Abstract
Lead (Pb) concentrations in whole blood and δ-aminolevulinic acid (ALA) concentrations in plasma and whole blood from 37 cattle with suspected Pb exposure were determined in order to investigate the usefulness of ALA as a biological indicator for Pb poisoning in cattle. Cows were divided into 4 groups based on blood Pb, as follows: <30 ppb (group 1), 30–100 ppb (group 2), 100–300 ppb (group 3), and >300 ppb (group 4). The derivatization reaction for ALA was improved by a greater than 2-fold measure in whole blood and by a 10-fold measure in plasma by adding 75 and 50 μl of 0.1 N HCl, respectively. Blood Pb concentrations ranged from <25 ppb to 1,006 ppb (185.5 ± 254.9 ppb), with 17 samples containing >50 ppb Pb. Delta-aminolevulinic acid concentrations in whole blood and plasma ranged from <62.7 ppb to 96.9 ppb (77.4 ± 8.4 ppb) and from <5.0 ppb to 24.0 ppb (4.6 ± 3.8 ppb), respectively. Whole blood ALA did not correlate with blood lead concentrations in any group. Increase in plasma ALA concentration was dependent on blood Pb concentration. There was no correlation between blood Pb concentration and plasma ALA concentration in group 2 (
Introduction
Lead (Pb) is a common environmental contaminant and source of poisoning in veterinary medicine. Lead poisoning in cattle and other food-producing animals is a public health issue because of the potential for human exposure to Pb through ingestion of contaminated meat and milk products. 16 One mechanism by which Pb acts is by inhibition of erythrocyte δ-aminolevulinic acid dehydratase (ALAD), an enzyme involved in the production of erythrocytes, which dimerizes δ-aminolevulinic acid (ALA) to form porphobilinogen. 19
It is useful to understand the biological response in order to use biomarkers to assess exposure to Pb and to study the health effects of Pb and other toxins. As one of the biomarkers of Pb exposure, the whole blood Pb concentration is commonly used in human beings and domestic and wild animals to assess exposure and expected clinical effects. 3,20,23 Delta-aminolevulinic acid dehydratase and free erythrocyte protoporphyrin are indicators of biological response to Pb that have been suggested11, 15,18,25 to be good indicators of Pb exposure in cattle and other species. Although blood ALAD activity was inhibited by blood Pb, there were significant differences in ALAD activity among asymptomatic cattle and dogs with elevated blood Pb concentrations. 11,12 Urinary ALA concentrations have been studied1, 2,8,21,22,24 as a sensitive exposure biomarker for Pb exposure in rats, cattle, and humans because they increase at very low-level Pb exposure. However, urinary ALA concentrations also vary with diet and time of urine collection. Plasma and whole blood ALA concentrations have been suggested 19 to be sensitive indicators of Pb exposure in human beings. The minimum blood Pb concentration associated with increased plasma ALA is <10 ppb in Pb-exposed human beings 9 ; therefore, plasma ALA is a useful parameter for determining trace Pb exposure. 19 Since the baseline ALA concentration in plasma is relatively low compared to the concentration found in urine or blood, analytic methods must be very sensitive for plasma ALA analysis.
The current study investigates the determination of Pb concentrations in whole blood and ALA concentrations in whole blood and plasma from exposed cattle from 3 herds in the United States. Plasma and blood ALA concentration were compared to blood Pb concentrations, and plasma ALA was determined to be a useful biomarker for Pb exposure in cattle.
Materials and methods
Animals
Venous blood samples were collected in 2 sodium—ethylenediamine tetra-acetic acid blood collection vials a (by veterinarians) from 37 Holstein, Angus cross, and Hereford cross cattle living on privately owned farms in the United States. Lead poisoning was diagnosed in dead cattle on these farms before blood collection, and the sources were determined to include lead paint chips, broken car batteries, and various hardware sources found in the pastures. No clinical signs were apparent in surviving animals at the time of sampling. Whole blood from one tube was used for Pb analysis and ALA determination. Plasma was separated from the second tube by centrifugation within 2–24 hr of sampling for ALA determination. Whole blood and plasma samples were stored at −20°C for the determination of ALA.
