Abstract
Bovine trichomoniasis is a sexually transmitted disease in cattle that causes considerable economic loss due to abortions and infertility. In vitro culture of the organisms is the traditional method for diagnosis. However, culture cannot differentiate Tritrichomonas foetus from other, closely related nonpathogenic protozoa. Recently, a quantitative real-time polymerase chain reaction (qPCR) was developed for the differential diagnosis of trichomoniasis. The objective of the current work was to evaluate the effect of different simulated transport conditions on samples containing T. foetus for the diagnosis of trichomoniasis using culture and qPCR. Results indicate that transport temperatures of 4–20°C for 1–3 days before culture will reduce or temporarily inhibit parasite replication but maintain viability. Testing of samples by either culture or qPCR would be expected to give positive results. However, diagnosis of trichomonads by both methods was negatively affected when specimens were maintained at transport temperatures of 42°C for 24 hr or more. The current study stresses the importance of ensuring that clinical samples arrive to the diagnostic laboratory within 24–48 hr and of avoiding temperature transport conditions above 37°C in order to achieve an accurate diagnosis of trichomoniasis in cattle.
Bovine trichomoniasis is a venereal reproductive disease of cattle caused by the protozoon parasite Tritrichomonas foetus. 9 This disease causes early embryonic deaths with infertility in many parts of the world. Bulls are asymptomatic chronic carriers and can infect cows during insemination, while cows are usually able to clear the infection within a few months.13,16 The organisms are found primarily in the superficial layers of the preputial mucosa in bulls. 12 Preputial wash specimens for bulls and cervicovaginal mucus for cows are the desired samples for diagnosis of trichomoniasis.1,15 Samples collected from potentially infected animals are inoculated into culture medium and transported to the diagnostic laboratory. 12 At the laboratory, pouches of culture media are incubated at 37°C for 4–7 days post-collection, depending on the laboratory. Traditional diagnosis of trichomoniasis is carried out based on daily microscopic examinations of inoculated “Trich pouches” by identifying spindle-shaped trichomonads with characteristic rolling and tumbling motility. Microscopic evaluation of organisms is not sufficient to differentiate T. foetus from nonpathogenic intestinal or coprophilic trichomonads,3,8,14 therefore, several conventional and real-time polymerase chain reaction (PCR) assays have been developed for the definitive diagnosis of trichomoniasis.5-7,10 This methodology has demonstrated significant advantages over culture.5,10 However, accurate PCR results correlate with the quality of the sample, which can be affected by transport condition parameters such as temperature and time of transport to the laboratory. The effects of cold temperatures during transport on the survival of T. foetus using different transport media has been investigated previously by light microscope examination 2 and PCR 4 ; however, previous reports did not consider some of the more extreme high temperature conditions present in tropical and subtropical areas of the country or evaluate the effect on PCR.
Despite increased awareness of the disease and implementation of control strategies, trichomoniasis continues to cause major economic loss in the U.S. cattle industry. 1 Texas and many other states have recently implemented control programs in which bulls sold for breeding purposes must be certified as virgin bulls or have negative official test results. Approved tests currently recognized by the Texas Animal Health Commission are based upon culture or quantitative real-time PCR (qPCR). Under current regulations in Texas, bulls must be tested negative on 1 qPCR assay, or negative on 3 consecutive culture tests, each conducted at least 7 days apart. In support of the new regulatory program, qPCR protocol was evaluated and introduced for routine diagnostics. However, little information is available regarding the proper temperature and length of time for shipping that would ensure accurate identification of T. foetus DNA by qPCR. The objective of the current study was to evaluate transport time and environmental temperatures that may affect the outcome of culture and qPCR assays for the diagnosis of T. foetus in cattle.
