Abstract
Actinobacillus pleuropneumoniae is the etiologic agent of porcine pleuropneumonia, a highly contagious pulmonary disease in pigs with major economic losses for pig producers worldwide. Whereas A. pleuropneumoniae isolates are divided into 15 serotypes, the isolates secrete 4 types of exotoxins (ApxI, ApxII, ApxIII, and ApxIV), which are known as major virulence factors. In the current study, the ApxIA, ApxIIA, and ApxIIIA genes were amplified and their recombinant proteins expressed in Escherichia coli M15 cells. The antigenicity of each recombinant protein was demonstrated by Western blot and enzyme-linked immunosorbent assay (ELISA) using sera from pigs vaccinated with a subunit vaccine. When ELISAs using the recombinant antigens were optimized and then applied to sera from 320 randomized pigs in Korea, an observed increase in seroprevalence was found among sows in comparison with weaned piglets and growing pigs, indicating an age-dependent seroprevalence. The results obtained in the study suggest that the developed ELISAs may be useful for A. pleuropneumoniae vaccination strategy as a screening tool for pig herds as well as for detection of specific antibodies to Apx exotoxins.
Introduction
Actinobacillus pleuropneumoniae causes a highly contagious pleuropneumonia in pigs with major economic losses in pig farming worldwide. 6,9,10,14,22 Fifteen serotypes of A. pleuropneumoniae have been described based on the differences of capsular polysaccharides (CP) and lipopolysaccharide (LPS) composition. 2,3,8 There are several virulence factors such as exotoxins, LPS, CP, membrane proteins, and adhesion factors in A. pleuropneumoniae. 1,10,18,24 Among these virulence factors, the exotoxin, called Apx toxins, is strongly correlated with virulence. 4,5,10,20 Actinobacillus pleuropneumoniae produces 4 different Apx toxins belonging to the pore-forming RTX (Repeats in the structural ToXin) toxins, which include the strongly hemolytic and cytotoxic ApxI, the weakly hemolytic and moderately cytotoxic ApxII, the nonhemolytic and strongly cytotoxic ApxIII, and the moderately hemolytic recombinant ApxIV. 10,20,21 The ApxI, ApxII, and ApxIII toxins, which are secreted by different serotypes in various combinations, mainly determine the virulence of the 15 serotypes of A. pleuropneumoniae. 3,5,11 The ApxIV toxin is produced by all serotypes of A. pleuropneumoniae. 21 The Apx toxin is encoded by the apx operon, which contains 4 genes: the activator gene apxC, the pretoxin structural gene apxA, and the secretion-apparatus–encoding genes apxB and apxD, in the order apxCABD. 10
Actinobacillus pleuropneumoniae can result in various clinical syndromes, which vary in degrees of severity from peracute to chronic, with infected pigs typically showing a hemorrhagic pneumonia often associated with necrosis and fibrinous pleuritis. 14,22 Carrier animals can be the origin of propagation of A. pleuropneumoniae. Diagnosis of porcine pleuropneumonia depends on the isolation, identification, and serotyping of A. pleuropneumoniae, and serodiagnosis. There are several methods used in the serological diagnosis of A. pleuropneumoniae, which include radioimmunoassay, tube or microplate agglutination tests, complement fixation test, hemolysin neutralization, and the enzyme-linked immunosorbent assay (ELISA). 8,17 Although ELISA requires selecting and purifying an antigen, it is more sensitive than other assays. 15,17 Commercial ELISA kits were developed for selected serotypes such as serotypes 1-9-11, 2, 2-6, 3-6-8, and 4-7 in A. pleuropneumoniae. Each ELISA tests for a specific serotype only, and it does not offer options to test for the other serotypes. The Apx toxins are major immunogenic factors and stimulate the production of antibodies to such toxins in infected animals. 12,13 Moreover, because of its wide distribution, vaccination against A. pleuropneumoniae is commonly practiced in Korea. Because it is recognized as a major virulence factor, an ELISA test that uses a native antigen from the Apx toxin may be a practical approach for the detection of antibodies and control of porcine pleuropneumonia. 8,19 The current report describes the development of an ELISA based on the recombinant ApxIA, ApxIIA, and ApxIIIA antigens and evaluated the application of the developed assays on the prevalence of anti-Apx toxin antibodies among pigs in Korea.
Materials and methods
Bacterial strains and growth conditions
A Korean isolate A. pleuropneumoniae serotype 2 was used for the apxIIA gene as previously described. 23 Actinobacillus pleuropneumoniae serotypes 5 and 2 isolated from infected Korean pigs with pleuropneumonia were used for apxIA and apxIIIA genes, respectively. The organism was grown in lysogeny broth (LB) a with 0.01% β-nicotinamide adenine dinucleotide b for 24 hr at 37°C. Escherichia coli M15 cells were grown in LB with ampicillin c (100 µg/ml) and kanamycin c (25 µg/ml) for 16 hr at 37°C.
