Abstract
Coxiella burnetii is an obligate intracellular bacterium that is responsible for the zoonotic disease Q fever. The distribution of this agent is worldwide except for New Zealand, and infection can be asymptomatic in both human beings and animals. Chronic exposures can produce abortions, stillbirths, and infertility issues in animals and endocarditis in human beings. A commercial enzyme-linked immunosorbent assay (ELISA) kit marketed in the European Union was purchased to compare C. burnetii antibody detection methods. The current study examined the agreement of ELISA and complement fixation results in over 668 diagnostic ruminant sera submitted to the National Veterinary Services Laboratories for Q fever serologic testing. The majority of combined sera (548) were negative on both tests. Fifty-seven of the combined sera were positive on both tests. There were 45 combined sera with low complement fixation titers at 1:10 and negative ELISA results. The results were surprising given the expectations that ELISA methods, by nature, amplify detection of antibody–antigen interactions leading to higher sensitivity. Potential mechanisms for these discrepant results are discussed.
Coxiella burnetii, the etiologic agent for Query fever (Q fever) was first described in the 1930s in Australia. The organism has since been identified in countries worldwide except for New Zealand. 6 Large amounts of the zoonotic bacteria are shed from infected animals in birthing products and also, to a lesser extent in feces, urine, milk, and vaginal mucus. 6 The bacteria are resistant to heat, drying, and light making them very environmentally stable. 8 This agent replicates in a diverse range of animal hosts including cattle, sheep, and goats, which can act as reservoirs for human disease. 6 The disease in human beings and animals can be asymptomatic. 6 Chronic exposures can produce abortions, stillbirths, and infertility issues in animals, and endocarditis in human beings. Infectious material can travel on the wind up to 10 miles dispersing the bacteria far from the initial source. 5 This obligate intracellular bacteria is a risk as a bioterrorism agent. 6
Submissions to the National Veterinary Services Laboratories for Q fever complement fixation (CF) testing included import and/or export testing that is prescribed for animal movement, diagnostic serum samples, and screening of ruminants used in research facilities. There has been an increase in submissions from research facilities seeking seronegative goats and/or sheep to minimize the risk to staff from Q fever exposure. The current study was undertaken to compare 2 C. burnetii antibody detection test methods. The traditional CF test was compared to an enzyme-linked immunosorbent assay (ELISA) kit manufactured in Europe with samples submitted to National Veterinary Services Laboratories for Q fever serologic testing.
According to the World Organization for Animal Health (OIE) Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, the Q fever CF test is specific, but less sensitive than the ELISA. 6 The ELISA has high sensitivity and good specificity, 6 and eliminates the ambiguous anticomplementary and nonspecific results from CF testing that require the client to resample. In many laboratories, Q fever ELISA has replaced both the indirect florescent antibody test (IFAT) staining and CF as the test of choice because it can easily handle large numbers of sera and is applicable across many species. 6 For the current study, a commercial ELISA kit a employing the phase I/II Nine Mile antigen strain of C. burnetii was used according to manufacturer’s directions. CF tests using the phase II Nine Mile C. burnetii antigen b were performed according to ISO17025 accredited National Veterinary Services Laboratories method for Q fever CF testing. The National Veterinary Services Laboratories method closely follows the OIE described CF method for Q fever testing.
Briefly, for the ELISA kit, the wells of a microtiter plate are coated with phase I and II inactivated C. burnetii antigen by the manufacturer. Serum Q fever antibodies bind to the Q fever antigens on the wells. The plate is washed to remove unbound antibodies. A peroxidase-labeled anti-ruminant immunoglobulin (Ig)G is added to the Q fever antigen–antibody complex, incubated to allow binding, and the unbound reagent is removed by washing. A substrate solution of tetramethylbenzidine is added to the plate for color development. The color development is measured as optical density (OD) with a spectrophotometer at 540 nm. The cutoff for positivity is determined by the %OD of the sample calculated as %OD = 100 × (OD value of test sample – OD value of negative control)/(OD value of positive control – OD value of the negative control). Samples with a %OD value less than 30% are considered negative for Q fever antibodies, those between 30% and 40% are considered suspect for Q fever antibodies, and those greater than 40% are positive. The ELISA kit manufacturer tested 81 goats from 3 separate herds that were experimentally or naturally infected to determine the ELISA kit’s sensitivity of 100% and specificity of 100% when compared to CF test sensitivity of 93% of the naturally infected herd.
