Abstract
To characterize Feline parvovirus (FPV) circulating in domestic cats in Brazil, 51 fecal samples from unvaccinated domestic cats were collected during 2004–2005. Six parvoviruses were characterized by hemagglutination (HA) assay at different pH values and temperatures and by polymerase chain reaction (PCR) using different pairs of primers. However, data obtained from HA and PCR did not allow the discrimination between FPV and Canine parvovirus (CPV). Two regions of the VP2 capsid gene (1,171-bp fragment) involved in controlling canine and feline host range were sequenced; 9 synonymous and 10 non-synonymous nucleotide substitutions were detected. All samples were confirmed as FPV by nucleotide sequencing, but 3 feline samples had amino acid changes at residues 93, 375, and 426, which are present in canine strains. The phylogenetic tree built based on nucleotide sequences showed that Brazilian feline samples form a cluster distinct from other parvoviruses deposited in GenBank. Taken together, the findings reinforce the importance of monitoring the continuous evolution of CPV and FPV in the feline population in Brazil.
Feline parvovirus (FPV) and Canine parvovirus-2 (CPV-2) are responsible for a highly contagious enteric disease in cats and dogs. Feline parvovirus–associated disease, also recognized as feline panleukopenia, has been recognized since the 1920s. 17 A host-range variant of FPV, CPV-2 emerged as a new virus in dogs during the late 1970s. Soon after the appearance of CPV-2, the original virus was replaced by new variants, CPV-2a and CPV-2b. 17 In the early 2000s, a novel CPV mutant (CPV-2c) emerged in Italy. 2
Feline parvovirus and CPV-2 are more than 98% identical at the nucleotide and amino acid sequence level.5,17 Six amino acid changes distinguish FPV from CPV-2 in the open reading frame encoding VP1/VP2 structural proteins (residues 80, 93, 103, 323, 564, and 568). 5 Feline parvovirus or CPV-2 host range is determined by combinations of residues 93, 103, and 323, 5 and another 5 amino acid changes (residues 87, 300, 305, 426, and 555) distinguish the new variants CPV-2a, -2b, and -2c from the original CPV-2. 2 Feline parvovirus may also differ from CPV-2 in pH and temperature dependence of hemagglutination (HA). The FPV-like viruses hemagglutinate porcine and rhesus macaque erythrocytes only in buffers with a pH less than 6.4, while CPV-2 isolates hemagglutinate at all pH values between 6.0 and 8.0. The CPV-2 isolates are also able to hemagglutinate at 4°C or 37°C.5,14Amino acid residues 323 and 375 have been reported to control the pH and temperature dependence of HA. 5
Canine parvoviruses are the most common viral agents associated with acute enteric clinical signs in young dogs up to 6 months of age in Brazil.4,19 However, data regarding parvovirus infection in domestic cats has not yet been reported. To characterize FPV circulating in domestic cats in Brazil, 51 fecal samples from 46 diarrheic and 5 asymptomatic unvaccinated domestic cats were collected in Rio de Janeiro from 2004 to 2005.
The 51 samples were screened for the presence of parvovirus by HA tests using borate buffered saline and “virus-adjusting diluent” (pH 6.0) as diluents for antigen and porcine erythrocytes, respectively. 15 In addition, positive samples were tested by HA at different pH values (6.0 and 7.4) with incubation at 4°C and 37°C. 14
For genome detection, viral DNA was extracted from 10% fecal suspensions in Tris–calcium using a combination of phenol–chloroform–isoamyl alcohol a and silica–guanidine thiocyanate. 6 A polymerase chain reaction (PCR) was performed on each DNA extract using the primer pair VPF/VPR (2285-4530), which amplifies FPV and all genotypes of CPV. 10 Each amplicon was then subjected to a nested PCR assay using 3 pairs of primers to differentiate FPV from CPV-2: FMF/FMR (3113-3810) to identify FPV, P2 (3025-3706) to distinguish CPV-2, and P2ab (3025-3706) to amplify both CPV-2a and CPV-2b.9,16
For sequence analysis, 3 different pairs of primers were used for partial amplification of the VP1/VP2 capsid protein gene as previously described2,17: primers M1/M2 (2949-3150), M13/M14 (3629-4111), and 555F/555R (4003-4561). The PCR products were purified using a PCR purification kit b and subjected to direct sequencing c in both directions. 11
A partial consensus sequence of VP2, obtained by assembling the nucleotide sequences of different amplicons was aligned and analyzed by BioEdit Sequence Alignment Editor version 7.0.5. d The nucleotide sequences generated in the current study have been deposited in GenBank under the following accession numbers: RJF606/04 (DQ507872, DQ658146, GQ415003), RJF616/04 (DQ507873, DQ658147, GQ415004), RJF617 (DQ507874, DQ658148, GQ415008) RJF618/04 (DQ507875, DQ658148, GQ415005), RJF620/04 (DQ507876, DQ658149, GQ415006), RJF628/04 (DQ507877, DQ658150, GQ415007). The FPV sequences from GenBank were selected for comparison from the United States (M38246), Japan (AB000070), France (AY606131), Germany (AY742937), Argentina (EU018145), China (FJ936171), and Italy (EU498693). Mink enteritis virus and Blue fox parvovirus from China (HM0158241 and EU698028, respectively) and CPV sequences from the United States (M38245, M24000, and M74849) were also selected for comparison (Table 1).
