Abstract
A 15-year-old gelding presented with a progressive lameness of the left forelimb of 2.5 months duration. Clinically, a dilation of the deep flexor tendon sheath with a firm elastic consistency and a pronounced tenderness was noted. Ultrasonically, a marked swelling of the flexor tendon sheath with an irregular density of the mesotendineum was observed. The white, firm material forming a nodular distension of the flexor tendon sheath with a diameter of approximately 1 cm was excised and sent for histopathological examination. Biopsies of the deep flexor tendon and corresponding tendon sheath were sent for histopathological evaluation. Histologically, the mass consisted of clefts and numerous anastomosing vascular channels extending between the collagen fibers of the deep flexor tendon. These capillary-like spaces were lined by neoplastic cells that were flattened to polygonal and contained few erythrocytes. There was 0 to 1 mitotic figure per 10 high power fields (400×). Immunohistochemically, the neoplastic cells stained positive for vimentin and factor VIII–related antigen. Adjacent to the neoplastic endothelial cells located pericytes expressed α-smooth muscle actin antigen. Based on the histopathological and immunohistochemical features, synovial hemangioma was diagnosed. One year after surgery, the horse has shown no lameness.
Synovial hemangiomas are uncommon benign lesions that have been described in human beings, dogs, and horses.1,2,5,12 The lesions have been reported in tendon sheaths, mainly in young horses of different breeds between 1 and 3 years of age. 12 In human beings and dogs, synovial hemangiomas are most commonly reported in the stifle joint.1,5,10 In the literature, it is unclear whether these lesions represent true neoplasms or vascular malformations (hamartomas), but the occurrence in young horses favors the classification as vascular hamartomas.2,11 The lesions are characterized by slow growth and, in some cases, infiltration of adjacent structures. 12 Histologically, the lesion consists of abnormal vascular structures varying from cavernous spaces to disorderly arranged vascular channels lined by plump endothelial cells, but showing no convincing features of malignancy. 12 The present report describes a 15-year-old gelding with a synovial hemangioma in the deep flexor tendon of the left forelimb.
A 15-year-old gelding presented with a progressive lameness of the left forelimb of 2.5 months duration. Clinically, a marked dilation of the deep flexor tendon sheath was noted proximal to the palmar annular ligament. At palpation, the tendon sheath displayed a firm elastic consistency and a pronounced tenderness. Ultrasonically, a marked swelling of the flexor tendon sheath with an irregular density of the mesotendineum was observed (Fig. 1). During surgery, a nodular firm white mass, measuring approximately 1 cm in diameter, was found in the mesotendineum and the flexor tendon sheath. Additionally, the deep flexor tendon was frayed (Fig. 2).
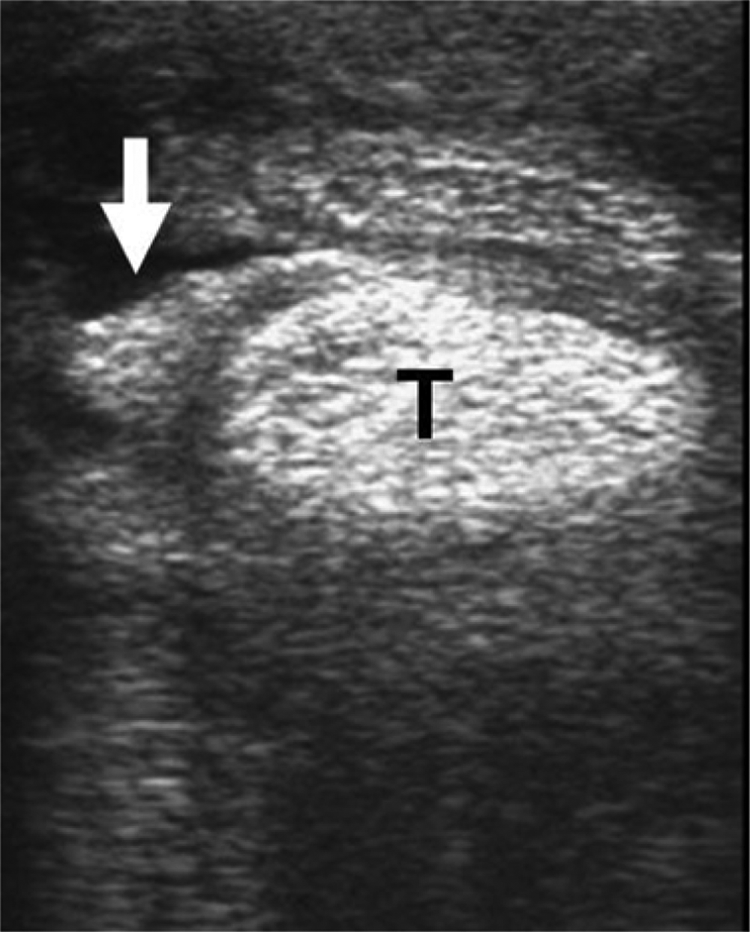
Fifteen-year-old gelding. Ultrasonic investigation of the left forelimb revealed a marked thickening of the flexor tendon sheath with alterations of the outline and density (arrow) next to the superficial flexor tendon and impairment of the deep flexor tendon (T).