Serum tested for Q fever by CF is applied at an initial 1:10 dilution to 3 round-bottom wells and serially diluted in the microtiter plate. Q fever–positive antigen, Q fever–negative control antigen, or buffer control were added to the test serum. Following an overnight, refrigerated incubation with guinea pig origin complement, sensitized sheep red blood cells (RBC) are added to each well on the microtiter plate as an indicator system. The plate is centrifuged to pellet any remaining intact sheep RBC. A positive response for Q fever antibodies is indicated by the RBC pellet in the Q fever–positive antigen wells and lysis of RBC in the buffer and negative antigen control wells. Titers are recorded as the highest serum dilution that binds the guinea pig complement in the antigen well. The CF testing was run according to the laboratory’s general testing schedule, which processed CF submissions twice weekly. After testing by CF, the sera were stored at 4°C, and ELISA testing was conducted within 2 weeks.
Sera submitted were separated into 3 groups composed of 243 sera from cattle, 154 sera from goats, and 271 sera from sheep. No samples submitted were identified as part of an outbreak situation. The majority of sera were negative by ELISA and CF. Many ruminants show no clinical signs when infected with Coxiella, and there were animals in the present study with Coxiella antibodies that were detected by both ELISA and CF. There was a regular sheep producer whose flock had 7 animals positive at 1:80 in the CF test for the prior year, which included the current study. Kappa as a measure of inter-test agreement was calculated for these groups to compare ELISA and CF methods. 4
Table 1 shows the number of samples for each test combination: CF+/ELISA+, CF+/ELISA–, CF–/ELISA+, and CF–/ELISA–. For cattle tested, there were 7 sera positive by both tests, 218 sera negative by both tests, and 5.3% (13/243) with discrepant results at 1:10. The kappa for cattle was poor at 0.38 (95% confidence interval [CI]: 0.10, 0.67). For sheep tested there were 37 sera positive by both tests, 193 sera negative by both tests, and 11% (29/271) with discrepant results at 1:10. The kappa for sheep was satisfactory at 0.56 (95% CI: 0.43, 0.86). For the goats tested there were 13 sera positive by both tests, 137 sera negative by both tests, and 2% (3/154) with discrepant results at 1:10. The kappa for goats was excellent at 0.85 (95% CI: 0.71, 0.99). A visual representation of these results is found in Figure 1. A majority of points are clustered around the CF-negative (<1:10) and CF-positi/ve 1:10 titers, which all tested negative by ELISA in cattle, sheep, and goats. With increasing CF titers, more of the sera are aligned in the positive portion of the graph. Only a few sera in these sample groups exhibited CF titers ≥1:20.
