Abstract
Diagnosis of canine parvovirus type 2 and feline panleukopenia virus infection in dogs and cats may be hampered by the severity of enteric lesions, secondary bacterial overgrowth, and rapid onset of autolysis. In contrast to small intestine, tongue epithelium is less sensitive to postmortem changes. Sections of tongue and small intestine from 11 dogs and 11 cats with a clinical history and gross and microscopic lesions compatible with canine and feline parvoviral infection were examined for parvoviral infection using real-time polymerase chain reaction (PCR), immunohistochemistry (IHC), and direct fluorescent antibody testing (FA). Parvoviral DNA was detected by PCR in both small intestine and tongue of all but 1 dog. Nineteen of 22 animals (86%) with suspect or positive FA staining in the small intestine also had positive FA and IHC staining in the tongue. Three of 3 dogs (100%) whose carcasses had been frozen and thawed prior to necropsy had more consistently positive staining in tongue than in small intestine by FA and IHC. These data confirm tongue as an excellent complementary sample for parvoviral testing in dogs and cats, especially in cases in which postmortem autolysis has occurred.
Canine parvovirus type 2 (CPV-2), which emerged as an important enteric pathogen in dogs in the late 1970s, 9 and feline panleukopenia virus (FPV), the etiologic agent of feline distemper, panleukopenia, and feline ataxia in cats, 4 are closely related members of the family Parvoviridae. 2,8 Canine and feline parvoviral infections are common in dog and cat populations. The high level of virus shedding and prolonged survival in the environment make identification of active disease extremely important, especially in high-density animal housing facilities such as shelters or kennels. Parvoviral replication is dependent on host-cell DNA polymerases produced in the S phase of the cell cycle and therefore requires actively dividing cells. 11,12 Virus replication results in the destruction of germinal epithelial cells in the intestinal crypts, leading to villus shortening, vomiting and diarrhea, and lymphoid necrosis and destruction of myeloproliferative cells. Adequate samples of intestinal mucosa are often not available for evaluation because of either postmortem changes or freeze-thaw artifacts.
Previous studies have identified intranuclear viral inclusions and pseudocytoplasmic inclusions within the epithelium of the tongue of dogs infected with CPV-2. 3,6 These studies indicate that tongue appears to support virus replication and therefore could be used as a complementary sample to the diagnosis of parvovirus disease. Based on these previous reports and cases observed at the Diagnostic Center for Population and Animal Health at Michigan State University, the authors hypothesized that parvovirus infection is associated with epithelial lesions in the tongues of cats and dogs, that tongue is a good sample for the diagnosis of parvovirus, and that compared to small intestine, tongue may be a superior sample to detect canine and feline parvovirus in cases with postmortem autolysis.
Study animals included 11 dogs and 11 cats between the ages of 1 month and 2 years that had been submitted to the Diagnostic Center for Population and Animal Health for necropsy with a clinical history of vomiting and/or bloody diarrhea, gross evidence of segmental enteric hyperemia with distended and fluid-filled intestines, and histologic evidence of small intestinal crypt necrosis, consistent with parvoviral disease. 1 Three dogs, ranging in age from 4 to 6 months, and 3 cats, ranging in age from 7 to 13 years, submitted to the Diagnostic Center for necropsy examination with no clinical, gross, or histologic signs suggesting parvoviral disease were used as negative controls. Control animal tissues contained no detectable parvovirus based on all diagnostic testing, including fluorescent antibody testing (FA), immunohistochemical staining (IHC), and polymerase chain reaction (PCR). Tongues collected from all animals with sterile instruments were sectioned into approximately 2.0-cm-thick sections from tip to base, including the tonsillar margin. Three to 4 small intestinal sections were collected randomly from grossly affected areas of the distal jejunum and ileum with sterile instruments. Alternating sections of tongue and small intestine were either placed in 10% neutral buffered formalin or frozen at −20°C.
Of the study animals examined, 4 of 11 dogs (36.3%) and 5 of 11 cats (45.5%) had been vaccinated at least once for parvovirus (Table 1). The carcasses of 3 of the dogs (27.3%) had been frozen and thawed prior to necropsy (Table 1). Of the negative control animals, 2 of the 3 dogs and all 3 cats had not been vaccinated within 4 weeks of necropsy examination. One of the 3 control dogs was moderately autolyzed at necropsy examination.