Polymerase chain reaction amplification and cloning of apxIA, apxIIA, and apxIIIA genes
Total genomic DNA was provided for gene amplification from A. pleuropneumoniae serotypes 2 and 5 using a commercial bacterial genomic DNA kit. d The sequences of apxIA, apxIIA, and apxIIIA and primers were designed on the basis of GenBank (Table 1; C-terminal part of apxIA: accession no. AF363361; full sequences of apxIIA: accession no. AF363362; N-terminal part of apxIIIA: accession no. AF363363). The ApxIIA gene (2,883 base pairs [bp]) was amplified and cloned as previously described. 23 Amplified apxIA and apxIIIA genes were purified with a gel extraction kit. e The polymerase chain reaction (PCR) products were electrophoresed in a 1.0% agarose gel and observed under ultraviolet light after ethidium bromide staining. The ApxIA (801 bp) and apxIIIA (615 bp) genes were cloned into the pQE30UA and the pQE31 vectors, e respectively. To confirm gene insertion, DNA sequencing was carried out with an automated DNA sequencer. f The cloned apxIA and apxIIIA were then transformed into competent E. coli M15 cells for expression.
Primer sequences used for cloning and expression of the Actinobacillus pleuropneumoniae ApxIA, ApxIIA, and ApxIIIA toxins.*

PCR = polymerase chain reaction. GenBank accession number in parentheses.
Expression and purification of the recombinant proteins
When the culture reached an optical density of 0.6 nm (OD0.6nm), isopropyl β-D-1-thiogalactopyranoside c (IPTG, 1 mM) was added and then cultured continuously for 4 hr. The harvested cells were re-suspended in lysis buffer (20 mM Tris–hydrogen chloride, 500 mM sodium chloride, 8 M urea, 40 mM imidazole, pH 7.0). Nickel–nitrilotriacetic acid (Ni-NTA) chelate affinity chromatography g was then performed according to the manufacturer’s instruments. The bound protein was eluted with elution buffer (20 mM Tris–hydrogen chloride, 500 mM sodium chloride, 8 M urea, 500 mM imidazole, pH 7.0). The predicted size of the purified recombinant proteins matched the results on the sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and Western blot, based on amino acids constitution. The purified ApxIA, ApxIIA, and ApxIIIA fusion proteins were analyzed by 12% SDS–PAGE with Coomassie blue staining and transferred to a nitrocellulose membrane; the membrane was immunoblotted with mouse anti-histidine antibodies h at a 1:2,000 dilution and an alkaline phosphatase–conjugated goat anti-mouse immunoglobulin G (IgG, H+L) d at a 1:1,000 dilution. Blots were developed with an alkaline phosphatase kit. i The purified recombinant ApxIA, ApxIIA, and ApxIIIA were immunoblotted with the serum of a pig vaccinated with a subunit vaccine containing ApxI, ApxII, and ApxIII toxoids and outer membrane proteins at a 1:2,500 dilution and an alkaline phosphatase–conjugated rabbit anti-pig IgG (H+L) i at a 1:2,000 dilution for the purpose of evaluating diagnostic antigens. Blots were developed with an alkaline phosphatase kit. i
Development of ApxIA, ApxIIA, and ApxIIIA enzyme-linked immunosorbent assays
The assays were performed using 96-well microplates coated with respective antigen at 4°C overnight. Plates were washed with solution of phosphate buffered saline (PBS) containing 0.01% Triton X-100 (Tween-20 in PBS [PBST]) after antigen coating and blocked with 10% normal horse serum by incubation at 37°C for 2 hr. Test reagents (antigen and conjugate) were checkerboard titrated utilizing representative positive and negative sera. Sensitization of recombinant ApxIA, ApxIIA, and ApxIIIA antigens was performed by a serial of two-fold dilution of each antigen ranging from 2.4 ng/well to 5,000 ng/well. A serum from a vaccinated pig and a serum from a nonvaccinated pig were used as positive and negative control sera for discrimination, respectively. The plates were incubated at 37°C for 2 hr with 100 µl of two-fold serial dilution of each control serum, followed by incubation with horseradish peroxidase–conjugated goat anti-pig IgG j (100 µl; 1:1,000 or 1:2,000 diluted in PBST) at 37°C for 1 hr, and washed between incubations with PBST. Color formation was done with 2,2’-Azino-bis(3-ethylbenzthiazoline)6-sulfonic acid d as substrate. Optical density was measured at 405 nm using a microplate reader. k
To determine optimal cut-off values, 40 sera from pigs vaccinated with Porcilis APP l and 25 sera from 10 colostrum-deprived piglets and 15 nonvaccinated piglets were used. The sensitivity, specificity, efficiency, and Youden index were obtained to evaluate diagnostic accuracy of the ApxIA, ApxIIA, and ApxIIIA ELISAs. The following formulas were calculated:

Youden index is the measure of the probability of correct classifications that is invariant to prevalence. 7 Values for the area under the receiving operator characteristic (ROC) curve of the ApxIA, ApxIIA, and ApxIIIA ELISAs were evaluated using a statistical program m at the 95% confidence interval.
