Abstract
Seven juveniles and 3 adults from a closed group of 19 rock hyraxes (Procavia capensis) housed in a zoo's indoor rock exhibit died or were euthanized after developing blepharoconjunctivitis and orofacial ulcers over a 2-week period. Histopathologic examination of dermal ulcers and ulcerated tongues revealed amphophilic to basophilic intranuclear inclusion bodies in epithelial cells bordering ulcers. Epithelial cells with inclusion bodies were often characterized by cytomegaly and karyomegaly, and many cells had formed syncytia. Examination of inclusion bodies in tongue epithelium by transmission electron microscopy revealed icosahedral nucleocapsids, approximately 80–95 nm in diameter, with morphologic features consistent with herpesvirus. Cytopathic effect (CPE) typical of alphaherpesvirus infection was seen in bovine turbinate, equine dermal, and Vero cell monolayers after inoculation with homogenates of the skin lesions, but CPE was not seen after inoculation onto Madin-Darby canine kidney or swine testicle cell monolayers. Polymerase chain reaction analysis using degenerate primers that targeted a portion of the herpesvirus polymerase gene generated a product of approximately 227 base pairs. The product was cloned, sequenced, and then analyzed using BLAST. At the nucleotide level, there was 86%, 77%, and 76% shared identity with Eidolon herpesvirus 1, Human herpesviruses 1 and 2, and Cercopithecine herpesvirus 2, respectively. Herpesvirus infections in rock hyraxes have not been characterized. The data presented in the current study suggest that a novel alphaherpesvirus caused the lesions seen in these rock hyraxes. The molecular characteristics of this virus would tentatively support its inclusion in the genus Simplexvirus.
Herpesviruses are double-stranded, enveloped DNA viruses with a worldwide distribution. The viruses infect a wide variety of mammals, birds, fish, reptiles, amphibians, and invertebrates, and they establish lifelong latent infections in their hosts. The family Herpesviridae is divided into 3 subfamilies: Alphaherpesvirinae, Betaherpesvirinae, and Gammaherpesvirinae. The subfamily Alphaherpesvirinae is divided into 2 major genera: Simplexvirus and Varicellovirus. The current report describes infections caused by a novel alphaherpesvirus with molecular characteristics of Simplexvirus in zoo rock hyraxes.
Seven juveniles and 3 adults from a closed group of 19 rock hyraxes (Procavia capensis) housed in a zoo's indoor rock exhibit died or were euthanized over a 2-week period after they developed blepharoconjunctivitis, orofacial ulcers, dyspnea, and weight loss and after they became nonresponsive to therapeutic treatment. Formalin-fixed specimens of skin, tongue, stomach, pancreas, small intestine, colon, lung, liver, spleen, heart, adrenal, kidney, and urinary bladder from 8 animals, and brain from 1 of these animals, were submitted for histopathologic examination. Two animals submitted for complete necropsies had focally extensive ulceration of periorbital and perioral skin (Fig. 1) and the tongue; there were no additional gross lesions. Histopathologic examination of dermal ulcers and ulcerated tongues from all animals revealed necrosis of scattered epithelial cells and amphophilic to basophilic intranuclear inclusion bodies in epithelial cells bordering ulcers. Epithelial cells with inclusion bodies were often characterized by cytomegaly and karyomegaly, and many cells had formed syncytia (Fig. 2). Examination of inclusion bodies in tongue epithelium by transmission electron microscopy revealed icosahedral nucleocapsids, approximately 80–95 nm in diameter, with morphologic features consistent with herpesvirus (not shown). One of the 3 brains examined histologically was characterized by mild nonsuppurative meningoencephalitis and trigeminal ganglionitis; no viral inclusion bodies were seen. There were no additional histopathologic lesions in any of the animals. No bacterial pathogens were isolated from swabs of the skin lesions or from specimens of lung, liver, kidney, or brain from the 2 animals submitted for necropsy.
Homogenates of the skin lesions were inoculated onto bovine turbinate (BT), equine dermal (ED), Vero (African green monkey kidney epithelial), Madin-Darby canine kidney (MDCK), and swine testicle (ST) cell monolayers. Cytopathic effect (CPE) typical of alphaherpesvirus infection was seen in BT, ED, and Vero cell monolayers, but CPE was not seen in MDCK or ST cell monolayers. Negative stain immunoelectron microscopy 2 using convalescent serum from a surviving hyrax revealed enveloped virions, 150–180 nm in diameter, with morphologic features consistent with herpesvirus morphology (Fig. 3).
