Abstract
Early diagnosis of Porcine reproductive and respiratory syndrome virus (PRRSV) is critically important for control of the disease. Two new commercially available enzyme-linked immunosorbent assays (ELISAs) based on different methodologies have been developed. In the present report, the 2 ELISAs were compared using blood samples from experimentally and naturally infected pigs. One of the 2 ELISAs was shown to be more sensitive than the other. The higher sensitivity of one of the ELISAs could pose a problem in PRRS diagnosis in endemic farms, because it can detect maternally derived antibodies for a longer time, overlapping with the detection of antibodies developed after PRRSV infection. However, the ELISA with higher sensitivity could be suitable for early detection of PRRSV antibodies in individual pigs, especially in PRRS-free herds.
Keywords
To date, both inactivated and attenuated vaccines against Porcine reproductive and respiratory syndrome virus (PRRSV; order Nidovirales, family Arteriviridae, genus Arterivirus) are available, but control and eradication of the disease are far from being achieved. 2 Early and accurate diagnosis is important for establishment of a control program. At present, the earliest way to detect a positive animal is by reverse transcription polymerase chain reaction (RT-PCR). If detection of antibodies was feasible at very early times postinfection, the cost of early diagnosis could be substantially lowered.
The most abundant viral protein in PRRSV is the nucleocapsid (N) protein that is highly immunogenic and induces early antibody responses in pigs. 3 This protein has been used extensively in the development of commercial enzyme-linked immunosorbent assays (ELISAs) for PRRSV diagnosis. Recently, 2 PRRSV ELISAs based on different methodologies (double-recognition ELISA and indirect ELISA) have been developed for the detection of antibodies against N protein. The principle of the double-recognition ELISA is based on simultaneous binding of an antibody sandwiched between precoated and enzyme-conjugated antigens. 4 The double-recognition ELISA used in the present study (E1, a hereafter) was developed with recombinant N protein from genotype I PRRSV serving as both the coating antigen and, in horseradish peroxidase (HRP)–conjugated form, the signal. Briefly, after coating with recombinant N protein, plates were washed 3 times and blocked with bovine serum albumin diluted at 1% in phosphate buffered saline. Then, 20 µl of sera were diluted 1:5 in 80 µl of the HRP-conjugated N protein and added in a one-step incubation. Then, the reaction was revealed by the addition of tetramethyl benzidine substrate and stopped with 0.5 M sulfuric acid. The indirect ELISA (E2, b hereafter) was designed to detect PRRSV antibodies using a recombinant protein as the coating antigen, and an anti-pig immunoglobulin (Ig)G-HRP conjugate in a second step. The diagnostic specificity of E1 is 99% (Ranz A, et al.: 2010, 4th Annual Meeting EPIZONE, 7–10 June 2010, Saint Malo, France), and the diagnostic specificity of E2 was 99.9% (Leathers V, et al.: 2009, International PRRS Symposium, 4–5 December 2009, Chicago, IL). The present report describes the comparison of these new commercially available ELISAs for detection of PRRSV antibodies in pigs experimentally and naturally infected with genotype I strains.
Four-week-old piglets (n = 36) obtained from a high-health farm historically free of PRRSV and reconfirmed to be free of PRRSV by ELISA and RT-PCR were experimentally infected with 6 selected strains of PRRSV genotype I subtype 1–6 (S1–S6). There were 6 piglets per subtype infection group. Sera were collected weekly from 0 to 84 days postinoculation (DPI). All PRRSV isolates were produced in porcine alveolar macrophages and corresponded to field strains circulating in Europe. Similarity of N protein among the 6 strains ranged from 90.6% to 96.0%, as determined by sequencing of open reading frame 7 and prediction of the corresponding protein’s amino acid sequence. In all cases except one (S3), animals were intranasally inoculated with 2 ml of ≥1 × 105.0 TCID50/ml. Pigs in S3 were infected with a lung homogenate of unknown virus titer from an infected pig. Sera for cross-sectional profiling of field cases were obtained from pigs from 3 PRRSV endemic farms sampled at different weeks of age; farm 1 (n = 5) at 9, 11, 15, and 24; farm 2 (n = 10) at 3, 5, 7, and 9; and farm 3 (n = 10) at 3, 6, 9, 12, 15, 18, and 21. Comparison of the performance of both ELISAs in a longitudinal follow-up was carried out in 2 PRRSV endemic farms (n = 45 in each farm; sampled at 4, 6, 8, 10, 12, 15, 17, and 20 weeks of age).
