Abstract
A 15-year-old Haflinger gelding presented with a mass in the left nictitating membrane. Two biopsies and the excised nictitating membrane were taken at different time points as a result of reoccurrence of the mass and submitted for histopathologic evaluation. The horse was euthanized as a result of poor prognosis following the reoccurrence of the mass after surgical removal. Histologically, the mass consisted of dilated, thin-walled vascular clefts and channels, lined by flattened to cuboidal endothelial cells with moderate cellular pleomorphism. There was up to 1 mitotic figure per high power field. The channels were empty or contained few erythrocytes. In the collagen-rich stroma, few lymphocytes, focal follicular lymphoid aggregations, and marked lymphangiectasia were observed. Immunohistochemically, the neoplastic cells stained positive for vimentin and partially positive for factor VIII-related antigen. Ultrastructural analysis revealed discontinuous endothelial lining vascular channels that partially lacked a basal membrane. Based on the histopathologic, immunohistochemical, and ultrastructural features lymphangiosarcoma was diagnosed.
Ocular tumors of vascular origin have been rarely described in horses. 1,4,9,13 Such tumors include hemangiomas, hemangiosarcomas, and lymphangiosarcomas. 1,13 In cases where the cell of origin remains undetermined, they are classified as angiosarcomas. 4,9
Lymphangiosarcoma is an extremely rare tumor originating from the lymphatic endothelium. 5 It occurs in various species and is most frequently described in dogs 16 and cats. 6 Additionally, single cases are reported in horses 7,12 and cattle. 11 In domestic animals, lymphangiosarcomas tend to be localized in the subcutis along the ventral midline and limbs. 5 Grossly, these neoplasms are poorly demarcated, soft to spongy masses, containing clear, serous fluid on cut surface. 3 Histologically, lymphangiosarcomas differ little from their benign counterpart, except for their increased cellular pleomorphism. Characteristic features include clefts and channels lined with cells displaying more rounded nuclei with hyperchromatism and a slightly increased mitotic activity. 5 The underlying cause triggering the development of lymphangiosarcomas remains undetermined in most cases. However, chronic lymphedema has been shown to play an important role in humans. 15
A 15-year-old Haflinger gelding showed a swelling of the left nictitating membrane of 4 weeks' duration. A biopsy of this cavernous, fluid-filled mass was taken and sent for histopathologic examination. Two weeks later, a total extirpation of the nictitating membrane was performed. Postoperatively, the horse developed a progressive periocular swelling and a sero-sanguinous nasal discharge over the following 5 months. Therefore, a second biopsy was taken. As a result of poor prognosis, the horse was euthanized but was not available for postmortem examination.
For histopathologic examination, biopsies were fixed in 10% neutral nonbuffered formalin and embedded in paraffin wax. Tissue sections were stained with hematoxylin and eosin, periodic acid Schiff (PAS), and elastic van Gieson (EVG). Additional sections were used for immunohistochemistry by applying the avidin-biotin complex method using primary antibodies directed against vimentin a and factor VIII-related antigen b as described. 8 Briefly, sections were dewaxed, and antigens were demasked by Triton treatment (vimentin; 0.25%, 30 min) and Pronase E treatment (factor VHI-related antigen; 20 min). 8 For electron microscopy, formalin-fixed tissue was postfixed in 5% glutaraldehyde and 1% osmium acid, and embedded in Epon. Thin sections were stained with lead citrate and uranyl acetate.
