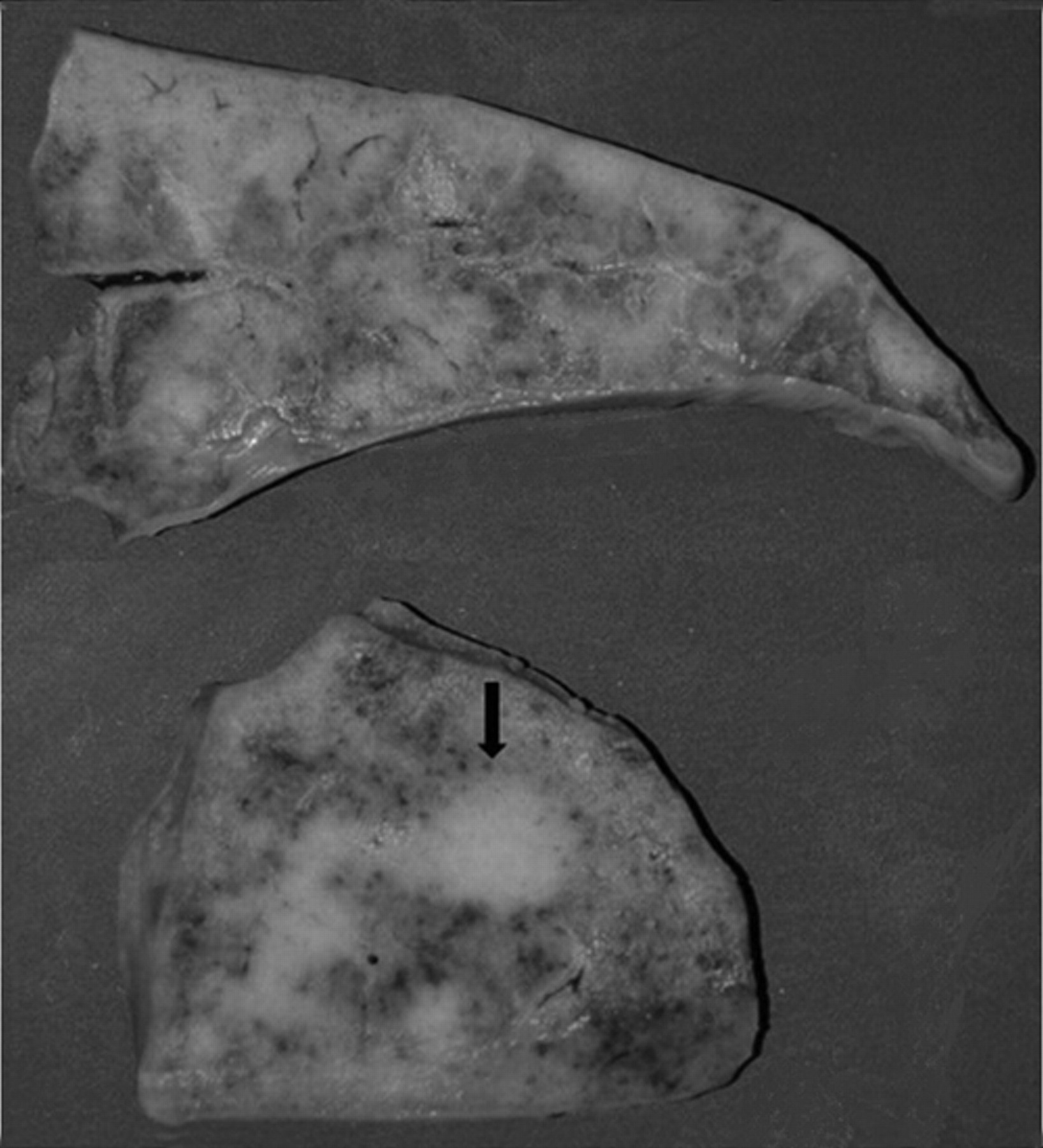
Lung, horse, multinodular pulmonary fibrosis. Multifocal to coalescing whitish to tan fibrous tissue, with focal nodular aggregate (arrow). Formalin-fixed specimen.
