Abstract
Sarcoidosis is a rare equine skin disease characterized primarily by an exfoliative and granulomatous dermatitis but also presenting granulomatous inflammation of multiple systems. The current report presents the clinical and histopathological findings of sarcoidosis in a 16-year-old American Quarter Horse gelding with nested polymerase chain reaction Mycobacterium spp. DNA detection within hepatic and skin samples. Mycobacterium spp. may play a role in the pathogenesis of equine sarcoidosis as has been proposed for human sarcoidosis.
Equine sarcoidosis is a rare disorder characterized by histopathological evidence of an exfoliative and granulomatous dermatitis as well as granulomatous inflammation of multiple systems. 16 The onset of this disease may be insidious or rapid, 20 and cutaneous signs are usually the first to be observed. 11 In addition to the skin lesions, virtually all described cases of equine sarcoidosis have internal organ involvement. 22 Although the exact etiopathogenesis of this disorder is unknown,16,19,22 the described pathology has many similarities to that noted in human sarcoidosis16,17 and, for this reason, the equine disease received the same denomination. 17 It has been proposed that sarcoidosis in horses may be the result of an exaggerated immune response to an exogenous infectious agent or allergen 20 as is described in the medical literature, 12 but attempts to find an etiologic agent in equine cases have been unsuccessful.19,22 The current report presents the clinical and histopathological findings of sarcoidosis in a horse and demonstrates the possible involvement of Mycobacterium spp. in this disease.
A 16-year-old American Quarter Horse gelding was referred to evaluation with a 20-day history of wasting and insidious dermatitis beginning with focal dorsal alopecia that spread after 12 days to the entire body. The referring veterinarian suspected a fungal dermatitis and applied antifungal shampoos, but no improvement was observed. The gelding was fed alfalfa (Medicago sativa L.), coast cross (Cynodon sp.) hay, and 4 kg/day of commercial ration. Furthermore, the gelding was never treated with Bacillus Calmette–Guérin extracts.
At presentation, the gelding was lethargic and in poor body condition. Physical examination revealed normal heart and respiratory rates, normal body temperature, mild bilateral mucopurulent nasal discharge, and abnormal tracheal sounds on auscultation. Laminitis was diagnosed in all 4 limbs, with depression along the coronary bands noted in the forelimbs. Generalized exfoliative dermatitis (Fig. 1) was observed and was characterized by severe scaling, crusting, alopecia, and erythema (Fig. 2). Pruritus and pain were not associated with the skin lesions. Skin scrapings revealed no arthropods or fungal organisms. Skin biopsies of the neck, chest, and the shoulder were performed, and histopathological evaluation revealed diffuse granulomatous dermatitis with aggregates of epithelioid macrophages, multinucleated histiocytic giant cells, and few lymphocytes. There were multifocal epidermal erosions, spongiosis, and mild hemorrhage.
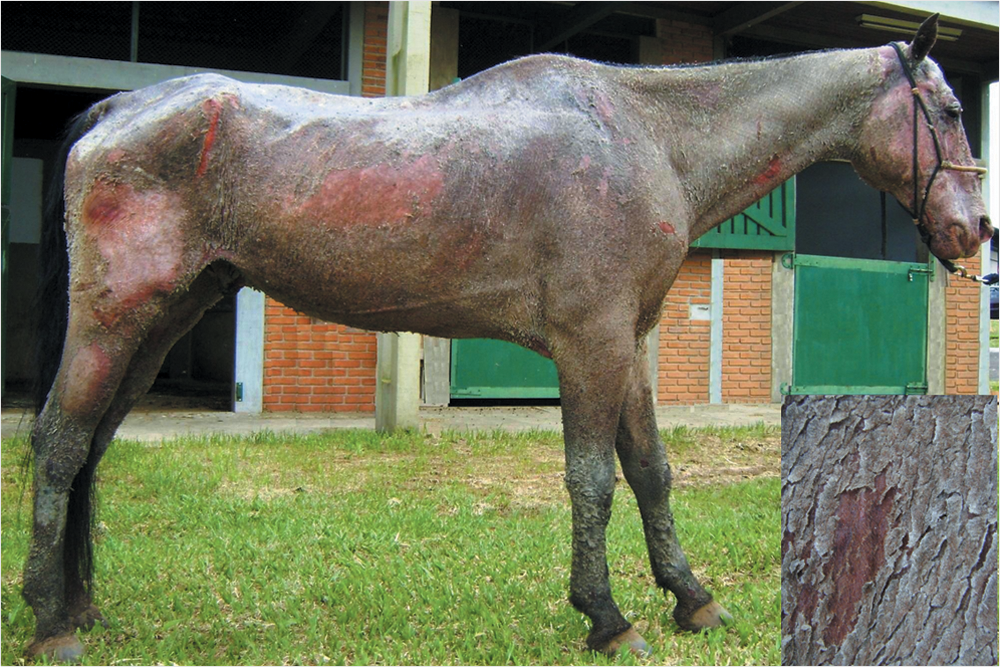
Generalized sarcoidosis–associated exfoliative dermatitis, with prominent scaling, crusting, and alopecia (inset).
