Abstract
Lead (Pb) poisoning remains a common cause of morbidity in dairy and beef cattle. Although Pb toxicosis is typically diagnosed in cattle with clinical signs of acute or subacute Pb poisoning, it has been hypothesized that subclinical chronic exposure of cattle to Pb, which often goes undiagnosed, poses more of a risk to the human consumer. There is not adequate information on Pb kinetics to determine when or if Pb-exposed cattle can safely enter the food chain. The objectives of the current study were to determine whether subclinical elevations in blood Pb (bPb) were present in cattle from herds where 1 or more individuals had clinical Pb poisoning and to determine the half-life (t1/2) of bPb in Pb-exposed cattle. Samples of blood were collected and analyzed for Pb from 126 cattle from 9 farms. Blood lead concentrations ranged from below the detection limit (2.50 µg/dl) to 423.0 µg/dl. Only 11 of the 94 cattle with detectable bPb had clinical signs such as diarrhea, blindness, bruxism, or seizures. When possible, cattle with detectable bPb had serial samples taken. The mean t1/2 calculated from 44 serially sampled cattle was 135 days (standard deviation: 125 days, range: 3–577 days). A source of Pb on the farm was determined for all but one herd.
Lead poisoning is the most common metal toxicosis in domestic animals; the domestic species most commonly affected is cattle.5,7,18 Lead poisoning in cattle usually occurs on pasture due to their indiscriminate eating habits and natural curiosity. However, Pb can also contaminate batches of feed and cause toxicosis in confined cattle.7,9 Lead poisoning in cattle is associated with economic losses due to mortality and treatment costs. 20 Also, as production animals, Pb contamination of either beef or dairy cattle is associated with risk to the human consumer.16,20
Lead is ubiquitous in the environment and has no known biological role. 18 Some environmental Pb is from geochemical sources, but most comes from anthropogenic sources (e.g., mining, smelting, battery production and recycling, and other commercial uses). Lead paint and Pb-acid automotive batteries are the most common sources of Pb for cattle. Paint and batteries are usually point sources of Pb (with short-term exposure and rapid onset of clinical signs likely and leading to either death or culling or appropriate diagnostics and treatment). 16 However, Pb paint removal can cause widespread contamination of soil and forage, and batteries have been accidentally baled into feed; these sources could cause chronic exposure to Pb in many animals.3,14,16 Other potential sources of lead are hardware cloth, wire and solder, linoleum, and lubricants. The agricultural insecticide lead arsenate is no longer available in the United States. 10
Though less common,2,7,12 chronic subclinical Pb exposure is likely to remain undiagnosed and therefore to be important to the human consumer.12,21 Livestock can be a source of Pb contamination for the human food chain, although clinical Pb poisoning from ingestion of contaminated animal products is unlikely. 16 Subclinical Pb exposure in children is associated with intellectual and cognitive deficits, aggressive behavior, renal impairment, hypertension, and cataracts.11,16 Miscarriages, stillbirths, and other fertility problems are also associated with human subclinical Pb poisoning. 11 However, even in herds with a history of exposure, cattle without clinical signs of Pb poisoning are not usually tested for Pb. A previous study of herds with clinical cases of Pb poisoning found that 7–40% of asymptomatic cattle had blood Pb (bPb) concentrations ≥10 µg/dl. 20
Elimination of Pb is different in ruminants compared to monogastrics due in part to the retention of Pb particles in the reticulum and rumen. 15 Blood Pb concentrations at any given time depend on absorption of Pb remaining in the gastrointestinal tract and mobilization from bone. 15 There is some excretion of Pb into milk, which is another possible mode of entrance into the human food chain. 15 Values for bPb half-life (t1/2) in the literature vary from 6 to 19 days (for experimental cattle given intravenous lead acetate) to between 48 and 2,507 days (for cattle exposed in the field).15,19 The differences in t1/2 in cattle exposed in the field did not depend on the source of lead, sex, or age of the animal, although the t1/2 was shorter in lactating cattle than non-lactating cattle.9,15 Elevated bPb has been detected in Pb-exposed cattle for up to 2 years. 15
Testing of Pb-exposed cattle in the absence of clinical signs is not routine. One objective of the current study was to determine whether there were elevated bPb concentrations in asymptomatic cattle on farms with a recent history of Pb poisoning in 1 or more herd mates. It was hypothesize in the present study that, in herds where individual animals were diagnosed with clinical Pb poisoning, many asymptomatic cattle also have elevated bPb concentrations. The other objective of the current study was to estimate the t1/2 of Pb in the blood of production cattle (including both dairy and beef cattle) exposed to various (unintended) sources of Pb in the field. A narrow range of estimated t1/2 would be useful in determining when and whether it is safe to market products such as meat and milk for human consumption, and in determining efficient Pb-testing protocols for exposed herds. Currently, in many U.S. states, serial bPb testing is used primarily on clinically affected cattle by some producers to determine the safety of exposed cattle to the food chain. However, testing is not a requirement in all states, is not typically done on asymptomatic cattle, and the disposition of many Pb-exposed cattle is unknown.