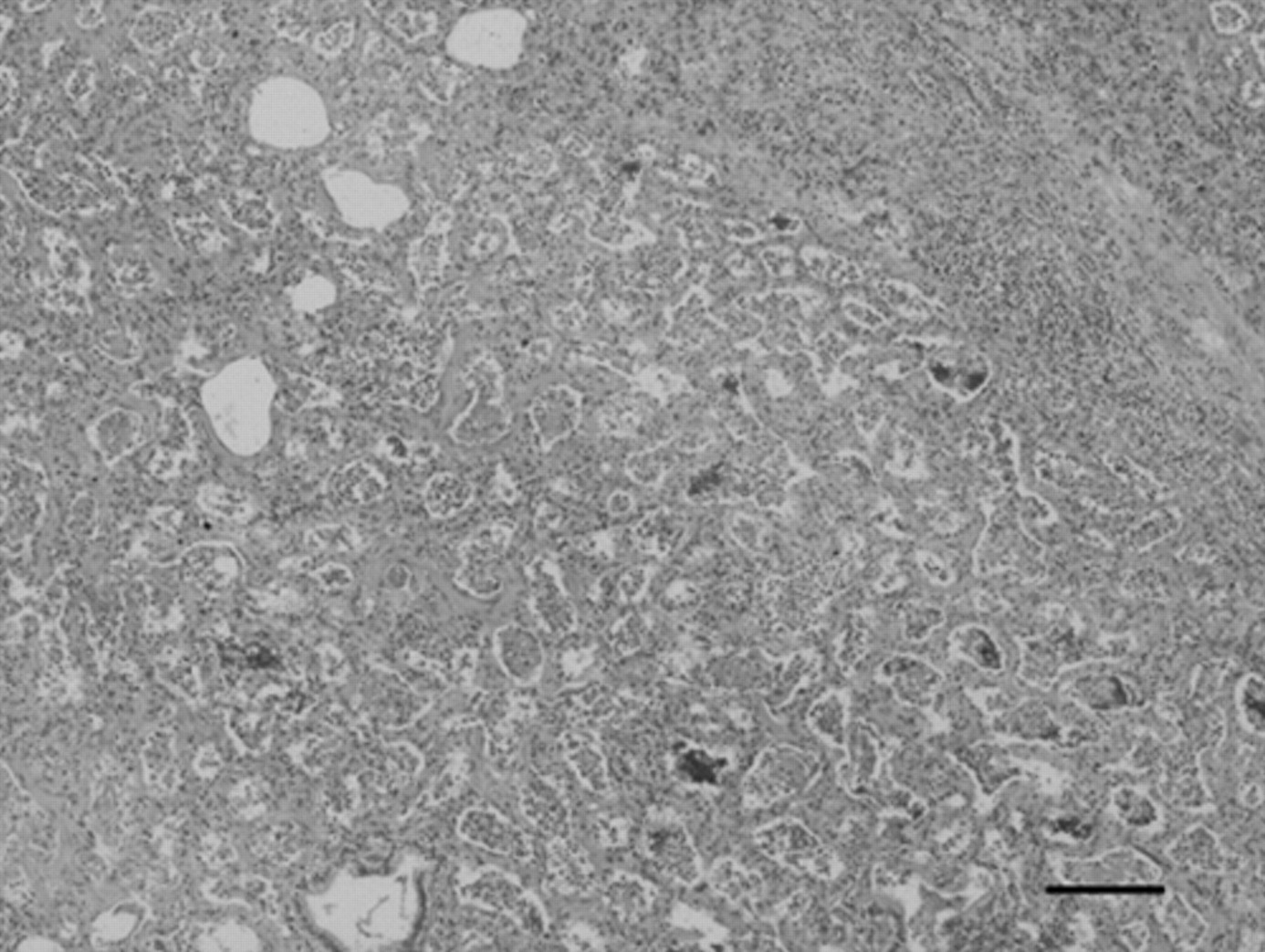
Lung, horse, multinodular pulmonary fibrosis. Diffuse interstitial and alveolar fibrosis with complete effacement of pulmonary architecture. Hematoxylin and eosin. Bar = 200 µm.