Isolates of T. foetus were obtained from routine clinical cases. Culture-based diagnosis for T. foetus was carried out by examining 37°C cultured Trich pouches a daily for 6 days for the presence of motile trichomonads. DNA extraction and subsequent qPCR was performed as previously described. 4 Master mix contained primers b and probes b at a concentration of 500 nM and 300 nM each, respectively, and 12.5 µl of TaqMan universal PCR master mix b in a 25-µl total volume reaction. Temperature and time of thermocycling parameters used in the present study were as recommended by the master mix manufacturer b : 1 cycle of 50°C for 2 min and 95°C for 10 min followed by 40 cycles of 95°C for 30 sec and 60°C for 1 min. Results obtained by qPCR equal to or higher than a threshold cycle (Ct) value of 35 were considered negative.
The analytical sensitivity of the qPCR was calculated by preparing serial dilutions of a field isolate of T. foetus that were quantified using a hemocytometer. Serial 10-fold dilutions were made, and triplicate pouches were spiked with 200 µl of each dilution. After culture at 37°C for 48 hr, a sample of 500 µl was taken and tested by qPCR. The limit of detection on day 0 of the qPCR assay was determined to be 57 cells/500 µl with an average Ct value of 33.79 (Table 1).
Detection limits of culture and quantitative real-time polymerase chain reaction (qPCR) results for the detection of Tritrichomonas foetus.*
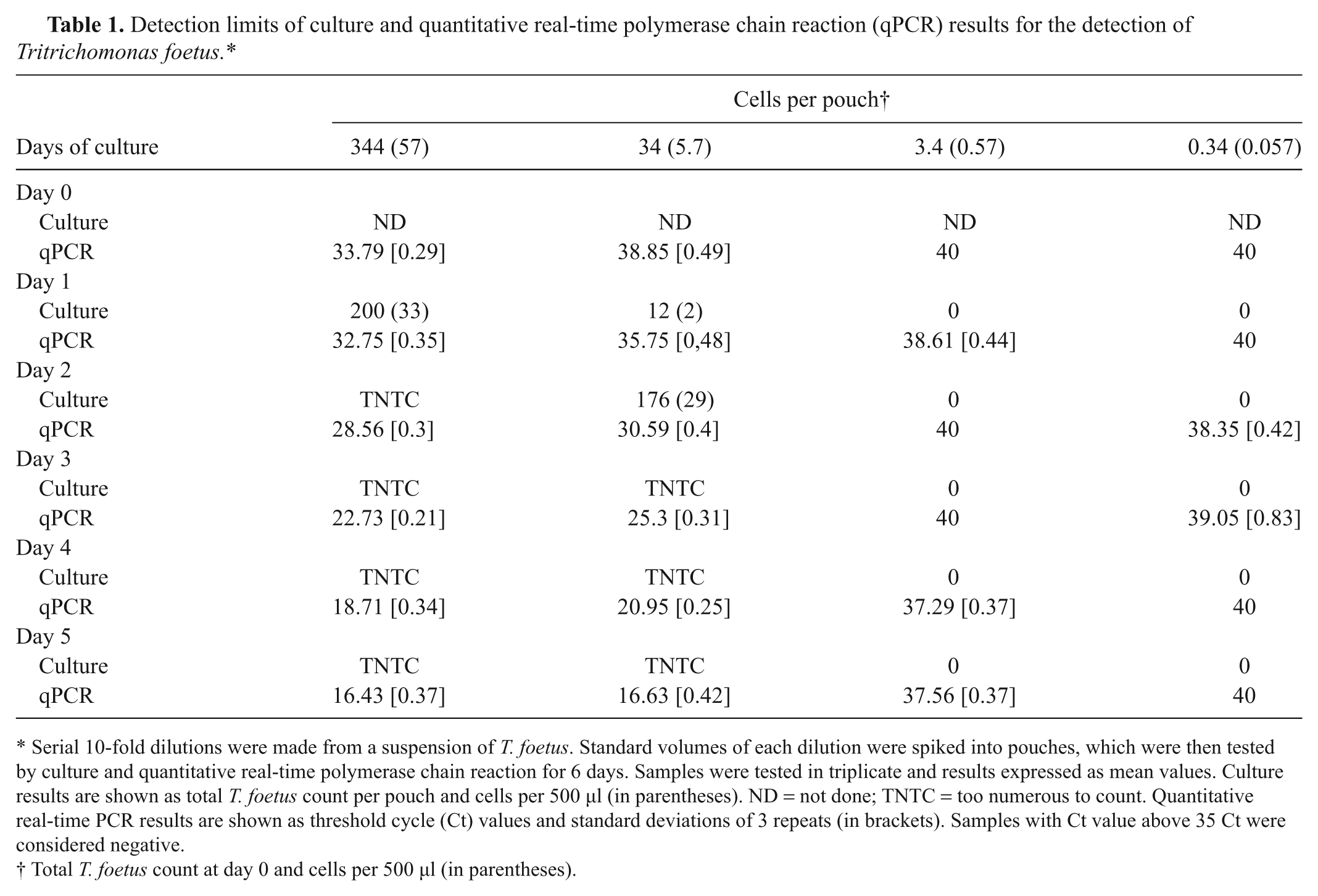
Serial 10-fold dilutions were made from a suspension of T. foetus. Standard volumes of each dilution were spiked into pouches, which were then tested by culture and quantitative real-time polymerase chain reaction for 6 days. Samples were tested in triplicate and results expressed as mean values. Culture results are shown as total T. foetus count per pouch and cells per 500 µl (in parentheses). ND = not done; TNTC = too numerous to count. Quantitative real-time PCR results are shown as threshold cycle (Ct) values and standard deviations of 3 repeats (in brackets). Samples with Ct value above 35 Ct were considered negative.
Total T. foetus count at day 0 and cells per 500 µl (in parentheses).
To evaluate the minimum incubation time necessary for reproducible PCR results, serial dilutions of T. foetus were inoculated into Trich pouches and then tested by culture and qPCR daily for 6 days. Starting with the day the pouches were inoculated (day 0), 500 µl of each dilution was aspirated from each pouch using a sterile plastic pipette, and qPCR was performed. Results indicate that qPCR after 48 