Abstract
Infections with Classical swine fever virus (CSFV) are a major economic threat to pig production. To combat CSF outbreaks and to maintain trade, new marker vaccines were developed that allow differentiation of infected from vaccinated animals (DIVA principle). The chimeric pestivirus CP7_E2alf was shown to be safe and efficacious. Its DIVA strategy is based on the detection of CSFV Erns-specific antibodies that are only developed on infection. However, for the new marker vaccine to be considered a valuable control tool, a validated discriminatory assay is needed. One promising candidate is the already commercially available enzyme-linked immunosorbent assay, PrioCHECK CSFV Erns ELISA (Prionics BV, Lelystad, The Netherlands). Four laboratories of different European Union member states tested 530 serum samples and country-specific field sera from domestic pigs and wild boar. The ELISA displayed a good robustness. However, based on its reproducibility and repeatability, ranges rather than single values for diagnostic sensitivity and specificity were defined. The ELISA displayed a sensitivity of 90–98% with sera from CSFV-infected domestic pigs. A specificity of 89–96% was calculated with sera from domestic pigs vaccinated once with CP7_E2alf. The ELISA detected CSFV infections in vaccinated domestic pigs with a sensitivity of 82–94%. The sensitivity was lower with sera taken ≤21 days post-challenge indicating that the stage of CSFV infection had a considerable influence on testing. Taken together, the PrioCHECK CSFV Erns ELISA can be used for detection of CSFV infections in CP7_E2alf-vaccinated and nonvaccinated domestic pig populations, but should only be applied on a herd basis by testing a defined number of animals.
Introduction
Together with Border disease virus (BDV) and Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2), Classical swine fever virus (CSFV) belongs to the genus Pestivirus within the family Flaviviridae. 31 Pestiviruses are small, enveloped viruses possessing a single-stranded RNA genome (~12.3 kb) in positive orientation. 24 The single open reading frame encodes for 1 polyprotein, which is cleaved during viral maturation into 4 structural (C, Erns, E1, and E2) and 9 nonstructural (Npro, p7, NS2, NS2-3, NS3, NS4a, NS4b, NS5a, and NS5b) proteins.7,22,24 The CSFV proteins NS3, Erns, and E2 induce antibodies on viral infection. Antibodies developed against Erns and E2 have virus-neutralizing abilities and confer protective immunity against viral infection.12,21 Because the E2 protein is the major immunogen, it represents the basis for vaccine development.
The ruminant pestiviruses BDV and BVDV are able to infect all cloven-hoofed animals. In contrast, CSFV is restricted to domestic pigs and wild boar in which it causes the highly contagious disease classical swine fever (CSF). As CSF has a major economic impact on pig production, member states of the European Union have dedicated great efforts to its eradication.15,25 With the implementation of mass vaccination and culling of infected animals in endemic regions, most areas of the European Union are currently considered CSF free. In order to maintain this status and to eradicate and control sporadic CSF outbreaks, strict control measures are applied (EU Council Directive 2001/89/EC, Commission Decision 2002/106/EC).2,3 These are based on a nonvaccination strategy combined with the culling of infected, contact, or neighboring pig herds in case of CSF outbreak. While this strategy has been proven to be effective, the destruction of animals and their products is increasingly considered unethical.16,32 An alternative to the present CSF eradication strategy is emergency vaccination. Under the current legislation, live attenuated CSFV strains and E2-based subunit vaccines can be applied. The use of E2 subunit vaccine allows serological differentiation of infected from vaccinated animals (DIVA principle), which is a prerequisite for the demonstration of a CSF-free status to maintain the international trade of pigs and their products. 28 However, a major drawback of this vaccine is that it induces a late onset of immunity after single vaccination.9,13 Moreover, vertical transmission is not prevented, and the vaccine can also not be used for oral immunization of wild boar as it has to be applied intramuscularly.
Several new CSF vaccines have been developed which combine an early onset of immunity, the possibility of oral vaccination and DIVA properties.8,13 The chimeric pestivirus CP7_E2alf was proven to be a safe and efficacious marker vaccine candidate.9,14,18,26 This vaccine is based on the BVDV-1 strain CP7 in which the E2 protein coding sequence was replaced by the corresponding sequence of the CSFV strain Alfort/187. 27 Using CP7_E2alf, the DIVA strategy can be based on the detection of CSFV Erns-specific antibodies that are developed on infection with naturally occurring CSFV isolates but not after vaccination. In a previous study various commercially available diagnostic tests used for the detection of CSFV Erns antibodies were tested for DIVA purpose. The PrioCHECK CSFV Erns ELISA a was favored. 29 However, the test displayed disadvantages in its sensitivity, specificity, and especially in its robustness. Responding to these test results the manufacturer modified the test system.
The aim of the present study was to evaluate the modified PrioCHECK CSFV Erns ELISA (hereafter, PrioCHECK assay) with a statistical approved number of serum samples to provide evidence and data for its applicability. For this purpose, a panel of 530 serum samples derived from domestic pigs and wild boar was tested by 4 partners of different European Union member states. In addition, all participants added negative field serum samples from domestic pigs or wild boar of their country. The diagnostic sensitivity and specificity, repeatability, and reproducibility of the PrioCHECK assay, as well as its field application as DIVA test in combination with an emergency vaccination strategy using CP7_E2alf marker vaccine is discussed.
Material and methods
CSFV antibody ELISA
In the present study, a slightly modified version of the PrioCHECK assay was evaluated. This version corresponds to the commercially available test to date. ELISA kits were derived from 1 batch. According to the manufacturer’s recommendations, percent inhibitions (PIs) were calculated on the basis of optical density values. Validity of the test was given if the validation control of the PrioCHECK assay kit had a PI value of <55%, and the positive control had a PI value >65%. Test samples with a PI <40% were classified as negative (= absence of CSFV Erns-specific antibodies). Test samples with a PI of ≥40% were classified as positive (= presence of CSFV Erns-specific antibodies).
Participating laboratories
The evaluation of the PrioCHECK assay was organized by the EU and OIE Reference Laboratory for CSF, Hannover, Germany (EURL). In addition, 3 other institutes participated: Institute of Diagnostic Virology, Friedrich-Loeffler-Institute, Island of Riems, Germany (FLI); Institute of Virology and Immunoprophylaxis, Mittelhaeusern, Switzerland (IVI); and Department of Virology, Immunobiology and Parasitology, Uppsala, Sweden (SVA). During the study, the laboratories were referred to as laboratories 1–4 to blind the results.
Experimental design
Validation of the PrioCHECK assay was performed according to the criteria of the OIE. 33 To meet requirements for statistical analysis, a panel consisting of 530 serum samples was compiled and subdivided into 8 categories (Table 1). All serum samples were derived from individual animals. Only category 4c comprised 19 serum samples that were taken from 4 individual domestic pigs and 1 wild boar at different time points (serially acquired serum samples). Selection criteria for the serum samples were the sample results using a commercial CSFV Ab ELISA b (detection of CSFV E2-specific antibodies) and/or virus neutralization (VN) test. Sera that tested positive were classified as CSFV infected. For specific criteria of each category, see below. The serum panel was randomized and blinded. Each laboratory tested the serum sample panel in a single approach once except for laboratory 1, which tested the panel 3 times independently to generate data for statistical analysis of repeatability.