Nine beef and dairy cattle herds (from 4 U.S. states) with histories of accidental Pb exposure were identified for the present study. A brief description of each herd is given in Table 1. Diagnosis of Pb poisoning was based on testing at the New York State Animal Health Diagnostic Center or another veterinary diagnostic laboratory in the United States. Sample collection began in 2005 for 2 farms. The owner of one farm agreed to participate in serial sample collection under conditions of anonymity. Serial testing was required for the other farm by state officials. Animal owners and veterinarians from 6 farms recruited after October 2006 agreed to participate in the study under conditions of anonymity, and owners signed consent forms. Serial collection data were submitted to a laboratory for evaluation from one other farm in 2009 and 2010, and its results were incorporated into the study.
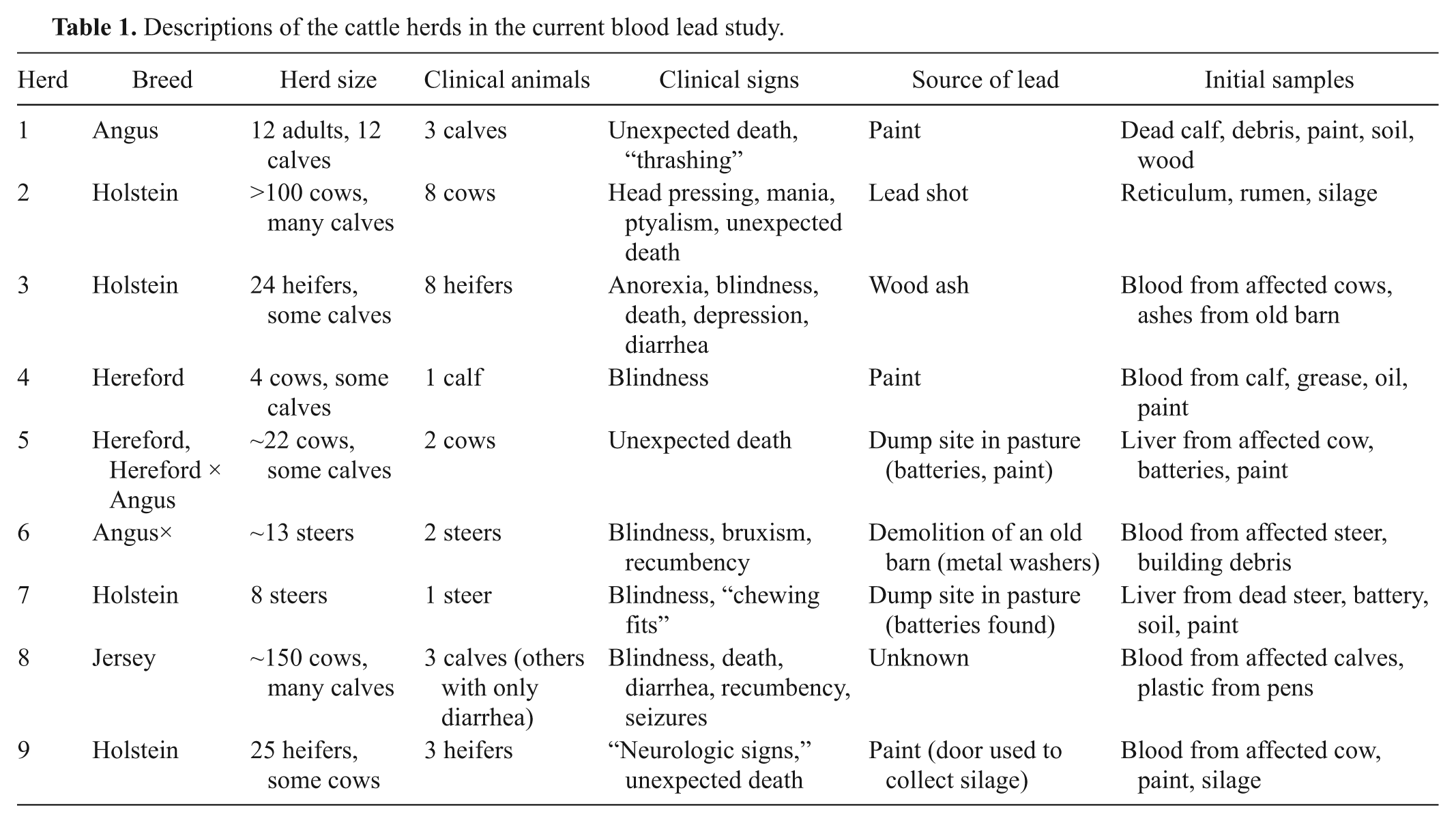
Descriptions of the cattle herds in the current blood lead study.
