Abstract
The objective of this study was to develop an indirect enzyme-linked immunosorbent assay (ELISA) using a sonicated pure culture of Lawsonia intracellularis as the antigen (So-ELISA). A total of 332 serum samples, consisting of 232 experimentally infected animals and 100 animals naturally infected with L. intracellularis, were used to assess the diagnostic sensitivity. Three hundred and fifty-five sera from uninfected animals were used to determine the diagnostic specificity. The receiver operating characteristic and mean +3 standard deviation of optical density (OD) values from uninfected animals were used for selecting cut-off points. The diagnostic accuracy of So-ELISA was considered to be high as the area under the curve index was 0.991 with 0.0029 standard error. The optimal cut-off for So-ELISA was set at 0.45 OD with 89.8% sensitivity and 99.4% specificity based on a combination of good sensitivity and high specificity. No cross-reactivity was found in sera from pigs exposed to Brachyspira pilosicoli, B. hyodysenteriae, Campylobacter mucosalis, C. jejuni, or C. coli. Inter- and intracoefficient of variation of all control sera tested with So-ELISA was less than 10%. The observed agreements between So-ELISA and the immunoperoxidase monolayer assay tested with experimental challenge animals and field samples were 95.08% with 0.88 kappa and 90.65% with 0.74 kappa value, respectively. So-ELISA was able to detect the seroconversion of infected animals at 2 to 4 weeks after exposure to L. intracellularis. Based on the validation results, So-ELISA could be used as an alternative serology for proliferative enteropathy diagnosis.
Introduction
Proliferative enteropathy (PE) caused by Lawsonia intracellularis, a Gram-negative, curved-shaped, obligate intracellular bacterium, is an important enteric disease of grower/finisher pigs. The disease occurs in various forms. One is an acute infection known as proliferative hemorrhagic enteropathy (PHE) in which the infected pigs may have bloody diarrhea and/or sudden death. Another form of infection, known as porcine intestinal adenomatosis, is a chronic form that causes slow growth, chronic diarrhea, and thickening of the ileum mucosa. 12 Diagnosis of PE can be achieved antemortem by various techniques such as fecal polymerase chain reaction, fecal immunoperoxidase, and serology tests. For monitoring the disease distribution and detecting a herd's exposure to L. intracellularis, serological tests are more suitable than other antemortem tests as they are less expensive and allow processing of a larger number of samples in a shorter time.
The most commonly used serologic tests for diagnosis of PE are the indirect fluorescent antibody test (IFAT) and the immunoperoxidase monolayer assay (IPMA). Both methods of producing the IFAT, a tissue culture method and a conventional slide test, demonstrate equal sensitivity (91.2%) and agree highly with one another (94.4% agreement). 7 A more recently developed test, IPMA, presented a similar sensitivity to IFAT (88.9%), but its specificity is estimated to be 100% using a 1:30 sera dilution as the cut-off. 5 However, reader subjectivity is the main limitation in widespread availability of either procedure.
Recent ELISA studies used various methods for antigenic preparation of pure culture whole cell L. intracellularis, such as a hot aqueous phenol method on lipopolysaccharide extraction 11 and sodium deoxycholate extraction format. 1 However, the investigations of indirect ELISA tests using purified sonicated whole cell L. intracellularis as antigen have not been undertaken. Therefore, the objective of this study was to develop an indirect ELISA based on a sonicated whole cell preparation of purified L. intracellularis as antigen (So-ELISA). Furthermore, the authors validated the accuracy and test performance of this ELISA by comparing it to the IPMA test for diagnosis of PE in experimentally and naturally infected pigs.