hr of incubation at 37°C gave a positive result on a pouch initially inoculated with approximately 34 trichomonads (average Ct value of 30.59; Table 1) or the equivalent to 29 organisms per PCR reaction (500 µl). At this concentration, incubation of pouches at 37°C for more than 48 hr allowed further replication of T. foetus organisms and reduction of Ct values as would be expected. At higher dilutions, with a theoretical concentration of initial cell count of less than 3.4 organisms per pouch, culture was negative and qPCR was unable to detect T. foetus DNA, giving no Ct value or Ct values above the negative threshold of Ct >35 (Table 1).
The effect of temperature during transport was investigated using Trich pouches inoculated with T. foetus to a final calculated concentration of 10 parasites per ml from the average of 3 replicates. Four temperature conditions were evaluated: 4°C, 20°C, 37°C, and 42°C, representing various temperatures to which diagnostic samples may be exposed during transport to a veterinary diagnostic laboratory. In addition to temperature conditions, 3 different time durations of transport were evaluated (24 hr, 48 hr, and 72 hr). Pouches were prepared for each temperature condition and transport duration. Pouches were stored at their respective temperature and time combinations in a controlled temperature environment. Pouches were then transferred to a 37°C incubator, simulating arrival at a diagnostic laboratory and designated as day 0. After mixing, a 500-µl aliquot from each pouch was removed and examined microscopically once a day for 6 days. Aliquots from each pouch were aspirated after microscopic examination and tested in triplicate by qPCR. Pouches stored at 4°C for 24 hr, 48 hr, and 72 hr revealed a steady growth of organisms following 2 days of incubation at 37°C (Table 2), indicating that organism viability was not compromised. The Ct values decreased until day 4, indicating that the parasites replicated well upon further incubation at 37°C. After day 4, the growth of parasite plateaued, possibly because of the reduction of available nutrients in the culture media and by the toxic by-products of parasite growth. Samples stored at the 20°C simulated transport conditions resulted in a similar pattern of growth. However, samples stored at 37°C showed a rapid decrease of Ct values and reached plateau after day 2 (Table 2). In contrast, samples stored at 42°C were consistently negative after culture in microscopic examination and qPCR. Few qPCR results gave a positive mean value close to the cutoff value of the assay.