Categories of the serum sample panel used in the current study.*
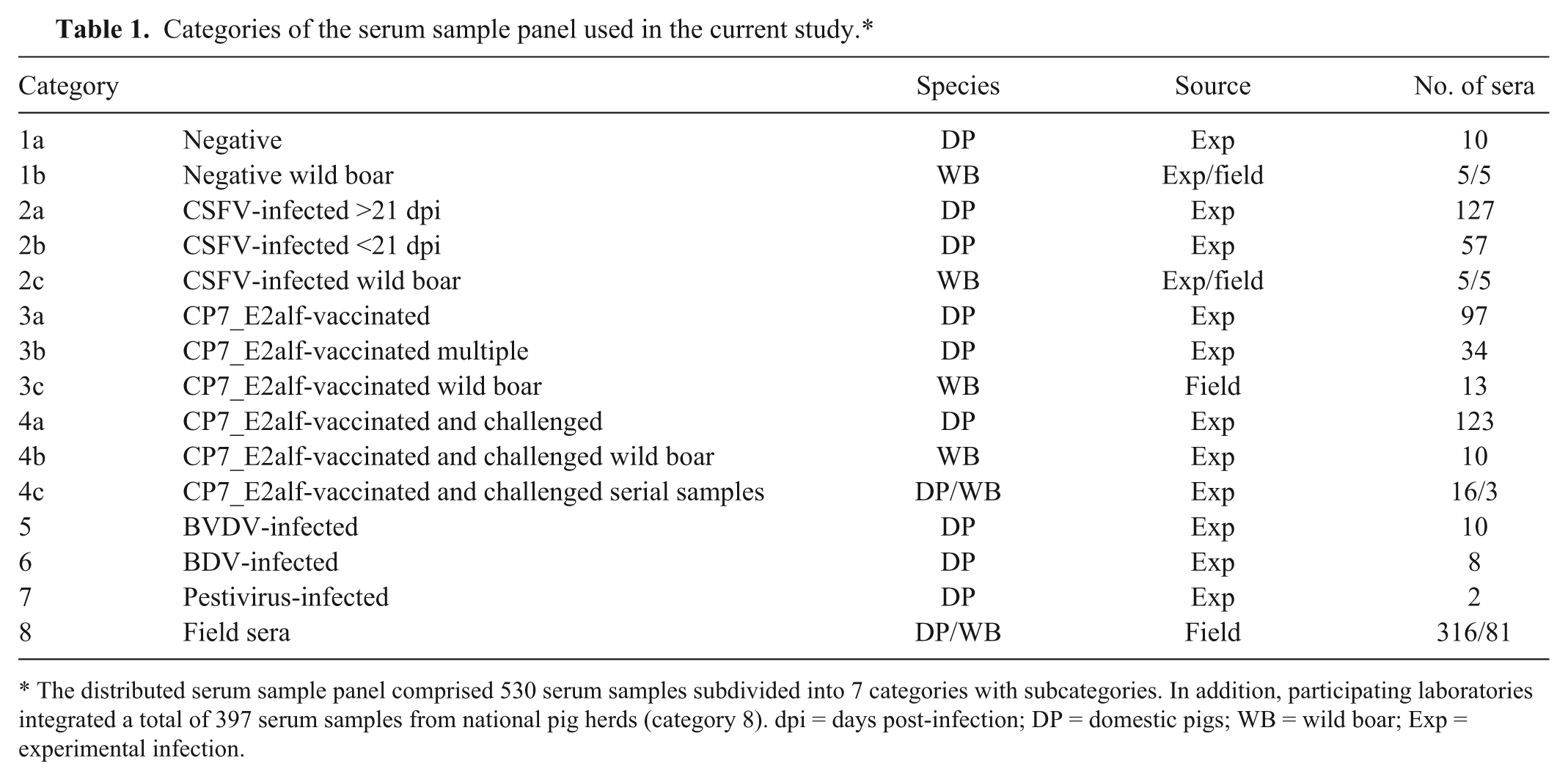
The distributed serum sample panel comprised 530 serum samples subdivided into 7 categories with subcategories. In addition, participating laboratories integrated a total of 397 serum samples from national pig herds (category 8). dpi = days post-infection; DP = domestic pigs; WB = wild boar; Exp = experimental infection.
Statistical analysis
Percent inhibition values of the validation control and positive control were used for determination of repeatability and reproducibility. Repeatability was defined as test–retest reliability of the same test method in a given laboratory. Reproducibility was defined as the ability of the test method to provide consistent results of the same sample tested in different laboratories. To determine reproducibility, PI values of the validation and positive control obtained in different laboratories were compared by pairwise tests. A total of 24 PI values from laboratory 1, 9 PI values from laboratory 2, and 8 PI values from laboratory 3 and 4 of both controls were taken into account. Each PI value was obtained on a separate ELISA plate. Diagnostic sensitivity and specificity were estimated for each serum category and laboratory using the 95% confidence interval.
Sample size calculations were done in analogy with 2009 European Food Safety Authority (EFSA) scientific reports, which use the sampling procedures (design prevalence and 95% confidence) defined in Commission Decision 2002/106/EC (Chapter IV).2,4,5 Briefly, the corrected sample size was calculated on the hypothesis of given design prevalences, test properties (sensitivity and specificity) of the PrioCHECK assay, and usual decision making. It was designed in such a way that the number of positive test results should be zero for the decision of “CSFV free.” If there is at least 1 positive test result, then true prevalence, which is higher than the design prevalence at the 95% confidence level, is confirmed. The influence of herd size on the corrected sample size was shown for 50 and 1,000 animals per farm. The conservative sample was calculated for an infinite population and with specificity and sensitivity of 100%. Sample size calculations were performed with software written by FLI, as described in a 2009 EFSA scientific report.4,5 Furthermore, the results were cross-checked with FreeCalc version 2 (http://www.freecalc.com/) using a threshold of 1 for the number of positive reactors to determine the sample size. 10
Characterization of serum samples
An overview of all serum samples is given in Table 1. CSFV antibody-negative sera (category 1, n = 20) were obtained during animal trials conducted at FLI and EURL. Field serum samples of wild boar were provided by FLI. All sera tested negative for antibodies against CSFV in the CSFV Ab ELISA as well as in the VN test using the test virus CSFV Alfort/187. In addition, 397 serum samples from national pig herds were tested (category 8). The samples were subdivided into 216 serum samples from domestic pigs that were supposedly negative for CSFV antibodies, and 181 negative serum samples collected and investigated during routine diagnosis for CSFV antibodies. The latter were derived from national domestic pig herds (n = 100) and from a monitoring program of wild boar in Germany (n = 81).
CSFV antibody-positive sera (category 2a/b, n = 184) were obtained from domestic pigs during experimental CSFV infections at EURL, FLI, IVI, National Veterinary Institute at the Technical University of Denmark, Lindholm, Denmark (DTU-Vet), Central Agricultural Office, Directorate of Veterinary Medicinal Products, Budapest, Hungary (CAO-DVMP), and Department of Virology at the Central Veterinary Institute of Wageningen UR, Lelystad, The Netherlands (CVI). CSFV antibody-positive sera from wild boar (category 2c, n = 10) were derived either from experimental studies or from the field. The sera were provided by FLI. Sera collected from animals infected with CSFV isolates of various genotypes reflect the genetic and antigenic diversity of CSF (Table 2).
Overview of viruses used for production of positive sera.*
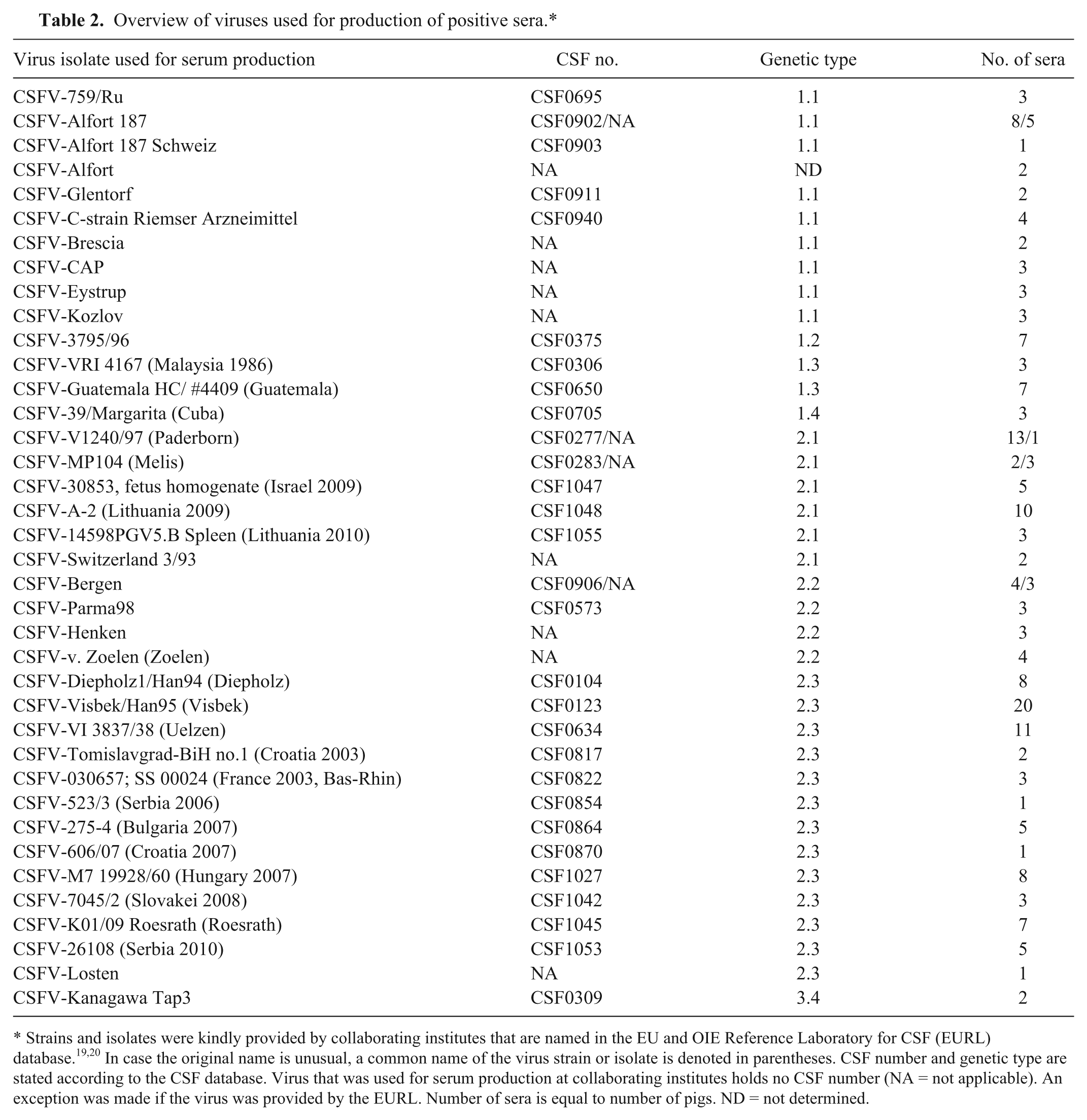
Strains and isolates were kindly provided by collaborating institutes that are named in the EU and OIE Reference Laboratory for CSF (EURL) database.19,20 In case the original name is unusual, a common name of the virus strain or isolate is denoted in parentheses. CSF number and genetic type are stated according to the CSF database. Virus that was used for serum production at collaborating institutes holds no CSF number (NA = not applicable). An exception was made if the virus was provided by the EURL. Number of sera is equal to number of pigs. ND = not determined.