Nucleotide (nt) and amino acid (aa) differences in the VP2 sequences of 6 Feline parvovirus strains from Rio de Janeiro, Brazil and other Feline and Canine parvovirus sequences obtained from the GenBank database.*
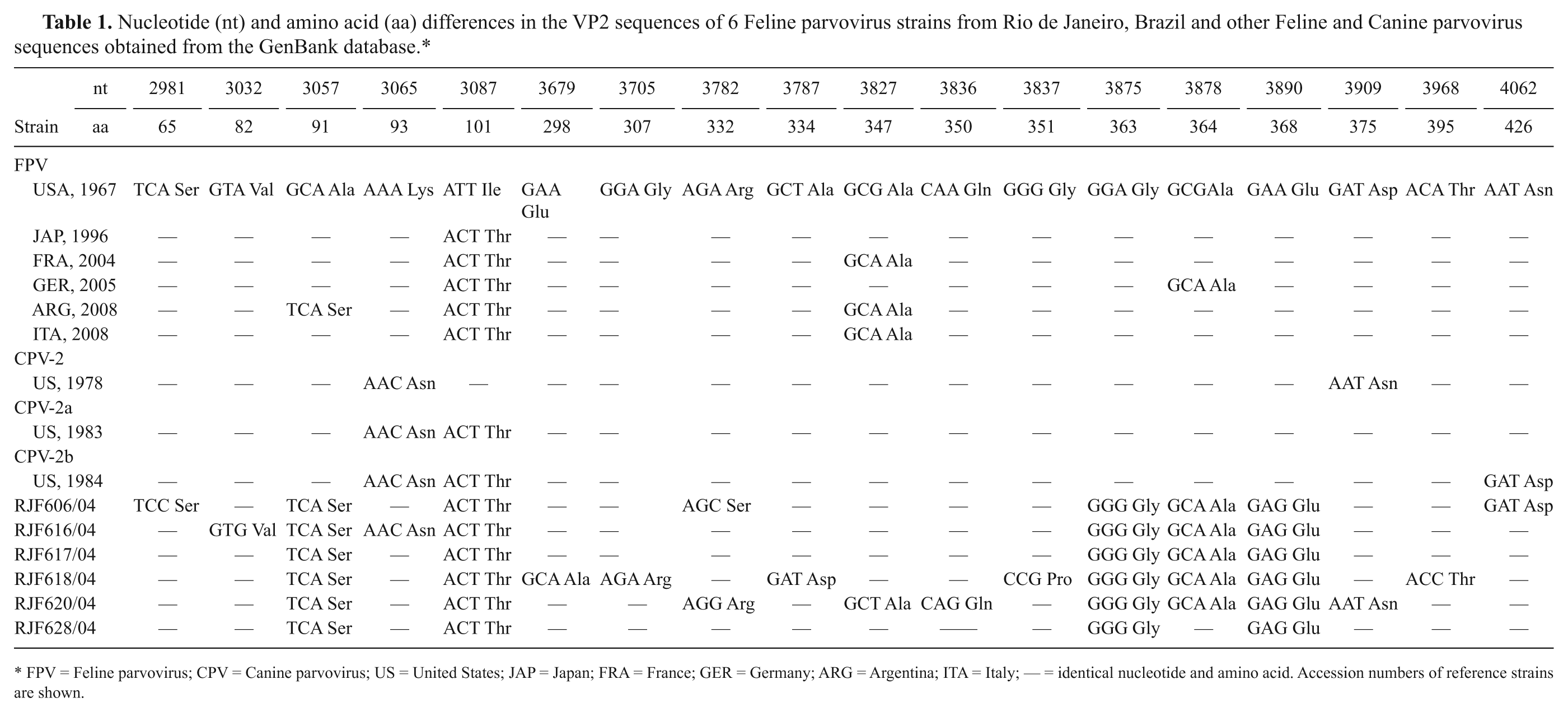
FPV = Feline parvovirus; CPV = Canine parvovirus; US = United States; JAP = Japan; FRA = France; GER = Germany; ARG = Argentina; ITA = Italy; — = identical nucleotide and amino acid. Accession numbers of reference strains are shown.