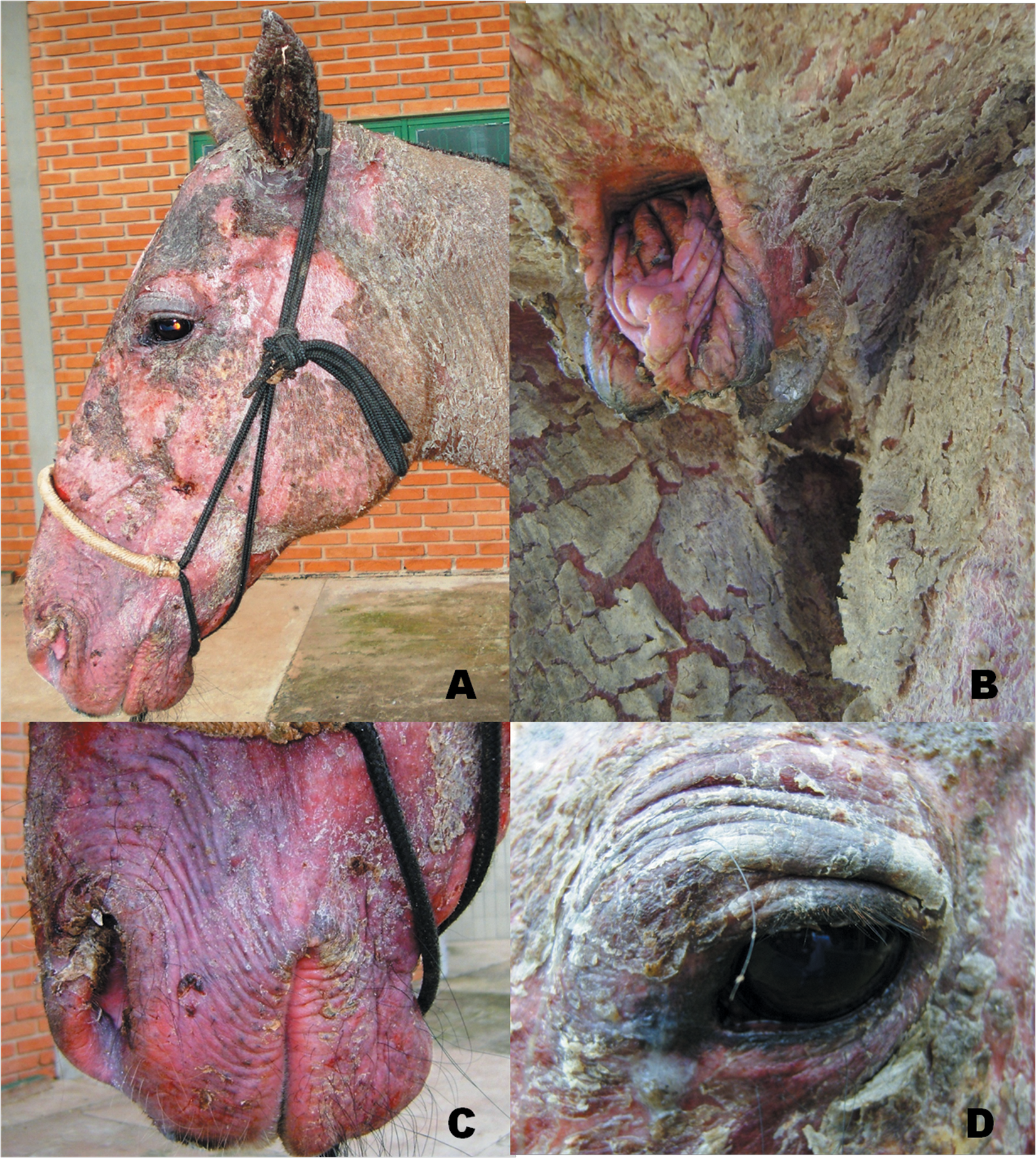
Perineal (
The gelding was in lateral recumbency for 2 days following referral to the hospital. Considering the severe wasting and worsening clinical evolution, generalized skin lesions, and laminitis with radiological evidence of rotation of the coffin bone in all limbs, the owner opted for euthanasia. Postmortem examination revealed peripheral subcutaneous ventral edema and lymphadenopathy. Lymph nodes were diffusely moderately swollen and firm. The liver contained multiple, white, firm, 0.5–3 cm in diameter nodules. Tissue samples of skin, liver, spleen, kidney, lymph nodes, stomach, lung, large and small intestine, and brain were fixed in 10% buffered formalin.