Measurement of Pb in whole blood
Lead analysis was performed using a transversely heated graphite furnace atomic absorption spectrometer b with longitudinal Zeeman-effect background correction. Analysis was based on a published method. 13 A 100-μl subsample was taken of each sample, placed in a 2-ml cup, b and mixed with 900 μl of matrix-modifier solution. Matrix-modifier solution is composed of NANOpure deionized water, 0.02% analytical-grade ammonium phosphate, d 0.05% analytical-grade magnesium nitrate, d 1.0% analytical-grade nitric acid, d and 0.1% Triton X. d Standards containing 2.50, 5.00, 10.00, and 50.00 μg/dl Pb (to construct a calibration curve) were prepared by diluting certified atomic absorption standard solution e in matrix modifier and placing the solution in cups. c The cups containing a matrix-modifier blank, standards from low to high concentrations, and blood samples were placed in the autosampler tray. All blanks, standards, and samples were analyzed in duplicate. The calibration curve gave a linear response across this range with a correlation coefficient of 0.999. Two replicates of each blood sample were analyzed, and the average was taken for statistical analysis. Blood samples containing >50 μg/dl of Pb were diluted 1:1 in matrix-modifier solution.
Measurement of ALA concentration in whole blood and blood plasma
A modified method was used to analyze blood and plasma for ALA by acidifying the samples.
10
Briefly, 50 μl of the whole blood (or plasma) sample was added to a glass tube, and then 75 μl of 0.1 N hydrochloric acid (HCl
d
; 50 μl for plasma) was added to decrease the pH of reaction solution. After vortexing for a few seconds, 1 ml of acetylacetone
f
(2 ml for plasma) was added, followed by 200 μl of 10% formaldehyde solution. The sample was again vortexed for a few seconds. The mixture was heated to 100°C for 10 min in a heating block, then immediately cooled with cold tap water and centrifuged at 3,000 ×
Grouping and statistical analysis
Cattle were divided into 4 groups based on blood Pb concentrations, as follows: <30 ppb (group 1), 30–100 ppb (group 2), 100–300 ppb (group 3), and >300 ppb (group 4). To calculate the correlation between groups, a trend line equitation was obtained using Excel.
h
To compare the difference between group 1 and other groups, a
Results
Effect of acidification on the derivatization of ALA in whole blood and plasma
Derivatization of ALA was increased by a 2-fold measure in whole blood and by a 10-fold measure in plasma by the addition of 0.1 N HCl. Maximum derivatizations were achieved at a volume of 75 μl for whole blood and a volume of 50 μl for plasma (Fig. 1).
Lead and ALA in whole blood and ALA in plasma
The Pb concentration in whole blood from the 37 cows ranged from <25 ppb to 1,006 ppb (185.5 ± 254.9 ppb). Seventeen samples had lead concentrations of >50 ppb. The ALA concentrations in whole blood ranged from <62.7 ppb to 96.9 ppb (77.4 ± 8.4 ppb). The ALA concentrations in plasma ranged from <5.0 ppb to 24.0 ppb (4.6 ± 3.8 ppb; Table 1).
Correlation between Pb in whole blood and ALA in plasma
The concentration of ALA in whole blood did not change significantly between groups. Increases in ALA concentrations in plasma were dependent on the concentration of Pb in whole blood, but there was no correlation between blood Pb and plasma ALA in group 2. Correlation coefficients were 0.736 and 0.807 for groups 3 and 4, respectively, and the correlation coefficient increased to 0.851 for groups 3 and 4 combined (Fig. 2).
Discussion
Whole blood Pb is frequently used as an indicator of Pb exposure and toxicosis. The whole blood Pb concentration represents both recent exposure to Pb and release of Pb from tissue storage. 6 Whole blood Pb is widely used as a biomarker of Pb exposure because blood is an easy antemortem sample to collect and because most veterinary diagnostic laboratories are capable of performing the analysis. However, the blood Pb concentration does not necessarily reflect previous or intermittent exposure because blood Pb concentrations are strongly influenced by recent exposure. 4 Furthermore, individual differences in response to elevated blood Pb concentrations have been documented. A study 4 of dogs with elevated blood Pb concentrations found that some individuals with high blood Pb concentrations did not have the expected clinical signs of Pb poisoning. Because one mechanism by which Pb acts is by inhibition of erythrocyte ALAD, which is an early step in the synthesis of heme for erythrocytes, the substrate of this enzyme, ALA, has been proposed 7 as another biomarker for Pb exposure.