Samples of paint, soil, feed, hardware (metal washers), battery plates, or other material suspected as sources of Pb were collected from the environment of the exposed cattle (either by herd veterinarians or by New York State Animal Health Diagnostic Center veterinarians) and were analyzed by inductively coupled plasma–atomic emission spectroscopy (ICP-AES)
a
to determine the sources of Pb. Analysis was based on the following published methods
1
: approximately 0.5 g of feed or plant tissue, or 1–500 mg of paint chips, soil, battery plate material, or hardware, was placed into polytetrafluoroethylene-coated microwave vessels with 5 ml of trace mineral–grade concentrated nitric acid
b
and rested for 10 min before 3 ml of 30% reagent-grade hydrogen peroxide
b
was added. Vessels were capped and microwaved
c
at 210°C for 20 min and, after a 20-min ramp time, were allowed to cool, and then were rinsed into 125-ml conical flasks with distilled deionized water. The flasks were boiled until the volume decreased to 2–3 ml. Cooled samples were transferred to 15-ml centrifuge tubes and were distilled deionized water was added to a total volume of 5 ml and centrifuged at 700 ×
Suspected sources of Pb exposure were determined for 8 out of 9 herds in the current study (Table 1). The suspected sources were composed of paint (5/9, 56%), Pb-acid batteries (1, 11%), and metallic Pb (2, 22%) including shot (1 herd) and, possibly, building material (metal washers, 1 herd). The source of Pb for calves in herd 8 could not be determined. Plastic from calf pens in that herd contained approximately 1.2% (12,000 ppm) Pb, but minimal Pb leached out when the plastic was soaked for approximately 1 hr in warm water or an acidic solution; therefore, Pb from this source appears unlikely to have been biologically available. The most likely source of Pb was undetermined in 1 (11%) herd, which is within the 9–51% range given in the literature. 16
Lead absorption from the gastrointestinal tract depends upon the form ingested and the age and nutritional status of the animal. Lead salts (present in paint and Pb-acid battery plates) are biologically available—though less so than organic forms of Pb. Lead salts are ionized in the abomasum for absorption. Metallic Pb is the least absorbed form and can remain in the rumen and reticulum for a long time. 17 Mature animals only absorb 1–3% of ingested Pb, but young animals can absorb up to 50%.2,5,15,18 Factors that increase Pb absorption include a milk-based diet, mineral deficiencies including calcium, iron, and zinc, and high dietary fat.5,17
Lead shot as a source of Pb poisoning in cattle has been reported.4,14 Although metallic Pb in shot is unlikely to dissolve in the environment of the rumen and reticulum (which is only mildly acidic), it is soluble in the more acidic environment of the gastric stomach (abomasum) and under the acidic conditions produced in silage and haylage, as observed in the case of herd 2 and previous studies.4,8 The pH of the haylage in a previous study ranged from 3.9 to 4.4. 4 The Pb concentration in a silage sample from herd 2, after removal of visible Pb particles, was 649 ppm on a dry-matter basis. A previous study detected 900–4,100 ppm lead (dry matter) in silage, and another study found 3,800 ppm (dry matter) in haylage fed to cattle.4,14 Metal washers were found in an area of the herd 6 pasture from which a pole barn had been removed. This metallic Pb was not expected to be biologically available. There was a small area painted with paint containing a moderate concentration of Pb, but no other source of Pb for this herd was found after extensive investigation.