Materials and methods
Bacterial antigen preparation
The purified sonicated L. intracellularis antigen used in the present study was obtained from L. intracellularis strain PHE/MN1–00. The bacteria were grown as described elsewhere. 3 Extracellular L. intracellularis was harvested from the supernatant of T-175 cm 2 flasks, and centrifuged at 8,000 × g for 20 min. The resulting supernatant was discarded, and the bacterial pellet was resuspended with sterile phosphate buffered saline (PBS) and filtered through 5-μm filters to remove any remaining eukaryotic cell debris. The bacterial suspension was centrifuged 3 additional times, each time washing and resuspending the pellet with 15 ml of sterile PBS. The purified L. intracellularis was sonicated on ice through 3 cycles of 30 sec with 2 min downtime between cycles. The sonicated suspension was centrifuged at 5,000 × g for 20 min, and the supernatant was used as antigen to coat ELISA plates. a Total protein contents of the sonicated antigen were measured according to the Lowry method using a commercial protein assay kit. b
Source of reference control sera
Four positive control and 2 negative control sera were included on every ELISA plate. Each positive control serum was a pool of 6 sera from pigs previously experimentally infected with L. intracellularis. Four levels of positive control sera were established by IPMA results: high (> 1:960), middle high (1:480), middle (1:120–1:240), and low (1:30–1:60). Two negative sera were selected from either a pool of 6 nonchallenged, L. intracellularis-free pigs or a pool of 6 cesarean-derived and colostrum-deprived (CDCD) pigs. All reference sera were divided into 100-μl aliquots and stored at −20°C until use, at which time the sample was thawed and used for all development and standardization testing of the protocol.
Development of the So-ELISA
To obtain the maximum optical density (OD) with the lowest background, all influencing factors such as time of incubation, incubation temperatures, and type or level of pH of blocking buffers were optimized as described. 2 The concentrations of sonicated whole cell antigen, serum, and conjugate (secondary anti-pig horseradish peroxidase [HRP] conjugated) c were titrated to achieve the optimal concentration using checkerboard titrations. The final So-ELISA protocol is summarized as follows: the sonicated antigen was diluted in a carbonate buffer (15 mM Na2CO3, 35 mM NaHC03, pH 9.6) to a final protein concentration of 2.5 μg/ml. One hundred μl of working antigen was added to 96-well microplates, a resulting in 250 ng/well, and incubated overnight at 4°C. The following day, the coating solution was discarded, and the plates were washed twice with washing buffer (0.05% Tween-20 in PBS [PBST], pH 7.3 to 7.4) using an Immuno Wash 8-channel manifold. d The coated plates were blocked with 200 μl/well of blocking buffer (5% [wt/vol] skim milk in PBST, pH 9.6) for 2 hr at room temperature.
Test sera were diluted 1:120 in a blocking buffer (5% [wt/vol] skim milk in PBST, pH 7.4), and 100 μl of diluted sera was transferred to the corresponding wells of the coated plates. After 1-hr incubation at room temperature, the sera were discarded and the wells were washed 3 times with washing buffer. The plate contents were then decanted and the residual liquid blotted out. One hundred μl of 1:4,000 diluted goat anti-pig IgG HRP c conjugated in blocking buffer (pH 7.4) was transferred to each well and incubated for 1 hr at room temperature. After washing 4 times with washing buffer, 100 μl of 3,3′,5,5′-tetra-methylbenzidine c was applied to each well and incubated at room temperature for 15 min. The reaction was stopped with 100 μl of 1 M phosphoric acid, and all plates were read with an ELISA plate reader e at an OD of 450 nm.
Validation methods
Analytical specificity of the So-ELISA. Cross-reactivity and specificity of the So-ELISA test were assessed by testing sera from pigs that had been exposed to related enteric pathogens such as Brachyspira pilosicoli (n = 2), B. hyodysenteriae (n = 2), Campylobacter mucosalis (n = 1), C. jejuni (n = 2), and C. coli (n = 2). Additionally, 18 CDCD pigs were tested with the So-ELISA as negative controls.
Determination of cut-off value for the ELISA
To determine the diagnostic sensitivity (DSn) and the specificity (DSp) of the So-ELISA test, the sample size of positive and negative sera required to give an acceptable margin of error was calculated based on the formula: n = {(DSn or DSp)[1 - (DSn or DSp)]c2}/e2, where n = number of known exposed/unexposed pigs, DSn = the expected test sensitivity, DSp = the expected test specificity, e = the acceptable marginal of error, and c = the confidence interval for the estimates. 8
The prior estimation of DSn and DSp ranged from 90% to 93%, respectively. 7,10 With the expectation of 92% sensitivity and specificity using a 95% confidence interval (1.96 representing ±2 SD), the number of positive and negative sera required at 2%, 3%, and 4% margins of error were 707, 314, and 177 samples, respectively. Based on these calculations, the total sample size of 687 individual sera, with a 3% margin of error, was used for the cut-off determination.