Anti-ApxI, -ApxII, and -ApxIII prevalence in domestic pigs
A total of 320 field pig sera was chosen randomly according to age, which was divided into suckling piglet (0–4 weeks), weaned piglet (4–10 weeks), growing pig (10–20 weeks), and sow classes. Prevalence of antibodies against ApxI, ApxII, and ApxIII was investigated with the ELISA in domestic pigs in Korea. Each pig sera were measured 3 times. The absorbance of samples was measured at 405 nm and analyzed.
Results
Production of the recombinant ApxIA, ApxIIA, and ApxIIIA
The nucleotide sequences of the cloned genes have confirmed the identity of each toxin (data not shown). The transformed bacteria were cultured and harvested after IPTG induction. The sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis showed that the molecular masses of recombinant ApxIA, ApxIIA, and ApxIIIA fusion proteins were approximately 29 kDa, 106 kDa, and 23 kDa, respectively, as shown in Figure 1, in good agreement with the software-predicted molecular weights. Western blot analysis also revealed the presence of a clear protein band with an estimated molecular mass of each purified recombinant protein, as presented in Figure 2.
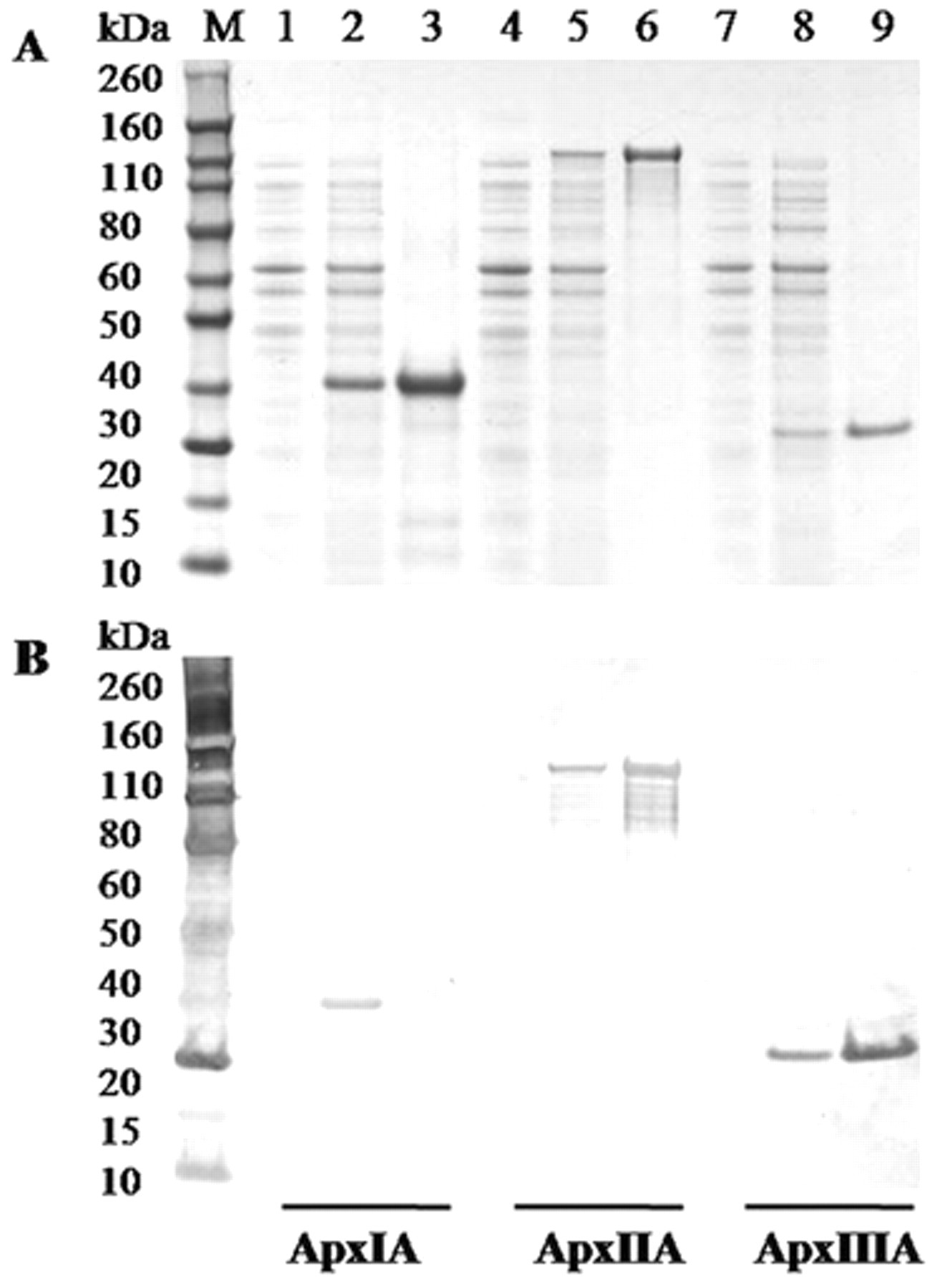