Blood samples were collected from dairy and beef cattle from the 9 herds. When possible, blood was collected from all potentially exposed animals, but collection was done at the discretion of the referring veterinarian and farm owner. Initial samples from clinically affected and unaffected cattle were usually collected by the attending veterinarian within a week of the diagnosis of Pb poisoning in the herd. Because samples were often collected at the convenience of animal owners, a flexible collection protocol was required. Blood samples were collected into blood tubes containing ethylenediamine tetra-acetic acid (EDTA) or heparin as an anticoagulant and were labeled with the animal’s name or identification number. Treatment options were left to the discretion of the veterinarians and animal owners. Days of serial blood sampling are listed in Table 2. Access to the potential source of Pb was restricted (by removing the source of Pb such as feed or by placing the cattle on a new pasture) upon its determination.
Animals sampled per herd by day for serially blood-sampled cattle.*
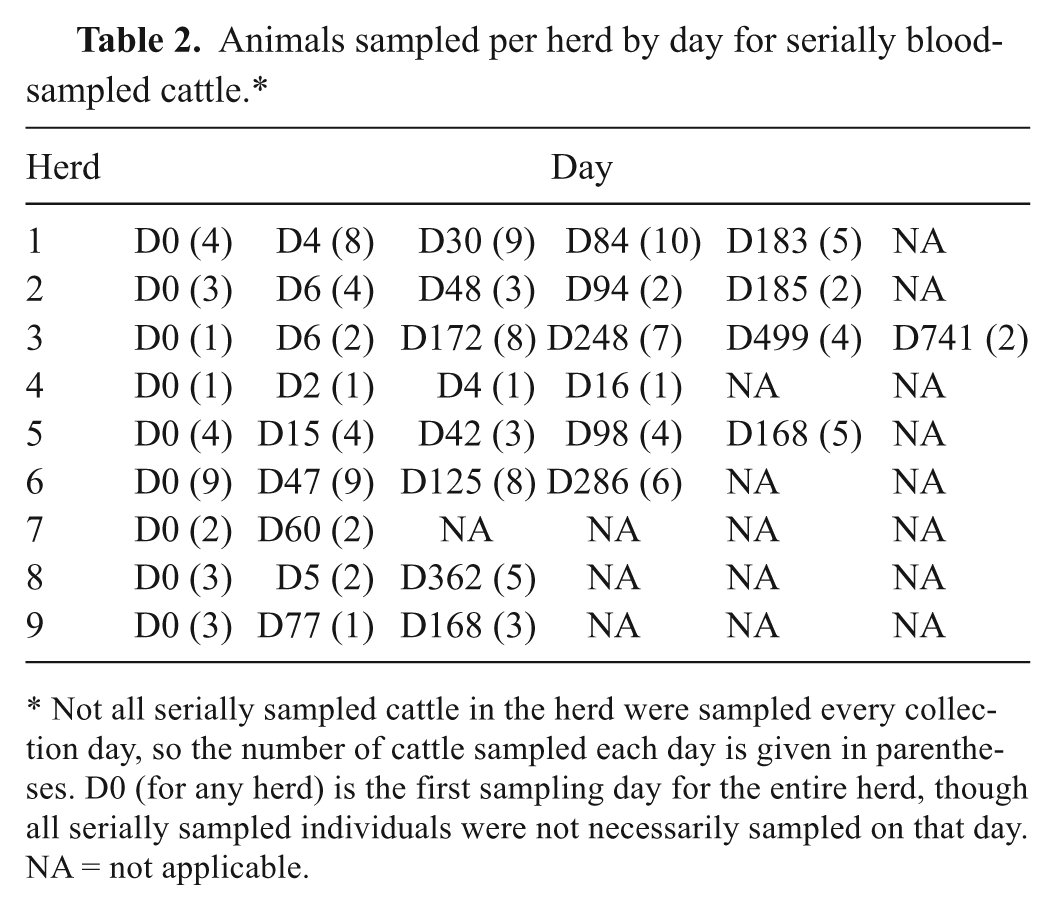
Not all serially sampled cattle in the herd were sampled every collection day, so the number of cattle sampled each day is given in parentheses. D0 (for any herd) is the first sampling day for the entire herd, though all serially sampled individuals were not necessarily sampled on that day. NA = not applicable.