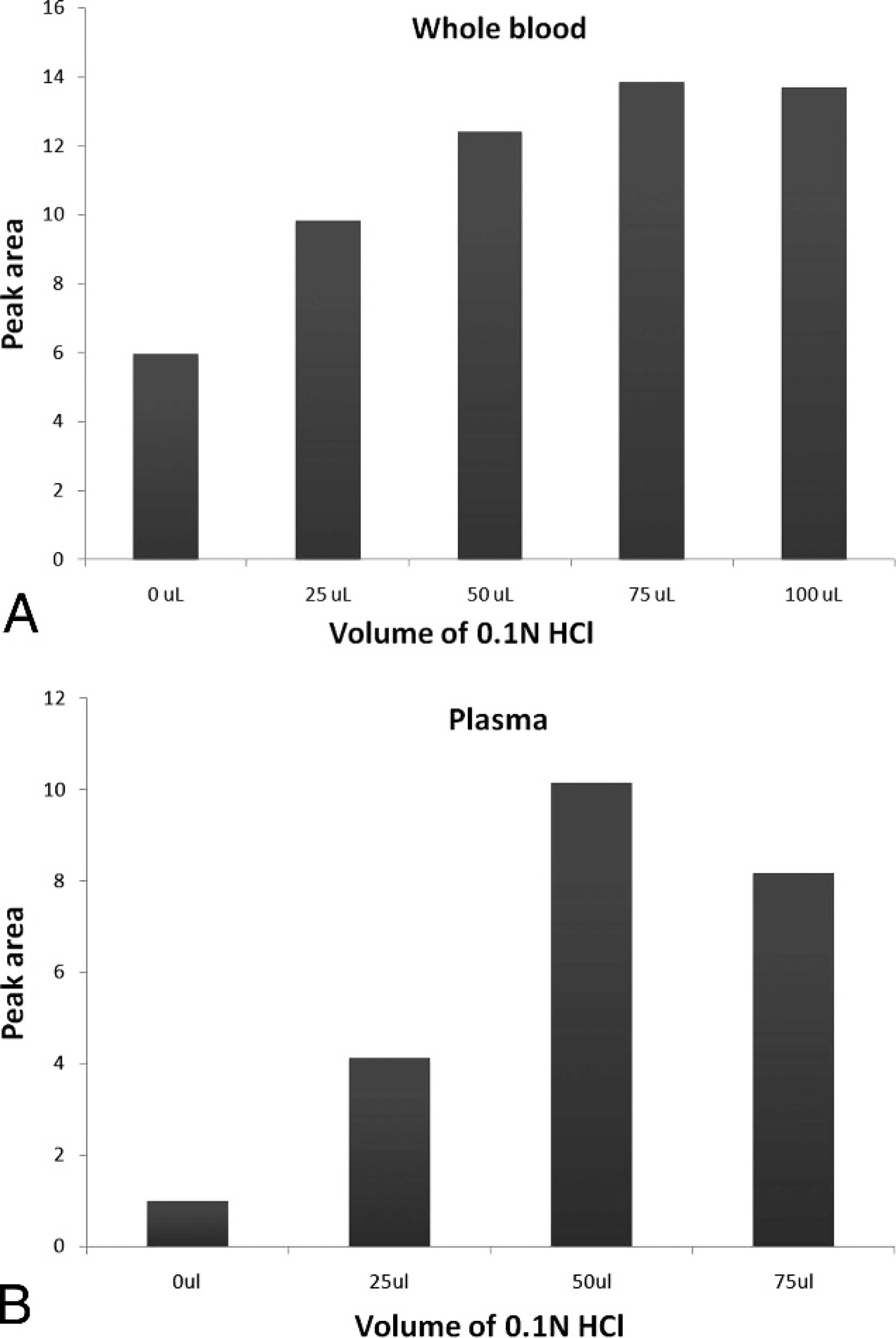
Effect of 0.1 N HCl on the derivatization of δ-aminolevulinic acid in whole blood (
Whole blood lead (Pb) compared to blood and plasma δ-aminolevulinic acid (ALA) concentration in cattle (in ppb). *
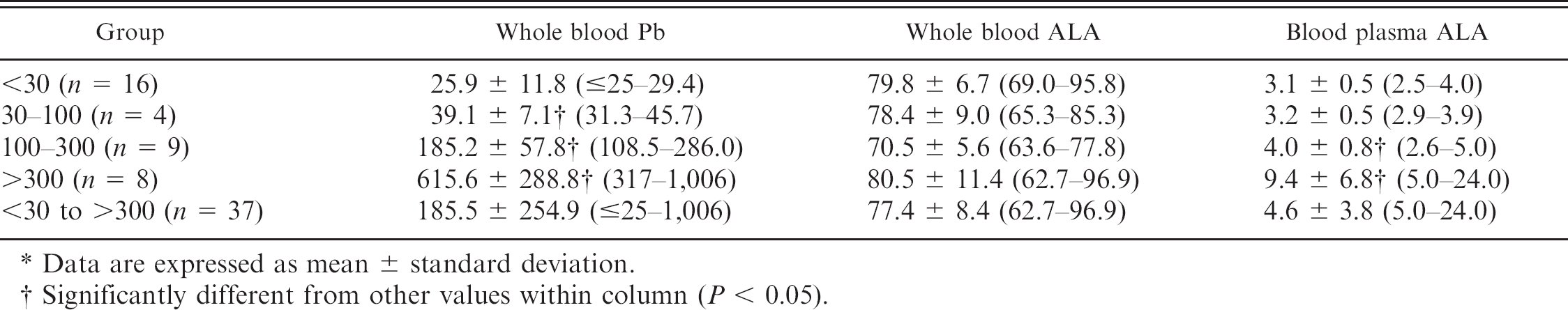
Data are expressed as mean ± standard deviation.