For determination of the DIVA potential, the panel included sera from domestic pigs that were experimentally vaccinated with CP7_E2alf once (category 3a, n = 97) or multiple times (category 3b, n = 34). Category 3b included 25 serum samples of day 14 post–last vaccination from pigs that were vaccinated twice, as well as 6 sera of day 21 and 3 sera of day 28 post–last vaccination from pigs that were vaccinated 3 times. The animal trials were performed in the context of the CSFV_goDIVA project (KBBE-227003) at CVI, FLI, EURL, French Agency for Food, Environmental and Occupational Health & Safety, Ploufragan, France (ANSES), and Pfizer Olot S.L.U. (Zoetis), Vall de Bianya, Spain. Additionally, 13 serum samples were derived from CP7_E2alf field-vaccinated wild boar (category 3c). The field study was conducted by Instituto Zooprofilattico Sperimentale dell’ Umbria e delle Marche, Perugia, Italy.
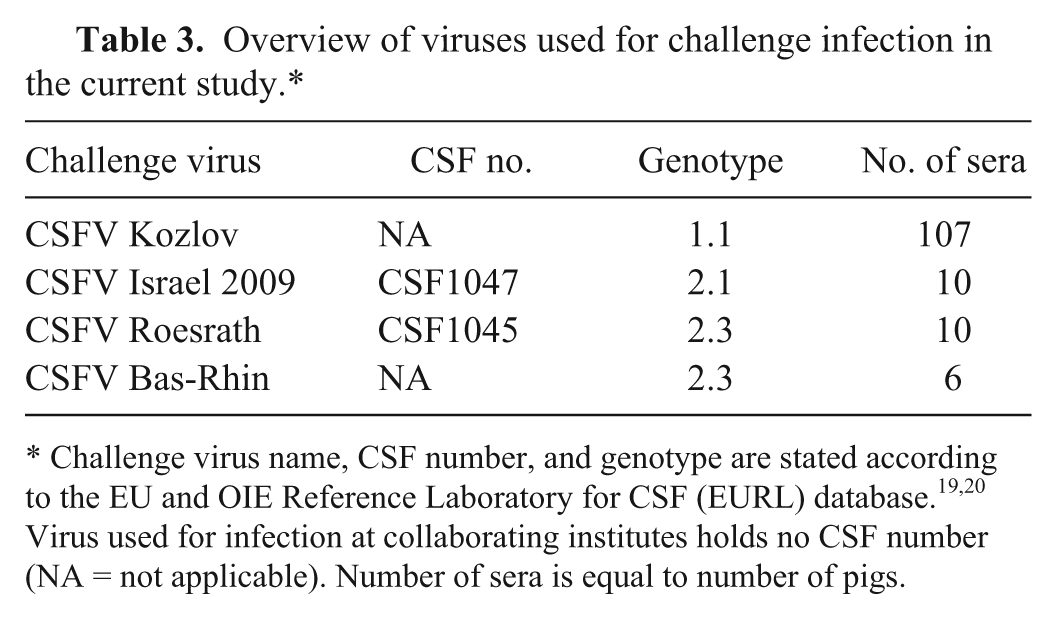
Serum samples from CP7_E2alf-vaccinated and challenged animals (category 4, n = 133) were collected during animal trials of the CSFV_goDIVA project (KBBE-227003) at DTU-Vet, ANSES, CAO-DVMP, CVI, FLI, and EURL. Animals were vaccinated once with the marker vaccine CP7_E2alf and subsequently infected with different CSFV strains at different days post-vaccination (dpv; 2–21 dpv and 182 dpv; Table 3). Sera were collected 10–35 days post-challenge (dpc).
Overview of viruses used for challenge infection in the current study.*
Serially acquired serum samples of 4 domestic pigs originated from an immunity trial conducted by FLI. 18 Within this trial, pigs were vaccinated orally or intramuscularly and challenged with CSFV Kozlov 6 month later. Serially acquired serum samples of 1 wild boar, which was challenged with CSFV Roesrath (CSF1045) 21 dpv at FLI, were also included. Ruminant pestivirus antibody–positive sera were obtained from domestic pigs experimentally infected with BVDV, BDV, and with the unclassified pestivirus Aydin/04-TR, respectively. 6 Sera were collected during animal trials at FLI and EURL.
The CSFV antibody-positive status of each serum sample in categories 2–4 was determined at the institute of origin or at EURL with the CSFV Ab ELISA based on detection of antibodies raised against glycoprotein E2. Antibody titers were determined by VN test using the test virus CSFV Alfort/187. Presence of pestivirus-specific antibodies in serum samples from pigs infected with ruminant pestiviruses of categories 5–7 was evaluated by VN test using the homologous virus.
Results
Analysis of the PrioCHECK CSFV Erns ELISA test method
Practical aspects
Performing the PrioCHECK assay did not cause handling problems in any laboratory. Furthermore, in 49 out of 50 performed runs, the validity of the test was within the range of the manufacturer’s specifications. Only 1 run was invalid.
Repeatability and reproducibility
For calculation of repeatability, PI values of the positive and validation control, respectively, were analyzed for each laboratory separately. The validation control tested at laboratory 1 showed a mean PI value of 25.8 with a variation range of ± 8 PI units. The positive control was tested with a mean PI value of 72.1 with a variation range of ± 6 PI units (Fig. 1). A variation of the measured PI values of both controls was also seen in the 3 other laboratories (Fig. 1). A comparison showed that the dispersions were comparable.