The negative sera, 355 individual samples, were obtained from pigs known to be free of L. intracellularis infection in 4 experimental challenges. Pigs were 4 to 6 weeks old at the time of blood collection. Eighteen CDCD pigs were included as the source of pigs truly negative for antibody against PE infection. As a result of the high level of prevalence of PE in the field, no negative sera from field populations were included in the cut-off validation.
The positive sera, 332 samples, were obtained from 2 previous mucosal homogenate experimental challenges (n = 232 samples) and positive samples from the field population (n = 100 samples). In both experimental studies, all animals were 5 to 6 weeks old when the experiments were conducted and the blood samples were collected 3 weeks and 6 weeks after challenge, respectively. IPMA-positive sera from the field were obtained from sera submitted through the University of Minnesota Diagnostic Laboratory. These field samples were randomly selected to represent different production systems, including ages: gilts, sows, and grower/finisher pigs. The mean and standard deviation of the OD were calculated, and the receiver operating characteristic (ROC) was defined to determine the cut-off of the So-ELISA using Medcalc software version 8.1.1.0. f
Measurement of repeatability
The repeatability, an estimate of intra- and interprecision of the So-ELISA test, was measured by running 5 replicates of 10 positive sera from pigs previously experimentally infected with L. intracellularis and 8 negative control sera from CDCD pigs on 1 plate. Ten plates were operated per run and on 10 different occasions. For the intra- and interprecision of the So-ELISA test, the mean ± SD of each control replicate was calculated. The coefficient of variation (% CV) of normalized data from the replicates of each control was checked to determine whether it exceeded 10% (expressed as SD/mean × 100).
Detection of seroconversion
To determine the seroconversion of pigs after L. intracellularis infection using So-ELISA and IPMA, IPMA plate preparation and staining procedure was performed as described previously. 6 The collections of serum samples from 2 experimental challenge studies and field PHE outbreaks were used. In the first study, 48 nursery pigs at 5 weeks of age were challenged with mucosal homogenate containing L. intracellularis. The inoculum antigen was prepared as described elsewhere. 14 All experimentally challenged pigs received 60 ml of the inoculum, approximately 6.6 × 109 L. intracellularis organisms, via stomach tube. All pigs were bled 13 days before experimental challenge, and then 14 days and 21 days after challenge.
In the second study, 15 nursery pigs at 6 weeks of age were inoculated with a similar mucosal homogenate protocol. 14 All infected pigs were bled every 2 weeks following challenge until 20 weeks of age.
In the third study, the sera from 18 piglets were collected as a longitudinal follow-up from a sow herd natural PHE outbreak. Fourteen piglets from 5 seropositive gilts and 4 piglets from 2 seronegative sows were ear-tagged and bled. Piglets were bled once or twice during the lactation period and bled every 3 weeks until 22 weeks of age.
Measurement of agreement between IPMA and ELISA tests
Agreement between IPMA and So-ELISA was determined using sera from both experimentally challenged animals and field samples. A total of 264 serum samples were obtained from 2 previous experimental challenge studies. Pigs were 5 to 6 weeks old when the animals were challenged with a mucosal homogenate. All pigs were bled 13 days prior to receiving the inoculum and bled again at 14 and 21 days postinoculation. A total of 1,583 sera obtained from the Veterinary Diagnostic Laboratory at the University of Minnesota between March and September 2005 were used. All sera were tested in parallel using IPMA and So-ELISA, and agreement between the 2 tests was analyzed using kappa statistic.