Histopathological examination of the tissues revealed superficial and deep granulomatous dermatitis characterized by epithelioid histiocytes and Langhans giant cells, often forming granulomas with occasional central necrosis and mineralization (Fig. 3). Multifocally, the epidermis contained large subcorneal pustules. There were also extensive epidermal ulcers with overlying serocellular crusts, composed of degenerate and necrotic neutrophils. Within the underlying skeletal muscle was a mild multifocal lymphohistiocytic rhabdomyositis. Histopathological examination of the liver revealed a moderate multifocal granulomatous and lymphoplasmacytic hepatitis with fibrosis and mild mineralization (Fig. 4). Multinucleated Langhans giant cells were also present. Multifocal moderate granulomatous lesions containing multinucleated Langhans-type giant cells were also identified within spleen, kidney, and lymph nodes. In the kidneys, there was a predominantly corticointerstitial lymphoplasmacytic and granulomatous nephritis admixed with multinucleated giant cells. In the spleen and lymph nodes, the granulomatous infiltrate was multifocal to coalescing with fewer lymphocytes, plasma cells, giant cells, and prominent hemosiderosis. The stomach had mild diffuse lymphoplasmacytic gastritis with rare multinucleated giant cells. No significant lesions were observed in the intestine, lungs, and brain. No Mycobacterium spp. antigens were detected via indirect immunohistochemistry. a Ziehl–Neelsen acid-fast, Grocott–Gomori methenamine silver, and periodic acid–Schiff (PAS) stains did not reveal microorganisms.
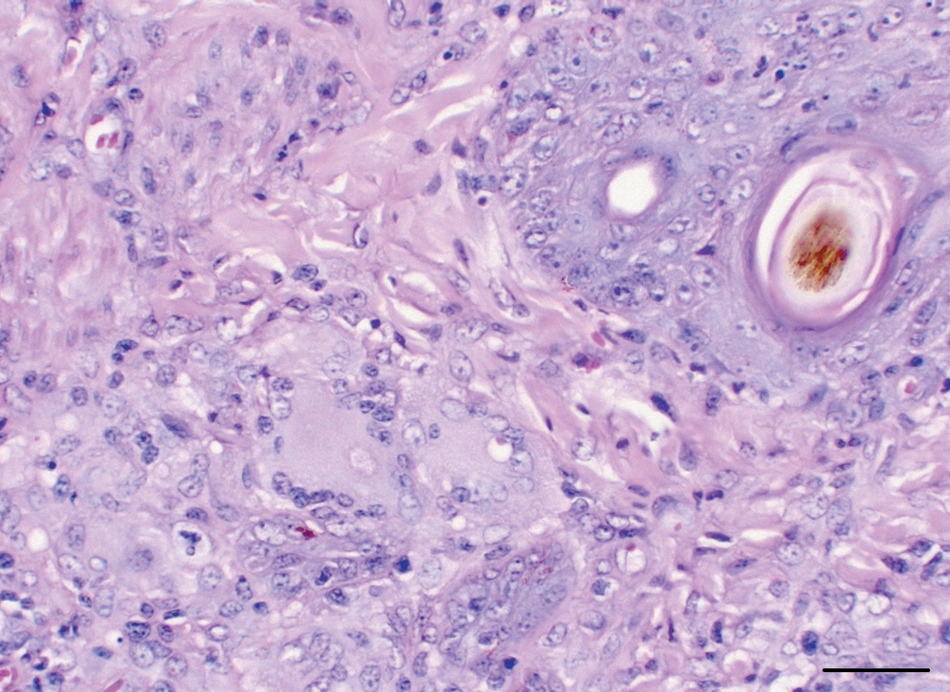
Skin; sarcoidosis; horse. Granulomatous dermatitis with Langhans giant cells, epithelioid macrophages, and a few lymphocytes and plasma cells. Hematoxylin and eosin. Bar = 100 µm.