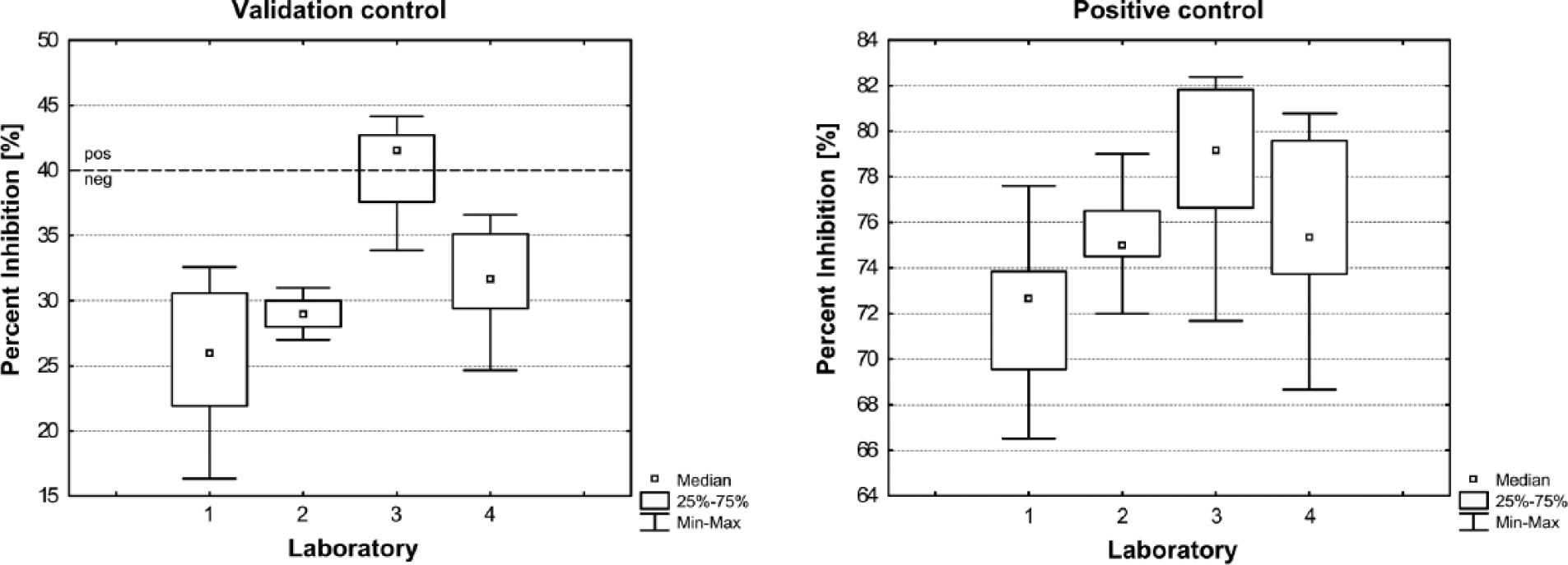
Variation of the validation and positive control obtained by 4 participating laboratories. The validation and positive control are components of the PrioCHECK CSFV Erns ELISA kit.a Each box plot referred to more than 8 percent inhibition (PI) values obtained in separate runs in the given laboratory using a single measurement instrument. Cutoff value is marked by a dotted line.
To determine reproducibility, the PI values of the positive and validation control, respectively, were compared between the laboratories. The analysis revealed that the PI values displayed a laboratory-dependent local accumulation (Fig. 1). Therefore the values were not comparable.
Despite intralaboratory differences (repeatability) and interlaboratory differences (reproducibility), the PI values of the positive control scored inhibition values >40% in all runs. The PI values of the validation control scored a clear inhibition of <40% for 3 laboratories (classified as negative). However, for 1 laboratory, PI values were similar to the cutoff, resulting in an equivocal classification between different runs (Fig. 1). This demonstrated clearly that intra- as well as inter-laboratory differences can significantly influence negative or positive classifications. Thus, the classification of individual serum samples obtained in different runs was not comparable. Consequently, diagnostic specificity and sensitivity were separately calculated for each run and laboratory.
Determination of diagnostic measurements
Specificity and sensitivity with samples from nonvaccinated domestic pig populations
All negative serum samples (categories 1a and 8) from domestic pigs scored negative in all runs. Based on this, diagnostic specificity was calculated collectively using data of category 8, which comprised a sufficient number of serum samples for statistical analysis. For 216 serum samples that were not pretested for antibodies against CSFV, the estimated specificity was >98.6% (confidence level of 95%) while for 100 pretested serum samples a specificity of >97% (confidence level of 95%) was estimated.
For determination of sensitivity, 127 positive sera from pigs in a convalescent stage of CSFV infection (>21 dpi; category 2a) and 57 sera taken from pigs in the early phase of CSFV infection (≤21 dpi; category 2b) were analyzed separately according to the diagnostic manual. 2 For the individual laboratories, the sensitivity for sera taken >21 dpi ranged from 90% to 98% (Table 4). Three serum samples of this category scored univocally negative. Nine sera of the same category were classified as either negative or positive. For these equivocally classified sera, the mean of the PI values was 45%, which is near the cutoff. The sensitivity of serum samples taken <21 dpi ranged from 82% to 95%. One serum sample scored negative in all runs. For 10 serum samples, equivocal results were obtained. Again, the mean of the PI values of these sera (37%) was very close to the cutoff value. Sera from C-strain “Riems”-inoculated pigs taken 32 dpi were classified univocally negative.
Sensitivity of positive sera.*
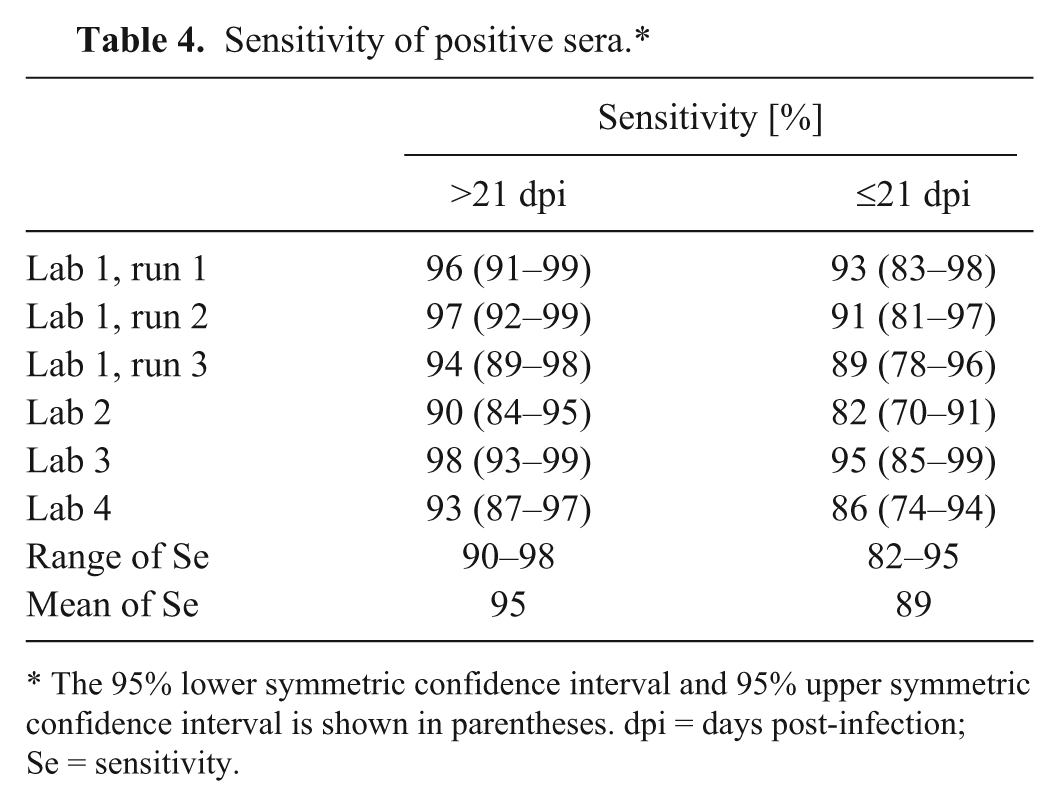
The 95% lower symmetric confidence interval and 95% upper symmetric confidence interval is shown in parentheses. dpi = days post-infection; Se = sensitivity.
DIVA potential
To prove the DIVA property of the ELISA, 97 serum samples derived from CP7_E2alf-vaccinated domestic pigs were analyzed. For sera from single vaccinated animals, a specificity of 89–96% was calculated (Table 5). Four sera scored univocally positive leading to a false interpretation of CSF positive. The mean PI value of these positive tested sera was 51.48. A total of 14 serum samples gave equivocal results (mean PI value: 33.62). False-positive and equivocal results could not be assigned to a certain time period post-vaccination. For sera taken after multiple vaccinations, the specificity was 79–85%. Five serum samples were falsely interpreted as CSF positive (mean PI value: 57.82), while a total of 4 sera were equivocal (mean PI value: 27.33).