Parvovirus test results for dogs included in the study.
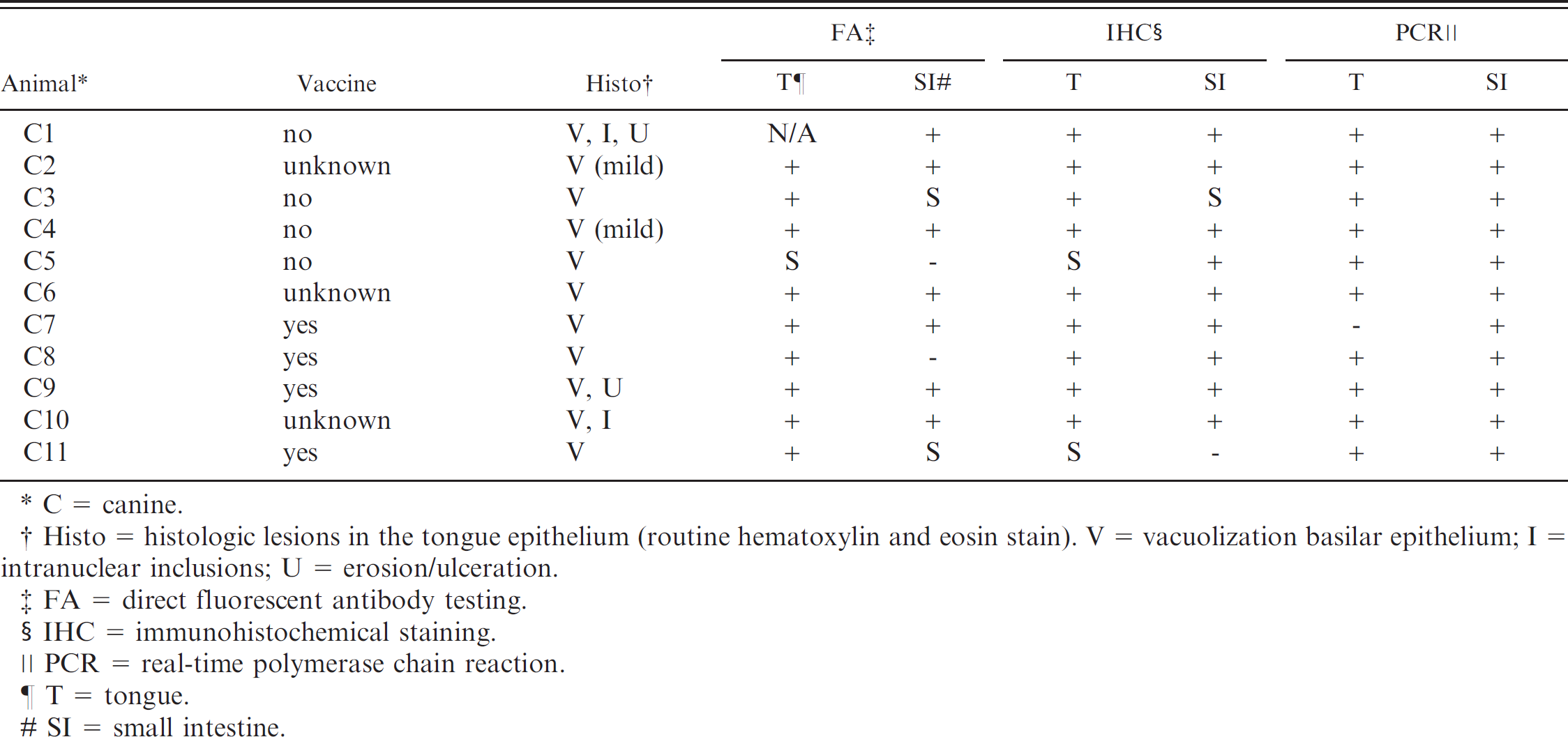
C = canine.
Histo = histologic lesions in the tongue epithelium (routine hematoxylin and eosin stain). V = vacuolization basilar epithelium; I = intranuclear inclusions; U = erosion/ulceration.
FA = direct fluorescent antibody testing.
IHC = immunohistochemical staining.