Effects of transport temperature on culture and quantitative real-time polymerase chain reaction (qPCR) results for the detection of Tritrichomonas foetus.*
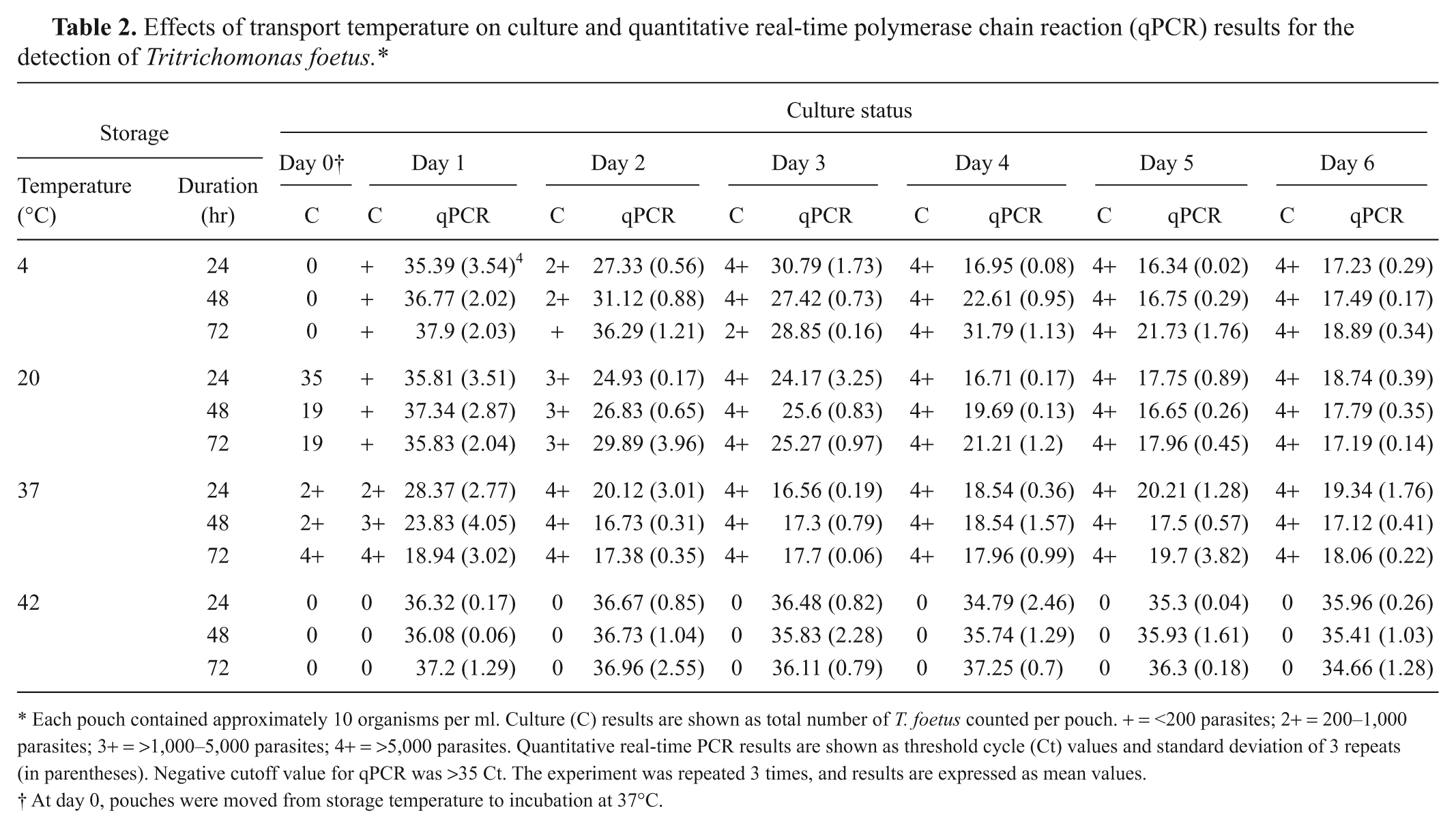
Each pouch contained approximately 10 organisms per ml. Culture (C) results are shown as total number of T. foetus counted per pouch. + = <200 parasites; 2+ = 200–1,000 parasites; 3+ = >1,000–5,000 parasites; 4+ = >5,000 parasites. Quantitative real-time PCR results are shown as threshold cycle (Ct) values and standard deviation of 3 repeats (in parentheses). Negative cutoff value for qPCR was >35 Ct. The experiment was repeated 3 times, and results are expressed as mean values.