Differentiation of infected from vaccinated animals (DIVA) potential.*
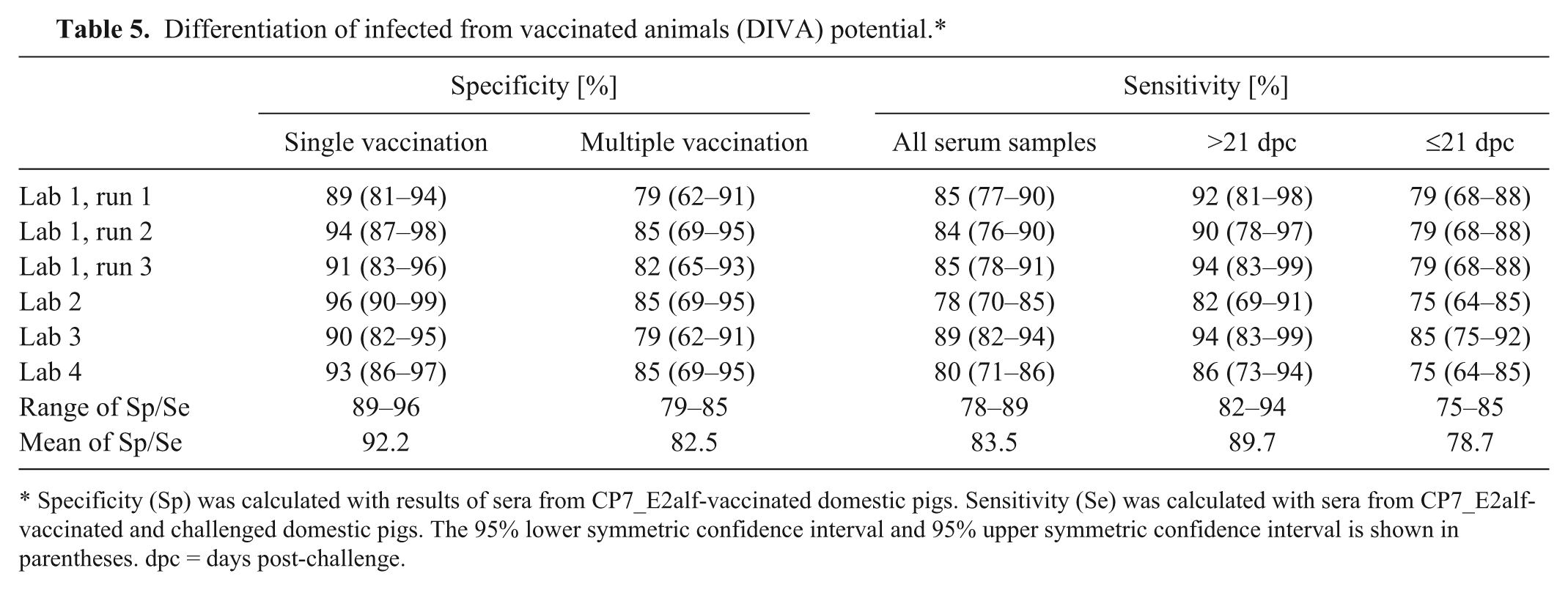
Specificity (Sp) was calculated with results of sera from CP7_E2alf-vaccinated domestic pigs. Sensitivity (Se) was calculated with sera from CP7_E2alf-vaccinated and challenged domestic pigs. The 95% lower symmetric confidence interval and 95% upper symmetric confidence interval is shown in parentheses. dpc = days post-challenge.
Furthermore, the sensitivity for samples taken from vaccinated and subsequently challenged pigs was calculated. Using 123 serum samples of category 4a, a total of 13 scored univocally negative (mean PI value: 2.42), and 14 serum samples gave equivocal results (mean PI value: 38.67). The sensitivity ranged from 78% to 89% and was higher for samples taken >21 dpc (82–94%), while the sensitivity of serum samples taken ≤21 dpc ranged between 75% and 85% (Table 5).
Using the mean of the calculated range of specificity from single vaccinated pigs (92.2%) and the mean of the calculated range of sensitivity from vaccinated and challenged pigs in a convalescent stage of infection (89.7%), sampling sizes were calculated for CP7_E2alf-vaccinated populations (Table 6). The calculation of the sampling size was done in analogy with a 2009 EFSA scientific report, which in turn uses the sampling procedures mentioned in manual 2002/106/EC.2,4,5 The corrected sample size calculated for different reasons in Table 6, gives the number of animals that should be tested to detect an infection and attest to freedom of disease. For the test results of the sample size, a rule of inference applies: If 1 sample of the random sample size tests positive, the population will be considered infected. In accordance with manual 2002/106/EC, design prevalences of 5% and 10% were used for calculation. As the prevalence in vaccinated populations is considered very low, a design prevalence of 1% was also used.4,5 Table 6 shows that farm size and design prevalences have an influence on the sample size.
Calculation of sampling sizes in CP7_E2alf-vaccinated pig populations.*
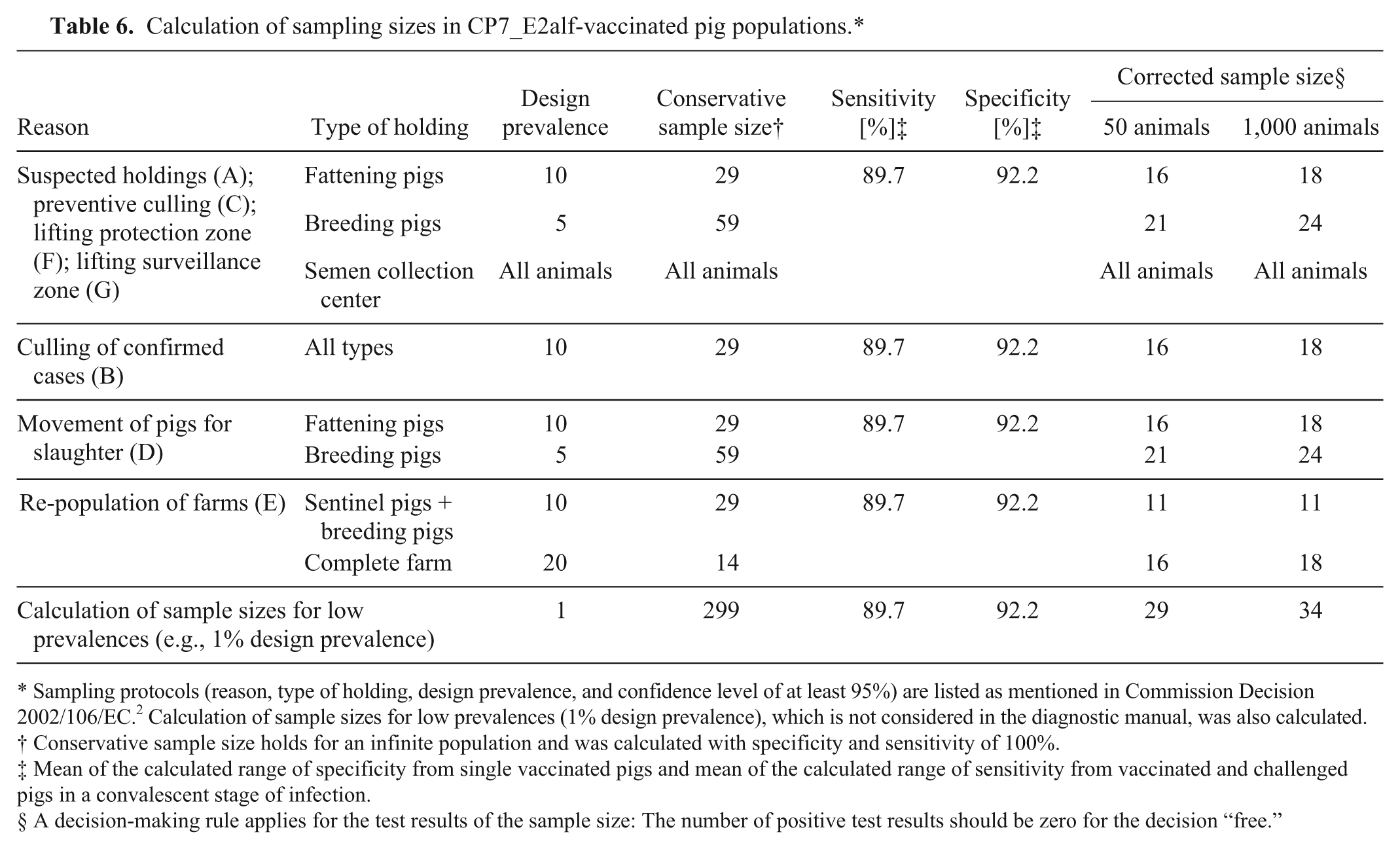
Sampling protocols (reason, type of holding, design prevalence, and confidence level of at least 95%) are listed as mentioned in Commission Decision 2002/106/EC. 2 Calculation of sample sizes for low prevalences (1% design prevalence), which is not considered in the diagnostic manual, was also calculated.