PCR = real-time polymerase chain reaction.
T = tongue.
SI = small intestine.
Serial sections of frozen small intestine and tongue were cut on a cryostat at a thickness of 3–5 μm. Sections were placed on slides, flooded with FITC-conjugated anti-CPV-2/FPV antibody at a ready-to-use dilution a or with canine coronavirus conjugate as a negative control, and examined with ultraviolet illumination for specific fluorescence. Samples were considered positive if more than 1 cell had a positive fluorescent signal and suspect if only a single fluorescent cell was detected.
Immunohistochemical staining was performed on formalin-fixed paraffin-embedded tissue sections, following a previously described protocol. 10 A rabbit-polyclonal anticanine/feline parvovirus antibody b was applied for 30 minutes at a 1: 400 concentration at room temperature. Sections were stained with an autostainer using a labeled streptavidin-immunoperoxidase staining procedure c as previously described. 5 Sections of small intestine with parvoviral lesions that had tested positive by PCR served as positive controls. For the negative controls, the primary antibody was replaced with Tris-HCl buffer. Tissues were evaluated as positive if more than 1 cell had positive red- brown intranuclear and intracytoplasmic staining for parvovirus. Suspect tissues had only 1 cell with positive red-brown intranuclear and intracytoplasmic staining for parvovirus.
A pair of primers was designed from the viral capsid protein gene (VP2) sequence of CPV-2 and FPV using the OLIGO 6 primer analysis software. d The primer pair, 5' AGC TGA AGG AGG TAC TAA CTT 3' (forward primer) and 5' GCC TCA AAA GAA TAA TAT GGT 3' (reverse primer), targeted a 153-base pair (bp) segment of the VP2 gene (888-1040; nucleotide position in VP2 gene, Genbank Accession no. AB115504). DNA was extracted from sections of fresh or formalin-fixed, paraffin-embedded tongue and small intestinal samples using the QIAGEN DNeasy Tissue Kit. e Citrisolv f was used in the deparaffinization step. DNA was eluted in 100 μl of the kit elution buffer. The sensitivity of the test was determined using plasmid pB1625 containing the entire CPV genome, obtained from Dr. C. Parrish, Cornell University. The detection limit of the real-time CPV2 PCR assay was determined to be 485 plasmid copies. The specificity of the real-time PCR was confirmed based on the sequencing of amplification products obtained from 4 different field strains of CPV2.
Real-time PCR was performed using the Quantitect SYBR Green PCR Kit g with an optimal primer concentration of 0.5 μM in a final reaction volume of 50 μl, containing 5 μl of template DNA. A thermocycler with an integrated real-time optical detection system h was used for PCR amplification. The optimized cycling conditions were as follows: predenaturation at 95°C for 15 minutes, followed by 40 cycles of 94°C for 30 seconds, 53 °C for 30 seconds, and 72°C for 30 seconds. A postamplification step at 55°C for 1 minute was followed by an 80-cycle melt curve analysis consisting of raising the incubation temperature from 55°C to 95°C in 0.5° increments every 10 seconds, to determine the amplicon melting temperature. A positive control (canine parvovirus or feline parvovirus DNA) and a negative control (sterile water) were included with every run. Samples with a melting temperature at 77.5°C were considered positive based on the verified melting temperature of the sequenced positive control amplicons. The expected size (153 bp) of all realtime PCR positive samples was verified by gel electrophoresis.
Gross and histologic lesions consistent with parvoviral disease were observed in the intestinal samples from all 22 study animals (100%) (Fig. 3). Test result comparisons for all study animals are summarized in Tables 1 (canine) and 2 (feline). All study animals were positive for parvoviral DNA by PCR on sections of small intestine and tongue, except for 1 dog (C7) who tested positive in the small intestine only. The small intestine from the exception was positive for parvoviral DNA by PCR, and the tongue was negative by PCR, but IHC and FA were positive. Negative PCR results for a parvovirus-infected animal may have been due to the absence of viral DNA, a sample with undetectable amounts of parvoviral DNA, or a sample containing PCR inhibitors.