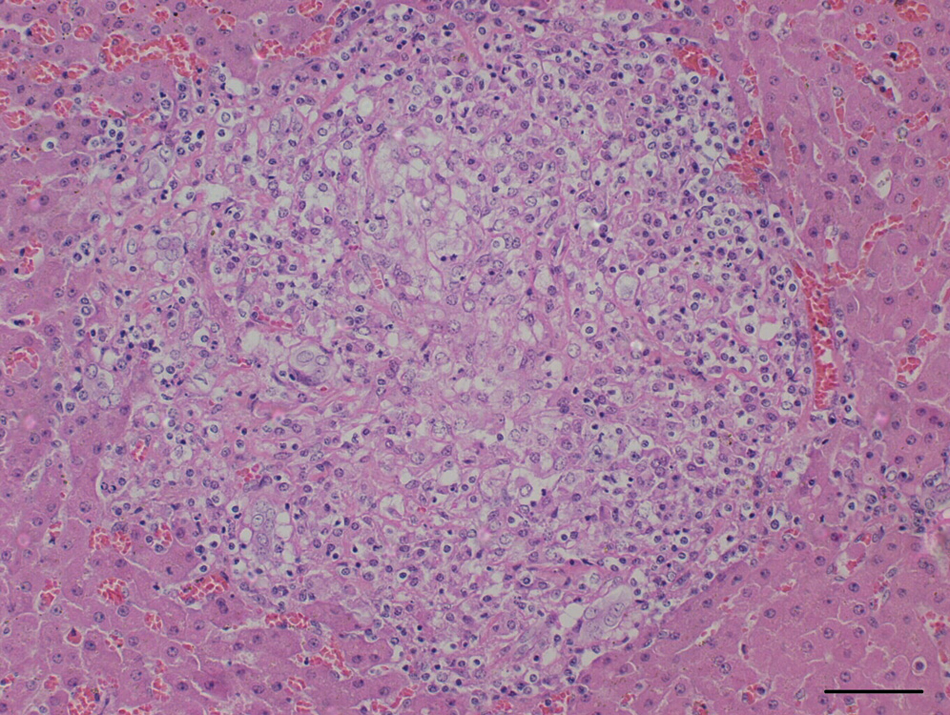
Liver; sarcoidosis; horse. Granuloma with peripheral lymphocytes and plasma cells. Hematoxylin and eosin. Bar = 100 µm.
Hepatic and skin samples with compatible histopathological diagnoses of sarcoidosis were evaluated for the presence of mycobacterial DNA by nested polymerase chain reaction (nPCR) and DNA sequencing. Five-µm-thick sections were obtained from formalin-fixed, paraffin-embedded tissue and immediately placed into 1.5-ml sterile plastic microtubes, DNA/RNA free, with 1 ml of xylol analytical grade reagent b to remove paraffin. Residual xylol was removed by the addition of 1 ml of 100% ethanol. b Bacterial DNA was extracted from samples using a genomic DNA tissue kit, c following the manufacturer’s instructions. DNA samples were also obtained from hepatic and skin formalin-fixed tissues using a genomic DNA mini kit. d The relative purity and quality of the isolated DNA was determined by spectrophotometry, e and the ratio of A260/A280 nm exceeded 1.8 for all preparations. Nested PCR amplification was performed using sets of primers previously described from Mycobacterium spp. 16S ribosomal RNA (rRNA) gene region. 8 First-round primer sets used in nPCR, amplifying 590 base pairs (bp), were forward 246 (5′-AGAGTTTGATCCTGGCTCAG-3′) and reverse 247 (5′-TTTCACGAACAAGCCCAGAA-3′). First PCR was set in duplicate in a total of 25 µl each, which contained 0.4 µM of each forward 246 and reverse 247 primers, 5.0 µl of template DNA, 12.5 µl of PCR master mix, f and 5.5 µl of nuclease-free water. In addition, a “no-template” control in duplicate was performed to show that contamination was absent. Furthermore, all handling, extraction, and amplification procedures were performed in separate laboratories using disposable tips with barrier protection. Cycling conditions g were as follows: 1 denaturation cycle (95°C, 2 min); 30 cycles with denaturation (95°C, 30 sec), annealing (60°C, 60 sec), and extension (72°C, 60 sec); and a final extension (72°C, 7 min). Second-round primers set (amplifying 455 bp) were forward M1 (5′-AGTGGCGAACGGGTGAGTA AC-3′) and reverse R7 (5′-TTACG CCCAGTAATTCCGGA CAA-3′). Ten microliters from the first PCR at a 1:20 dilution was added to 40 µl of nPCR master mix (0.4 µM of each forward M1 and reverse R7 primer, 25 µl of PCR master mix, e and 11 µl of nuclease-free water). Cycling g parameters were: 1 denaturation cycle (95°C, 2 min); 25 cycles with denaturation (95°C, 30 sec), annealing (68°C, 60 sec), and extension (72°C, 30 sec); and a final extension (72°C, 5 min). Negative control was prepared, using the first blank sample (first PCR). The PCR products were analyzed after electrophoresis in 1.5% agarose gels. The PCR products with predicted size of 455 bp were purified using a PCR purification kit. h As laboratory control, equine liver and skin tissue samples from 2 horses that died of the noninflammatory disease were used. These control samples were subjected to the same procedures performed in positive sarcoidosis samples (i.e., formalin-fixation, DNA purification, PCR, and nPCR).