Histologic examination of both the biopsies and the extirpated nictitating membrane revealed a densely cellular, poorly demarcated, diffusely infiltrating, unencapsulated mass, which in some areas extended to the cut borders. The tumor cells were arranged in intimate association to collagen bundles, which stained red with EVG. The neoplastic cells formed clefts and numerous, anastomosing, in some areas markedly dilated, vascular channels. An incontinuity and multifocal absence of the basal lamina were observed in the PAS stain. Tumor cells were flattened to polygonal, measuring 10 to 25 μm in diameter. They had indistinct cell borders and a moderate amount of a homogeneous, eosinophilic cytoplasm. The eccentrically located, round to oval nucleus showed a finely stippled chromatin pattern with hyperchromasia in single nuclei. Each nucleus contained 1 to 2 basophilic nucleoli. There was a moderate cellular pleomorphism present (Fig. 1) and up to 1 mitotic figure per high power field detectable. The periphery of the neoplasm showed a moderate, diffuse infiltration of lymphocytes and macrophages with lymph follicle-like lymphoid aggregates in the peritumoral area. Additionally, there was a marked lymphangiectasia, characterized by flattened endothelia, in the nontumorous area of the nictitating membrane.
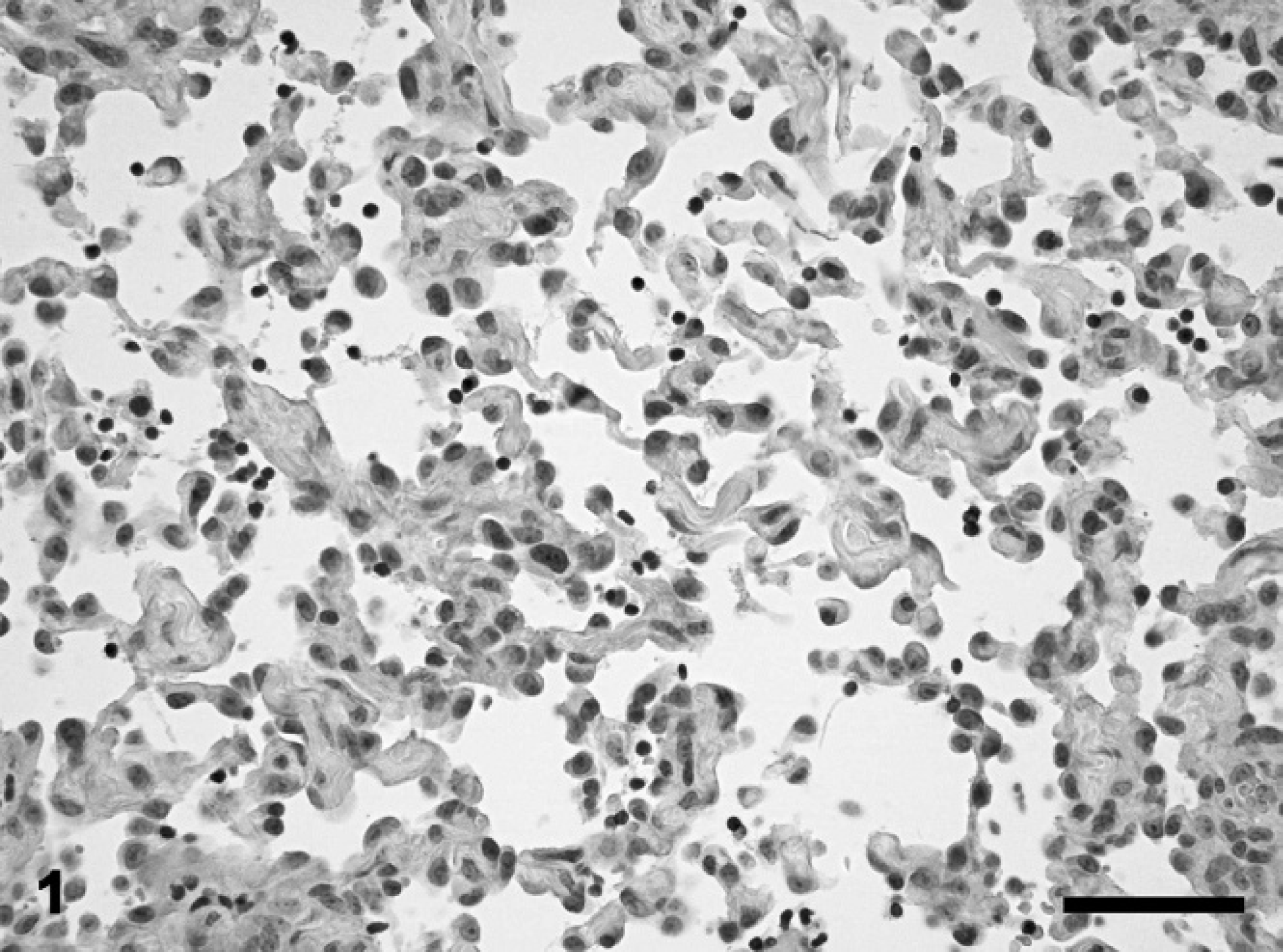
Nictitating membrane, equine lymphangiosarcoma. Tumor cells with a moderate cellular pleomorphism forming clefts and anastomosing vascular channels. Hematoxylin and eosin. Bar = 50 μm.