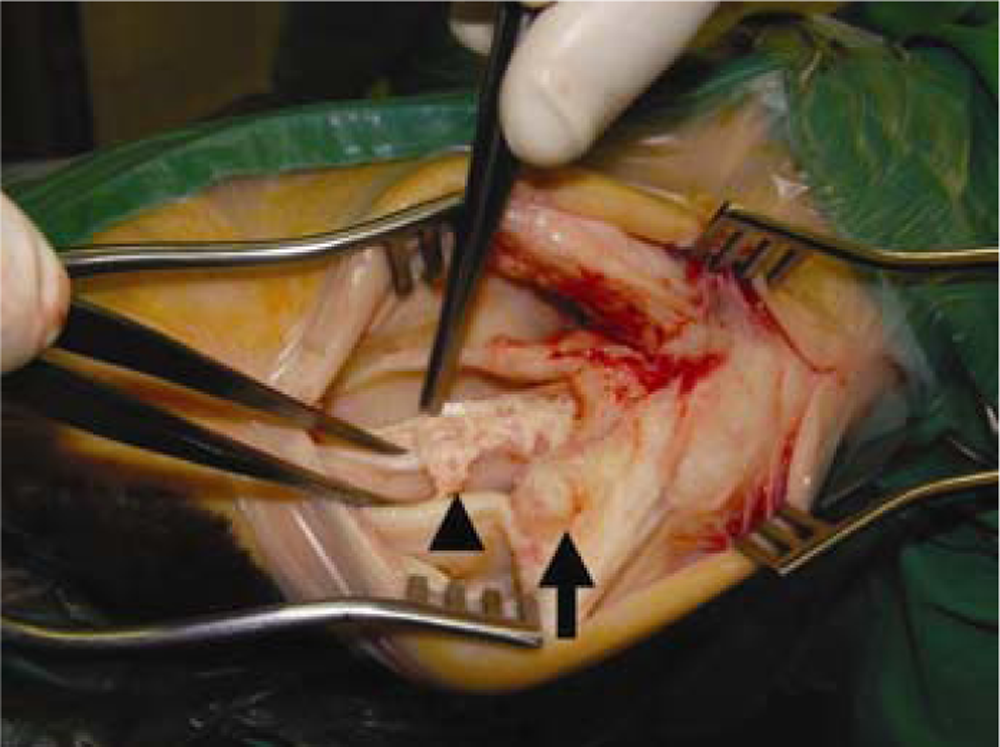
Fifteen-year-old gelding. During surgery, a mass at the flexor tendon sheath of the left forelimb (arrow) and an irregular fraying of the deep flexor tendon (arrowhead) were found.