Purification of the recombinant Actinobacillus pleuropneumoniae toxins ApxIA, ApxIIA, and ApxIIIA. The recombinant ApxIA, ApxIIA, and ApxIIIA were analyzed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (
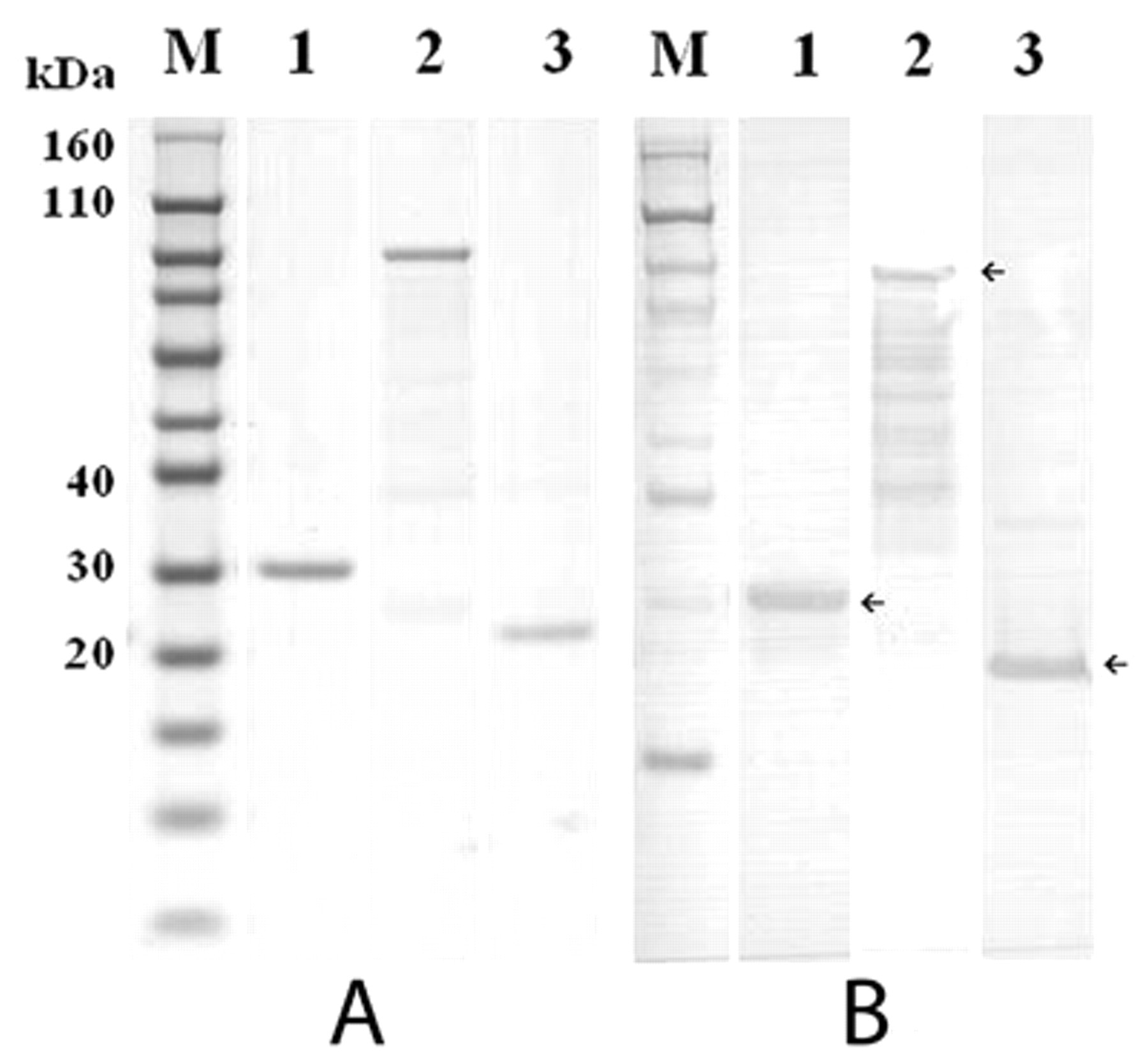
Immunoblots of the purified recombinant Actinobacillus pleuropneumoniae toxins ApxIA, ApxIIA, and ApxIIIA for its antigenicity. The proteins were characterized by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (
Development of ApxIA, ApxIIA, and ApxIIIA enzyme-linked immunosorbent assays
The results indicated that the optimal concentration of recombinant ApxIA antigen used to coat was 625 ng per well using a 1:100 dilution of serum, and also revealed that the concentrations of recombinant ApxIIA and ApxIIIA antigens were 9.8 and 156.3 ng per well, respectively, using a 1:200 dilution of serum. The largest differences between the absorbance of positive and negative control sera were obtained by the addition of a 1:1,000 dilution of the conjugate for wells coated with the ApxIA antigen under optimized conditions. In cases of using wells coated with the ApxIIA antigen or the ApxIIIA antigen, a 1:2,000 dilution of the conjugate made a significantly clear distinction between positive and negative control sera.