Escherichia coli and Klebsiella pneumoniae were isolated from different organs; Candida spp. was cultured from lung tissue after enrichment. No viral isolation was observed on RK13 cells seeded with lung tissue and pulmonary lymph nodes after 5 passages.
Qualitative semi-nested PCR for EHV-1, -2, -4, and -5 was carried out on different tissues (Table 1). All clinical samples were repeated in triplicate.
To estimate the EHV-5 DNA loads in the tissues, a real-time quantitative PCR (qPCR) was performed by targeting the gB gene (F: ATGTAGATTTCCAGGTGAT; R: TTTGGGTGTAGTAGTTGTA) and by using SYBR Green chemistry. Briefly, a 105-bp EHV-5 gB gene–specific DNA fragment was amplified and inserted into a vector sequence a to obtain the recombinant plasmid. To ensure that the 105-bp DNA fragment was correctly inserted, the cloned plasmid was sequenced. A standard curve, relating the PCR cycle number as a function of the viral DNA concentration, was constructed by serial dilution of the plasmid containing the 105-bp DNA insert, and each dilution was quantified by qPCR and used as the copy reference to construct the standard curve. A total of 7 reference points were utilized starting from 2.15 × 108 viral genome equivalents serially diluted 1:10 until 2.15 × 100. Genome equivalents were calculated by using the single plasmid molecular weight. Efficiency of the standard curve result was 94.6 % (R2 = 0.993).
For the quantification of EHV-5 concentrations in tissue samples, DNA was extracted with a commercial kit, b and the EHV-5–specific DNA fragments were amplified by qPCR using the EHV-5–specific primer pair previously described. The qPCR reactions were carried out as follows: 50 ng of DNA template were added to the master mix c with the 5-carboxy-X-rhodamine fluorochrome internal check. The PCR reactions were performed in a volume of 25 µl on a real-time PCR cycler, d and conditions were the same for all primer pairs: initial denaturation at 95°C for 10 min followed by 40 cycles of denaturation at 95°C for 30 sec, annealing at 58°C for 30 sec, and extension at 72°C for 30 sec. Fluorescence data were collected at the end of the extension step. Following cycling, the melting curve was determined to be in the range of 58–95°C, with a temperature increment of 0.01°C/sec. Each reaction was run in triplicate with appropriate negative controls. The EHV-5 concentration (viral genome equivalents/µl) was quantified by correlation with the standard curve. The detection limit was approximately 200 viral genome equivalents/50 ng. All clinical samples were screened blindly. Quantitative PCR found different EHV-5 DNA load in different organs, and the higher quantities were in lung, especially in the fibrotic nodule. The mean results of 3 experiments in the different organs are reported in Table 1.
Chronic interstitial pneumonia is rather common in horses, and can be related to viral, bacterial, or toxic causes; among viral causes, the role of EHV-5 is yet to be fully elucidated. EHV-5 has been associated with chronic interstitial pneumonia characterized by prominent interstitial to coalescing fibrosis and a slight interstitial inflammation; gross and histological lesions are suggestive, demonstrating intranuclear inclusion bodies in large macrophages and multinucleated giant cells. 15 The current report describes the EHV-5 DNA loads in tissues of a horse affected by EMPF. The EHV-5 status was assessed by qualitative EHV-5 PCR and quantified by EHV-5 qPCR. The quantitative real-time detection limit applied in the current study (200 viral genome equivalents/50 ng) was higher than those used in conventional qualitative semi-nested PCR assays (1–10 virus particles); for this reason, some of the tissues that were positive for EHV-5 in qualitative PCR tested negative by qPCR assay, showing viral burden below the cut-off value. On the other hand, the determination of viral load by qPCR offers the advantage of a better characterization of infection, as already indicated for EHV-1. 7
The viral load was higher in lung tissue and lung-related samples (TTW, BAL) than in other tissues. The 3 samples from the lungs had the highest viral loads, with the highest one detected in the sample collected from fibrous nodule. The data indicate that EHV-5 is more abundant in the most severely affected tissues, which in the present case was the fibrotic nodule.