At day 0, pouches were moved from storage temperature to incubation at 37°C.
Samples stored at 4°C for up to 72 hr and then culture at 37°C for 2 days, gave positive results in both culture and qPCR. Similar culture results were reported in a previous study. 2 The same study reported that cultures samples kept more than 5 days at 4°C gave negative culture results, stressing the assertion that practitioners and diagnosticians must reduce the period in which samples are maintained at 4°C to less than 3 days. Another study found that pre-storage of samples for up to 5 days at 4°C before qPCR would reduce the sensitivity up to 31%. 11 However, qPCR was performed directly from Trich pouches without previous incubation for 2 days as reported in the current work, which could explain the observed reduction in sensitivity. Although there may be some reduction of cell viability at 4°C, it was compensated by an increase in the number of parasites after a 2-day incubation period at 37°C.
In general, temperatures above 37°C are detrimental for T. foetus. The present study indicated that storage or transport of samples for as little as 24 hr at 42°C before arrival at the laboratory will kill the parasites, and no further growth was achieved after 2 days incubation as evidenced by culture negative results. However, some T. foetus DNA remained in the media giving weak positive results (near the threshold), or negative results with Ct values of 35–37 (Table 2). A 2010 study reported that pouches incubated at 42°C or 54°C for 24 hr were negative by culture but positive by qPCR. 4 These results are in conflict with what is reported herein, as qPCR results at 42°C were mostly negative. This disparity may be explained by the reduced cut-off of 35 Ct in the present study, which was selected to increase the specificity of the assay. In addition, current experiment started with a limited concentration of parasites close to the detection limit of the assay of 30 organism/pouch, as compared with 4,000 organism/pouch used in the previous study. 4 The average amount of T. foetus in a sheath scraping collected from experimentally infected bulls is estimated to be 50 organisms per ml and 141 organisms per ml in naturally infected bulls. 11 In actual practice, since most pouches are typically inoculated with 1 ml or less of sample, the experimental work reported herein used a number of parasites close to what will be found in a naturally infected bull, and therefore may illustrate results closer to what would be expected with actual diagnostic specimens. The previous study 4 concluded that high temperatures affect culture results of T. foetus but do not negatively influence the ability of qPCR to detect the organism. 4 Data gathered in the current study indicate that when a limited concentration of parasites is present, as may occur in naturally infected bulls, high temperatures will have a detrimental effect in as little as 24 hr, and that these samples will likely give negative results by culture as well as qPCR.
In summary, appropriate measures should be taken to protect clinical samples from high temperatures. Field veterinarians should strive to ensure that samples are maintained at temperatures of 4–37°C during shipment and are delivered to the laboratory within 24 hr of collection or following an incubation period at 37°C to allow amplification of low numbers of organisms.
Footnotes
Acknowledgements
The authors wish to thank the Molecular and Bacteriology sections at the Texas Veterinary Medical Diagnostic Laboratory for their technical assistance.
a.
InPouch™ TF Media, Biomed Diagnostics Inc., White City, OR.
b.
Applied Biosystems, Foster City, CA.
This work was supported by the funds from the Texas Veterinary Medical Diagnostic Laboratory.
The authors declared that they had no conflicts of interests in their authorship and publication of this contribution.