Cut-off values
The mean OD and standard deviation (SD) of 25 serum samples (from 10 colostrum-deprived piglets and 15 nonvaccinated piglets) were 0.191 and 0.108 for ApxIA ELISA, 0.107 and 0.058 for ApxIIA ELISA, and 0.15 and 0.074 for ApxIIIA ELISA. A cut-off value was defined as the average value plus SD of the 25 sera (i.e., 0.299 for ApxIA ELISA, 0.165 for ApxIIA ELISA, and 0.225 for ApxIIIA ELISA). Such cut-off values were determined to be optimal by ROC analysis of sensitivity and specificity at different cut-off levels, as shown in Figure 3, using the 25 control sera noted above and 40 sera from pigs vaccinated with the subunit vaccine. The specificity, specificity, efficiency, and Youden index of each ELISA at the corresponding cut-off value are summarized in Table 2.
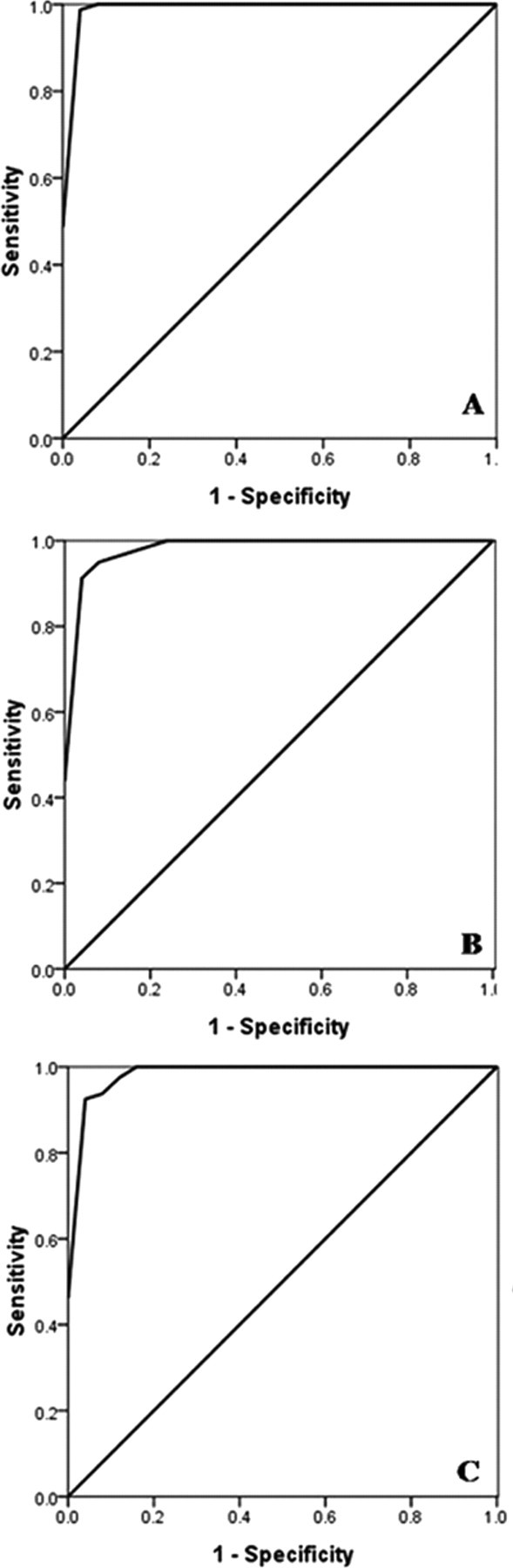
Receiver operating characteristic (ROC) curves of ApxIA (
Results of Actinobacillus pleuropneumoniae toxin ApxIA, ApxIIA, and ApxIIIA enzyme-linked immunosorbent assays (ELISAs) in control pig sera at each cut-off values.*
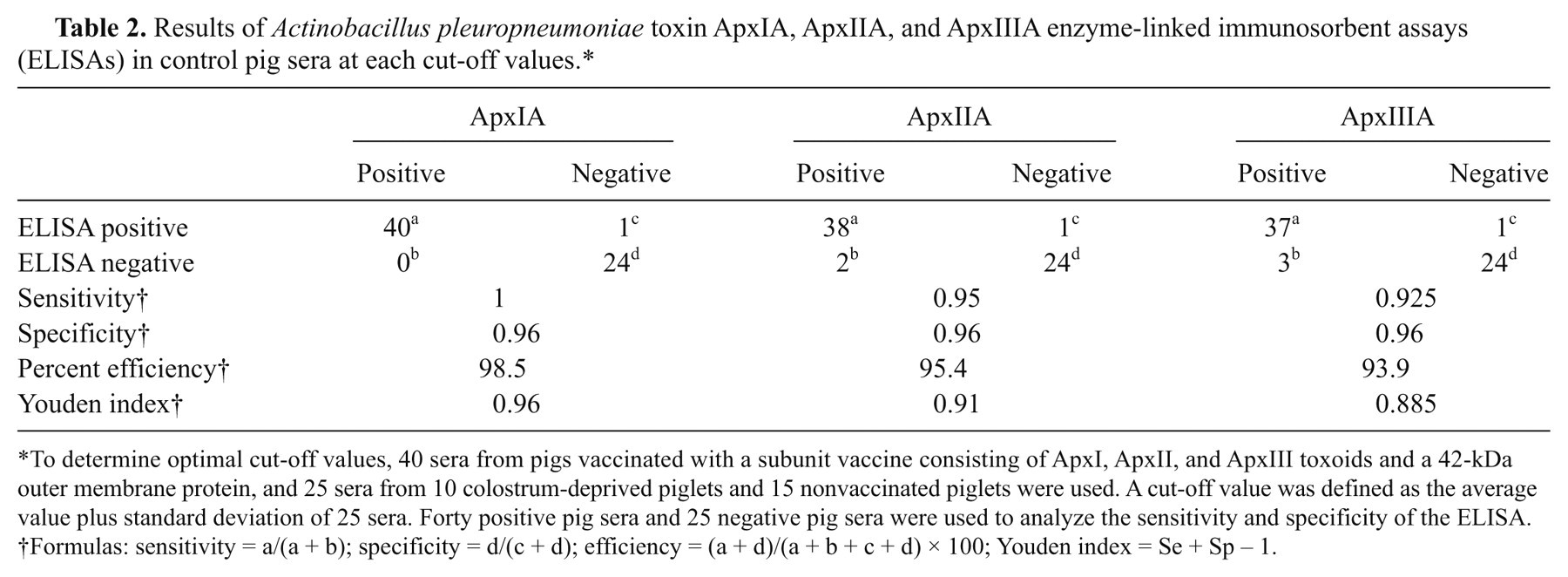
To determine optimal cut-off values, 40 sera from pigs vaccinated with a subunit vaccine consisting of ApxI, ApxII, and ApxIII toxoids and a 42-kDa outer membrane protein, and 25 sera from 10 colostrum-deprived piglets and 15 nonvaccinated piglets were used. A cut-off value was defined as the average value plus standard deviation of 25 sera. Forty positive pig sera and 25 negative pig sera were used to analyze the sensitivity and specificity of the ELISA.