DNA was purified from isolated virus using a commercial kit, a and nested polymerase chain reaction (nPCR) was performed using degenerate primers that target a portion of the herpesvirus polymerase gene. 14 This particular nPCR has generated products from 215 to 315 bp in most herpesviruses tested. 14 The outer PCR used 2 forward primers (5′-GAYTTYGCNAGYYTNTAYCC-3′ and 5′-TCCTGGACAAGCAGCARNYSGCNMTNAA-3′) and 1 reverse primer (5′-GTCTTGCTCACCAGNTCNCANCCYTT-3′). The nPCR used 1 forward primer (5′-TGTAACTCGGTGTAYGGNTTYCANGGNGT-3′) and 1 reverse primer (5′-CACAGAGTCCGTRTCNCCRTADAT-3′). The outer and nPCR reaction mixtures (50 μl) each contained 1 μmol of their respective primers, 200 μmol deoxynucleotide triphosphate, 1.25 U Taq polymerase, b 2 mmol MgCl, and 2 μl of viral DNA template (or outer PCR product in the nPCR) in the commercially supplied buffer. b Cycling parameters for both methods included denaturation at 94°C for 30 sec, annealing at 46°C for 60 sec, and 60 sec of extension at 72°C for a total of 45 cycles. After cycling was completed, an additional 7 min of extension was performed at 72°C. The nPCR product (15 μl) was analyzed by ethidium bromide–stained gel electrophoresis in a 2% agarose gel, c and it was visualized and photographed with an ultraviolet transilluminator. c Bands of approximately 220 bp were obtained from hyrax virus DNA, Equid herpesvirus 1, Human herpesvirus 1 (HHV-1), and Ovine herpesvirus 2. Amplified products were excised from the gel, purified using a gel extraction kit, a and cloned into pCR2. 1 using a commercial Taq-amplified cloning kit. d Clones were sequenced in both directions at the Iowa State University Sequencing Facility (Ames, IA) using a commercial DNA analyzer. e Plasmid sequenceswere edited out, and sequences (231 bp) were submitted to the Basic Alignment Sequence Tool (BLAST 2.2.18+; http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) to identify sequence homologies within GenBank. Sequences were translated using the Translate+ tool of the University of Wisconsin Package GCG program f and were submitted for BLASTP analysis. At the nucleotide level, there was 86%, 77%, and 76% shared identity with Eidolon herpesvirus 1 (EiHV-1), HHV-1 and −2, and Cercopithecine herpesvirus 2 (CeHV-2), respectively. At the amino acid level, there was 92% shared identity with EiHV-1; 83% shared identity with HHV-1, CeHV-2, and CeHV-16; and 81 % and 79% shared identity with CeHV-1 and HHV-2, respectively. The sequence of the rock hyrax herpesvirus was deposited in GenBank under accession number EU760899.