The unrooted phylogenetic tree from a 932-bp fragment of the VP2 gene (3629-4561) was constructed according to the neighbor-joining method and subjected to 2,000 bootstrap samplings under the Tamura–Nei model with MEGA software, version 4.0. e The FindModel program 13 was used to identify the optimal evolutionary model that best fit the sequence dataset.
Six feline fecal samples were positive by HA. An HA pH dependence was observed in only 1 sample, RJF618, for which the HA titer at pH 7.4 was decreased 32-fold when compared to the HA titer at pH 6.0. Five out of 6 viruses tested did not exhibit the FPV HA pH dependence and requirement for low temperature.
In addition, a PCR typing approach was used to characterize the parvoviruses in feline fecal samples. All 6 samples were amplified with the primer pairs FMF/FMR and P2. The DNA from 4 samples (RJF606/04, RJF616/04, RJF618/04, and RJF620/04) was also amplified with the primer pair P2ab, which is considered specific for detection of the new types of CPV. 9
Amino acid sequences were inferred from DNA sequences, and all 6 viruses were identified as FPV. All amino acid residues used to differentiate between FPV and CPV-2 (80-Val, 93-Lys, 103-Val, 323-Asp, 564-Asn, and 568-Ala) were conserved. 5 No changes were observed at residues 323 and 375 in the only strain displaying HA pH dependence (RJF618).
Of the nucleotide changes identified in the VP2 partial sequence, 9 were synonymous and 10 were non-synonymous substitutions. All FPV strains had 2 non-synonymous substitutions at residues 91 (Ala→Ser) and 101 (Ile→Thr) and 2 synonymous substitutions at residues 363 and 368 (Table 1). Also, amino acid changes at residues 93 (Lys→Asn), 375 (Asp→Asn), and 426 (Asn→Asp) were found in 3 feline samples: RJF616/04, RJF620/04, and RJF606/04 (Table 1). The phylogenetic tree built based on the nucleotide sequences showed that Brazilian feline samples formed a cluster distinct from the other parvovirus DNA sequences deposited in GenBank, which probably reflects their geographic origin (Fig. 1).
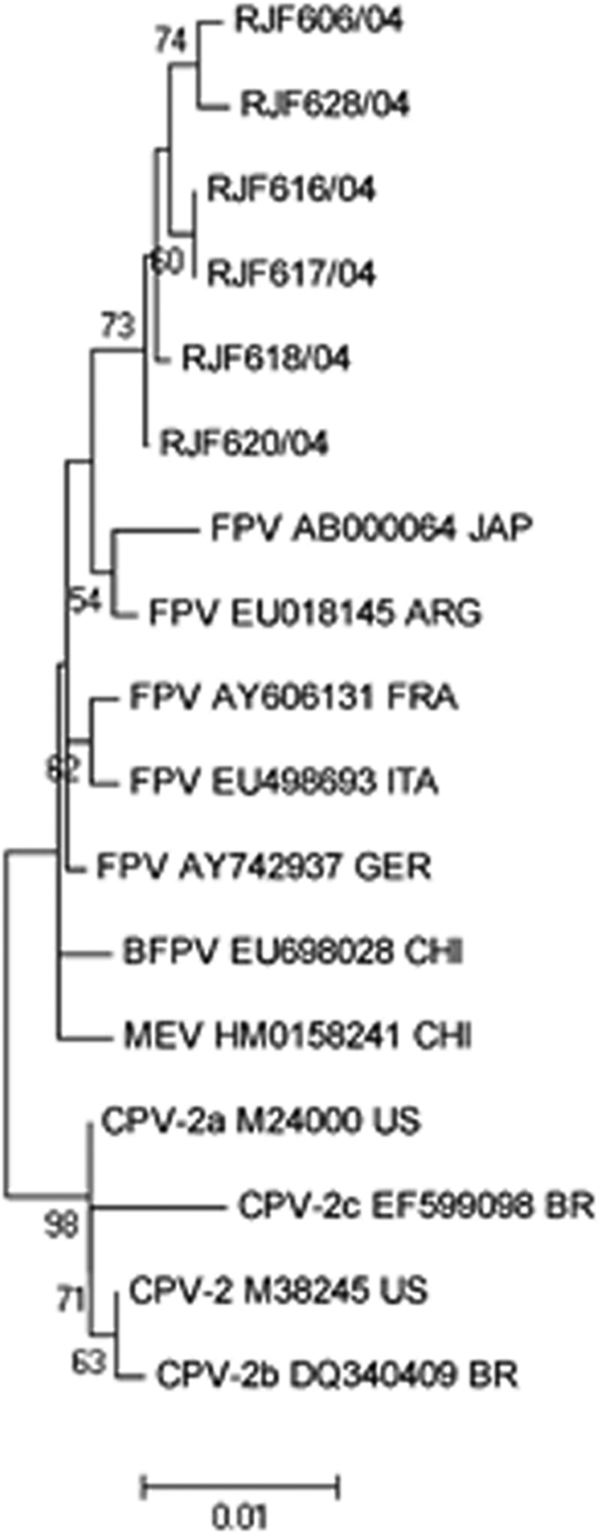
Phylogenetic tree based on the 932-bp fragment of the VP2 gene nucleotide sequences (nucleotide [nt] 3629-4561) of the Brazilian Feline parvovirus (FPV) strains characterized in the current study and other sequences obtained from the GenBank database. Accession numbers of reference strains are indicated. Bootstrap percentages are shown above the branches that are supported in at least 50% of the 2,000 replicates.