Nested PCR from skin and liver tissue amplified a product with the expected size. Automated direct sequence analysis i performed in quadruplicate using a DNA sequencing kit j using 5 µl each of M1 or M7 primers and 10 µl of nPCR product showed that both sequences were 100% similar. Sequence obtained was deposited in GenBank (accession no. JF518933), and BLAST (http://blast.ncbi.nlm.nih.gov/) was used to verify sequence homology against previously available sequences deposited in GenBank and Ribosomal Differentiation of Medical Microorganisms (http://rdna.ridom.de/). Sequence results of the PCR products presented 100% homology with the 16S rRNA sequence of Mycobacterium sacrum (GU201853.1), Mycobacterium frederiksbergense (AF544629.1), and other Mycobacterium sp. (HM107174.1, EU836191.1, EU167969.1). Lower identity degree was also observed with Mycobacterium smegmatis (NC008596.1, 96%), Mycobacterium brasiliensis (EU165538.1, 96%), Mycobacterium abscessus (NC010397.1, 95%), Mycobacterium tuberculosis (AELF01000052.1, 90%), Mycobacterium bovis (AEZH01000004.1, 90%), Mycobacterium avium (AFPI01000125.1, 90%), and Mycobacterium intracellulare (ABIN01000139.1, 90%). No amplification was obtained from the control samples.
Equine sarcoidosis, also known as equine idiopathic granulomatous disease 7 and generalized or systemic granulomatous disease,2,16,20 is a rare dermatitis in horses. 16 Previous cases investigated via molecular techniques did not detect etiologic agents in association with equine sarcoidosis.19,22 The current study, using molecular techniques, demonstrated the presence of Mycobacterium spp. in tissues with lesions compatible with equine sarcoidosis. No age, sex, or breed predilection of sarcoidosis has been described in horses14,20; however, a higher incidence of sarcoidosis was observed in geldings, and castration, therefore, is considered a possible risk factor. 19 The gelding in the present case was castrated several years prior to the onset of the clinical signs. Hairy vetch (Vicia villosa) intoxication has been associated with a similar generalized granulomatous disease in horses1,23 and cattle. 9 However, the majority of the horses described in the literature with sarcoidosis had no reported exposure to hairy vetch.16,20 As this was also the case with the gelding in the present study, and as equine sarcoidosis is observed in geographic regions and pastures where hairy vetch is not present, 19 hairy vetch intoxication was not considered to be a potential differential in the current case. Diffuse granulomatous inflammation has been reported in the kidneys of horses infected with Halicephalobus gingivalis, 10 a nematode affecting horses and human beings.4,10,21 However, in such previous reports, adult and larval nematodes were typically found in association with renal granulomatous lesions, 10 and there was no evidence of parasitic infection in any of the tissues examined in the present case. In addition, H. gingivalis was not considered to be a potential differential in the current case, due to the absence of central nervous system pathology in this animal.4,10,21 Also, H. gingivalis infection has a low incidence in Brazil. 21
In cases of equine sarcoidosis, cutaneous signs are usually the first to be detected. 11 In the current case, clinical evidence of wasting was associated with a rapidly spreading generalized exfoliative dermatitis. The cutaneous scaling, crusting, and alopecia of the face, thorax, and limbs were similar to other described cases14,19 and are commonly reported in horses with sarcoidosis. 20 Rarely skin lesions in sarcoidosis may also include nodules or larger tumor-like masses.2,11,20 Nodular and exfoliative forms may occur simultaneously,16,20 a feature that was not observed in the present case. Pruritus and pain are common signs in equine cases 19 but were not observed in this animal. Lethargy and severe wasting are frequently observed,7,16,17 but normal body condition also occurs in some cases. 14 Other reported systemic clinical signs include intermittent fever, lymphadenopathy,7,16,17 peripheral edema, diarrhea, and icterus.11,19,20
The skin, lungs, lymph nodes, and gastrointestinal tract are also commonly affected in cases of equine sarcoidosis,2,7,14,19 whereas organs less commonly affected include the liver, spleen, kidneys, musculoskeletal system, heart, adrenal and thyroid glands, pancreas, and the nervous system.11,16,19,20 Unlike the equine and bovine disease, human sarcoidosis is associated with hypercalcemia and soft tissue mineralization. 15 In the present case, the cutaneous and hepatic granulomas were also mildly mineralized.