Immunohistochemical examination of the tumor cells revealed immunoreactivity for vimentin. Factor VIII-related antigen could be detected in variable intensities in approximately 30% of the neoplastic cells.
Ultrastructural analysis showed empty clefts lined by neoplastic cells without a basement membrane between the tumor cells and the adjacent stroma (Fig. 2). The cells consisted of a round to oval nucleus, a paranuclear zone rich in intermediate filaments, and slightly eccentrically placed mitochondria (Fig. 3). Based on the histopathologic, immunohistochemical, and ultrastructural features a lymphangiosarcoma was diagnosed.
Lymphangiosarcoma is a rare occurring neoplasm in domestic animals. 3 In horses, so far few cases of lymphangioma and 2 cases of lymphangiosarcoma have been described. 2,7,10,12 One case of lymphangiosarcoma is reported in the visceral trunk with extension through the diaphragm into the mediastinum. 7 A metastatic lymphangiosarcoma affecting variable lymph nodes, spleen, lungs, and kidneys has been observed in another horse. 12
In humans and in dogs, most cases of lymphangiosarcoma had a history of prolonged lymphedema at the tumor site, suggesting that the constant protein-rich interstitial fluid might be a stimulus for neoplastic transformation. 15,16 In the present case there was no known history of chronic lymphedema. The differentiation between lymphangiomas and lymphangio sarcomas can be critical as they only differ in their cellular pleomorphism. 5 Another challenge is the discrimination between hemangiosarcomas and lymphan-giosarcomas, which is mainly based on the apparent close apposition of neoplastic cells to collagen bundles and the lack of blood cells in the channels and clefts in the latter. 3 As in the present case, the expression of factor VHI-related antigen and vimentin has been reported in lymphangiosarcomas of various species, including the horse. 7,12 Similarly, as in the present case, a lack of the basement membrane between the neoplastic cells and the underlying collagen bundles as determined by electron microscopy represent a key feature to confirm the lymphatic histogenesis. 7,14 In conclusion, this report describes for the first time a lymphangiosarcoma in the nictitating membrane of a horse.