Results
Cut-off determination, diagnostic sensitivity, and diagnostic specificity
The cut-off value of the So-ELISA test was calculated as either the mean of negative control +2 SD and +3 SD or the results of the ROC curve distribution, which was based on a combination of good sensitivity with high specificity. The OD values of the negative control population ranged from 0.08 to 0.46. The mean and standard deviation of the negative population were 0.19 and 0.073, respectively. Consequently, mean +2 SD and +3 SD of the negative control were 0.33 and 0.41, respectively. A single ROC curve analysis was used to validate the overall accuracy of the assay, as indicated by the area under the curve (AUC) of the plot. So-ELISA results showed that the AUC was 0.991 with 0.0029 standard error (95% confidence interval [CI] of area: 0.986–0.997, P = 0.0001). This result indicates excellent separation of positive control from the negative control population with So-ELISA. The distributions of all positive and negative control sera tested with So-ELISA are shown in Figure 1. The cut-off for the So-ELISA test was determined to be 0.45 at 450 OD, based on a high sensitivity with greatest specificity. The DSn was 89.8% with a 95% CI of 86.0–92.8 and the DSp was 99.4% with a 95% CI of 98.0–99.9. Therefore, if the OD value of tested pigs is greater than 0.45, the sample is considered positive for L. intracellularis antibodies. In contrast, if the sample OD is lower than 0.45, the sample is considered negative.
Analytical specificity
No positive reactions were found in the So-ELISA test with the sera from pigs exposed to B. pilosicoli, B. hyodysenteriae, C. mucosalis, C. jejuni, or C. coli. In addition, all 18 CDCD pigs were also negative when tested with So-ELISA.
Repeatability validation