Significantly different from other values within column (
The pH of a solution affects the solubility of solutes, so pH adjustments are needed to move chemicals from the aqueous phase to the organic phase. In the present experiment, the derivatization of ALA was increased by a >2-fold measure in whole blood and by a 10-fold measure in plasma by adding 0.1 N HCl. Delta-aminolevulinic acid is present in the nonionized form at low pH and can therefore move to the organic layer (acetylacetone), where the derivatization reaction occurs.
Delta-aminolevulinic acid in serum or plasma has been suggested
18
as a biological indicator of human Pb exposure and biological effects of Pb in bone marrow. Plasma ALA is useful for discriminating between low- and high-level Pb exposure.
19
The critical concentration of Pb in blood associated with increased plasma ALA in human beings is <100 ppb.
14
Because of the higher affinity of human ALAD2 for Pb, Pb is likely to have a lower bioavailability in
Cattle in heavily polluted areas and urban areas of some developing countries have elevated blood Pb concentrations. 5,12 The Pb concentrations in whole blood of cattle in the current study ranged from <25 ppb to 1,006 ppb, with 17 out of 37 containing >50 ppb Pb. Blood ALA concentrations were not significantly different between groups of cattle classified based on blood Pb concentrations in the present study (Table 1). There was no difference in plasma ALA concentration from cattle with blood Pb concentrations of >30 ppb compared to those with blood Pb concentrations between 30 and 100 ppb. Thus, ALA is relatively more sensitive than ALAD or free erythrocyte protoporphyrin, based on a previous study 5 that found no change in other Pb biomarkers (ALAD activity and free erythrocyte porphyrin) in cattle with blood Pb concentrations of <100 ppb. However, only 4 samples were available in the current study for cattle with blood Pb concentrations between 30 and 100 ppb; thus, more samples are required for a conclusive result.
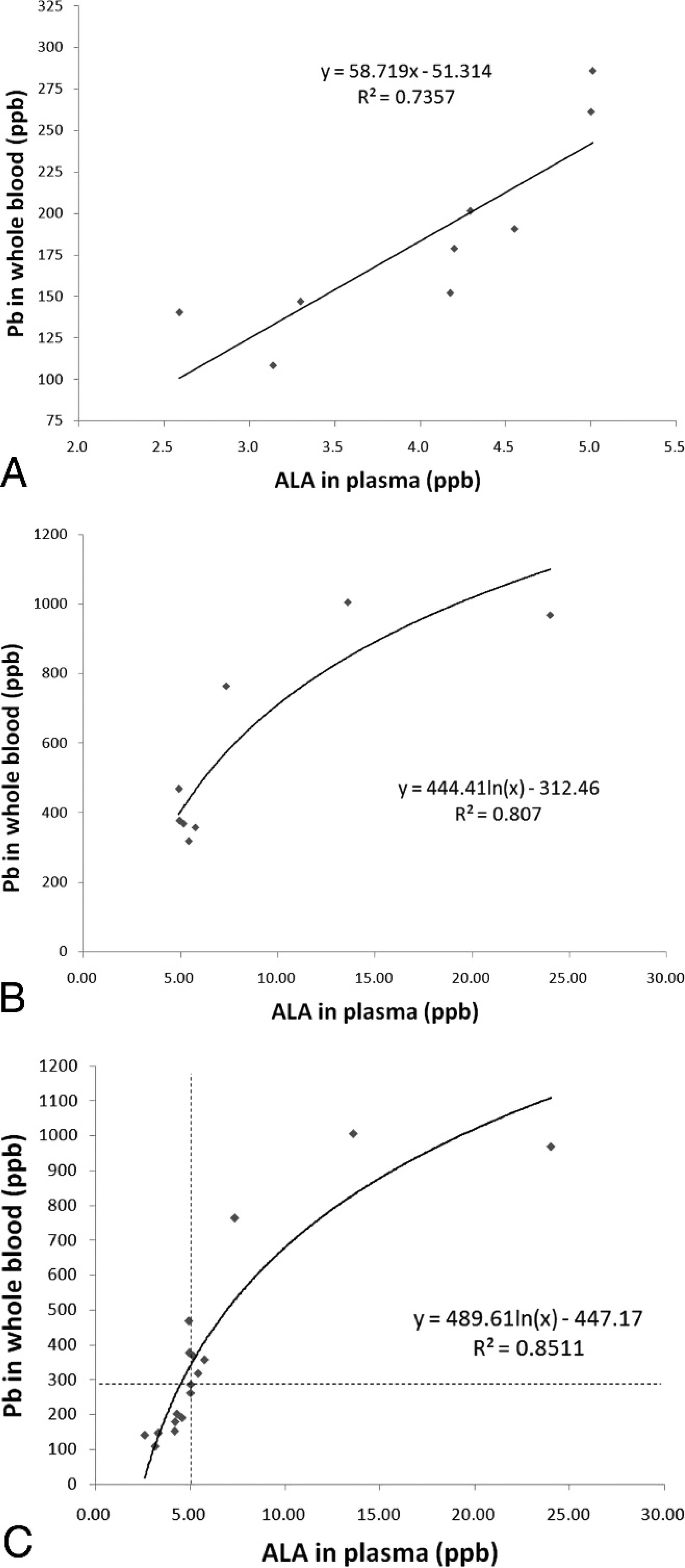
A, correlation between whole blood lead (Pb) concentration (100–300 ppb,