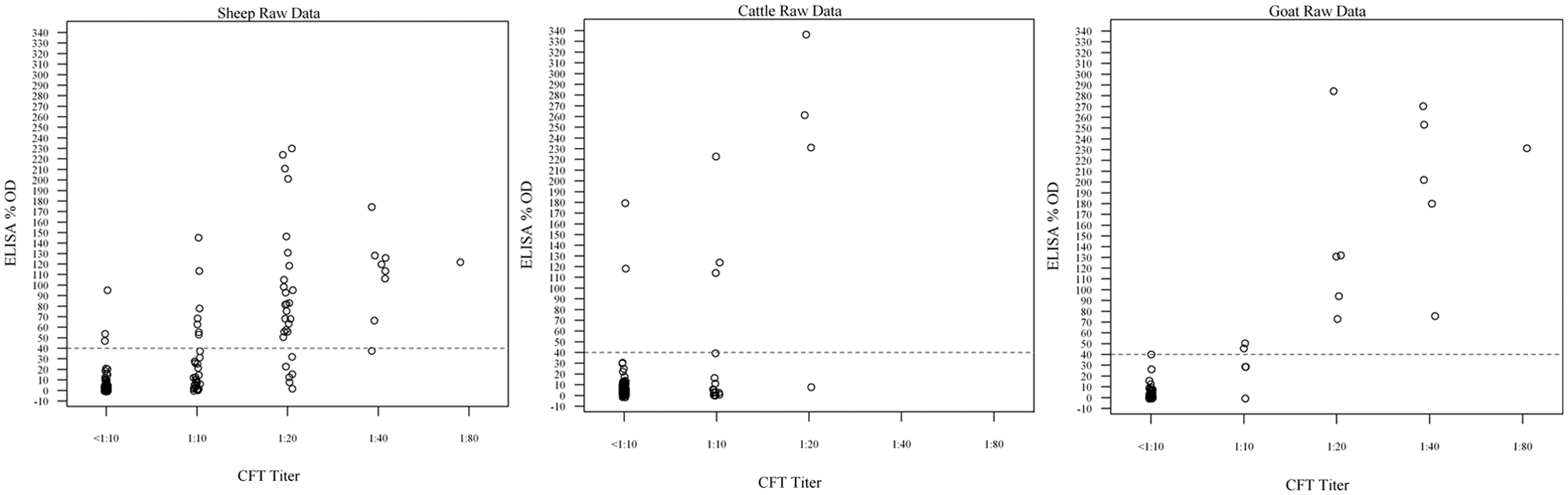
Graph examines the percent optical density (%OD) for each CF test titer. The dotted line divides the graph into 2 sections (negative below and positive above) by enzyme-linked immunosorbent assay (ELISA) results.
Comparison of complement fixation (CF) titers with Q fever enzyme-linked immunosorbent assay (ELISA) results in diagnostic ruminant Q fever sera.*
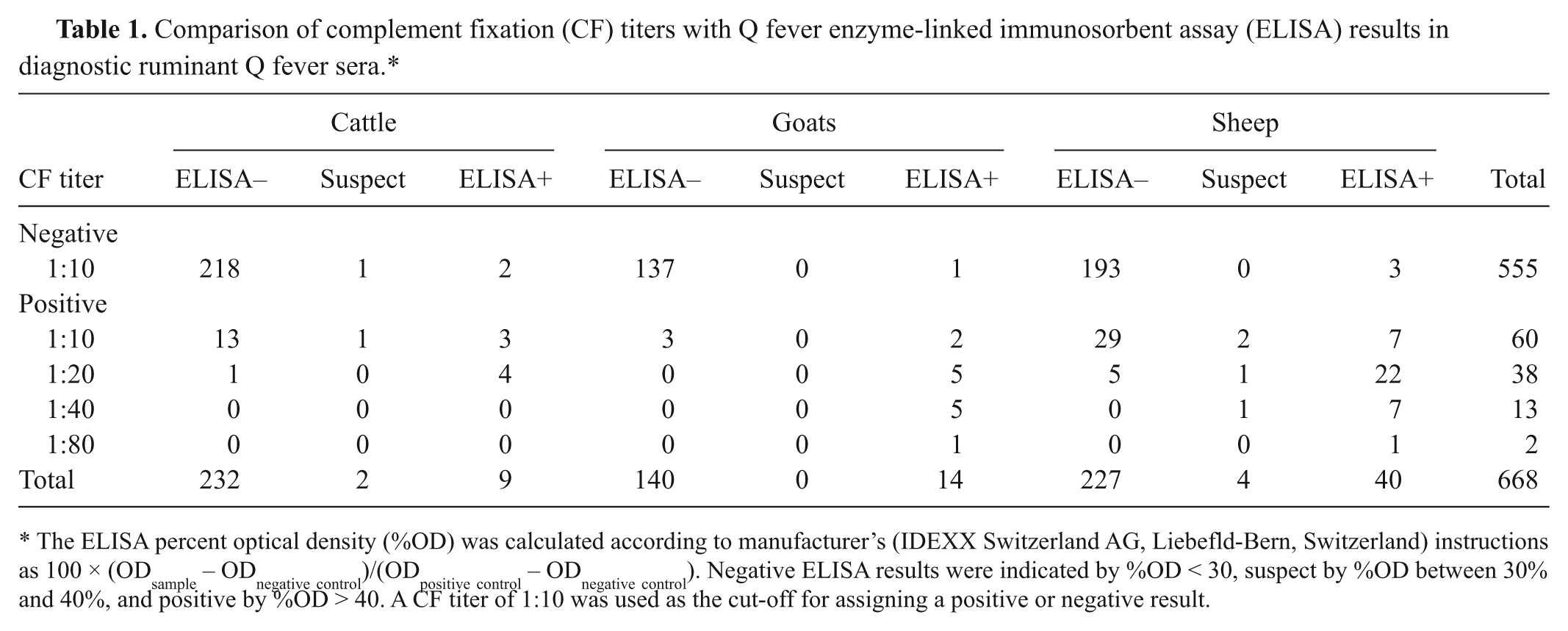
The ELISA percent optical density (%OD) was calculated according to manufacturer’s (IDEXX Switzerland AG, Liebefld-Bern, Switzerland) instructions as 100 × (ODsample – ODnegative control)/(ODpositive control – ODnegative control). Negative ELISA results were indicated by %OD < 30, suspect by %OD between 30% and 40%, and positive by %OD > 40. A CF titer of 1:10 was used as the cut-off for assigning a positive or negative result.
The current study supports earlier work of French researchers using the same ELISA kit but with a different CF antigen (phase II mixture of C. burnetii Henzerling and Nine Mile strains). In the French study, there were 14.8% (31/209) sera positive at 1:10 by CF and negative by ELISA, which were classified as a dubious result. 7 In the National Veterinary Services Laboratories study, there were 5.3% (13/243) of cattle, 2% (3/154) of goats, and 11% (29/271) of sheep that could fit into the same dubious result classification. This was an unfortunately high number, but well documented in both studies.
Several authors have described examples in cattle (1/28) and sheep (1/12) that have coupled low CF titers with ELISA-negative results.2,3 Both studies concluded this observation was due to differences between IgG and IgM reactivity. The studies postulated that elevated IgM antibodies could produce a positive CF result and negative ELISA response. A recent publication examined serum from Bavarian cattle from a Q fever–infected herd with 3 ELISA platforms. 