Parvovirus test results for cats included in the study.*
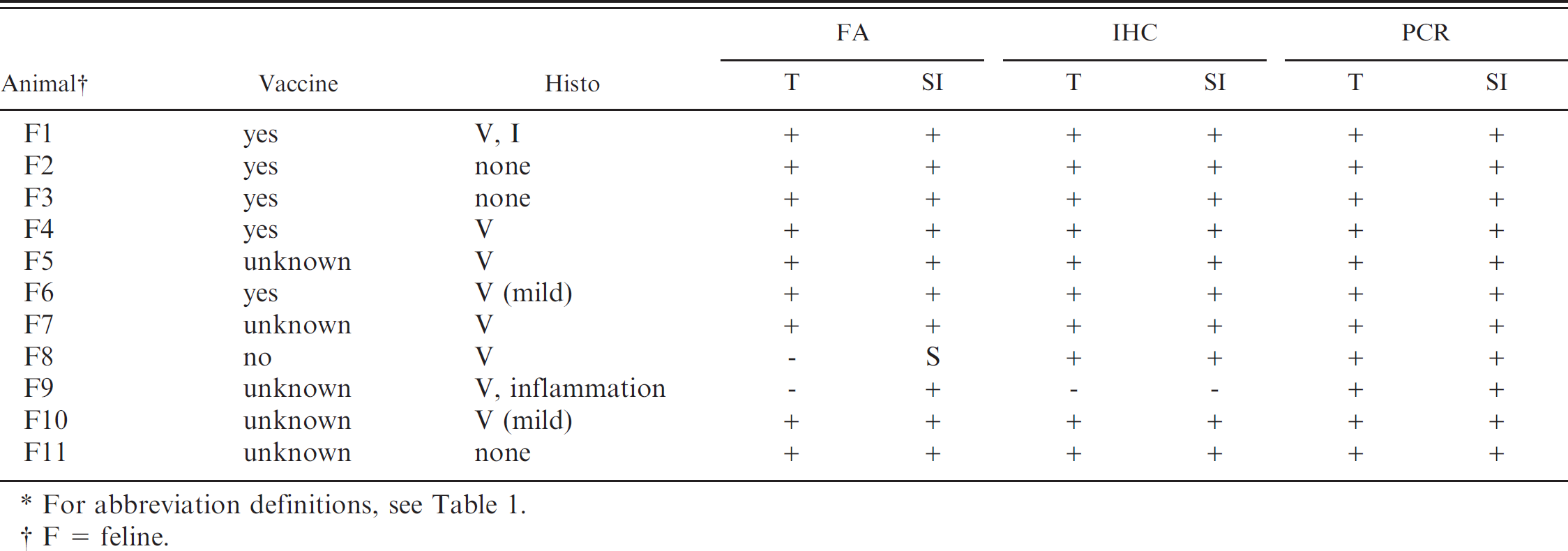
For abbreviation definitions, see Table 1.
F = feline.
Three study cats (27.3%) had focal erosions and ulcerations affecting the dorsal aspect of tongue epithelium (Fig. 1), which were associated with intralesional parvoviral antigen, observed with IHC. All animals tested negative for feline calicivirus by IHC and did not have evidence of renal disease. Microscopic lesions, characterized by focal to diffuse vacuolization and hydropic degeneration of epithelial keratinocytes, particularly along the basal layer (Figs. 2 and 5; Tables 1 and 2), which were often associated with parvoviral antigen observed by IHC (Fig. 6), were identified in the tongue of 11 study dogs (100%) and 8 study cats (72.7%).
Two study dogs (18.1%) and 1 study cat (9.1%) had intranuclear eosinophilic inclusions in affected keratinocytes (Fig. 2, Tables 1 and 2). Two study dogs (18.1 %) had epithelial attenuation and loss, consistent microscopically with erosion or ulceration, respectively (Table 1), associated with parvoviral antigen observed with IHC. Control animals had no gross or histologic lesions consistent with parvovirus, and no ulcerative lesions were observed in tongue tissue.
Both CPV-2 and FPV are transmitted orally and initially replicate in the lymphoid tissues of the oropharynx. 2,4,7 Tongue has been shown to contain viral inclusions in dogs and cats affected by canine and feline parvovirus, respectively, indicating active viral replication. 6 If parvovirus can actively replicate in tongue epithelium, then similar changes to those seen in intestinal crypt epithelial cells could be expected to occur in the tongue. Vacuolar changes are often present artifactually within keratinocytes of samples from carcasses, particularly those that have been frozen and thawed or have marked postmortem autolysis, whereas parvovirus-induced vacuolization should be accompanied by the presence of detectable antigen within affected cells.