Plasma ALA concentrations in groups 2 and 3 of the present study were significantly different from those in group 1, and increase in plasma ALA correlated with increased blood Pb concentration up to approximately 1,000 ppb. Similarly, plasma ALA concentrations increased continuously with blood Pb concentration up to 1,000 ppb in a study 19 of exposed people. The correlation in cattle was linear at blood Pb concentrations ranging from 100 to 300 ppb, and there was a nonlinear correlation at blood Pb concentrations of >300 ppb. Studies 17 of human plasma ALA concentrations found a strong linear correlation with blood Pb concentrations of between 300 and 600 ppb. Based on the results presented in the current study, the plasma ALA concentration in cattle increases linearly to 5 ppb as the blood Pb concentration approaches 300 ppb, then the response appears to plateau at higher blood Pb concentrations (Fig. 2C).
The results of the present study demonstrate that the measurement of plasma ALA using a fluorometric HPLC method is useful for monitoring Pb exposure in cattle. The analytical method is simple, sensitive, and specific over a wide range of blood Pb concentrations. The cattle in the current study were recently exposed to Pb. Further work is needed to determine plasma ALA concentrations in unexposed cattle as well as to determine how plasma ALA concentration compares to blood Pb concentration as a biomarker in chronic or intermittent Pb exposure. Further studies are also needed to determine how blood Pb and plasma ALA correlate with Pb concentrations in edible tissues from cattle, such as meat and milk, as well as in other tissues.
Acknowledgements
This project was supported by grants from the National Veterinary Research and Quarantine Service, Republic of Korea, and the U.S. Department of Agriculture. The authors would like to acknowledge Ms. Kerry Manzell and Drs. Belinda Thompson and Paul Virkler for their assistance with sample collection and testing.
Footnotes
a.
Vacutainer®, BD, Franklin Lakes, NJ.
b.
Perkin-Elmer 800, PerkinElmer Inc., Waltham, MA.
c.
Auto Analyzer Cup, Krackeler Scientific, Albany, NY.
d.
Fisher Scientific Co., Pittsburgh, PA.
e.
EMD Chemicals Inc., Gibbstown, NJ.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Agilent Technologies Inc., Santa Clara, CA.
h.
Microsoft Corp., Redmond, WA.