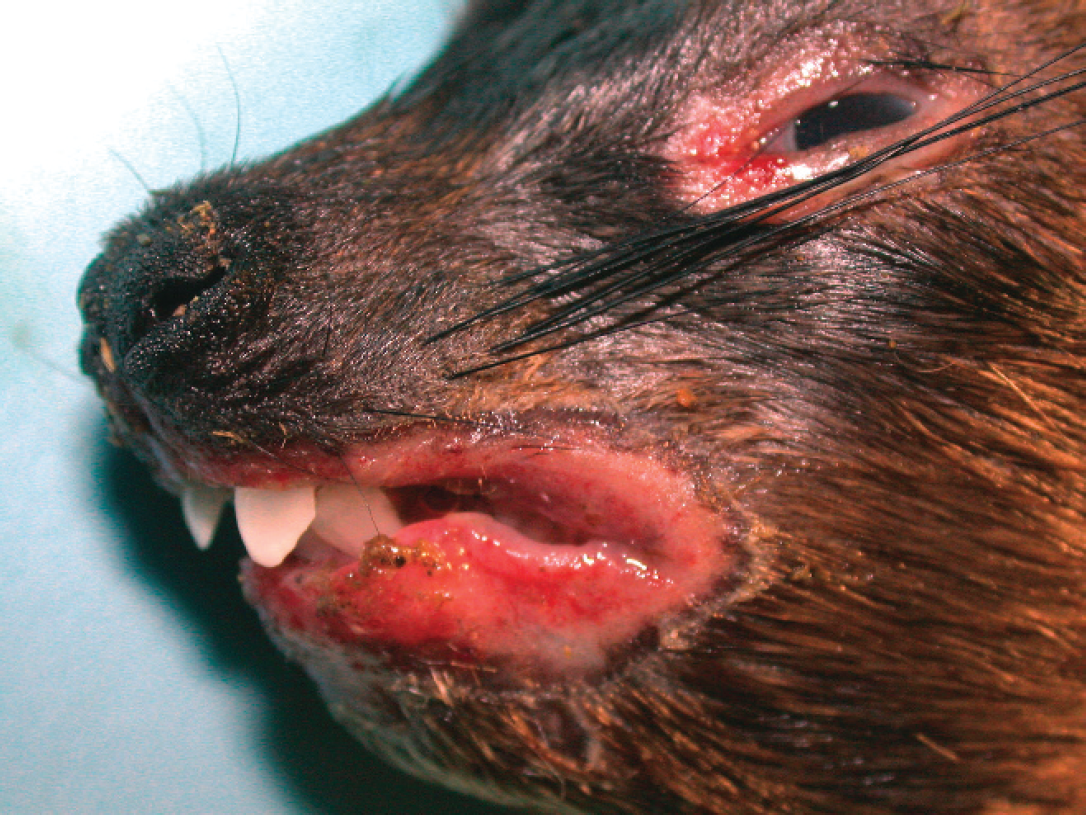
Rock hyrax (Procavia capensis) with focally extensive ulceration of periorbital and perioral skin.
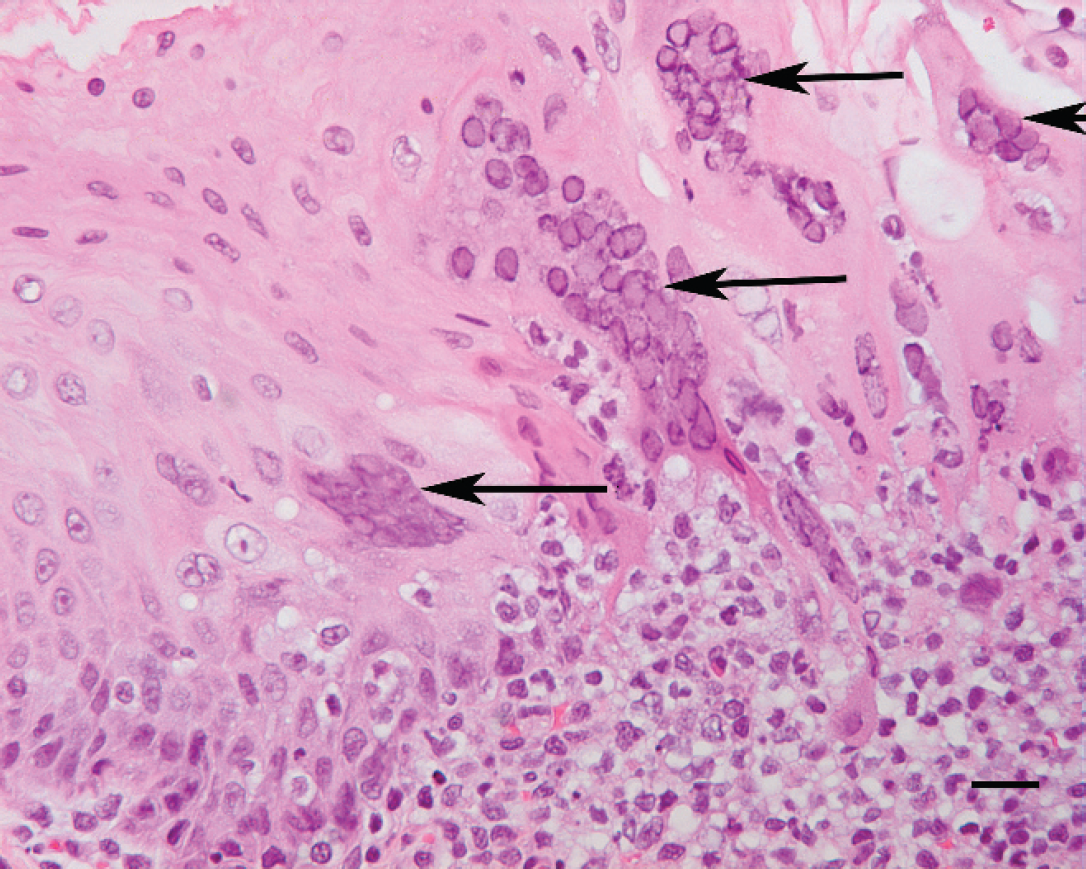
Tongue, rock hyrax (Procavia capensis). Amphophilic intranuclear inclusion bodies are in epithelial cells that have formed syncytia (arrows), and mixed inflammatory cells have infiltrated the adjacent propria-submucosa. Hematoxylin and eosin. Bar = 20 μm.