Conservative sample size holds for an infinite population and was calculated with specificity and sensitivity of 100%.
Mean of the calculated range of specificity from single vaccinated pigs and mean of the calculated range of sensitivity from vaccinated and challenged pigs in a convalescent stage of infection.
A decision-making rule applies for the test results of the sample size: The number of positive test results should be zero for the decision “free.”
Cross-reactivity
The potential of the PrioCHECK assay to differentiate CSFV Erns-specific antibodies from serological reactions against ruminant pestiviruses was analyzed with sera from domestic pigs that were infected with BVDV-, BDV-, and Aydin/04-TR, respectively. Six out of 10 BVDV-positive sera scored negative, 1 gave an equivocal result, and 3 scored positive. Three out of 8 BDV-positive sera were classified as negative, 2 were equivocal, and 3 scored a positive result. Serum samples of animals infected with strain Aydin/04-TR were classified negative.
Wild boar serum samples
Based on the results of pretested negative field serum samples from wild boar, a specificity of 87.1% was calculated (Table 7). With regard to the CSFV antibody-positive field samples, 4 out of 5 were classified negative, even though they had a clear positive result in the E2 ELISA as well as in the VN test. Out of 13 sera obtained from a CP7_E2alf field trial, only 7 were correctly classified as negative. On the contrary, for experimentally derived samples, 4 out of 5 CSFV antibody-positive sera were correctly classified as positive. Sera were taken on days 15–28 post-infection. The animal that tested inconclusive was sampled on day 23. Similarly, 10 sera taken from CP7_E2alf-vaccinated and experimentally challenged pigs (category 4b) were all correctly classified as positive.
Analysis of wild boar serum samples.*
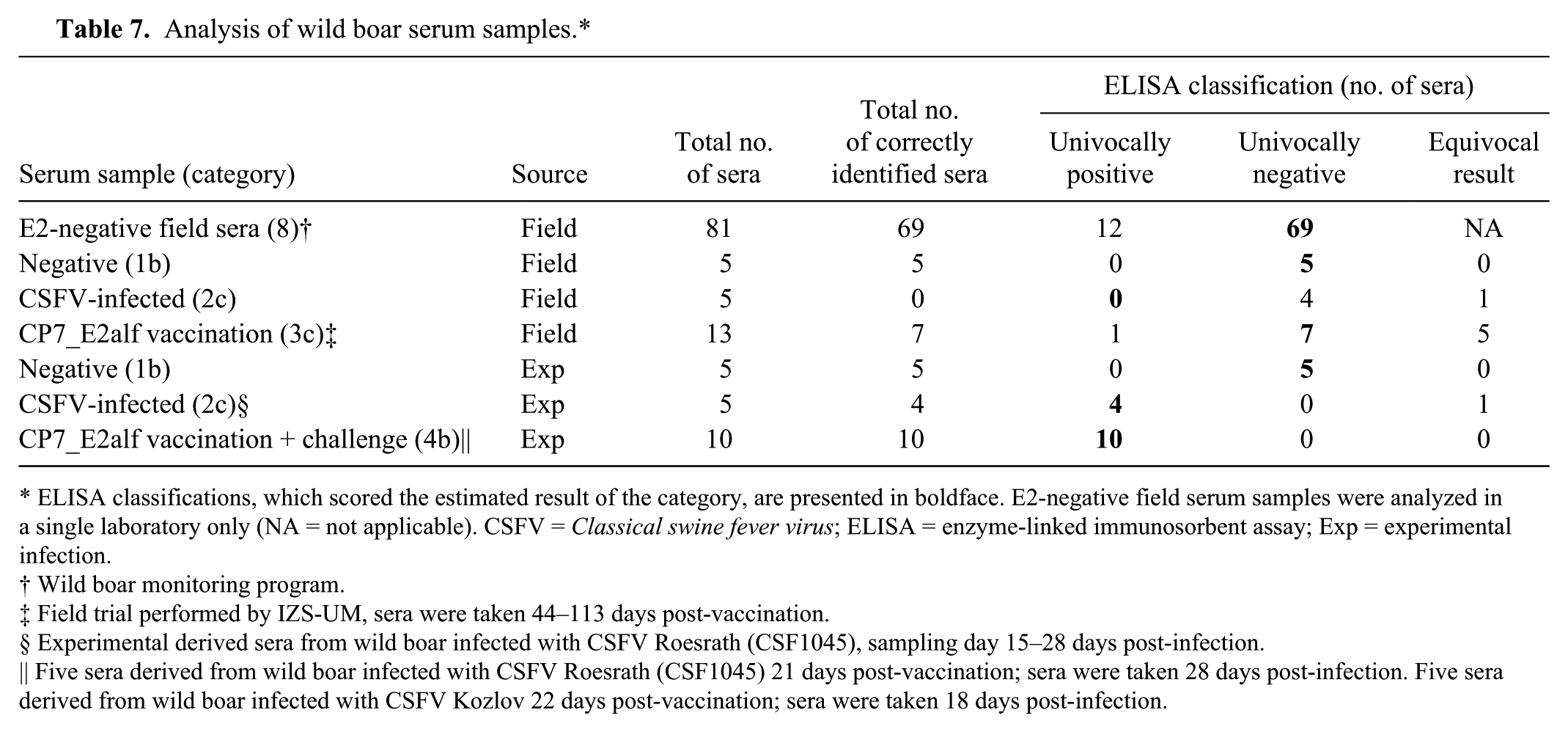
ELISA classifications, which scored the estimated result of the category, are presented in boldface. E2-negative field serum samples were analyzed in a single laboratory only (NA = not applicable). CSFV = Classical swine fever virus; ELISA = enzyme-linked immunosorbent assay; Exp = experimental infection.
Wild boar monitoring program.
Field trial performed by IZS-UM, sera were taken 44–113 days post-vaccination.
Experimental derived sera from wild boar infected with CSFV Roesrath (CSF1045), sampling day 15–28 days post-infection.
Five sera derived from wild boar infected with CSFV Roesrath (CSF1045) 21 days post-vaccination; sera were taken 28 days post-infection. Five sera derived from wild boar infected with CSFV Kozlov 22 days post-vaccination; sera were taken 18 days post-infection.
Serially acquired serum samples
To determine the onset of an Erns-specific immune response after infection, serially acquired serum samples of 5 vaccinated and subsequently challenged individuals were integrated into our study. Univocal results were achieved for sera taken from a domestic pig (Fig. 2, animal B) and a wild boar (data not shown). In both cases, Erns-specific antibodies were detected as early as 7 dpc in every run. For animal C, no Erns-specific seroconversion was detected until the end of the animal trial. Only at 4 dpc was this animal classified as positive at laboratory 3. However, on the following sampling days and also before the challenge (data not shown), this animal tested negative in this particular laboratory, which indicates a performance error (e.g., pipetting). In contrast to these animals, the results from animals A and D were equivocal (Fig. 2, equivocal results are marked by arrows). Animal A was interpreted as Erns antibody positive and therefore as CSF positive 10 dpc in 2 runs at laboratory 1 and also at laboratory 3. On the contrary, this animal was not detected to be infected with CSFV until 14 dpc in the third run by laboratory 1, laboratory 2, and laboratory 4. Intra- and interlaboratory differences were also observed for animal D. Here, Erns-specific antibodies were detected first on 7 dpc, 10 dpc, and 22 dpc depending on the run. At laboratory 4, the same animal was not classified as CSF positive.