The mass was excised surgically from the deep flexor tendon sheath of the left forelimb and, together with biopsies from the frayed deep flexor tendon, was sent for histopathological examination. The tissue was fixed in 10% nonbuffered formalin and embedded in paraffin wax. Tissue sections were stained with hematoxylin and eosin. Additional sections were used for immunohistochemistry by applying the avidin–biotin–peroxidase complex method using primary antibodies directed against vimentin, a cytokeratin, b α-smooth muscle actin, c factor VIII–related antigen, d and PROX-1 (prospero homeobox protein 1) e as previously described.7,9,14 Briefly, sections were dewaxed, and antigens were demasked by Triton treatment (vimentin; 0.25%, 30 min), Pronase E treatment (factor VIII–related antigen; 20 min), Pronase E and Tween treatment (cytokeratin; 20 min), or citrate buffer and pepsin treatment (PROX-1; 20 min and 10 min, respectively). For α-smooth muscle actin, no pretreatment was used. For each antibody, negative control sections with replacement of the specific antibodies by ascitic fluid from nonimmunized BALB/c mice (monoclonal antibodies) and serum from nonimmunized rabbits (polyclonal antibodies), respectively, were prepared. Positive controls included fibroblasts (vimentin), epidermis (cytokeratin), vascular smooth muscle cells (α-smooth muscle actin), endothelium (factor VIII–related antigen), and lymph node (PROX-1). For electron microscopy, formalin-fixed tissue was postfixed in 5% glutaraldehyde and 1% osmium acid and embedded in epoxy resin. Thin sections were stained with lead citrate and uranyl acetate.
Histologic evaluation of the biopsies revealed a moderately cell-rich, unencapsulated mass that multifocally extended between the collagen fibers of the deep flexor tendon and reached resection margins in some areas. The neoplastic cells lined clefts and numerous anastomosing, occasionally slightly dilated, vascular channels. These capillary-like spaces demonstrated a partially undirected growth with irregular branching within the mass, and the spaces were embedded in a moderate amount of a collagen-rich stroma (Fig. 3). Tumor cells were flattened to polygonal and measured approximately 10 µm × 5 µm. The cells had distinct cell borders, a nuclear-to-cytoplasmic ratio of approximately 1:0.5 and a scant amount of a homogeneous, eosinophilic cytoplasm. The centrally located oval-to-elongated nucleus showed a finely stippled chromatin pattern. Each nucleus contained 1–2 basophilic nucleoli. There was no anisocytosis or anisokaryosis. There was 1 mitotic figure per 20 high power fields (400×). Furthermore, there was a moderate, chronic, villous synovitis with moderate fibrosis of the tendon sheath; multifocal, mild, acute hemorrhages; and hemosiderin-laden macrophages in the flexor tendon. Immunohistochemical examination of the tumor cells revealed immunoreactivity for vimentin (Fig. 4) and factor VIII–related antigen (Fig. 5). Adjacent to the neoplastic endothelial cells located pericytes expressed α-smooth muscle actin antigen (Fig. 6). There was no immunoreactivity for cytokeratin or PROX-1. In addition to the tumor cells, stromal fibroblasts displayed immunoreactivity for vimentin. Ultrastructural analysis showed empty clefts lined by neoplastic cells with a basement membrane between the tumor cells and the adjacent stroma (Fig. 7).