Since its first identification in 1920, FPV has not undergone significant changes in antigenic and biological properties. 8 Although FPV displays HA pH and temperature dependence, which distinguishes it from CPV-2 isolates,14,15 the samples examined in the current study could not be characterized by these parameters. Likewise, a PCR typing approach was not able to distinguish FPV from CPV.
To confirm the genomic identity of Brazilian FPVs, 2 regions of the VP2 gene involved in controlling canine and feline host range were sequenced. The Brazilian strains displayed non-synonymous substitutions at residues 91 and 101, as described previously. The substitution at residue 91 has only been reported in FPVs after 2006 from cats in Italy and Argentina. 8 Although amino acid 91 is located in the vicinity of residue 93 on the top of the 3-fold spike of the capsid and is one of the residues involved in determining parvovirus host range,5,20 the exact phenotypic consequence of these mutations is not yet clear. The change at residue 101, which is also present in the new CPV variants, has been observed in FPV strains since the beginning of the 1990s. 8
Based on the viral DNA sequences analyzed in the current study, there was no evidence of CPV infection in kittens despite its widespread distribution in the canine population in Brazil.4,19 Nevertheless, amino acid changes at residues 93, 375, and 426, which are present in canine strains, were found in 3 feline samples, and these residues have been shown to affect parvovirus host range and HA.5,12,20
Feline parvovirus has a certain degree of genetic stability, 1 and the detection of anomalous strains with mutations characteristic of canine strains might indicate that recent FPV strains are undergoing changes in their biological properties. The CPV-2a, -2b, and -2c viruses have acquired the ability to infect, replicate, and cause clinical disease in cats as well as in dogs.1,7,9 CPV-2c has been detected in puppies with enteric disease in Brazil3,18 and may represent a threat to the cat population.
In summary, the current study reports the molecular characterization of FPV strains in Brazil. Although CPV infection is widespread in Rio de Janeiro, no evidence of CPV infection in domestic cats was detected. The detection of 3 feline strains with amino acid changes characteristic of CPV demonstrates the importance of monitoring these viruses in order to evaluate whether CPV will be able to replace FPV in the feline population in Brazil. The results also emphasize that sequence analysis constitutes an important tool for monitoring and characterizing FPV strains circulating in the feline population and represents a further contribution to the study of parvoviruses in Brazil.
Footnotes
Acknowledgements
The authors thank Dr. Paul M. Fricke (University of Wisconsin) for the English revision of the manuscript.
a.
UltraPure™ Phenol:Chloroform:Isoamyl Alcohol (25:24:1), Invitrogen, Sao Paulo, Brazil.
b.
QIAquick® DNA Mini Kit, Qiagen Ltd., West Sussex, United Kingdom.
c.
ABI 3130xl Genetic Analyzer, Big Dye® Terminator Cycle Sequencing Kit; Applied Biosystems, Foster City, CA.
d.
Sequence Alignment Editor 7.0.1, Ibis Biosciences Inc., Carlsbad, CA.
e.
Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0, Center of Evolutionary Functional Genomics, Biodesign Institute, AZ.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The current study was supported by the Brazilian National Council for Scientific and Technological Development (CNPq), Fluminense Federal University, Office of Research and Graduate Studies (UFF-PROPP), Carlos Chagas Filho Foundation for Research Support of the State of Rio de Janeiro (FAPERJ), and Oswaldo Cruz Institute (IOC-Fiocruz). T. X. Castro is a post-doc recipient of scholarship from CNPq and S.C. Miranda was a master degree student and recipient of scholarship from Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES).