The intra- and interassay precisions were expressed as a percentage of the coefficient of variation (CV). The mean, standard deviation, and range of % CV of each control sera are summarized in Figure 2. A total of 100 plates with 5 replicates per plate of each sample were evaluated. The % CVs of intraplate, interplate within a run, and between-runs in all control samples were less than a prior precision criteria of 10% CV. This indicates that the So-ELISA system is of adequate repeatable precision.
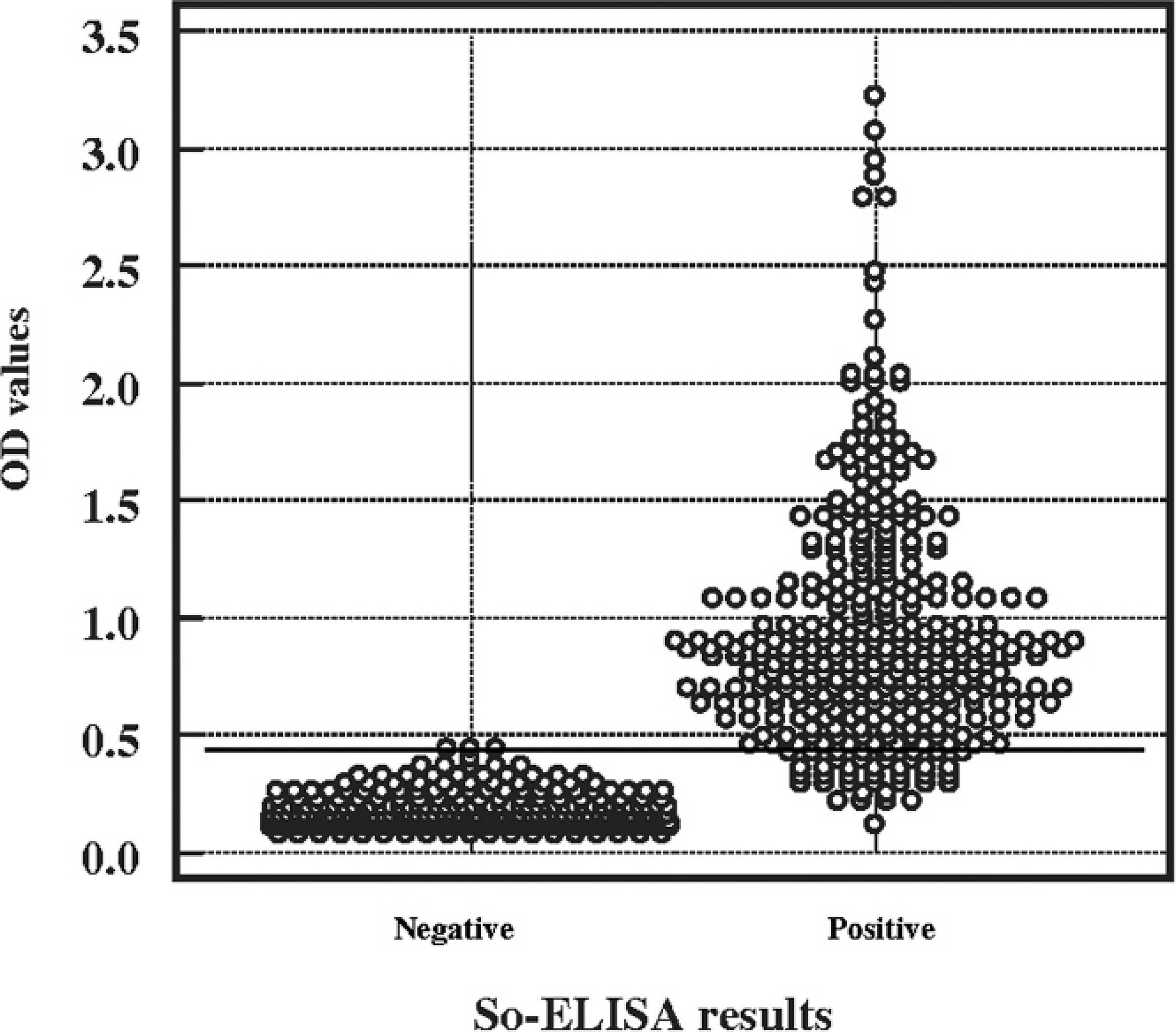
Distribution of the optical density values from 355 individual proliferative enteropathy (PE)-negative sera (known to be negative using disease challenge status of pigs) and 332 individual PE-positive sera after exposure to Lawsonia intracellularis obtained using the So-ELISA.
Detection of seroconversion
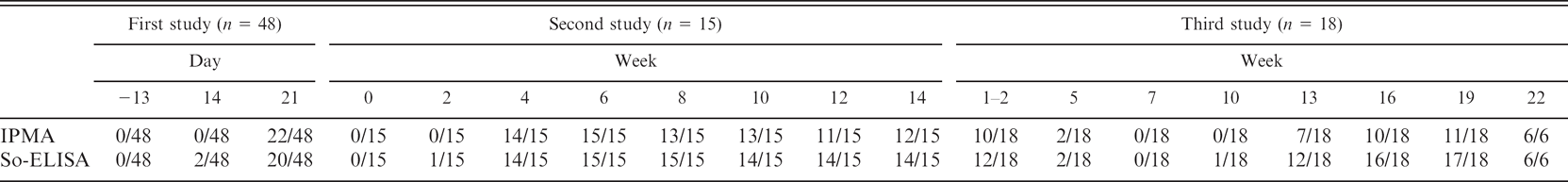
The sera samples from challenged pigs and from a natural PHE outbreak were used to detect seroconversion using both So-ELISA and IPMA. The summary of results is shown in Table 1. In the first study, 48 nursery pigs were inoculated with PE-infected intestinal mucosa. At 14 days postinoculation, 2 of 48 pigs had seroconverted as measured by So-ELISA, while IPMA determined all pigs to be negative. Three weeks' postchallenge, almost half of the challenged pigs, 22 and 20 pigs, were identified to have seroconverted by using IPMA and So-ELISA, respectively.
In the second study, the results were similar to the first study in that So-ELISA identified seroconversion of 1 of the 15 inoculated animals at 2 weeks' postchallenge while all were negative by IPMA. However, all challenged pigs had seroconverted by 6 weeks' postinoculation. These results indicated that both IPMA and So-ELISA detected the specific antibody against L. intracellularis infection 2 to 4 weeks after pigs were exposed.