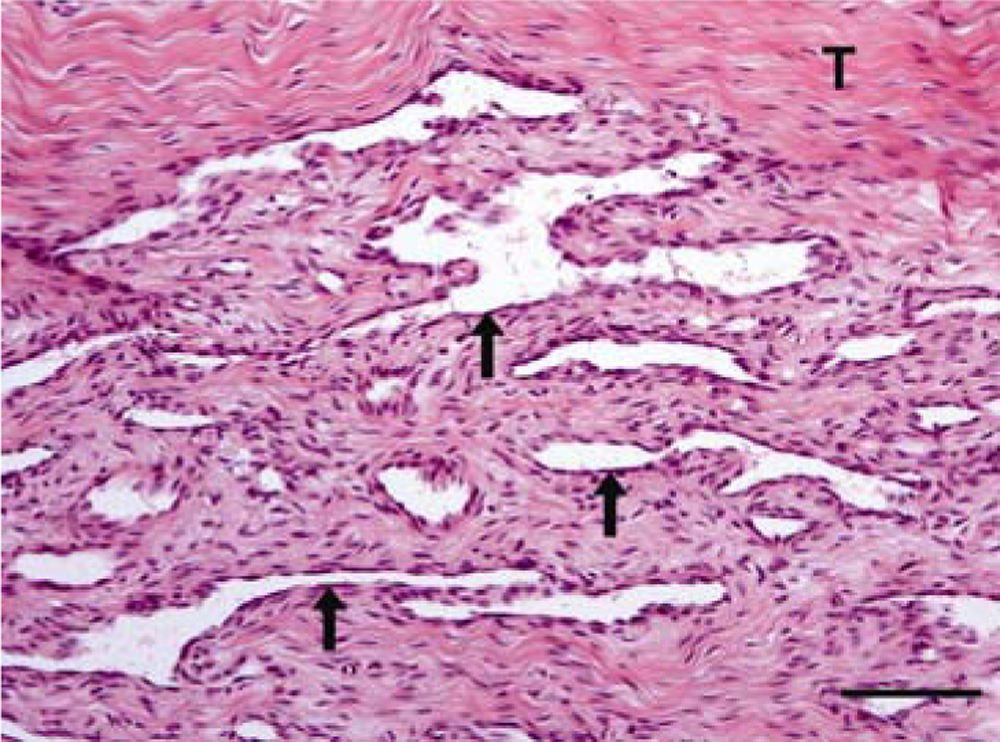
Fifteen-year-old gelding. Synovial hemangioma extending between the collagen fibers of the deep flexor tendon (T), forming clefts and vascular channels with a partially undirected growth (arrows). Hematoxylin and eosin. Bar = 100 µm.
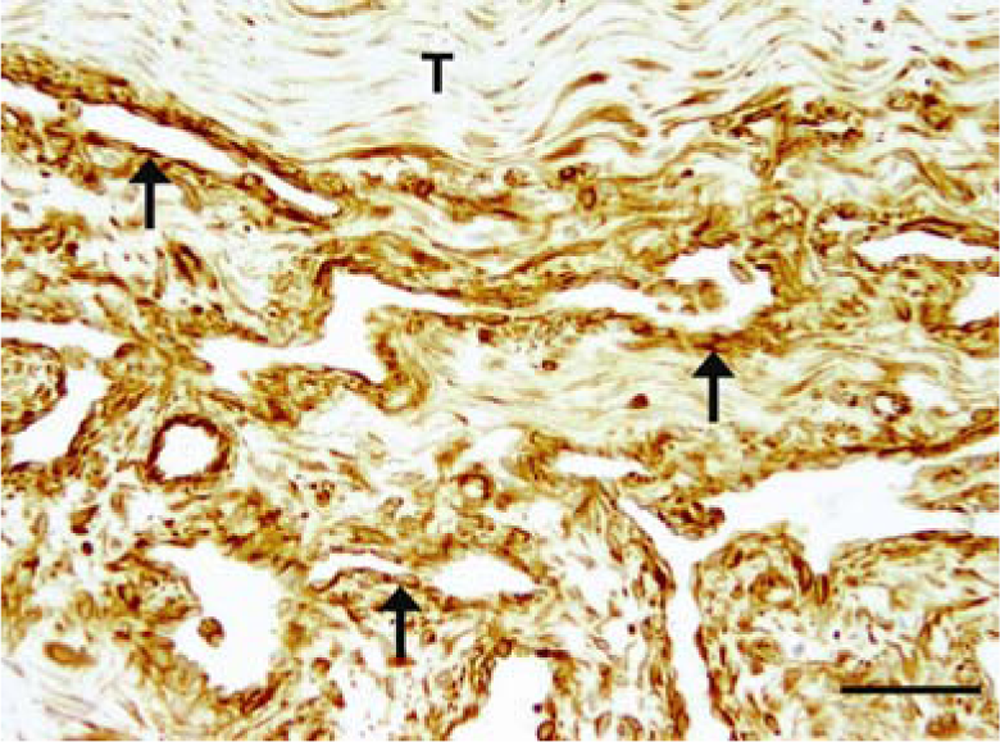
Fifteen-year-old gelding. Synovial hemangioma of the deep flexor tendon. Immunohistochemically, the tumor cells strongly expressed vimentin (arrows). Additionally, stromal fibroblasts displayed vimentin immunoreactivity. T = deep flexor tendon. Immunohistochemistry, avidin–biotin–peroxidase complex method. Bar = 100 µm.