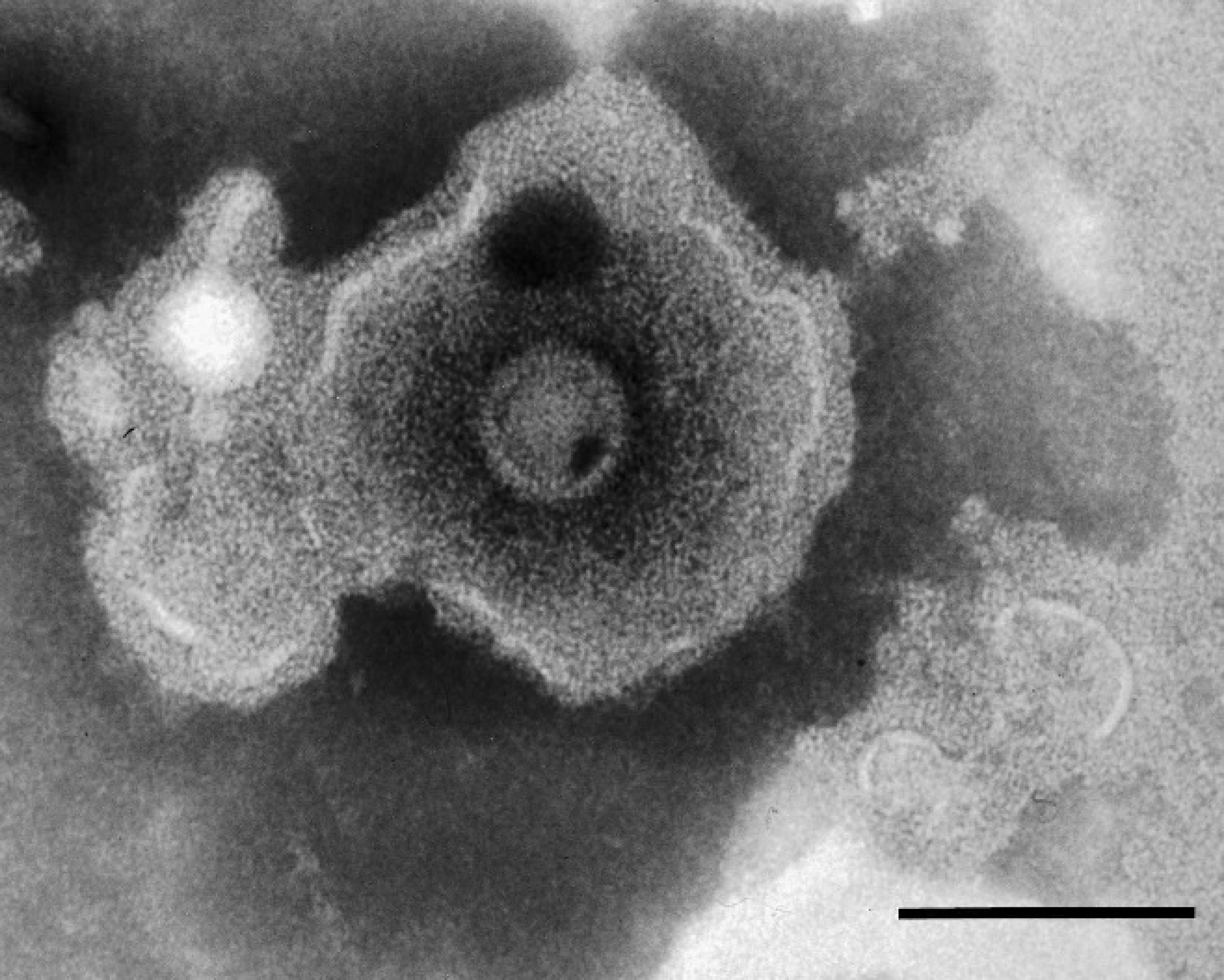
Cell culture isolate of rock hyrax herpesvirus. Negative stain immunoelectron microscopy photomicrograph of an enveloped virion. Bar = 100 μm.