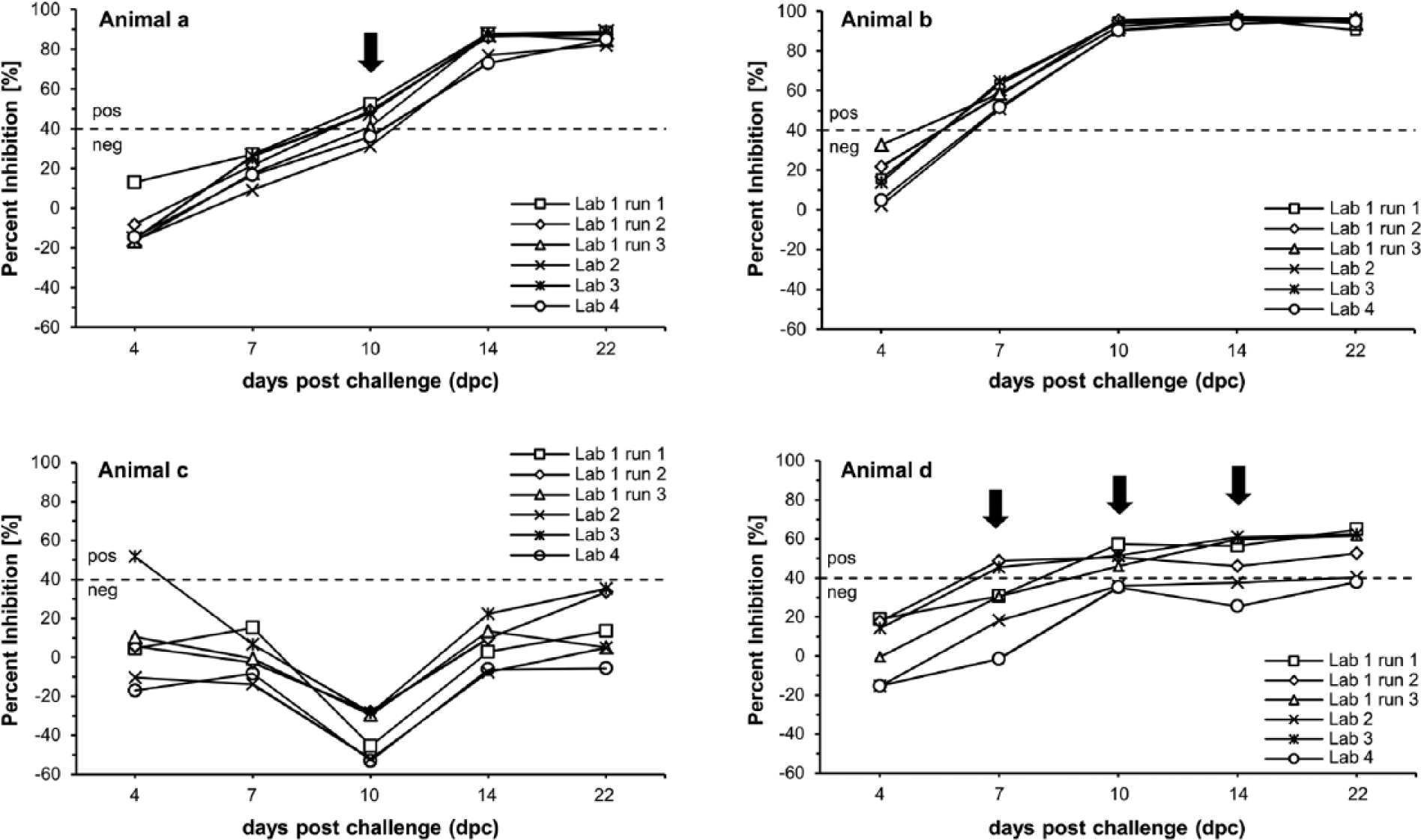
Detection of Erns-specific antibodies in 4 domestic pigs in different runs in the same or in different laboratories with the PrioCHECK CSFV Erns enzyme-linked immunosorbent assay (ELISA).a Depicted is the detection of the antibody response in serially acquired serum samples of 4 domestic pigs (animals a–d) on challenge infection after vaccination with CP7_E2alf. Cutoff value is marked by a dotted line. Arrows indicate differences in Erns antibody detection between runs in a single laboratory and different laboratories (equivocal results).
Discussion
The applicability of emergency vaccination against CSF using the potential marker vaccine CP7_E2alf is highly dependent on the availability of a reliable assay for differentiation of infected and vaccinated animals (DIVA). To date, the commercially available diagnostic assays do not fulfil this requirement. One promising candidate is the PrioCHECK assay, which showed disadvantages in a previously conducted study testing a small-scale serum sample panel. Disadvantages in sensitivity, specificity, and robustness were observed. 29 Now, a modified version of the test system was evaluated focusing on the determination of its diagnostic specificity and sensitivity with a serum sample panel that fulfilled the requirements for statistical analysis. In addition, the unique opportunity of the acquisition of a huge data set from different laboratories was applied to determine the robustness, repeatability, and reproducibility of this test system.
The modified version of the PrioCHECK assay showed a good robustness, as only 1 out of 50 ELISA runs was invalid. As validation and positive control were tested several times by each laboratory, their PI values were used for determination of repeatability and reproducibility. PI values of both controls showed a variation (intralaboratory variability), which was comparable between the laboratories, but displayed a statistical approved laboratory-dependent local accumulation around the laboratory-specific median (interlaboratory variability). Whereas the intra- and interlaboratory variability had no impact on serum samples with PI values greater or smaller than the cutoff value, a considerably influence was seen on sera with PI values near 40% (see Figs. 1, 2). Subject to the ELISA run and the laboratory that tested the sample, such sera yielded different results in the negative or positive classification. Therefore, determination of overall valid diagnostic measurements was not possible. Consequently, diagnostic sensitivity and specificity were calculated for each laboratory separately, and respective ranges were defined. Based on the lack of standardization, the test system needs to be further improved. However, the range of sensitivity and specificity can be used to calculate the minimum sample size that had to be tested to exclude a CSFV infection in a pig herd.4,5 Despite the lack of a standardized diagnostic measurement, the PrioCHECK assay proved to be highly specific (>97%) in nonvaccinated domestic pig populations. The test system also displayed a satisfactory sensitivity (90–98%) for sera taken >21 dpi. The negative classification of sera that were supposedly positive was not linked to certain sera characteristics, such as the CSFV strain used for infection, time point after infection, titers of CSFV E2-specific Ab, or titers determined in VN testing. Alternatively, factors of individual pigs might have an influence on the correct detection of Erns-specific antibodies by the PrioCHECK assay. It should also to be noted that serum samples of this category were selected according to a positive E2 antibody titer. The true Erns antibody status was unknown and could not be tested beforehand, as there was no other diagnostic test available. An exception was the result of sera from C-strain “Riems”-infected pigs taken at 32 dpi. All tested samples scored univocally negative, even though they were clearly positive for E2 antibodies. A delayed production of Erns antibodies in pigs infected with C-strain “Riems” was described previously, and it was assumed that this delay is a unique feature of this virus and may be associated with its attenuation.1,17 As C-strain “Riems” is no longer used for area-wide vaccination of domestic pigs in the European Union, it has no influence on the performance of the Erns ELISA in a field application. Based on this, it was excluded when calculating this diagnostic measurement.
Overall, the CSFV Erns ELISA was less sensitive than the conventional E2 antibody ELISA that is recommended for detection of CSFV-specific antibodies in nonvaccinated pig herds.4,5 Nevertheless, the Erns ELISA can be used with an appropriate number of samples.
Besides the determination of specificity and sensitivity in nonvaccinated pig populations, the capability to differentiate antibodies raised after infections with ruminant pestiviruses was analyzed. Many of the sera collected from BVDV- and BDV-infected pigs tested positive. In a nonvaccinated pig population this has no significant importance, as positive ELISA results need to be confirmed by differential diagnosis using VN tests to exclude ruminant pestivirus infections. 2 In contrast, VN tests are not suitable to apply to sera from CP7_E2alf-vaccinated pigs as these animals develop not only specific antibodies with neutralizing abilities against the CSFV E2 protein, but also against the BVDV Erns protein. However, in the European Union, seroprevalence against BVDV and BDV is currently low in domestic pig populations. 23 Therefore, cross-reactivity is a minor problem, as it is not expected that ruminant pestivirus infections interfere with the ability of the ELISA to detect CSFV antibodies on a herd level.
With regard to the DIVA potential, the PrioCHECK assay had a specificity of 89–96% with sera from pigs vaccinated once with CP7_E2alf, while the specificity with sera from multiple vaccinated animals was lower and ranged between 79% and 85%. After a booster vaccination, the titer against the CSFV E2 protein, as well as against the BVDV Erns protein, may rise. As the Erns protein is highly conserved among pestiviruses, it is reasonable that differentiation is difficult for sera with high antibody titers. 1 In an emergency vaccination scenario, multiple vaccinations would not be applied in domestic pig populations. Therefore, the specificity obtained with sera from single vaccinated domestic pigs can be used to calculate the sampling and testing procedure. However, it should be pointed out that oral vaccination of wild boar can easily result in multiple vaccinations by repeated uptake of baits.