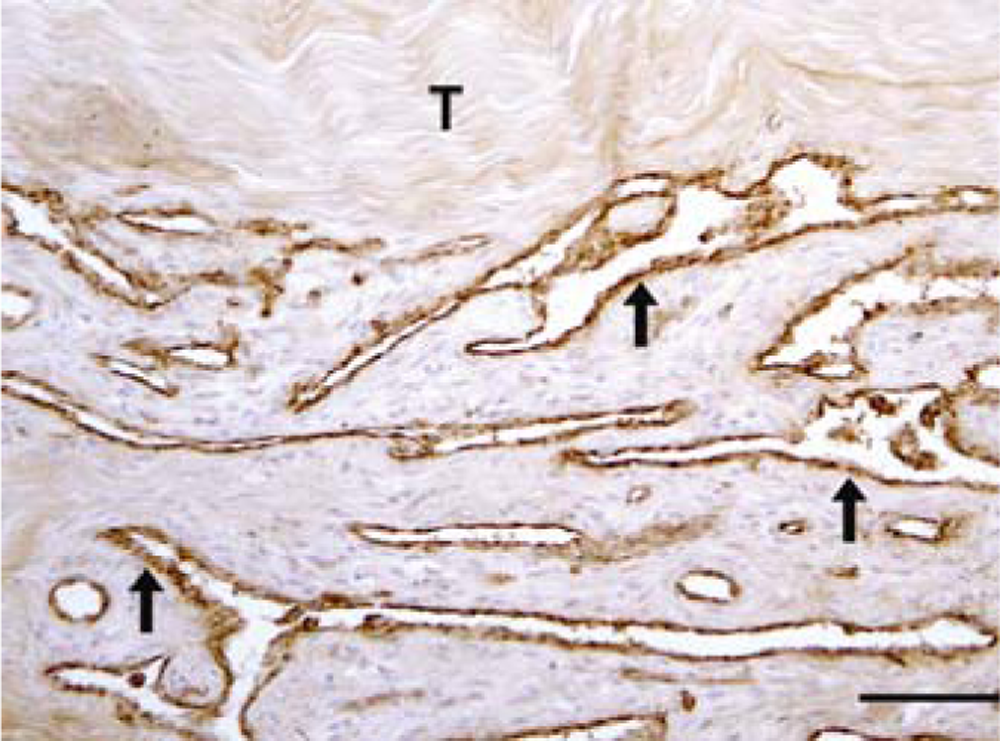
Fifteen-year-old gelding. Synovial hemangioma of the deep flexor tendon. Immunohistochemically, the tumor cells strongly express factor VIII–related antigen (arrows). T = deep flexor tendon. Immunohistochemistry, avidin–biotin–peroxidase complex method. Bar = 100 µm.
