Abstract
Central nervous system tumors are rarely reported in cervids. The current report describes gross and histopathologic oligodendrogliomas in 3 white-tailed deer (
With the exception of virally induced fibromas,29,30 neoplasia is a relatively uncommon finding in white-tailed deer (
Three adult female deer were euthanized after showing central nervous system signs characterized by disorientation and loss of fear of human beings. By tooth wear, deer 1 (from Greenwood, Kansas) was estimated to be over 9 years old, deer 2 (from East Baton Rouge Parish, Louisiana) was greater than 6 years old, and deer 3 (from Blackville, New Brunswick, Canada) was approximately 2 years old. Deer 2 had nasal and oral discharge. The deer cases exhibited many characteristic gross and histologic features of oligodendroglioma. In all deer, masses were locally invasive in the ventral cerebrum (thalamus and forebrain, extending into the midbrain in deer 3) and ranged from 2 to 5 cm in diameter. Grossly, the masses were light gray, variably circumscribed, and soft (Fig. 1) with gelatinous, friable areas in deer 1. In deer 2, the ventral surface of the mass appeared hemorrhagic.
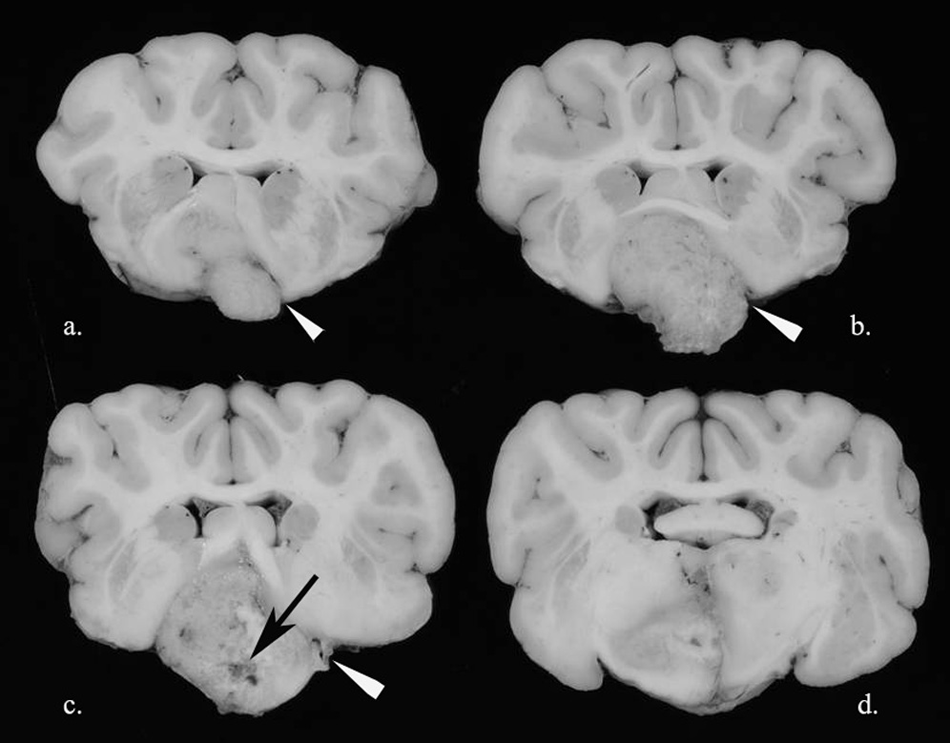
White-tailed deer (
After gross evaluation of the head and brain, representative samples of brain were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin for routine histopathologic examination. Immunohistochemical staining of paraffin-embedded sections was conducted with murine monoclonal antibodies specific for glial fibrillary acid protein (GFAP; 1:800), a vimentin (1:600), a and pancytokeratin (1:100), a and rabbit polyclonal antibody specific for S100 (1:2,000). b The only immunohistochemical tests performed were ones that have been validated and in routine use at the University of Georgia histology laboratory. Cytokeratin staining was conducted to rule out tumors of epithelial origin; vimentin (an intermediate filament protein) was performed to identify cells and/or tumors of mesenchymal origin; GFAP (an intermediate filament protein found in mature astrocytes) staining was used as a marker to rule out astrocytic tumors; and S100 (a calcium binding protein) was performed because oligodendrogliomas have been reported to have positive immunoreactivity to S100. Immunohistochemical tests were performed for GFAP and S100 in deer 1 and 2, and vimentin and pancytokeratin in deer 1. Sections of domestic dog tissue were used for positive controls for immunohistochemical stains. Immunohistochemical stains performed were considered adequate for staining of white-tailed deer tissues based on evaluation of tissues from other deer cases and the broad reactivity of these markers for many species. Immunohistochemistry was not performed on deer 3.