The 2 examined bronchial lymph nodes had different viral loads; this finding could also be related to the severity of the histological lesion, more prominent on lymph node #1, which showed severe hyperplasia with numerous multinucleated giant cells that were otherwise scattered in lymph node #2. Compared to other tissues, in the present case, BAL appeared to be the clinical sample that better reflects the medium viral loads of the 3 sites of lung collected at necropsy (12,224 ± 9,620 viral genome equivalents/50 ng of DNA template), even better than the lung biopsy, which is a more invasive procedure and can sample slightly affected tissue, as in the current case. Quantitative molecular assays for the assessment of viral load is routinely used to monitor many diseases related to human gammaherpesviruses, 12 and BAL fluid has been demonstrated to be a more sensitive diagnostic sample than other biological samples to detect Epstein-Barr virus DNA in thoracic transplant recipients. 1,9
In the present case, it is not possible to draw conclusions about the effect of antiviral therapy on viral load detected in different clinical specimens, because the samples submitted to qPCR before and after the beginning of antiviral treatment were different (TTW vs. BAL #2). Neither the results obtained from the lung tissue sampled intra vitam nor postmortem are comparable, as the viral load in lung is markedly variable among different regions, and the biopsy was taken from diffusely affected parenchyma in an earlier stage rather than terminally from fibrotic nodules. At the time of biopsy, no obvious nodule was ultrasonographically evident. However, the severe and progressive clinical course suggests a poor efficacy of acyclovir administered orally in the horse, as previously reported. 5,6,16 On the other hand, steroid therapy could have induced an immunosuppression, facilitating viral replication.
The positivity of the samples for EHV-2 could suggest a possible synergistic effect of EHV-2 and EHV-5 in EMPF. This association was previously reported in 10 out of 33 other cases. 6,10,15
The presence of Candida spp., suspected by cytological exam of BAL #1 and isolated from lung tissue after enrichment, was not confirmed by histology. This could be an overgrowth due to an impairment in the respiratory system.
In spite of the high EHV-5 viral nucleic acid load in the lung biopsy, no virus was isolated from this sample after 5 passages on RK13. The RK13 cells are considered to be permissive to EHV-5 infection but an adjuvant factor may be necessary to cultivate EHV-5 on these cells. EHV-5 isolation is reported rarely: in 2 out of 7 documented cases of EMPF in one report 15 and in zero cases of horses with EMPF in other studies. 6,10,14
In conclusion, the current report presents quantitative analysis of EHV-5 loads in several tissues of an EMPF-affected horse. The results indicate that the most severely affected tissues, namely the lungs, have the greatest number of viral DNA copies, and this reinforces a possible causal relationship between EHV-5 and development of EMPF. The BAL seems to be a good sample to estimate EHV-5 load in lung, as reported previously. In this scenario, demonstration of different virus burden in affected and unaffected tissues can be useful to increase the knowledge about this disease. Additional animals should be investigated to generalize these results and to study the effects of antiviral therapy and the role of EHV-2 in EMPF.
Footnotes
a.
pCR®4 TOPO® vector sequencing kit, Invitrogen SRL, San Giuliano Milanese, Italy.
b.
QIAamp® DNeasy Tissue kit, Qiagen SpA, Milan, Italy.
c.
FastStart SYBR Green Master, Roche Diagnostics GmbH, Mannheim, Germany.
d.
Mx3000P instrument, Stratagene, La Jolla CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