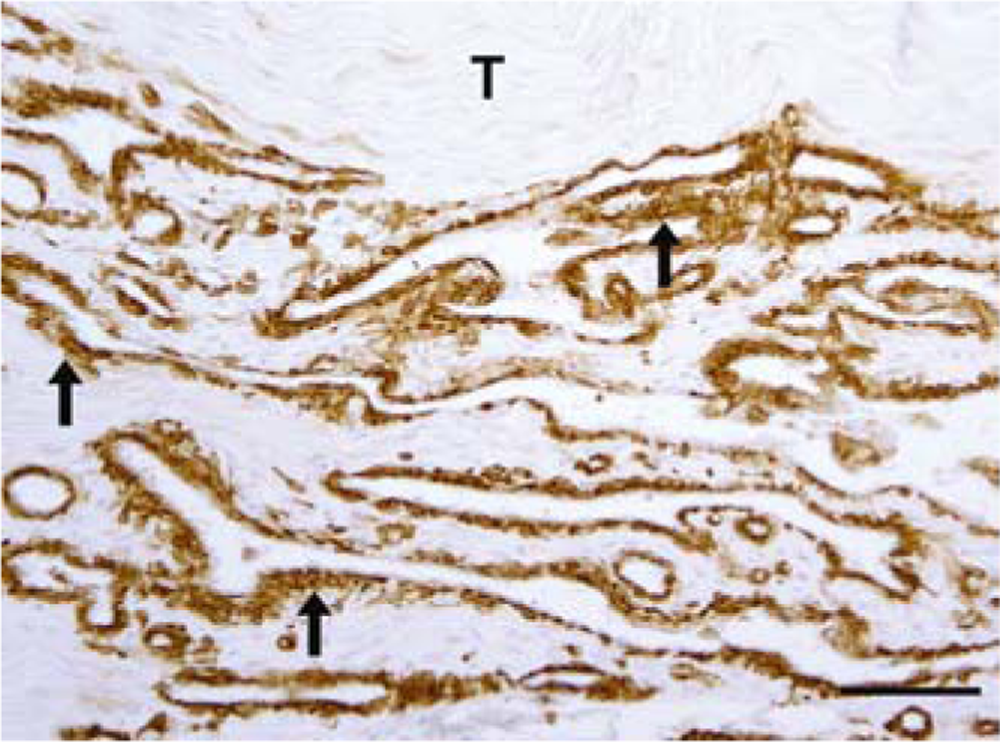
Fifteen-year-old gelding. Synovial hemangioma of the deep flexor tendon. Immunohistochemically, adjacent to the neoplastic endothelial cells located pericytes strongly expressed α-smooth muscle actin antigen (arrows). T = deep flexor tendon. Immunohistochemistry, avidin–biotin–peroxidase complex method. Bar = 100 µm.
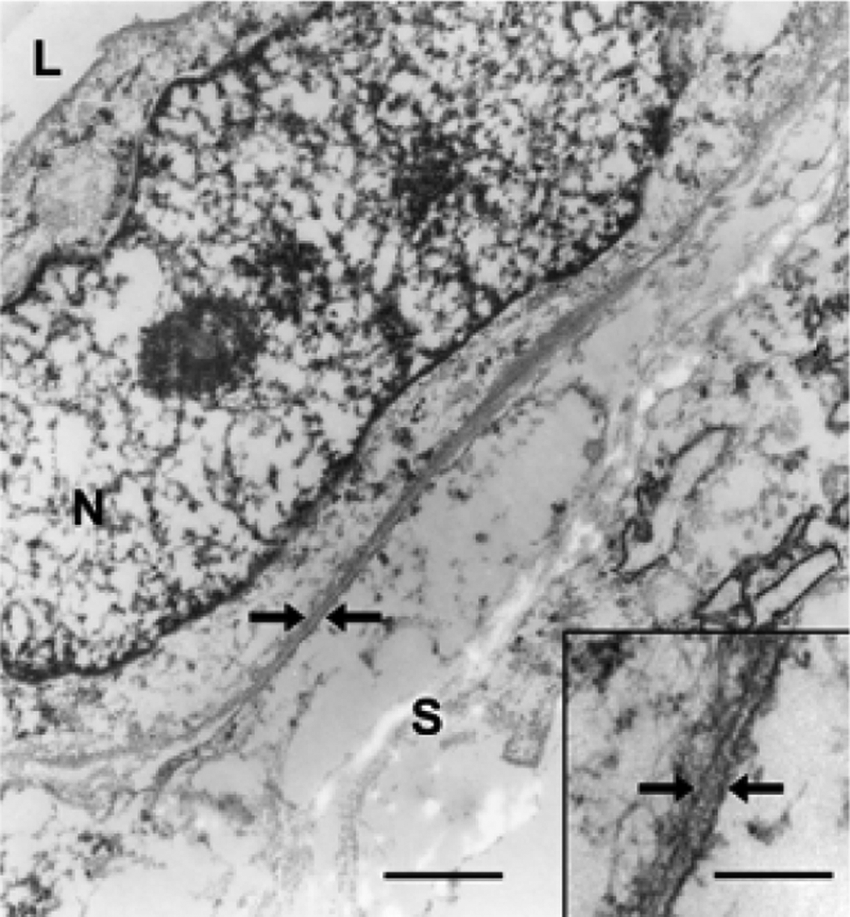
Fifteen-year-old gelding. Synovial hemangioma of the deep flexor tendon. Ultrastructurally, the tumor cells are separated from the adjacent stroma by a basement membrane (arrows). L = lumen; N = nucleus; S = stroma. Transmission electron microscopy. Bar = 0.72 µm. Inset: higher magnification of the basement membrane (arrows). Transmission electron microscopy. Bar = 0.17 µm.