Sera were examined by 2 ELISAs (E1 and E2) according to manufacturer’s instructions. Results were expressed as a ratio of the optical density (OD) of a given sample over the OD of the positive control provided (sample-to-positive [S/P] ratio). The positive S/P threshold values were >0.175 for E1 and ≥0.4 for E2. Statistical analyses were performed using chi-square testingc and by computing kappa values.d
After inoculation, viremia was demonstrated in all pigs by RT-PCR using serum samples (7–35 DPI). In pigs inoculated with strains 5 and 6, viremia was also demonstrated by viral titration in swine alveolar macrophages: 7–14 DPI for strain 5 (mean titer = 2.9 ± 0.5 TCID50/ml at 14 DPI) and 7–35 DPI for strain 6 (mean titer = 2.2 ± 0.2 TCID50/ml at 35 DPI).
Table 1 summarizes results obtained from the experimental infections. With both ELISAs, all pigs were always positive from 21 DPI onward, but at earlier times, sensitivity depended on the assay used and on the PRRSV strain inoculated. At 7 DPI, 64% of the infected pigs were detected as positive by E1 (95% confidence interval [CI]: 46–79%) versus 19% by E2 (95% CI: 8.8–36.5%; P < 0.05). Interestingly, differences between strains were noticed and, for S3 and S5, most animals were negative at 7 DPI in both tests. At 14 DPI, both tests showed sensitivity over 90%. The observed differences in early sensitivity could be attributable to the preferential detection of IgM, which appears before 14 DPI in the course of a PRRSV infection 1 using E1, the ELISA that is based on a double recognition. Because of the pentameric nature of IgM, binding of this class of antibodies to the coating and to the conjugated N protein is probably easier than for the monomeric IgG. However, both Ig classes could contribute to the results, albeit in different proportions. In contrast, ELISAs that mainly detect IgG antibodies would detect lower percentages of positive pigs at early stages of the infection. Later on, the E1 assay would still detect IgG but differences between ELISAs seen at the early infection phase should tend to disappear, and indeed, E1 had a similar performance to E2 when sera from convalescent experimentally infected pigs (84 DPI) were tested (Table 1).