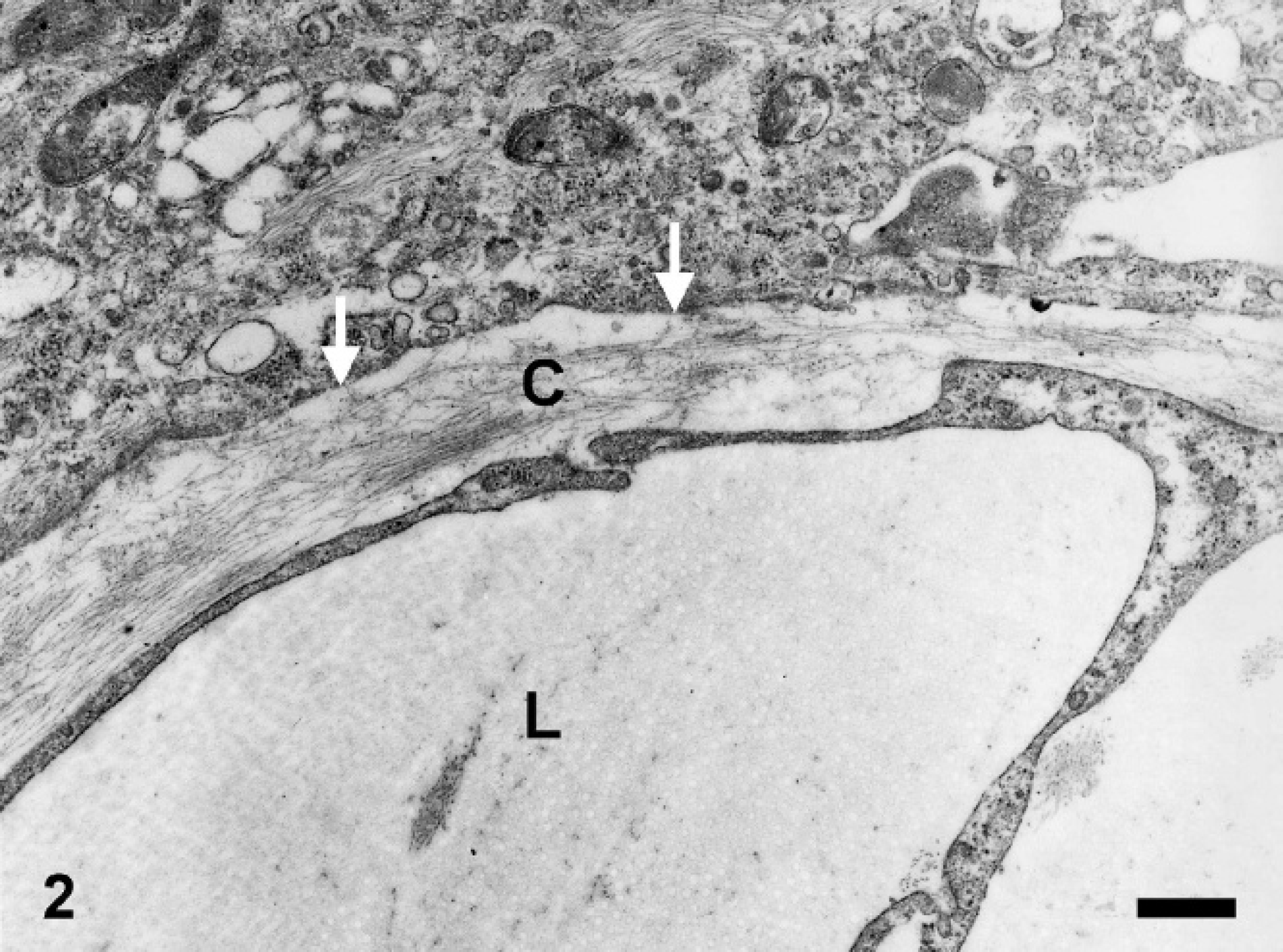
Nictitating membrane, equine lymphangiosarcoma. Clefts (Lumen = L) lined by neoplastic cells lacking a basement membrane (arrows) between the tumor cells and the adjacent stroma. The stroma consisted mainly of collagen fibers (C). Electron microscopy. Bar = 0.5 μm.
Acknowledgements. The authors would like to thank Mrs. P. Grünig, Mrs. B. Buck, and Mrs. K. Rohn for excellent technical assistance, and Mrs. B. Behrens for photographic support.