On the basis of the histopathological, immunohistochemical, and ultrastructural features, a synovial hemangioma was diagnosed. One year after surgery, the horse has shown no lameness and has continued to train regularly.
Synovial hemangiomas can arise from any synovium-lined surface; therefore, the lesions may be found along the course of tendons or in joint spaces. The origin from synovium in the case of tendon sheaths is only assumed because they may also involve superficial structures, and confinement by synovium is often not apparent. 15 Presenting symptoms of a synovial hemangioma can be recurrent and vary from a nonpainful mass, localized pain alone, or pain and swelling, depending on their size, location, and the alterations caused in neighboring structures.3,4,13 Despite their benign nature, synovial hemangiomas are not sharply marginated and have no capsule. 3 Their shape usually follows that of the anatomic structure where they are located but frequently extends insidiously across normal tissue barriers. 3 Synovial hemangiomas often become difficult to excise because they may spread throughout the entire synovium and infiltrate the surrounding muscles and fat, but often they demonstrate no significant displacement of surrounding skeletal muscles, fat, ligaments, and tendons. 3 Bone invasion is only rarely observed. 6 Regardless of their infiltrative growth, these tumors revealed no cytological features of malignancy like pleomorphism and high mitotic rate. Furthermore, they form no metastases. One other example of such tumors, in addition to synovial hemangiomas, is the infiltrative lipoma, which displays a marked infiltrative growth in many cases but exhibits histological features of mature adipocytes and does not metastasize. 8
In synovial hemangiomas, hemorrhages into the tendon sheaths are apparently due to the trauma of locomotion. 12 As in the present case with intratendinal invasion, surgical removal is often difficult, and hemostasis displays a challenge due to the location. 12 Following complete excision, recurrence is unlikely. 12 There is controversy in the literature whether synovial hemangiomas represent true neoplasms or are vascular malformations (hamartomas). The occurrence in young horses favors the classification as vascular hamartomas.2,11,15 In horses, synovial hemangiomas in tendon sheaths have been described in animals between 1 and 3 years. However, in 2 cases they remain undetected until several years of age. 2 Due to the advanced age of the animal in the current report, with no known history of lameness or swelling of the left forelimb, a hamartoma seems unlikely. Hemangiosarcoma was considered as a differential diagnosis, but histology revealed no signs of malignancy. Additionally, lymphangioma and lymphangiosarcoma were taken into account as differential diagnoses. However, a lymphatic origin of the tumor cells could be excluded due to the lacking PROX-1 antigen expression and the basement membrane separating the tumor cells from the adjacent stroma. The cause of synovial hemangiomas remains undetermined, but in human medicine it is considered that, in some cases, synovial hemangiomas could present posttraumatic lesions but is not interpreted as a common underlying pathogenesis. 4 In human medicine, synovitis secondary to chronic irritation due to hemorrhages caused by the hemangioma is described. 15 In the present case, it could not be determined if the synovitis was hemangioma associated or an unrelated lesion.
Footnotes
Acknowledgements
The authors would like to thank Mrs. B. Buck, Mrs. P. Grünig, and Mrs. K. Rohn for excellent technical support.
a.
Mouse anti-vimentin, clone V9, monoclonal, DAKO Deutschland GmbH, Hamburg, Germany.
b.
Mouse anti-human, clone AE1/AE3, DAKO Deutschland GmbH, Hamburg, Germany.
c.
Mouse anti-human, clone 1A4, monoclonal, DAKO Deutschland GmbH, Hamburg, Germany.
d.
Mouse anti-human, polyclonal, DAKO Deutschland GmbH, Hamburg, Germany.
e.
Rabbit anti-human, polyclonal, RELIATech, Wolfenbüttel, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