Samples of blood underwent graphite furnace atomic absorption spectroscopy (GFAAS) d Pb analysis using a transversely heated GFAAS with longitudinal Zeeman-effect background correction. Analysis was based on a published method. 13 A 100-µl subsample was taken of each sample, placed into a 2-ml cup, e and mixed with 900 µl of Pb matrix-modifier solution composed of distilled deionized water, 0.02% analytical-grade ammonium phosphate, f 0.05% analytical grade magnesium nitrate, c 1.0% analytical-grade nitric acid, c and 0.1% Triton X. f Standards containing 2.50, 5.00, 10.00, and 50.00 µg/dl Pb to construct a calibration curve were prepared by diluting certified atomic absorption standard solution b in matrix modifier and placing into 2-ml cups, e which were placed in the autosampler tray. All blanks, standards, and samples were analyzed in duplicate. The calibration curve gave a linear response across this range with a correlation coefficient of 0.999. The average of 2 replicates was taken for statistical analysis. Blood samples containing >50 µl/dl of Pb were diluted 1:1 in matrix-modifier solution and reanalyzed.
At least one sample was analyzed for bPb from 126 cattle from the 9 farms enlisted in the present study. The age and number of tested cattle with and without clinical signs suggestive of Pb exposure is given in Table 3. The number of cattle with detectable bPb in the absence of clinical signs is given in Table 4. Determination of clinical toxicosis was based on observation by study veterinarians or the history submitted by the referring veterinarian with blood samples. Out of the 102 cattle in both age groups with detectable bPb concentrations, only 14 (14%) had clinical abnormalities noted on the history. A previous study of 3 herds determined that 4–12% of cattle with no evidence of clinical signs had bPb concentrations high enough to be associated with toxicosis (35 µg/dl), and 7–40% of asymptomatic exposed cattle had “high normal” bPb concentrations (detection limit: 10 µg/dl). 