The data demonstrate that tongue epithelium is a comparable sample to small intestine for detecting parvoviral infection by PCR and antigen detection (FA or IHC). Sections of small intestine from 7 dogs (63.6%) and 10 cats (90.9%) were positive, and 2 dogs (18.1%) and 1 cat (9.1%) were suspect for parvoviral antigen by direct FA. Nine dogs (81.8%) and 9 cats (81.8%) were positive and 1 dog (9.1%) was suspect for parvoviral antigen in epithelial keratinocytes of the tongue by direct FA. Fresh tongue tissue from 1 dog (C1) was not available for FA testing.
Sections of formalin-fixed, paraffin-embedded tongue and small intestine from study animals were examined for parvoviral antigen by IHC. Nine dogs (81.8%) and 10 cats (90.9%) showed diffuse positive staining in intestinal crypt epithelial cells (Fig. 4). Nine dogs (81.8%) and 10 cats (90.9%) showed diffuse positive staining for parvoviral antigen in tongue epithelial cells (Fig. 6).
In dogs, FA and IHC tests performed on tongue had a higher diagnostic sensitivity compared to small intestine and yielded no false-negative results when compared to the PCR data (Table 1). In animals that were positive by PCR but negative by FA and IHC, variations in test results could be due to a very small amount of viral antigen in both tongue and small intestine samples; in addition, submission of an uninfected section of tissue could also have led to negative test results. The segmental nature of a viral infection should always be considered when submitting samples, and multiple smaller samples from different areas may further increase diagnostic sensitivity. In histologic sections, only small segments of epithelium are stained by FA and IHC testing; therefore, samples from infected animals may appear falsely negative.
Since the small intestinal mucosa easily undergoes autolytic postmortem changes and is subject to severe freeze-thaw artifacts, false-negative PCR results and difficulties in interpretation of FA or IHC staining for CPV-2 and FPV occur. Furthermore, marked crypt necrosis followed by complete loss of parvovirus-infected cells and mucosal collapse may decrease the diagnostic sensitivity of these tests. In this study, the carcasses of 3 dogs (C3, C5, C11) had been frozen and thawed prior to examination. In all 3 dogs, samples of tongue were more consistently positive for canine parvovirus by IHC and FA than samples of small intestine (Table 1). In these cases, in contrast to small intestine, the tongue epithelium had retained its cellular integrity and tissue architecture, making interpretation of FA and/or IHC staining much easier and more reliable. Tongue epithelium, which is more resistant to artifactual changes, is therefore a more appropriate tissue for confirmatory parvoviral testing in severely autolyzed or frozen/thawed carcasses.