Number of Porcine reproductive and respiratory syndrome virus–positive pigs and sample-to-positive values (mean ± standard deviation) in experimental infections until 84 days postinoculation using enzyme-linked immunosorbent assay 1 or 2.*
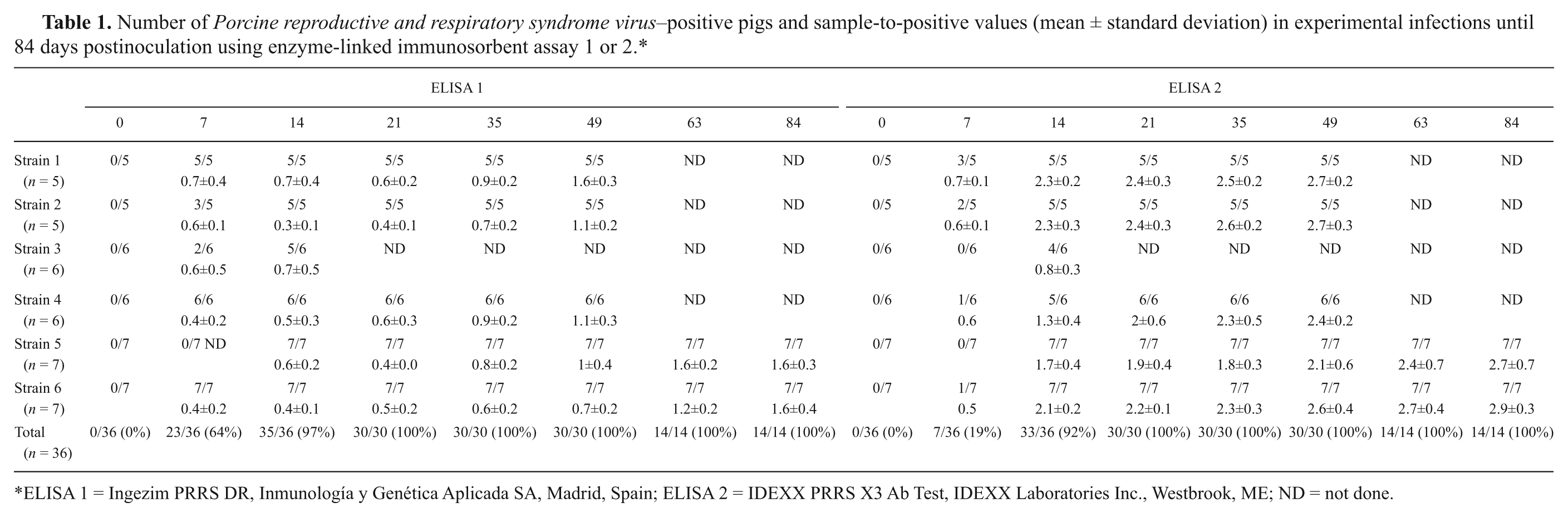
ELISA 1 = Ingezim PRRS DR, Inmunología y Genética Aplicada SA, Madrid, Spain; ELISA 2 = IDEXX PRRS X3 Ab Test, IDEXX Laboratories Inc., Westbrook, ME; ND = not done.
For sera from experimentally infected animals, agreement between the 2 ELISAs before 21 DPI was strongly associated to the strain used. Thus, for strains S1, S4, and S6, the discrepancies between the 2 tests arose from sera detected as positive in E1 but negative in E2, whereas no E2-positive sera were E1 negative. In contrast, the agreement was excellent for S5 (kappa = 1.0): for S2 and S3, kappa values were 0.5 and 0.7, respectively. Overall, kappa values varied from 0.24 (95% CI: 0.028–0.425) at 7 DPI to 0.48 (95% CI: 0.20–0.757) at 14 DPI and to 1.0 from 21 DPI onward.
When field sera were used, the performance of the 2 ELISAs was similar, with a few exceptions (Table 2). The double-recognition ELISA (E1) already detected PRRSV antibodies in most animals by week 9 of age in farm 1, whereas E2 detected infection by week 11. In farm 3, colostrum-derived antibodies were detected longer with E1; later on, both tests identified positive animals although E1 identified a higher proportion. To assess the performance of both tests under field conditions, animals from 2 endemic farms were followed from 3 weeks of age. Results are shown in Figure 1. Under those conditions, E1 detected maternally derived antibodies for a longer time than E2, probably reflecting higher analytical sensitivity. However, this caused some overlap between the detection of fading colostral antibodies and the development of antibodies after infection of piglets. This fact could complicate the interpretation of serological results when E1 is applied in longitudinal monitoring.