Formulas: sensitivity = a/(a + b); specificity = d/(c + d); efficiency = (a + d)/(a + b + c + d) × 100; Youden index = Se + Sp – 1.
Seroprevalence rate of the Actinobacillus pleuropneumoniae toxins ApxIA, ApxIIA, and ApxIIIA using the developed enzyme-linked immunosorbent assay in Korean domestic pigs.*
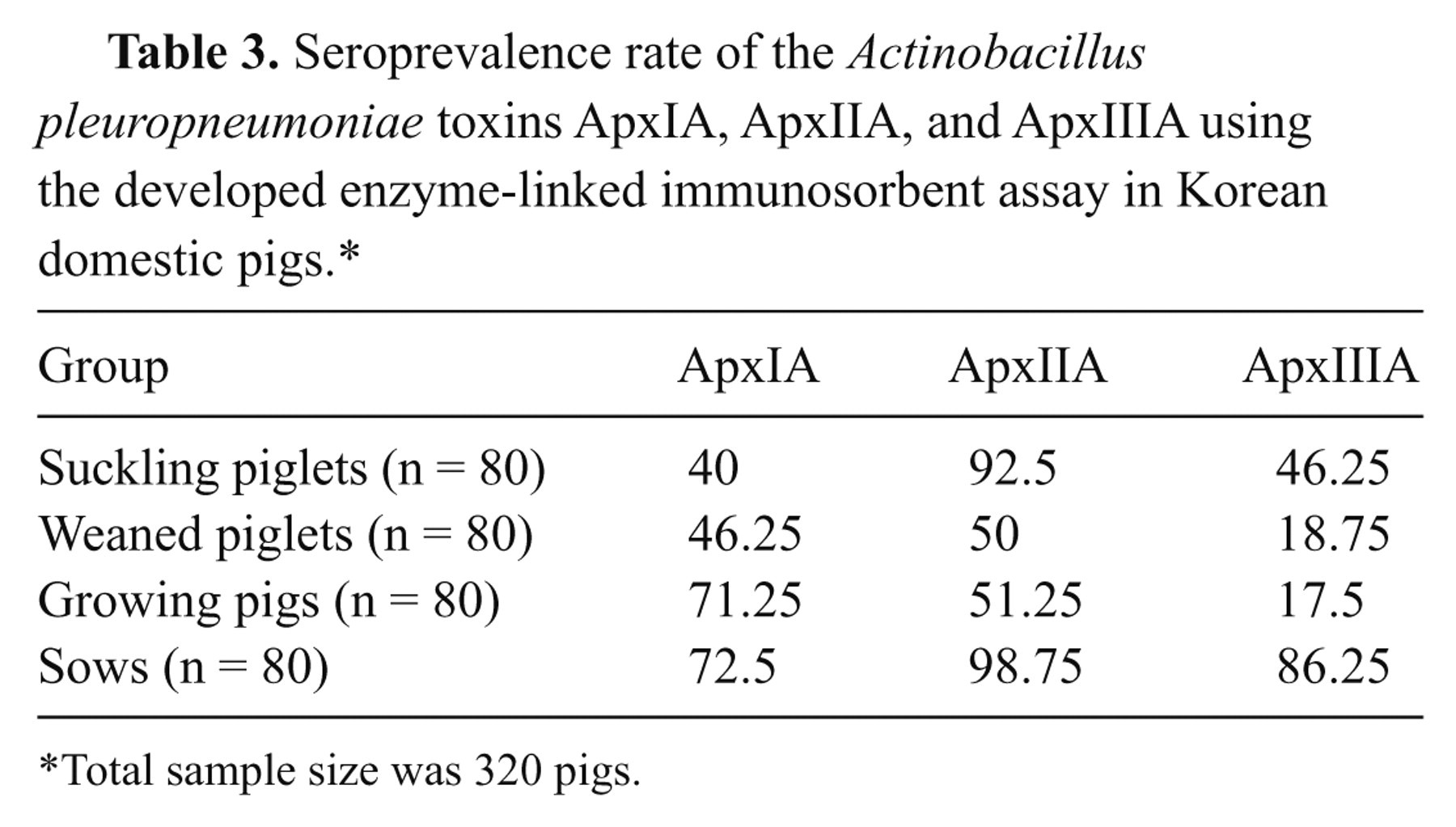
Total sample size was 320 pigs.
Anti-ApxI, -ApxII, and ApxIII -IgG prevalence in domestic pigs
High seroprevalence was found among domestic pigs in Korea, as shown in Figure 4. The rates of antibody positivity for ApxIA, ApxIIA, and ApxIIIA antigens were 57.5%, 73.13%, and 42.19%, respectively. The highest rate was seen for sows when the surveyed pigs were grouped in four herds: suckling or pre-weaning piglets, post-weaning piglets, growing pigs and sows.
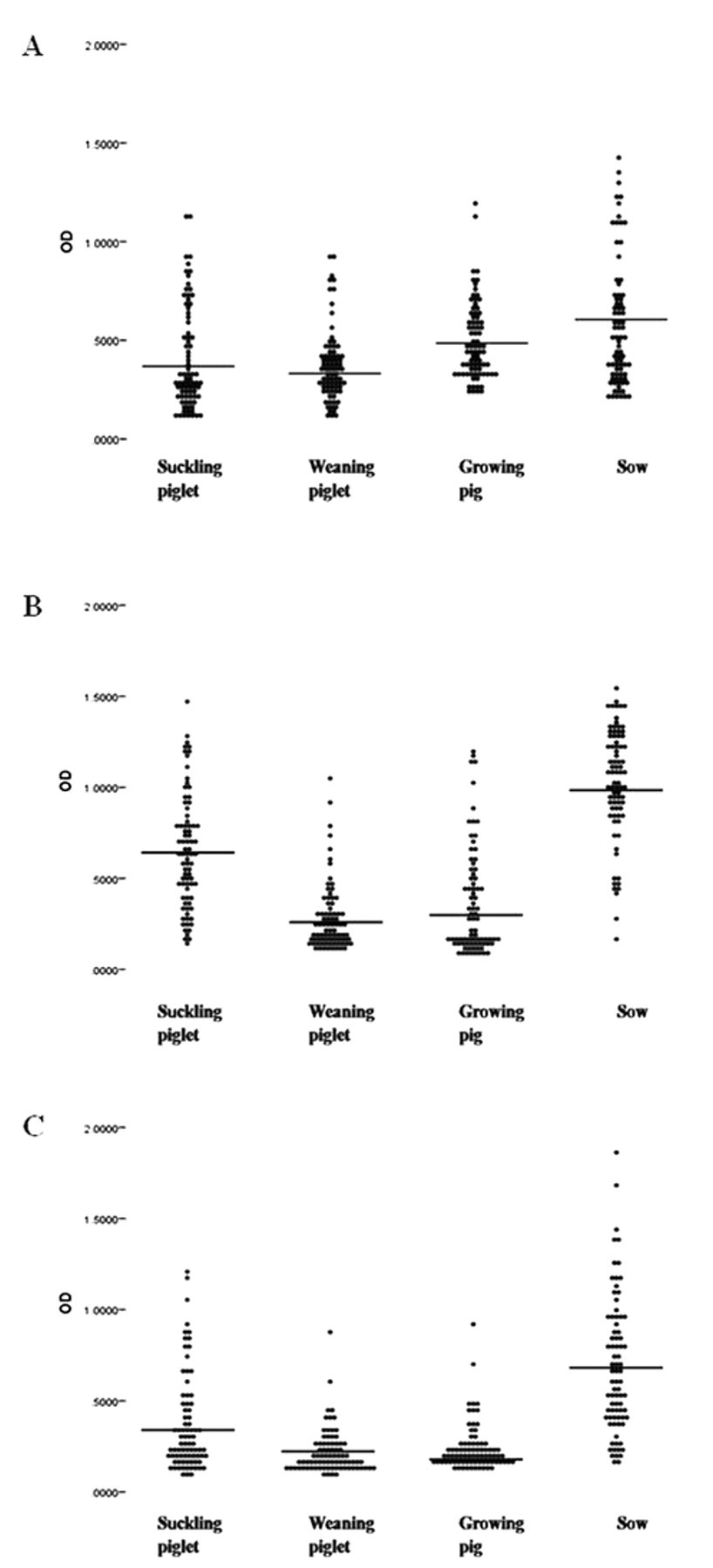
Distribution of anti–Actinobacillus pleuropneumoniae toxins ApxIA (
Discussion
In the current study, the recombinant ApxIA, ApxIIA, and ApxIIIA proteins were used as ELISA antigens instead of employing native proteins. Each recombinant protein was visible as one single band strongly reactive with vaccinated pig sera in Western blot analysis (Fig. 2), suggesting that the developed recombinant proteins produced and purified herein have effective antigenicity enough