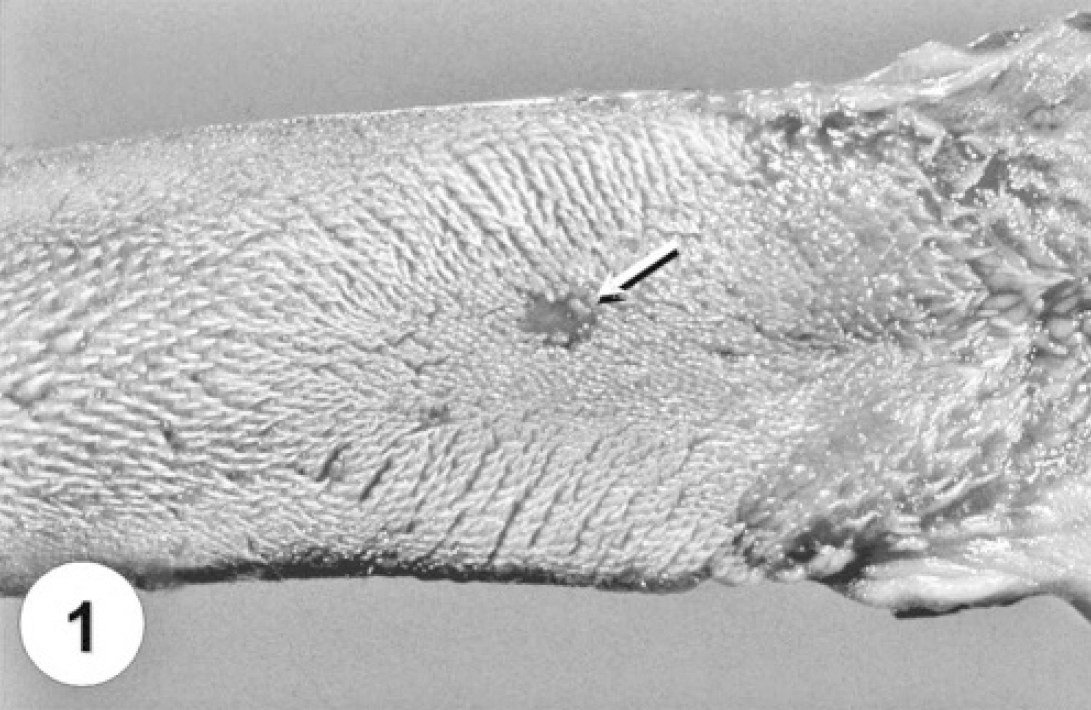
Tongue from a cat naturally infected with feline parvovirus. Note the focal area of ulceration on the dorsal surface, which is associated with intralesional parvoviral antigen identified by immunohistochemistry (arrow).

Tongue from a dog naturally infected with CPV-2. There is focal to diffuse vacuolization and hydropic degeneration of epithelial keratinocytes, particularly along the basal layer. Note the basophilic intranuclear inclusion within the epithelial cell (arrow). Hematoxylin and eosin staining. Bar = 50 μm.

Jejunum from a dog naturally infected with CPV-2. Characteristic histologic lesions of parvoviral enteritis, including mucosal collapse with severe crypt loss due to necrosis of lining epithelium (arrow). Hematoxylin and eosin staining. Bar = 150 μm.

Serial section of jejunum from the dog depicted in Fig. 3 stained with IHC for parvoviral antigen. Parvovirus antigen is present within degenerate crypt epithelial cells (white arrows) and superficial villus epithelium within affected areas. Immunohistochemistry for parvoviral antigen CPV-2a, hematoxylin counterstain. Bar = 150 μm.

Tongue from a dog naturally infected with CPV-2. There is marked epithelial cell vacuolar degeneration and nuclear pyknosis (arrows), which is particularly prominent within basilar epithelial cells. Hematoxylin and eosin staining. Bar = 40 μm.

Serial section of tongue from the dog depicted in Fig. 5 stained with IHC for parvoviral antigen. Parvovirus antigen is present within the nucleus of the basilar epithelial cells. Note that the cells with histologic evidence of vacuolar degeneration are staining positively (arrows). Immunohistochemistry for parvoviral antigen CPV-2a, hematoxylin counterstain. Bar = 40 μm.
Acknowledgements. We would like to acknowledge the hard work of the staff of the Histopathology and Immunohistochemical laboratories (T. Wood, K. Cicinelli, P. Schultz, S. Marsh), the Virology laboratory (C. Benson, M. Brown, J. Harmala) and the Anatomic Pathology Section (technicians: S. Thon, A. Wismer, B. VanKampen, C. Garrison; residents: Dr. M. Hoenerhoff, Dr. D. Jackson, Dr. Y. Jones, Dr. D. Rehagen, Dr. R. Smedley) at Michigan State University Diagnostic Center for Population and Animal Health.
Footnotes
a.
Canine parvovirus FITC conjugate, VMRD, Inc., Pullman, WA.
b.
CPV1-2a, Custom Monoclonals International, West Sacramento, CA.
c.
Labeled streptavidin-immunoperoxidase, Dako Cytomation, Carpinteria, CA.
d.
OLIGO 6 primer analysis software, Molecular Biology Insights, Cascade, CO.
e.
QIAGEN DNeasy tissue kit, QIAGEN, Inc., Valencia, CA.
f.
Fisherbrand Citrisolv Clearing Agent, Fisher Scientific, Pittsburgh, PA.
g.
Quantitect SYBR Green PCR kit, QIAGEN, Inc., Valencia, CA.
h.
iCycler iQ System with detection software v2.3B, BIO-RAD Laboratories, Hercules, CA.