In the third study, 14 piglets that originated from 5 seropositive gilts and 4 piglets from 2 seronegative sows were bled once or twice during the suckling period and then every 3 weeks until 22 weeks of age. These sets of sera were collected 12 weeks after an outbreak of PE in the sow herd. 4 So-ELISA detected 12 of 18 positive suckling piglets from seropositive gilts, whereas IPMA detected 10 positive. The number of seropositive piglets dramatically decreased to 2 and 0 positive pigs at 5 and 7 weeks of age, respectively, by both tests. So-ELISA detected the seroconversion again starting at 10 weeks of age (1 pig) and dramatically increased with peak seroconversion between 16 and 19 weeks of age (16 and 17 pigs, respectively) while IPMA detected 7 positive pigs at 13 weeks of age, with a positive peak at 16 and 19 weeks of age (10 and 11 pigs), respectively. The number of seropositive pigs increased again at 13 weeks through 22 weeks of age. This indicated that a real infection occurred in the herd at the age which PE is often observed in endemic herds. Using So-ELISA to determine amti-Lawsonia antibodies, higher and earlier seroconversion of pigs were identified than with the use of IPMA.
Comparative agreement of So-ELISA with IPMA
The percentages of observed agreement between IPMA, So-ELISA, and kappa value are shown in Table 2. IPMA found 80 samples positive and 184 samples negative, whereas So-ELISA found 77 samples positive and 187 samples negative. Agreement between IPMA and So-ELISA was 95.08% with 0.88 kappa value. The kappa statistic indicates that there is very high agreement between IPMA and So-ELISA when validated using sera from experimentally challenged pigs. With sera samples from field conditions, IPMA resulted in 323 samples positive and 1,260 samples negative, whereas So-ELISA found 397 samples positive and 1,186 samples negative, giving 90.65% agreement between the 2 tests with 0.74 kappa value. This kappa statistic indicates substantial agreement between the 2 tests when determined by use of field sera.
Discussion
Proliferative enteropathy has emerged as one of the most common enteric diseases in grower/finisher pigs. As a result of high disease prevalence in the field, better performing and more efficient diagnostic tools are required for continued monitoring and epidemiologic surveillance of the disease. Serological approaches, such as IPMA and IF AT, have been effectively used as complementary antemortem detection tools for PE diagnosis. 