20 A previous study sampled all animals from the 3 herds, but the current study could not. Still, the current study found that 36% of clinically unaffected cattle had bPb concentrations ≥35 µg/dl, 58% had bPb ≥10 µg/dl, and 81% had bPb ≥2.5 µg/dl. Diarrhea was the only clinical sign in 6 calves from one farm (accounting for 67% of the clinically affected cattle <1 year old). Diarrhea has been associated with acute Pb poisoning in cattle,3,17 but diarrhea is also a common cause of morbidity in calves and is caused by many pathogens. Indeed, bPb was not detected in 3 calves with diarrhea from the same farm.
Numbers, ages, and clinical signs of cattle (
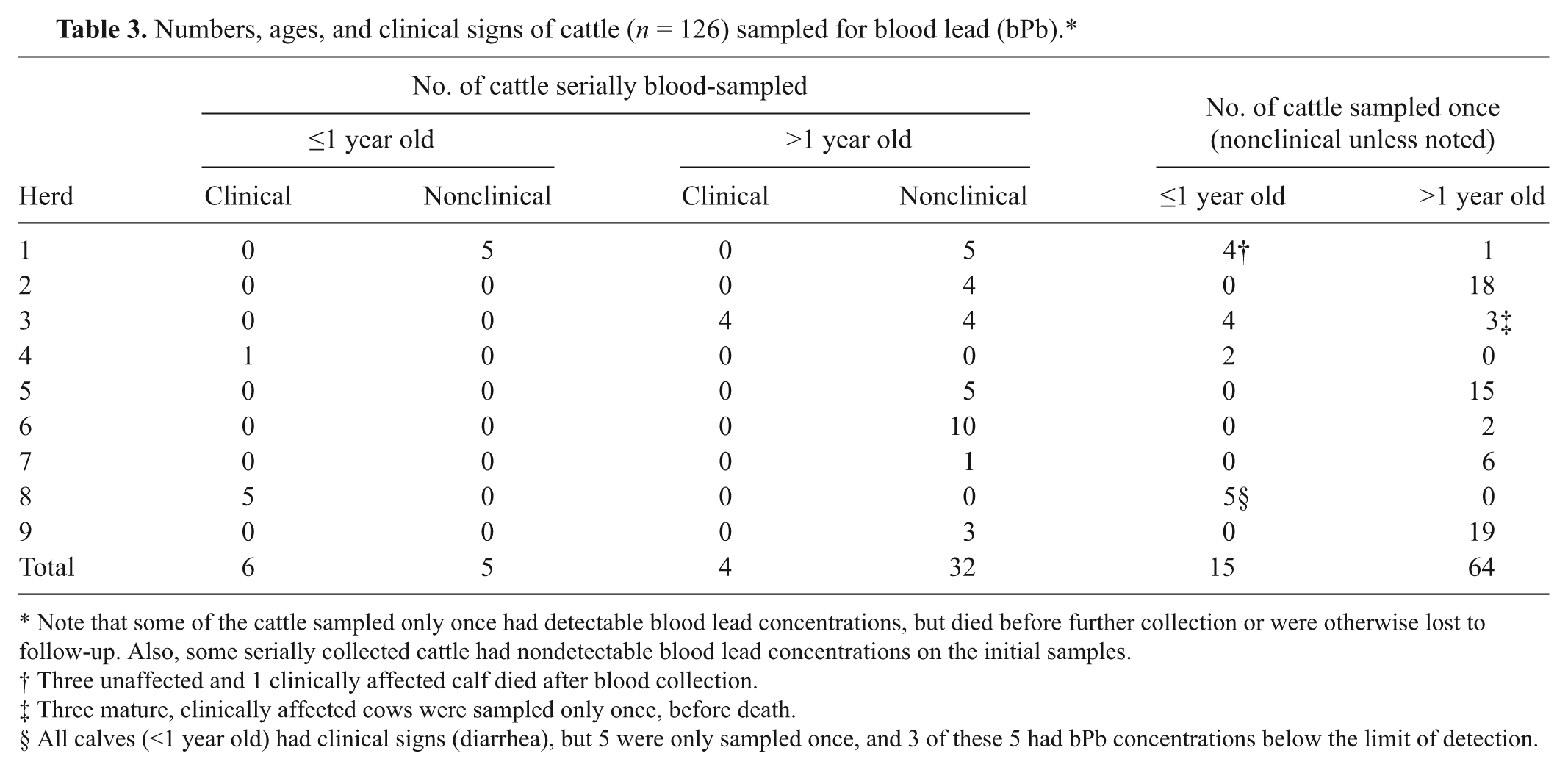
Note that some of the cattle sampled only once had detectable blood lead concentrations, but died before further collection or were otherwise lost to follow-up. Also, some serially collected cattle had nondetectable blood lead concentrations on the initial samples.
Three unaffected and 1 clinically affected calf died after blood collection.
Three mature, clinically affected cows were sampled only once, before death.
All calves (<1 year old) had clinical signs (diarrhea), but 5 were only sampled once, and 3 of these 5 had bPb concentrations below the limit of detection.
Blood lead (bPb) concentrations in 109 cattle that were not observed with signs of clinical lead toxicosis but which were from herds in which clinical lead toxicosis was diagnosed in other cattle.
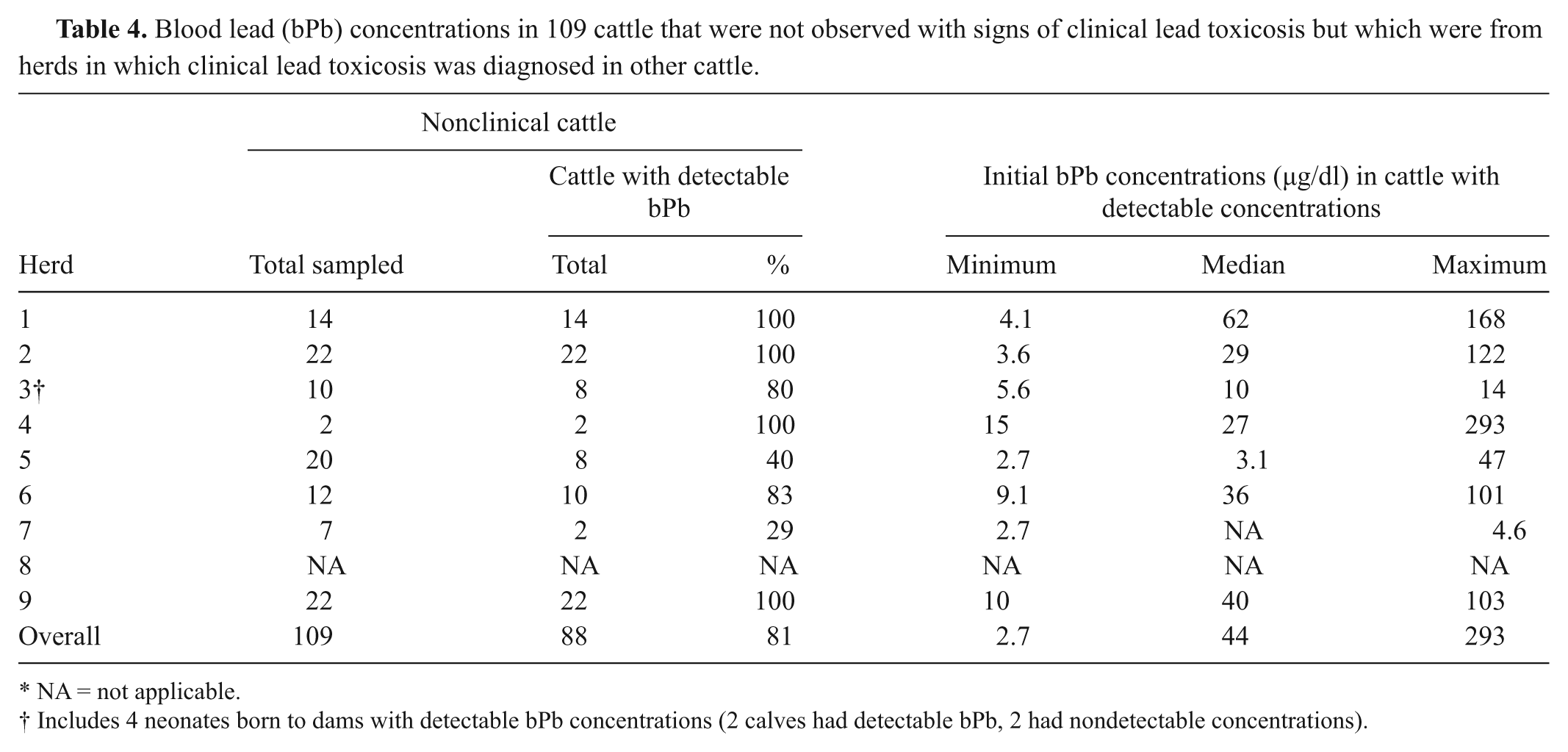
NA = not applicable.
Includes 4 neonates born to dams with detectable bPb concentrations (2 calves had detectable bPb, 2 had nondetectable concentrations).
Half-life estimations were calculated for all 47 cattle that had more than 2 blood samples with detectable bPb concentrations; each had samples collected over a period ranging from 7 to 741 days between April 2005 and December 2010 (Table 2). A spreadsheet program g was used to determine the slope of a line using the natural log of the bPb concentration as the Y axis and the collection day (starting with the first day as day 0) as the X axis. The formula for t1/2 calculation was: t1/2 = ln(2)/(–slope). Mean and standard deviation (SD) for all t1/2 were calculated using the spreadsheet. g The 3 individuals in which the t1/2 was a negative number (implying that the bPb concentration increased during the study period) were dropped from further calculations.