Histologically, all masses were variably circumscribed, invasive, and highly cellular. The tumors were composed primarily of oval, round, and polygonal cells arranged in sheets or poorly defined cords admixed with moderate numbers of thin-walled blood vessels (Fig. 2A). Neoplastic cells had a central oval to round normochromatic to hyperchromatic basophilic nucleus and pale cytoplasm with sparse, fine, pale, amphophilic cytoplasmic granules surrounded by a thin basophilic membrane (Fig. 2B). Together, many of these neoplastic cells exhibited a characteristic “honeycomb” appearance (Fig. 2C). Additionally, scattered throughout the masses of all deer was amorphous amphophilic to basophilic mucinous material. All masses also had variable amounts of hemorrhage and liquefactive necrosis (ranging from 20% to 40% of the tumor area on sections examined), characterized by nuclear pyknosis and karyorrhexis admixed with eosinophilic cell debris. Within and surrounding necrotic foci were variable amounts of inflammation, comprised of moderate numbers of neutrophils and lymphocytes, and few plasma cells. Dystrophic mineralization was associated with tumor necrosis in deer 1 and 2. In 1 animal (deer 2), the mass contained a moderate number of glomeruloid vascular tufts, particularly at tumor margins. Mitotic rates varied from 0 to 1 per 40× field (deer 1 and 3) to 3–5 per 400× field (deer 2).
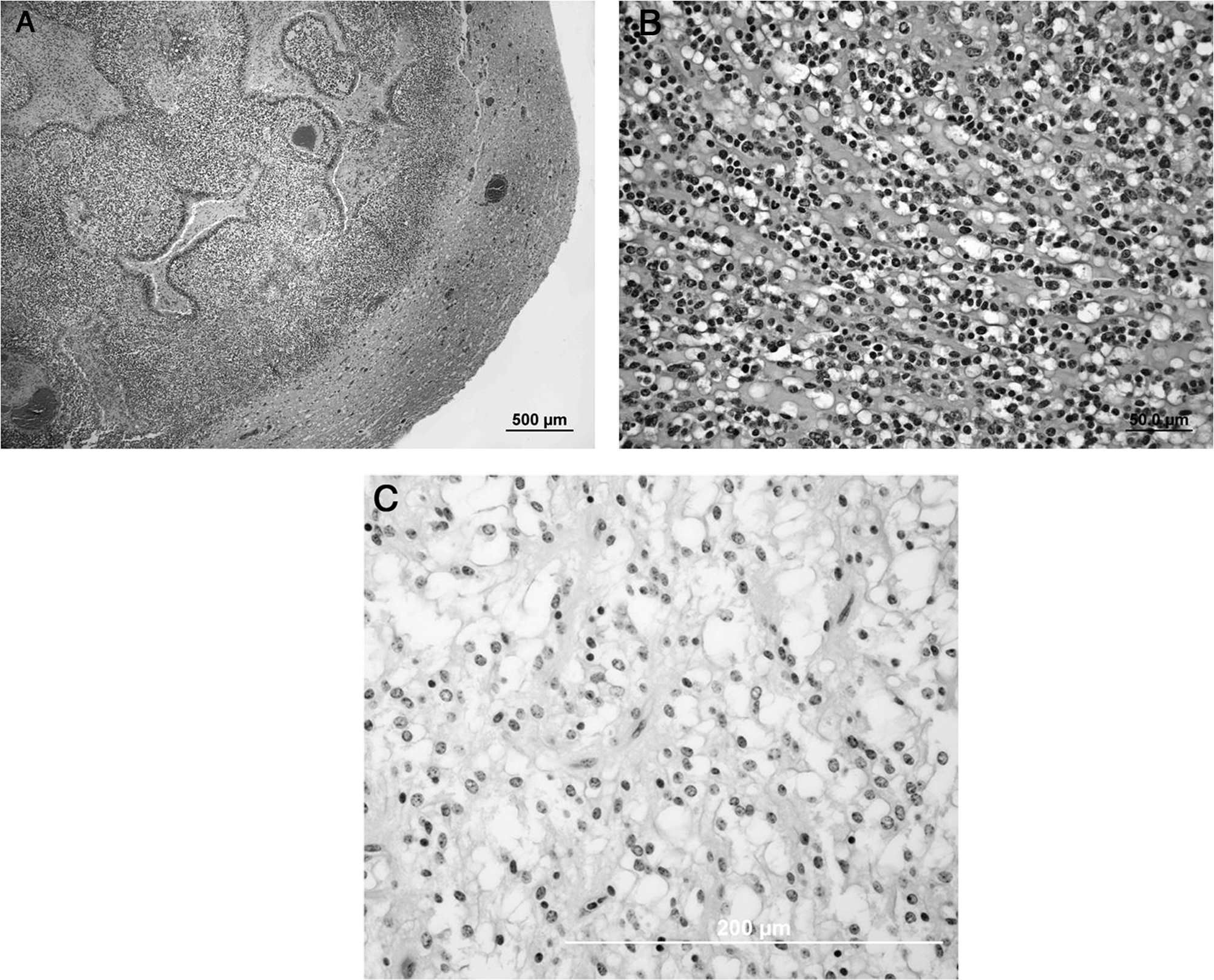
White-tailed deer (
Immunohistochemistry, performed in deer 1 (pancytokeratin, vimentin, S100, GFAP) and deer 2 (S100, GFAP) showed negative cytoplasmic staining of neoplastic cells for pancytokeratin and vimentin (deer 1), and positive cytoplasmic staining for S100 (deer 1 and 2).Neoplastic cells in deer 1 were GFAP negative, and only rare cells interspersed between neoplastic cells in deer 2 (interpreted as reactive astrocytes) were GFAP positive.