Percentage of positive pigs in cross-sectional profiling in Porcine reproductive and respiratory syndrome virus–endemic farms.*
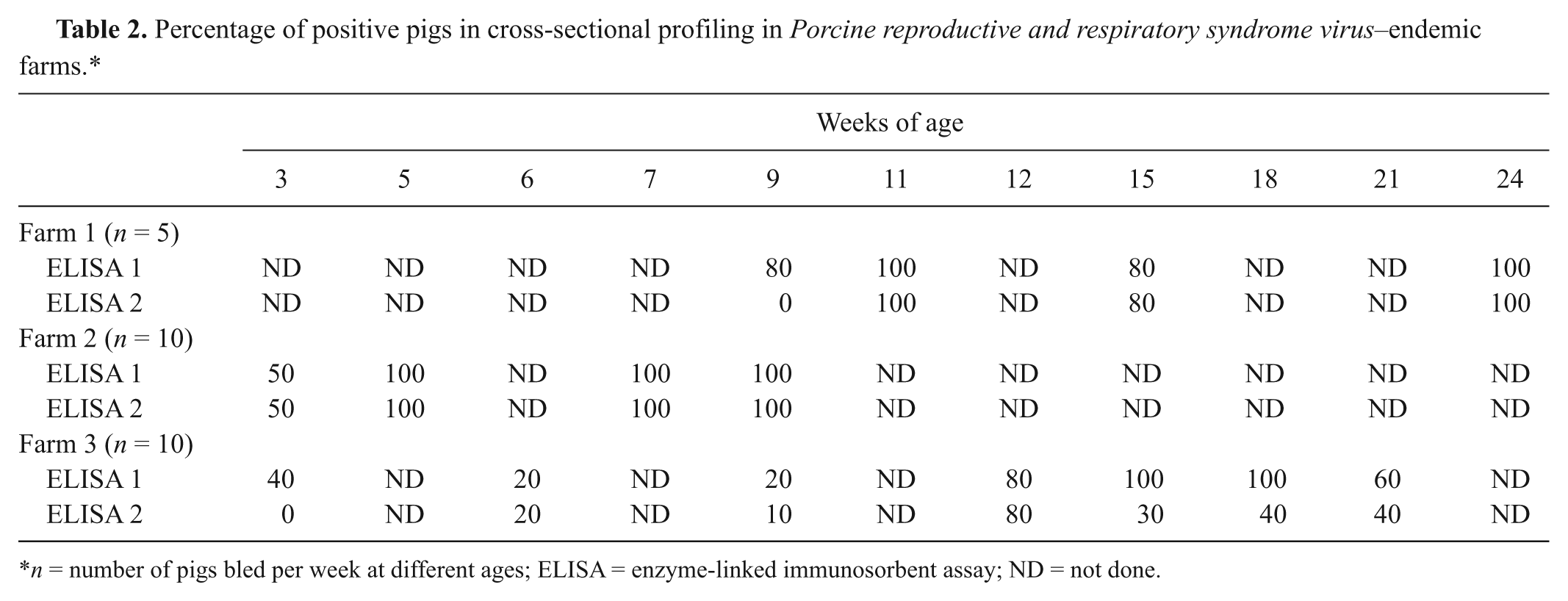
n = number of pigs bled per week at different ages; ELISA = enzyme-linked immunosorbent assay; ND = not done.
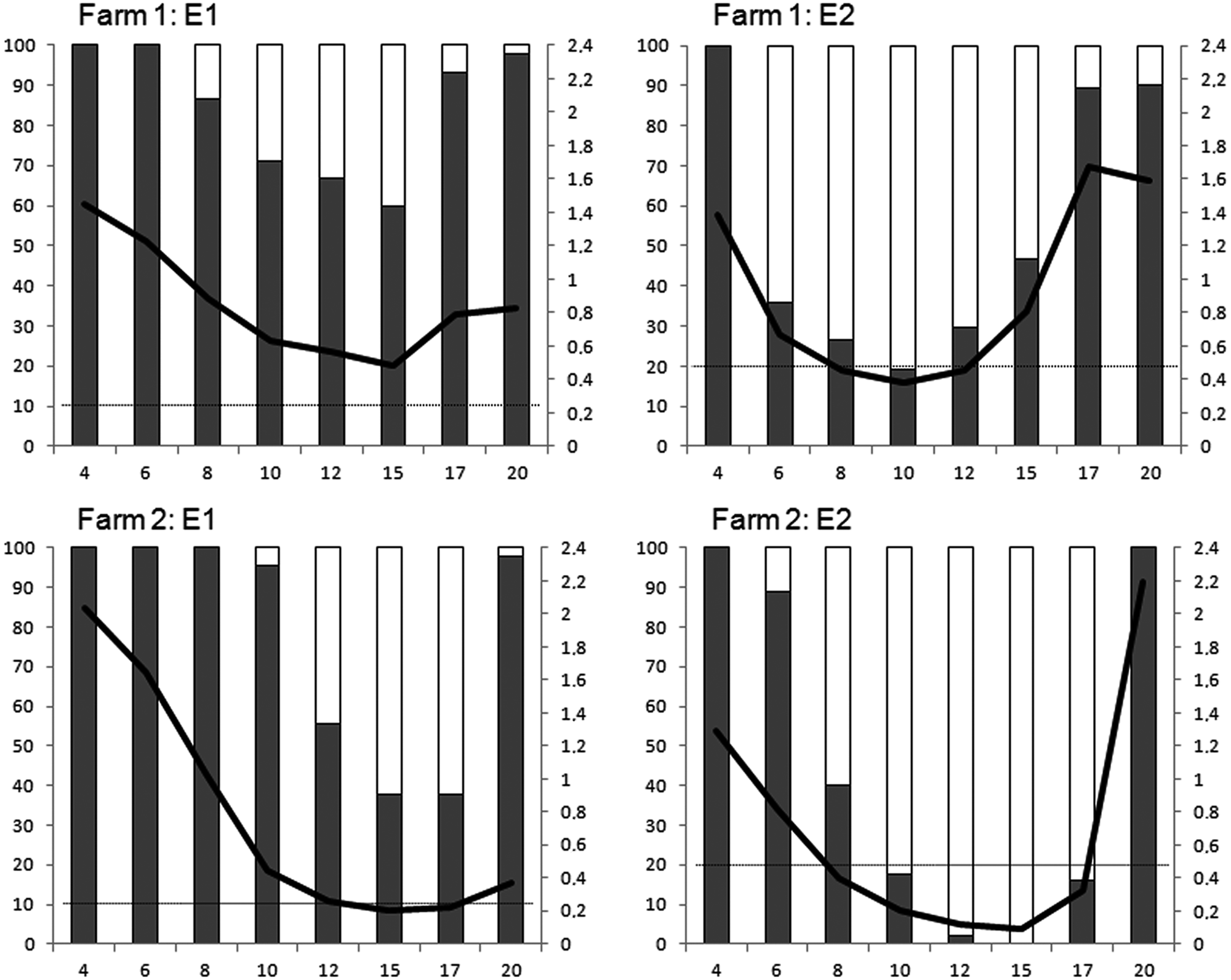
Longitudinal profiling. Percentage of positive (gray bars) and negative (white bars) pigs from 4 to 20 weeks of age. Results obtained with enzyme-linked immunosorbent assays (ELISAs) E1 and E2 in farm 1 and farm 2. The black line indicates the average sample-to-positive (S/P) ratio (secondary axis) at a given time. The dotted black line shows the positive threshold S/P value for each ELISA.
In summary, E1 had a higher sensitivity than E2, making E1 a more suitable test for the early detection of PRRSV antibodies and thus infection monitoring in negative swineherds (i.e., testing of boars or gilts entering a PRRSV-free herd). However, the higher sensitivity of E1 could also present a problem for infection monitoring after colostrum-derived antibodies have waned, as the test remained positive for a longer period in the longitudinal profiling experiment.
Footnotes
Acknowledgements
The authors would like to thank Anna Coll and Dr. Montse Tello for assisting in the obtaining of the samples in cross-sectional profiling in PRRSV endemic farms.
a.
Ingezim PRRS DR, Inmunología y Genética Aplicada SA, Madrid, Spain.
b.
IDEXX PRRS X3 Ab Test, IDEXX Laboratories Inc., Westbrook, ME.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Á. Venteo, B. Rebollo, and A. Sanz are employees of Ingenasa. All other authors did not receive any funding or honoraria from Ingenasa nor have financial or personal relationship with other people or organizations that could inappropriately influence or bias the content of the paper.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by project 245141 (PoRRSCon) of the 7th Framework Program of the European Union. Ivan Díaz was funded by project Consolider-Ingenio 2010 CDS2006-00007-Porcivir (MCINN). Gerard E. Martin-Valls was funded by a doctoral FPI grant from the Spanish Ministry of Science and Innovation, and Meritxell Simon-Grifé was funded by a pre-doctoral FI grant of the Government of Catalunya (Spain).