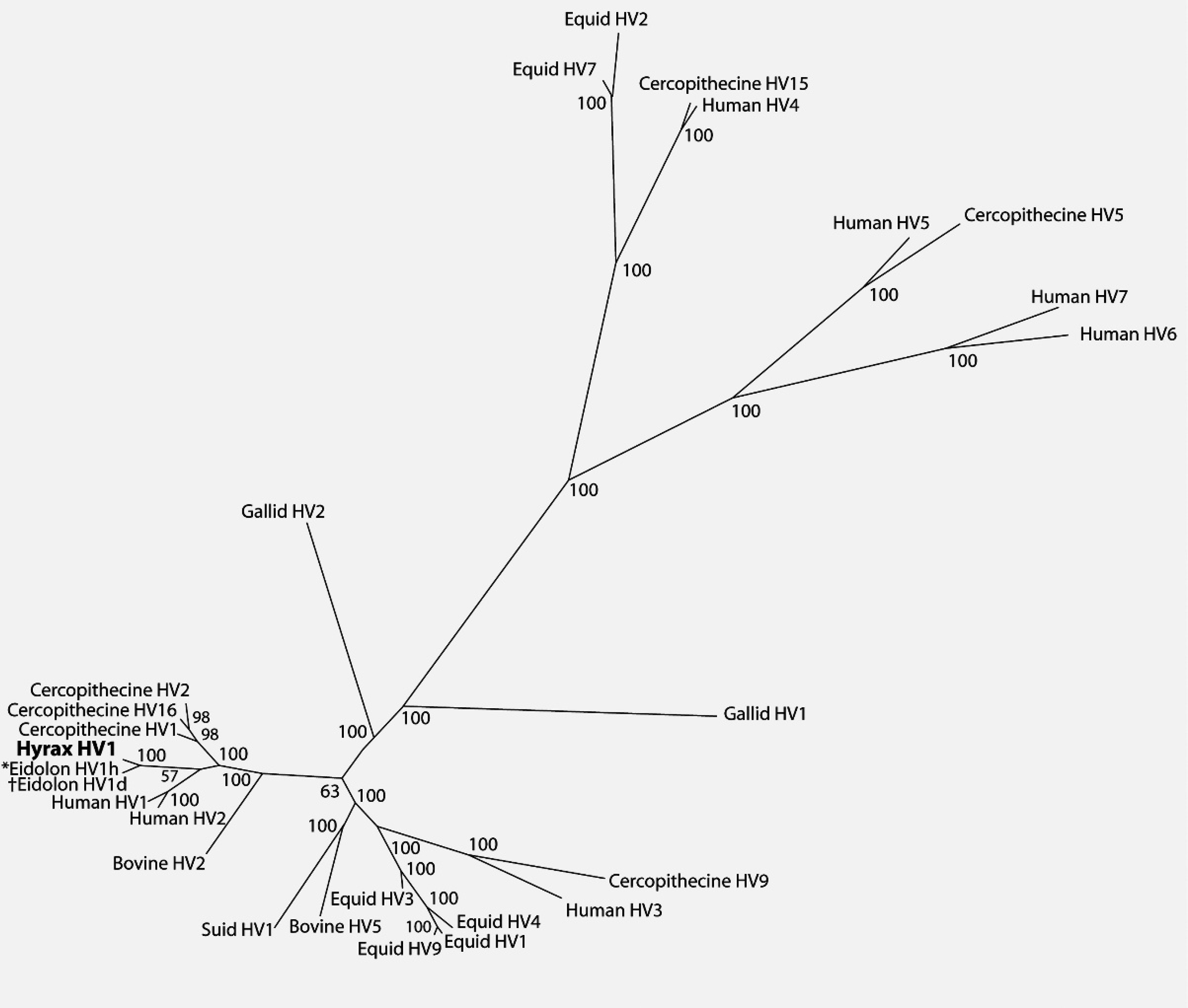
Bayesian phylogenetic tree of predicted herpesviral DNA-dependent–DNA polymerase (catalytic subunit) amino acid sequences based on MUSCLE alignment. Numbers indicate Bayesian posterior probabilities of the branching. *Eidolon HV1d and †Eidolon HV1h indicate alphaherpesvirus isolates recovered from frugivorous bats Eidolon dupreanum and Eidolon helvum, respectively.