1 The first ELISA was a mixed phase I/II C. burnetii Nine Mile strain antigen, the second was a phase I–specific Nine Mile ELISA antigen, and the third was a phase II–specific Nine Mile ELISA antigen. All 3 kits were produced by the same commercial Q fever ELISA kit manufacturer. These animals were tracked for approximately 2 years, and the multiple permutations of their antibody responses were recorded. Out of 115 sera described as phase I negative/phase II positive on the phase-specific ELISA kits, only 63 were positive on the mixed phase I/II ELISA kit, perhaps indication that the mixed-phase antigen kit lacks sensitivity to phase II response. As phase II antigens are produced early in infection, the antibody response to phase II has been associated with IgM class immunoglobulin.
The present study compared 2 Q fever serologic tests in 668 ruminant sera. Eighty-two percent of the combined sera were negative for Coxiella antibodies on both tests. There were 45 combined sera that had low CF titers with negative ELISA. With the highly skewed negative population, the 45 sera appeared discrepant. The results were somewhat surprising given the expectation that ELISA methods, by nature, amplify detection of antibody–antigen interactions. The veterinary literature has several examples of low CF titer with negative ELISA in populations of cattle, sheep, and goats. Further research should confirm the mechanism of these uniquely discrepant sera.
Footnotes
Acknowledgements
The authors thank David McFarling and Luanne McGonigle for technical assistance with ELISA and CF testing, and Monica Reising for statistical analysis.
a.
IDEXX Switzerland AG, Liebefld-Bern, Switzerland.
b.
Institut Virion\Serion GmbH, Wurburg, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