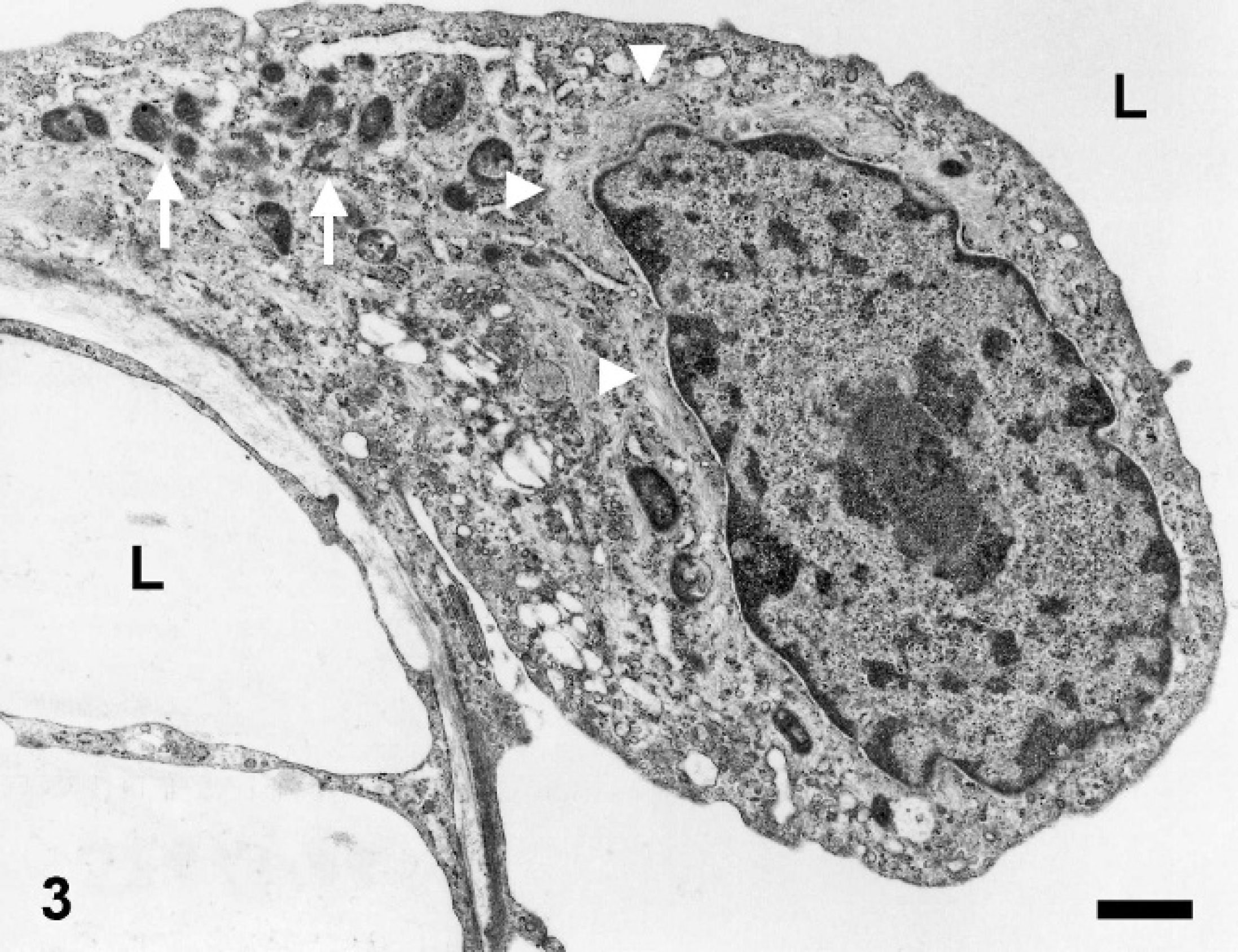
Nictitating membrane, equine lymphangiosarcoma. Tumor cells lining clefts (Lumen = L) showed characteristics of lymphatic endothelial cells including a round to oval nucleus, a paranuclear zone rich in intermediate filaments (arrowheads), and a slight dislocation of mitochondria to the periphery of the cell (arrows). Electron microscopy. Bar = 1 μm.
Footnotes
a.
Mouse-anti-vimentin, monoclonal, DAKO cytomation, Glostrup, Denmark.
b.
Mouse-anti-human, polyclonal, DAKO cytomation, Glostrup, Denmark.