The bPb t1/2 estimates ranged from 3 to 577 days in the current study. Previous studies of accidentally Pb-exposed cattle found t1/2 values ranging from 9 to 2,507 days.5,9,15 The mean t1/2 (and SD) calculated from the 44 individuals with positive calculated bPb t1/2 estimates was 135 days (SD: 125 days). The t1/2 values of animals <1 and ≥1 year old were compared by Student
Half-life (t1/2) ranges, days between first and last blood collection for the entire herd, and number of collections per animal in each herd.
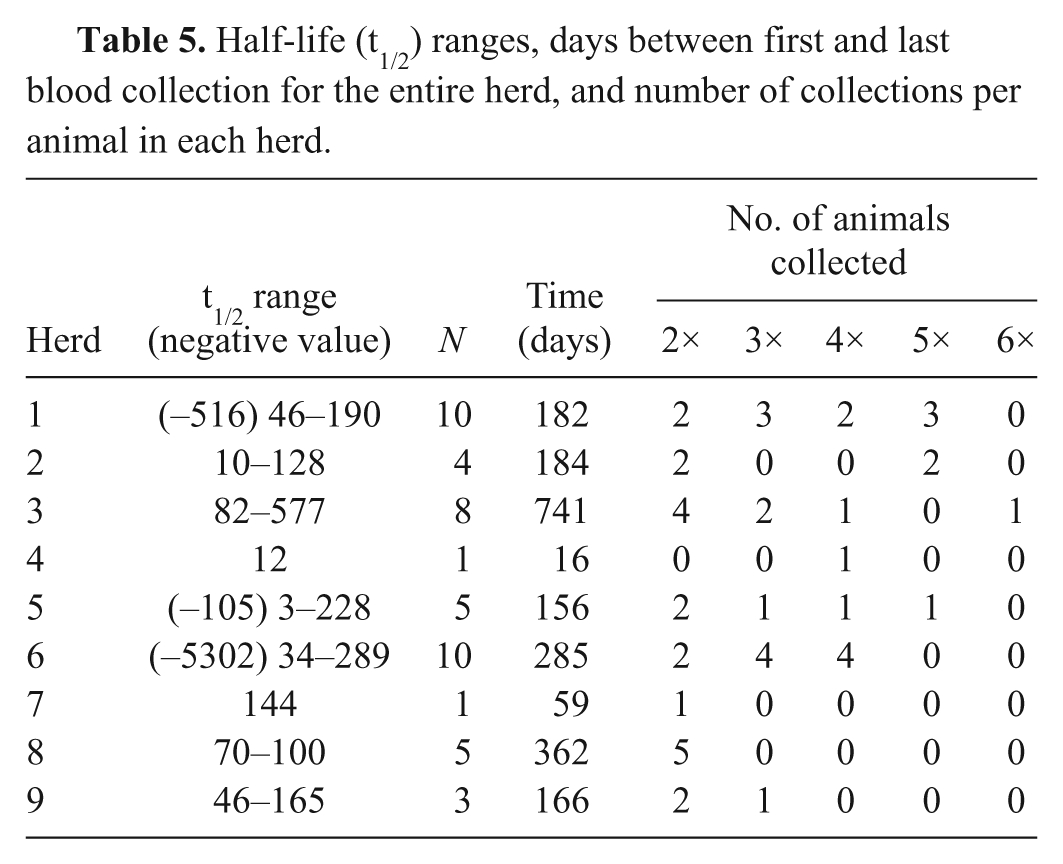
Variability in the estimated values for bPb t1/2 is likely to occur for a combination of reasons. Continued exposure to Pb is a contributing factor. Access to the source of Pb was terminated by removing either the animals from the source of lead or the source of lead from the environment as well as possible, according to herd owners and veterinarians. However, exposure might not have ended completely. Furthermore, Pb is retained indefinitely in the rumen and reticulum of cattle. 15 Included in these calculations were 2 calves from herd 1 that were treated by the herd veterinarian (one with a single dose of activated charcoal and one with a single dose of mineral oil), 2 surviving clinically affected mature heifers from herd 3 that were treated by chelation therapy with calcium EDTA (110 mg/kg intravenously, twice daily; t1/2 values of 101 and 186 days) and 1 calf from herd 4, which was treated with EDTA and magnesium sulfate as a cathartic. Removing the heifers and calf treated with EDTA changed the mean from 135 to 138 days (and reduced the SD to 127). There was no history of treatment for the other cattle, but many Pb-exposed cattle undergo minimal, if any, medical treatment (especially if clinical signs of toxicosis are not evident). Ending exposure to Pb by removing the environmental source or removing the cattle from the contaminated area is the most consistent method used to manage Pb-exposed cattle.