The diagnosis of equine sarcoidosis is largely based on the identification of the suggestive histopathologic lesions and the exclusion of other infectious agents. 20 Cutaneous differential diagnoses include dermatophilosis, dermatophytosis, pemphigus foliaceus, lymphoma, systemic lupus erythematosus, and eosinophilic dermatitis. 16 Histopathologic lesions observed in the horse in the current study were compatible with equine sarcoidosis.
In a previous report evaluating this disease, no Equid herpesvirus 1 (EHV-1) or EHV-2 DNA was detected in 8 horses with equine sarcoidosis. 22 However, EHV-2 has been detected via PCR in a case of equine granulomatous dermatitis. 18 Histochemistry, immunohistochemistry, and PCR assays on paraffin-embedded cutaneous specimens from the 8 horses with histopathological evidence of sarcoidosis did not reveal Mycobacterium spp., Coccidioides immitis, Cryptococcus neoformans, Corynebacterium pseudotuberculosis, and Borrelia burgdorferi. 19 Ziehl–Neelsen, Grocott–Gomori, and PAS stains did not reveal microorganisms in the present case. However, mycobacterial DNA was detected in the hepatic and skin lesions by nPCR, and the products obtained were sequenced demonstrating 100% homology with other Mycobacterium sequences deposited in GenBank. Sample contamination of environmental Mycobacterium spp. was unlikely because all negative and laboratory control cases were negative and did not amplify Mycobacterium spp. 16S rRNA sequence with PCR and nPCR. The absence of detectable mycobacteria via histochemistry and indirect immunohistochemistry does not rule out the presence of their antigens and nucleic acid and their possible role in this disease. Although the mycobacterial DNA detection may indicate a previous exposure instead of true infection, there was no history information confirming this hypothesis. Mycobacterial infections are rarely diagnosed in horses, 13 but some Mycobacterium species were already associated with skin disease in horses. Mycobacterium smegmatis has been isolated from cutaneous abscess, 3 and M. intracellulare was associated with avian mycobacterial dermatitis in a horse. 6
Although the etiology of sarcoidosis remains unknown, recent molecular, genetic, and immunologic studies in human beings seem to indicate the association of sarcoidosis with infectious agent antigens such as Propionibacterium and Mycobacterium species. 5 Although PCR assays failed to detect the DNA of Mycobacterium spp. in previous cases of equine sarcoidosis, 19 the detection of mycobacterial DNA in the current case indicates that Mycobacterium spp. may play a role in this disease.
Footnotes
a.
Rabbit polyclonal antibody against Mycobacterium spp., Dako North America Inc., Carpinteria, CA.
b.
Merck KGaA, Darmstadt, Germany.
c.
QIAamp® DNA FFPE tissue kit, Qiagen Inc., Valencia, CA.
d.
QIAamp® DNA Mini kit, Qiagen Inc., Valencia, CA.
e.
NanoDrop® 2000 Spectrophotometer, Thermo Scientific, Wilmington, DE.
f.
GoTaq® Green PCR Master Mix, Promega Corp., Madison, WI.
g.
Mastercycler® ep gradient S, Eppendorf AG, Hamburg, Germany.
h.
QIAquick® PCR Purification Kit, Qiagen Inc., Valencia, CA.
i.
3500 Genetic Analyzers, Applied Biosystems, Foster City, CA.
j.
BigDye® Terminator v3.1 Cycle Sequencing Kit, Applied Biosystems, Foster City, CA.
The author(s) declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The author(s) declared that they received no financial support for their research and/or authorship of this article.