for ELISA tests. Using each of the cut-off values, ApxIA, ApxIIA, and ApxIIIA ELISAs had sensitivities of 1, 0.95, and 0.925 and specificities of 0.96, 0.96, and 0.96, respectively, when 25 serum samples from 10 colostrum-deprived piglets and 15 nonvaccinated piglets and 40 serum samples from vaccinated pigs were used as test serum controls (Table 2 and Fig. 3). The ELISA results indicate that the developed assay can be used for the detection of serum antibodies to 3 A. pleuropneumoniae exotoxins (ApxI, ApxII, and ApxIII).
When the ELISAs were applied to a seroepidemiologic study of anti–A. pleuropneumoniae exotoxin antibodies in 320 randomized pigs in Korea, high rates of positive antibody were observed in animals of all groups (i.e., suckling piglets, weaned piglets, growing pigs, and sows), especially in sows, which gave the highest mean OD value in the ApxIIA ELISA (Fig. 4). Such finding may be related to the fact that serotypes 2, 5, and 6 are prevalent in Korea, of which all produce ApxIIA, as noted earlier. 10,16,19 Also, the results seemed to show an age-dependent prevalence, except for suckling piglets (Fig. 4). Considering that the mean OD value of weaned piglets in any one of ApxIA, ApxIIA, and ApxIIIA ELISAs was lower than that of suckling piglets (Fig. 4), anti-Apx toxin antibody activity in sera of preweaned animals may have been due to maternal antibodies that disappear gradually after