Another aspect of the current study design that contributed to t1/2 variability is the sporadic blood collection from Pb-exposed cattle, which depended on voluntary collection at the discretion of the herd owners and their veterinarians. With the exception of herd 2, herd owners and veterinarians were not required to do follow-up testing by the laws of their states. Each t1/2 calculation was based on 2–6 serial blood samples over 7–741 days. Lead is widely distributed in the soft tissues, and some variation depends on the form of Pb ingested.6,18 Lead kinetics in most species involves multiple compartments, and the bPb t1/2 is therefore likely to be multiphasic. After gastrointestinal absorption, there is a rapid initial rise in bPb, followed by a decline as Pb becomes distributed to other tissues, after which is a plateau.2,15 A later increase in bPb is associated with redistribution of Pb from soft tissues to bone9,15,17 More blood collections—spaced appropriately and starting soon after initial exposure—would be necessary for a better estimate of the multiphasic bPb t1/2 in cattle. Continued gastrointestinal absorption of ingested Pb is another cause of increased circulating blood concentrations over time and is likely to have occurred in some of the cattle with negative t1/2 estimates in the present study.
When Pb poisoning is diagnosed in cattle (which produce both meat and milk), serial blood testing of clinically affected cattle is necessary to determine when these animals can safely be returned to the human food chain. The bPb t1/2 is complicated and highly variable; thus, it cannot be accurately predicted. Surgical removal of Pb from the rumen and reticulum might increase the overall rate of elimination of Pb from the blood. Most importantly, it is imperative to test herd mates of exposed cattle, because only approximately 1 in 7 animals with detectable bPb concentrations (>2.5 µg/dl) had clinical signs that could be attributed to Pb toxicosis.
Footnotes
Acknowledgements
The authors thank Ms. Kerry Manzell for her analytical expertise, and the cattle producers and veterinarians who participated.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was funded in part by the United States Department of Agriculture (grant no. NYC-478425).