4,9,13 Unfortunately, the limitations of all currently available serological tests for PE diagnosis are that the tests are unable to identify the current disease status of the animals, or to differentiate the immune response antibodies resulting from natural infection or vaccination. For instance, if the results of serology tests are positive, it implies that the animals have been previously exposed to L. intracellularis or are demonstrating passive maternal immunity. If animals from a herd with no vaccination history are seropositive, it can be implied that the immune response occurred due to L. intracellularis infection. Therefore, a good record of vaccination status is important for accurate serological interpretation.
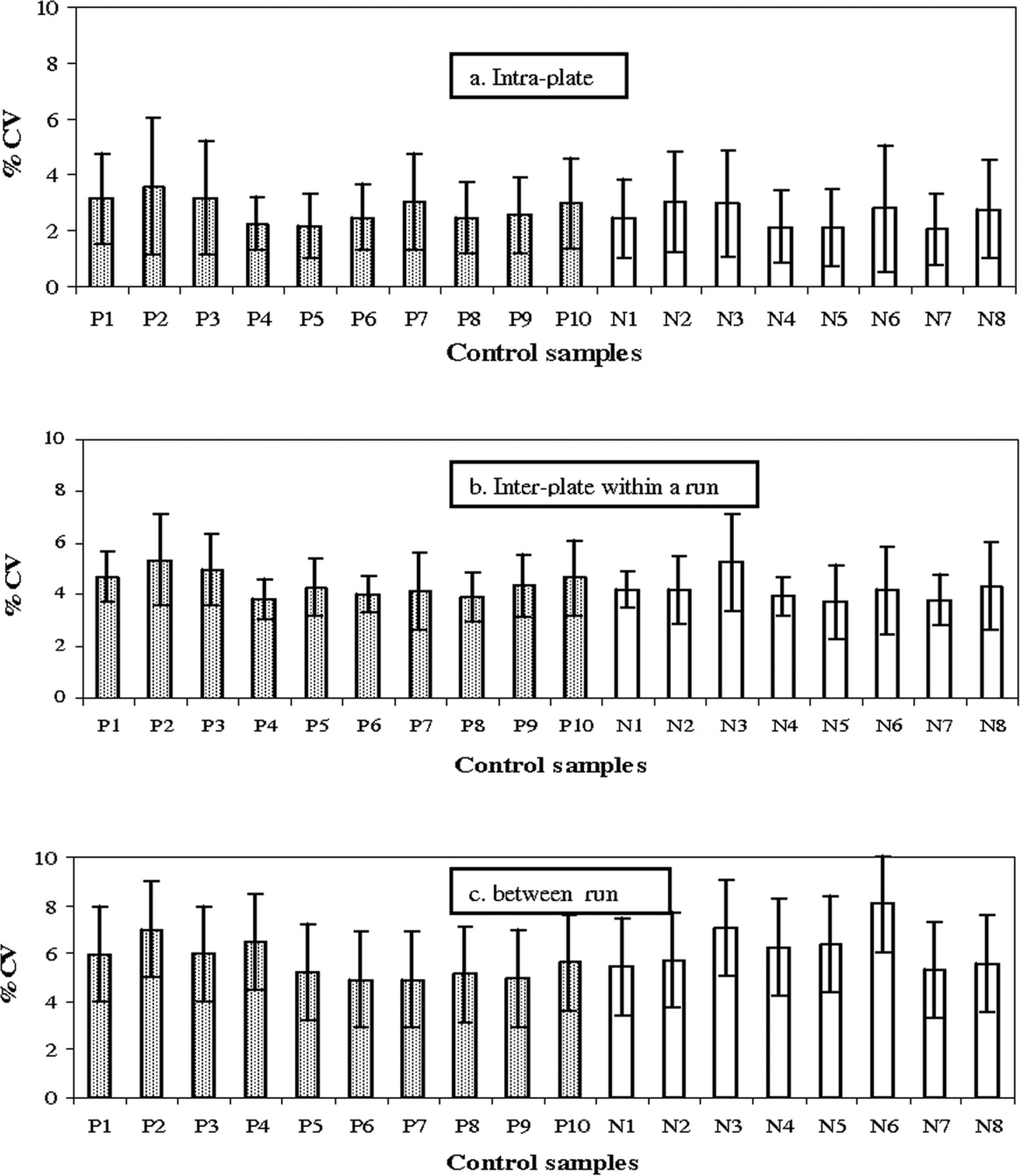
Precision (repeatability) assessment of the So-ELISA. These values are the average of coefficient of variation and its standard deviation of a variety of positive controls (P1–P10) and negative controls (N1–N8).
This study was conducted with the aim of developing and assessing the test performance of an indirect ELISA using sonicated whole cell L. intracellularis as an antigen for the diagnosis of PE. Previously, IPMA showed a 100% specificity. 5
Therefore, the present study used the IPMA test as an antemortem reference test for comparison to the newly developed ELISA test.
Seroconversion detection using immunoperoxidase monolayer assay (IPMA) and sonicated whole cell preparation of purified Lawsonia intracellularis as antigen (So-ELISA) were compared using samples from 2 previous experimental challenge studies (first and second study) and seroconverted field samples (third study) 3 months after an outbreak of proliferative enteropathy.
The method for optimization of the assay conditions such as antigen, sera, anti-pig conjugate, and other reagents was done by checkerboard titration. 2 Satisfactory So-ELISA conditions were based on the criteria that the assay showed a maximum OD signal difference between positive and negative controls with minimal OD background interference. Results showed that samples diluted 1:120 gave a maximum OD signal difference with low background.
At the serum sample dilution of 1:120 in blocking buffer, the high specificity was obtained with So-ELISA when using negative serum from CDCD pigs. Pigs experimentally exposed to other enteric swine pathogens were also determined to be negative using the same ELISA. In addition, 2 of 355 (0.56%) negative control samples had an OD at the borderline (0.46) above the cut-off. These results indicate that this So-ELISA was highly specific.