weaning. Although there is evidence for geographic boundaries of pig infection by particular serotypes of A. pleuropneumoniae, emergence of an unexpected serotype should always be taken into consideration in naive pigs. 15 An expanded serologic survey would be needed to understand the present status of A. pleuropneumoniae prevalence, which also is important in planning and evaluating A. pleuropneumoniae vaccination programs. The exotoxin-specific ELISA developed in the current study may provide a useful screening tool for pleuropneumonia control and prevention in swine farms.
Footnotes
a.
Difco Laboratories Inc., Detroit, MI.
b.
Tokyo Chemical Industry, Tokyo, Japan.
c.
Duchefa Biochemie, Haarlem, The Netherlands.
d.
GenElute™, Sigma-Aldrich, St. Louis, MO.
e.
QIAquick, Qiagen GmBH, Hilden, Germany.
f.
ABI 377L, Applied Biosystems, Foster City, CA.
g.
GE Healthcare, Buckinghamshire, United Kingdom.
h.
IG Therapy Co., Kangwon, Republic of Korea.
i.
Bio-Rad, Hercules, CA.
j.
Bethyl Laboratories Inc., Montgomery, TX.
k.
Emax Precision, MDS Inc., Sunnyvale, CA.
l.
Intervet International BV, Boxmeer, The Netherlands.
m.
version 17.0, SPSS Inc., Chicago, IL.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The BioGreen 21 Program (20080401034048), ARPC (107034-03-3-CG000), BK21 Program for Veterinary Science, and the Research Institute of Veterinary Science, Seoul National University, Korea, supported the present study.