An initial phylogenetic analysis comparing the hyrax herpesvirus sequence with representative alpha-, beta-, and gammaherpesviruses is shown in Figure 4. Full-length protein sequences of the representative species (Table 1) were retrieved from GenBank and aligned via MUSCLE 4 with the translated hyrax herpesvirus fragment. Bayesian analysis of the amino acid multiple alignment was performed using MrBayes 3.1 13 with gamma distributed rate variation and a proportion of invariant sites, and mixed amino acid models. Four chains were run and statistical convergence was assessed via the standard deviation of split frequencies. The first 25% of 1,000,000 generations were discarded as the burn in.
The biological characteristics of the hyrax herpesvirus would support its inclusion in the subfamily Alphaherpesvirinae, and its molecular characteristics would tentatively support its inclusion in the genus Simplexvirus. This is somewhat unexpected because the current model of herpesvirus evolution postulates that these viruses have co-evolved with their host species. 8,9 The genus Simplexvirus includes 7 human or primate viruses. 3 The remaining members include Bovine herpesvirus 2 (BoHV-2), 2 wallaby viruses, and recently described frugivorous bat (Eidolon) viruses, 3,5,7,12 but these viruses also do not fit the model of herpesvirus/host co-evolution. The data indicate that members of Simplexvirus co-evolved with their primate and human hosts approximately 78 million years ago, 8,9 whereas phylogenetic divergence of cattle and bats, hyraxes, and marsupials occurred approximately 87, 103, and 130 million years ago, respectively. 1,6,10,11 The discrepancy with BoHV-2 and the wallaby viruses has been explained as an interspecies transfer of viruses, 9 and this may be true for the hyrax virus. Although the hyrax herpesvirus sequence available from the current investigation is very short, it is an excellent target for more detailed phylogenetic analysis because it is contained in the highly conserved polymerase gene. It will be necessary to obtain a complete sequence of the polymerase gene and sequences from other regions of the genome to more closely examine the homologous relationship of this virus to the human and primate viruses. Future studies should also compare the hyrax herpesvirus sequence with sequences of alpha-herpesviruses recovered from phylogenetic relatives of the hyrax, such as elephants, manatees, and dugongs.
The data presented in the current study indicates that a novel alphaherpesvirus caused the lesions seen in these rock hyraxes. The severity of the lesions might suggest that an interspecies transfer of the virus occurred, but the source of the infections is unknown. The hyraxes were on exhibit by themselves with exposure only to a variety of free-flight birds. A variety of frugivorous bats were housed in an adjacent facility, and zookeepers serviced both facilities. Reactivation of latent infections is also possible because no new animals had been introduced into the group. The only stressors identified were changes in group dynamics, which juveniles born 3 months earlier could have created. All affected hyraxes were initially treated with antibiotics. They were then treated with antibiotics and acyclovir once the herpesvirus infections were diagnosed. Three treated hyraxes survived, but acyclovir therapy was perceived to have had no positive effect. Six hyraxes from this closed group remained clinically normal throughout the outbreak.
Acknowledgements. The authors thank Tim Moural and Drs. Alan Doster and Fangrui Ma for technical assistance.
NA = entire genome sequence not available.
Footnotes
a.
QIAamp® DNA Blood Mini Kit, QIAEX II® Gel Extraction Kit; Qiagen Inc., Valencia, CA.
b.
Promega Corp., Madison, WI.
c.
UVP LLC, Upland, CA.
d.
TOPO® TA Cloning® Kit, Invitrogen Corp., Carlsbad, CA.
e.
3730xl DNA Analyzer, Applied Biosystems, Foster City, CA.
f.
Copyright © 1982–2005 Accelrys Inc. All rights reserved.