The gross and histologic features in the deer cases in the current study were consistent with findings from other animals. In domestic animals, oligodendrogliomas are relatively straightforward to diagnose, 8 and are characterized grossly by a gray or gray–pink, soft to gelatinous mass mainly within the white matter.20,28 Histologically, oligodendrogliomas are composed primarily of uniform cells with a pale cytoplasm, cytoplasmic membrane staining, and centrally located chromatin-rich nucleus, giving most of the mass a “fried egg” or “honeycomb” appearance. Deposition of mucinous material in the mass is common. Occasionally, mineralization and vascular glomeruloid proliferation occur in association with the mass.8,20,28
Oligodendrogliomas are most commonly reported in human beings,33,34 domestic dogs, and very rarely in domestic cats.
28
There are few reports of oligodendrogliomas in cattle,2,14,32 and 1 report in a sheep.
5
Similarly, oligodendrogliomas have only been reported in 1 horse,
25
and a 2010 case of a horse describes oligodendroglioma-like cells within a glioblastoma.
9
In zoo animals, there is 1 report of an oligodendroglioma in an African lion (
Primary central nervous system tumors are rare in cervids. Aside from the present study, the only other reported oligodendroglioma in free-ranging wildlife is in a red deer (
Grossly, oligodendrogliomas cannot be distinguished from other central nervous system tumors, but common gross features can include a gelatinous appearance, with a pink–gray appearance and areas of hemorrhage or necrosis.16,28 Oligodendrogliomas are diagnosed by their characteristic histologic morphology, 16 rather than by gross appearance, immunostaining patterns, or ultrastructural features. 16 The primary diagnoses in all 3 deer cases in the current study were made by classical histomorphological features of oligodendroglioma.33,34
On immunohistochemistry performed on 2 deer, neoplastic cells were S100 positive and GFAP negative. Vimentin, performed only in deer 1, was negative. Accordingly, in many human oligodendrial tumors, vimentin expression is rare. 13 S100 stains most oligodendrogliomas, and stained the tumors in deer 1 and 2, but is not specific for neoplastic oligodendrocytes and can stain other glial cells. Differential diagnoses for oligodendroglioma include neurocytoma, a synaptophysin-positive tumor only reported in human beings, and oligoastrocytomas. 16 Oligoastrocytomas, unlike oligodendrogliomas, have a mixed oligodendrial and astrocytic component that is generally GFAP positive 16 with neoplastic astrocytes composing more than 25% of the neoplastic cells. 15 Unfortunately, there are no consistent markers to distinguish oligodendrogliomas by immunohistochemistry.18,23,33
Additional immunohistochemical stains used for characterization of neural tumors of domestic animals and human beings, such as EGFR and PDGFRα, 10 and Olig2 (used for distinguishing an oligodendroglioma from extraventricular neurocytomas 23 ), or tissue microarray profiling 13 may be useful for further characterization of oligodendrogliomas in white-tailed deer. Ultrastructure was not performed in the deer cases in the present study, but there are no pathognomonic ultrastructural features that are used for definitive diagnosis of oligodendroglioma. However, some ultrastructural characteristics reported in oligodendrogliomas include concentric arrays of membranes within the cytoplasm, round to oval nuclei that are not indented, and many ribosomes, microtubules, and glycogen granules within the cytoplasm. 7 Analyses of chromosomal mutations may also be a helpful means of characterizing and identifying oligodendrogliomas, as oligodendrogliomas in human beings have been associated with mutations in chromosomes 1 and 19. 1 However, a larger sample size of white-tailed deer neural tumors would be needed for further immunohistochemical characterization of oligodendrogliomas in this species.
When wild deer are observed with central nervous system signs, it is recommended that clinicians perform a full postmortem evaluation of the brain and spinal cord when possible, in addition to standard testing for chronic wasting disease, rabies, and other infectious etiologies. Increased surveillance and evaluation of central nervous system disease in white-tailed deer, in addition to chronic wasting disease evaluation, will allow for a better understanding of the incidence of brain tumors such as oligodendrogliomas in deer and their association with morbidity and mortality.
Footnotes
Acknowledgements
The authors wish to thank Lloyd Fox from the Kansas Department of Wildlife and Parks, Mike Perot of the Louisiana Department of Wildlife and Fisheries, and Stephen Coughlan and Reginal Manderville from the Blackville office of the New Brunswick Department of Natural Resources for submitting these cases. The authors thank the histology laboratory at the University of Georgia, College of Veterinary Medicine for histology and immunohistochemistry slides, and the histology section at the New Brunswick Provincial Veterinary Laboratory.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