An emergency vaccination with the marker vaccine CP7_E2alf would be applied after a confirmed outbreak of CSF with the goal to limit the spread of the disease. As a field virus infection in a vaccinated domestic pig herd might occur at any time point, pigs could be in any stage of CSFV infection at the time of sampling. On this basis, serum samples were analyzed from domestic pigs that were challenged at different time points after vaccination and were in different stages of CSFV infection. Sensitivity was higher with sera from a late-stage CSFV infection (>21 dpc), which indicated that the stage of CSFV infection had a considerable influence on this measurement. In order to obtain maximum sensitivity of the PrioCHECK assay in a field application, an appropriate number of vaccinated pigs should be sampled at the latest feasible time point. Under current European Union legislation, established measures in the 3-km radius zone around the outbreak are not lifted before 30 days have elapsed after completion of the preliminary cleaning and disinfection on the infected holding. 3 Erns-specific antibodies in sera from vaccinated and challenged animals integrated in the serum panel of this study were still present in 9 of 10 animals at day 35 after challenge (1 animal tested either positive or negative depending on the run). An even later time point was evaluated by a previous study. 9 Nine of 12 animals showed Erns antibodies at 49 dpi. In 1 animal, the Erns titer diminished under the cutoff of the Erns ELISA toward the end of the study. Erns-specific antibodies that developed after infection in CP7_E2alf-vaccinated pigs are therefore detectable to at least day 49 after infection.
Calculated sensitivity and specificity determine a herd application for the PrioCHECK assay, as both parameters are <100%. A reliable classification of individually tested animals as infected or not-infected is not possible. Therefore, application of this assay to classify individually tested pigs free of CSF for transportation and/or marketing of the pig or its products cannot be achieved with this testing method. The calculated specificity in a DIVA setting might be attributed to the detection of Erns-specific antibodies developed against the Erns protein of the vaccine. The given sensitivity of the PrioCHECK assay might be based on the capabilities of the test system itself, but it should also be noted that the detection of antibodies against the challenge virus in CP7_E2alf-vaccinated animals also has its biological limitations. As in previous studies, our study also showed (e.g., serially acquired serum samples) that vaccination with CP7_E2alf can lead to a delayed or no Erns-specific immune response after challenge infection.9,18 As speculated previously, a lack of challenge virus replication in CP7_E2alf-vaccinated animals might be responsible for this. 9
Because of decreased immune response after vaccination, the sensitivity of a serological test system used in DIVA strategies should be high. However, as even the highest sensitivity cannot overcome the limitation of a low or absent immune response in some pigs, the number of samples tested might need to be increased to compensate.
The sensitivity of the PrioCHECK assay in our study was calculated on the basis of serum samples derived from vaccinated pigs that were known to be infected and had a positive E2 immune response, whereas the Erns antibody status was unknown and not a parameter for selection. Based on this, the calculated sensitivity reflects and unites both the characteristics of the test system and the biological limitations. As this reflects to a great extent the reality of use in the field, it might be a good basis for the calculation of a minimum sample size needed to detect an infection and attest to freedom of disease. Using parameters mentioned in Commission decision 2002/106/EC and in analogy with the 2009 EFSA scientific report, sample sizes were calculated for populations vaccinated with CP7_E2alf.2,4,5 In general, positive tested samples can be evoked by true-positive results due to infection or by false-positive results due to the specificity of a test system. Taking into account the calculated test specificity of 92.2%, it is likely that testing of the whole herd or the calculated conservative sample size (see Table 6) will frequently include false-positive tested samples. Therefore, a method designed by FLI was implemented to calculate the corrected minimum sample size using the estimated specificity and sensitivity of the ELISA. The calculation method was designed in such a way that at least 1 positive test result in the random sample size is considered as an indication for infection. In such a case, the herd will be considered infected. However, the high probability of false-positive results based on the specificity of the ELISA represents a limitation for the application of this test system to attest to freedom of disease at the herd level. Altogether, there is a dire need of confirmatory assays; and epidemiological plausibility (e.g., clustering and level of positivity) has to be taken into consideration whenever single positive results are encountered. With regard to confirmatory tests, approaches have been published that could help to overcome this shortcoming.1,34
Besides the application of CP7_E2alf in domestic pig populations, this vaccine also allows for the possibility of oral immunization of wild boar. Estimation of specificity was possible for wild boar serum samples obtained from the field (specificity: 87.1%). If sera were taken from the field, a tendency was observed toward a not expected classification. As seen with the calculated specificity, a number of samples that were supposedly negative scored a positive result. An increase in the detected Erns-specific antibodies of such samples might occur with a ruminant pestivirus infection of wild boar. The prevalence of BVDV antibodies in wild boar is 0.8%, 1%, and 4.5%, in Germany, the Czech Republic, and Croatia, respectively.11,30,35 However, taking these numbers into account, ruminant pestivirus infections cannot be the only explanation for the PrioCHECK assay test result. It should also be noted that wild boar serum samples from the field usually exhibit a different quality than samples obtained under experimental conditions. The samples used for calculating sensitivity, for example, exhibit a very poor quality, as they were taken during hunting season. An improvement of the detection of true positive or true negative samples might therefore be achieved by enhancing the serum sample quality (e.g., by specific training and distribution of information to people who are responsible for taking blood samples). Furthermore, heat inactivation of sera, which will inactivate the complement, could be applied (Sandra Blome, personal communication, 2012).
As analyzed, the PrioCHECK assay showed no robustness with wild boar serum samples from the field. However, in a field application of the PrioCHECK assay after an emergency vaccination of wild boar with CP7_E2alf, it can be argued that determination of the rate of immunity (seroprevalence) and the detection of a trend may be sufficient to determine the stop of vaccination. Such a trend could be easily detected in wild boar younger than 1 year. In this population, the expected difference in the number of Erns-specific antibodies from a CSFV infection at the beginning of the vaccination campaign and a vaccinated population at the end is considered to be high. Furthermore, it must be noted that a lack of sensitivity and specificity of the accompanying DIVA diagnostic test in a vaccination campaign of wild boar might not have much impact as zero prevalence is not expected due to maternal immunity.
The aim of this study was to test the PrioCHECK assay with a statistical approved set of samples to determine whether it is suitable to apply in combination with the potential marker vaccine CP7_E2alf. The test showed a specificity of >97% and a sensitivity of 90–98% in a nonvaccinated population. In a CP7_E2alf single vaccinated population, specificity of 89–96% and sensitivity of 78–89% was calculated. While it is not possible to test individual animals free of CSF, CSF can be detected on a herd level in nonvaccinated as well as in CP7_E2alf-vaccinated populations. As only ranges of statistical measurements could be defined, possibilities to enhance standardization are being discussed in line with ongoing research to further enhance specificity and sensitivity of the test system.
Footnotes
Acknowledgements
We thank Jan Böhmer, Claudia Gabriel, Martin Hofmann, Sandra Juanola, Marie-Frédérique Le Potier, Réka Lévai, Willie Loeffen, Gian Mario De Mia, Patricia Renson, and Åse Uttenthal for providing serum samples and additional information on these samples. We also thank the Institute of Diagnostic Virology, Friedrich-Loeffler-Institute (Island of Riems, Germany), the Institute of Virology and Immunoprophylaxis (Mittelhaeusern, Switzerland), and the Department of Virology, Immunobiology and Parasitology (Uppsala, Sweden) for participating in the collaborative study. Likewise, we thank Gerard van de Wetering from Prionics (Lelystad, The Netherlands) for providing the modified version of the PrioCHECK CSFV Erns ELISA. In addition, we are also grateful to Gabriele Müller and Karin Ruthenberg for technical assistance.
Authors’ contributions
K Pannhorst, A Fröhlich, C Staubach, D Meyer, and S Blome contributed to conception and design of study. K Pannhorst, A Fröhlich, C Staubach, and S Blome contributed to acquisition, analysis, and interpretation of data. D Meyer and P Becher contributed to interpretation of data. K Pannhorst drafted the manuscript. All authors critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
PrioCHECK CSFV Erns ELISA, Prionics BV, Lelystad, The Netherlands.
b.
CSFV Ab ELISA, IDEXX Laboratories, Hoofddorp, The Netherlands.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or other publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Community’s Seventh Framework (FP7/2007-2013) under grant agreement 227003 CP-FP (CSFV_goDIVA).