The ROC analysis was calculated using 355 individual negative control sera and 322 individual positive control sera, based on experimental challenge, with a 3% margin of error. The OD value at 0.45 was selected as a cut-off for So-ELISA. The cutoff OD was selected based on the criteria of good sensitivity with high specificity. These gave the maximum DSn at 89.8% and DSp at 99.4%. With the OD values at 0.45 as a cut-off, 2 of the negative control sera (0.56%) had an OD value higher than 0.45 (0.46 both samples). In contrast, 34 of the positive control sera (10.55%) had ODs less than 0.45. The present study selected the positive control population based on known disease status of 2 experimental studies, 3 weeks and 6 weeks after exposure to the L. intracellularis inoculum, or field samples positive by IPMA. As seen in previous experimental challenges, about 80–90% of experimental pigs seroconvert by 3 weeks' postchallenge. Nearly 100% of pigs will seroconvert by 4 to 5 weeks' postchallenge. 10 This might be one explanation as to why some positive control pigs are classified as negative as a result of OD values less than the cutoff. The selections of positive control sera for cut-off determinations are recommended to include animals at different stages of infection, such as early, middle, and late infection, which could not be determined with the field samples. 8 Sensitivity calculations are not useful if only sera from the middle stage of infection are used because they may all be positive, leading to a 100% sensitivity.
Ten positive and 8 negative internal control sera were used in the assessment of the repeatability (precision) of the assay. Direct comparisons of the samples on the same and different plates were performed. The % CV values of all control samples for within plate, between plates within run, and between-run obtained in this study were less than 10%, which is within the standard repeatability criterion. 8 These low % CVs indicate that the sonicated whole cell ELISA is a precise and repeatable assay.
Agreement between the sonicated whole cell preparation of purified Lawsonia intracellularis as antigen (So-ELISA) and immunoperoxidase monolayer assay (IPMA) results using 264 individual experimentally challenged pig sera and 1,583 individual field sera.

95.08% observed agreement with 0.88 kappa value.
90.65% observed agreement with 0.7348 kappa value.
For testing agreement between IPMA and So-ELISA, the overall agreement, based on 264 serum samples from previous PE experimentally challenged pigs, showed an excellent agreement between tests (95.08% agreement with 0.88 kappa statistic). These results indicate that results obtained from samples tested with So-ELISA are very likely similar to IPMA results.
When a large number of field samples (1,583 samples) were used for determining agreement, the level of agreement obtained from the calculation was decreased to 90.65% agreement with 0.734 kappa statistic, indicating a substantial but reduced agreement between tests. So-ELISA has more positive and less negative results than IPMA. One hundred and eleven additional field samples were positive with the So-ELISA but these sera were negative by IPMA. The possible explanations for these phenomena are that either So-ELISA is more sensitive, thus finding more true positives, or the test is less specific than IPMA, thus resulting in a greater number of false positives. However, both tests showed similar diagnostic sensitivity and specificity, 88.9–89.8% and 99.4–100%, respectively.
So-ELISA appears to detect earlier seroconversion in infected animals than IPMA. However, both tests are equally able to detect the seroconverted animal between 2 to 4 weeks after exposure to L. intracellularis. In addition, both tests are equally able to identify the passive immunity passed from sow to piglets.
There are several advantages to using So-ELISA compared with the current IPMA: 1) So-ELISA carries less risk of other bacterial contamination, as the plates need not be cultured with live bacteria for assay preparation; 2) IPMA requires highly skilled readers to interpret the outcome, while ELISA gives more objective results due to automation of the reading process.
Presently, the only serologic test used in the United States for L. intracellularis is IPMA. This present study shows that So-ELISA can be used as an alternative to IPMA for the screening of sera for the presence of L. intracellularis antibodies. The results based on the standard validation protocol, the specificity, repeatability, detection of seroconversion, and test agreements validation indicate that So-ELISA is a reliable and precise test for the detection of L. intracellularis antibodies in pigs.
Acknowledgements
The authors would like to thank Professor Han Soo Joo for providing CDCD pig sera, Professor Sagar Goyal for laboratory guidance, and Molly Freese, Beth Thompson. Keith Kinsley, and Benjawan Wijarn for technical support.
Footnotes
a.
Immulon 2HB, Thermo Labsystems, Franklin, MA.
b.
Bio-Rad Laboratories, Hercules, CA.
c.
Kirkegaard & Perry Laboratories, Gaithersburg, MD.
d.
Nalgene Nuc International, Rochester, NY.
e.
BIO-TEK-ELx800, BIO-TEK Instruments, Inc., Winooski, VT.
f.
MedCalc Software, Mariakerke, Belgium.
