Abstract
The current study was undertaken to investigate the relation between serum C-reactive protein (CRP) concentrations and parameters of renal function in dogs with naturally occurring renal disease. Dogs were assigned to groups according to plasma creatinine concentration, urinary protein-to-creatinine ratio (UP/UC), and exogenous plasma creatinine clearance (P-ClCr) rates. Group A (healthy control dogs; n = 8): non-azotemic (plasma creatinine <125 µmol/l) and nonproteinuric (UP/UC <0.2), with P-ClCr rates >90 ml/min/m2; group B (n = 11): non-azotemic, nonproteinuric dogs with reduced P-ClCr rates (50–89 ml/min/m2); group C (n = 7): azotemic, borderline proteinuric dogs (P-ClCr rates: 22–67 ml/min/m2); and group D (n = 6): uremic, proteinuric dogs (not tested for P-ClCr). The serum CRP concentrations were measured via commercial enzyme-linked immunosorbent assay. The CRP concentrations in the clinically healthy dogs (group A) ranged from 2.09 mg/l to 8.60 mg/l (median: 3.21 mg/l). In comparison with dogs of group A, median CRP concentrations were significantly (P < 0.01) elevated in dogs of group B (17.6 mg/l, range: 17.0–19.2 mg/l), group C (24.8 mg/l, range: 18.0–32.5 mg/l), and group D (59.7 mg/l, range: 17.7–123 mg/l). Serum CRP was significantly related to P-ClCr (r = –0.83; P < 0.001), plasma creatinine (r = 0.81; P < 0.001), UP/UC (r = 0.70; P < 0.001), and leukocytes (r = 0.49; P < 0.01). The significant relations between serum CRP concentrations and biochemical parameters of kidney function in plasma and urine suggest that a stimulation of the acute phase response is implicated in the pathogenesis of canine renal disease.
Introduction
C-reactive protein (CRP), originally named for its capacity to precipitate the somatic C-polysaccharide of Pneumococcus pneumoniae, was the first acute phase protein to be described. 32 CRP belongs to the highly conserved pentraxin family of proteins characterized by a cyclic pentameric structure with radial symmetry, and calcium-dependent ligand binding. 16 Based on the primary structure of the subunits, the pentraxins are divided into short and long pentraxins. 26 CRP and serum amyloid P component are the prototype of the short pentraxin family, while pentraxin 3 is the prototypic long pentraxin. 26 The dog CRP molecule has a molecular mass of approximately 115 kDa and is composed of 5 subunits that are noncovalently linked to each other. 5 CRP is normally synthesized by hepatocytes at relatively low rates and retained in the endoplasmic reticulum. 47 Serum CRP concentrations may increase more than 1,000-fold in response to the release of proinflammatory cytokines, most prominently interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), from macrophages at the site of inflammatory lesions or infection. 45 Thus, CRP is well recognized as a sensitive systemic marker of the nonspecific acute phase response to most forms of tissue damage and inflammation in human beings, dogs, and pigs. 6,9,32 In dogs, several studies have described increased levels of serum CRP in response to a variety of pathological conditions, including infectious diseases, traumata, surgery, systemic inflammatory response syndrome, or immune-mediated diseases. 7,8,13,15,18,30 Thus, serum CRP has been suggested as a major acute phase protein in dogs, and its measurement may contribute significantly to the detection, prognosis, and/or monitoring of the underlying disease. 9
Recently, CRP was also detected in urine samples obtained from dogs with chronic renal disease. 37 Chronic renal disease in dogs is progressive and typically ends in uremia and death. 12 Therefore, the identification of risk factors and improved diagnostic methods are needed because the detection of early stages of renal disease could help to slow down the progression of the disease. 17,25 The determination of the glomerular filtration rate (GFR), usually measured as exogenous plasma creatinine clearance (P-ClCr) rate, is accepted as the best overall estimate of kidney function in dogs and can be used to evaluate onset and progression of renal disease in dogs. 19,20,34,42 Concentrations of plasma creatinine or plasma urea have been used as endogenous markers, but they provide only a crude estimate of GFR until 75% of kidney function has already been lost. 17 Moreover, proteinuria is not only an indicator of renal diseases but is also associated with the rate of progression of renal disease, and inflammation may further contribute to the risk of developing end-stage renal failure. 23,43 However, to the authors’ knowledge, no studies have investigated the relation of inflammatory biomarkers to renal function and/or proteinuria in dogs. Therefore, the present study was conducted to assess the CRP concentration in the serum of clinically healthy dogs and in dogs with naturally occurring renal disease in order to find out if there is a relationship between CRP in serum and established markers of kidney function such as the P-ClCr rate, plasma creatinine, and the urinary protein-to-urinary creatinine (UP/UC) ratio.
Material and methods
Dogs and sampling
Blood and urine samples were obtained from 32 client- or student-owned dogs that were presented for blood donation or that were treated at the Small Animal Clinic of the Freie Universität Berlin, Germany. Eighteen male and 14 female dogs of various breeds (median age: 6.2 years; range: 1–14 years) were included in the study. Blood and single voided urine samples (morning urines) were obtained from each dog after 12 hr of fasting. Dogs with underlying conditions (except renal diseases), such as infectious or inflammatory diseases, neoplastic diseases, endocrinopathies, and diseases of the lower urinary tract were excluded from the study. Blood samples were collected from a cephalic vein and placed in tubes a containing ethylenediamine tetra-acetic acid, lithium–heparin, or a clotting activator. A complete blood cell count b was performed, and plasma and serum was prepared by centrifuging blood samples (1,500 × g, 10 min at 4°C). The urine was centrifuged at 300 × g for 2 min for sediment analysis and for removal of cells and particulate matter. The supernatant, plasma, and serum were kept frozen at –80°C; the assays were performed within 4 months. The study protocol did not have to be approved by an animal welfare committee since samples were collected from dogs as part of their routine evaluation at the hospital. Owner permission was obtained for use of the samples.
Measurement of renal function and group allocation
Biochemical testing for plasma creatinine was performed by the use of an automated analysis system. c Concentrations of urinary protein (UP) were assessed by a colorimetric assay. d Urinary creatinine (UC) was determined by the standard Jaffé reaction. c The UP/UC ratio was calculated in order to estimate the degree of proteinuria. The GFR was determined by use of a modified exogenous plasma clearance (P-ClCr) test in dogs that did not have clinical signs of uremia (i.e., inappetence, lethargy, and vomiting) or proteinuria together with azotemia. 34 The exogenous P-ClCr test was carried out with a single intravenous injection of 5% creatinine solution e at a dose of 2.4 g/m2 body surface area. Three blood samples were obtained during 4–9-hr intervals after creatinine injection; plasma creatinine was measured as previously described. The P-ClCr was estimated as the amount of creatinine injected divided by the area under the curve calculated by the trapezoidal method by use of a noncompartmental model. 19 The calculations were performed by use of a commercially available computer software program. 38 An exogenous P-ClCr rate ≥90 ml/min/m2 was considered normal. 20,34
For the group assignment of dogs after the P-ClCr testing, reference values for plasma creatinine and UP/UC concentrations were based on the guidelines established by the International Renal Interest Society for dogs with chronic renal disease (Elliott J: 2006, Kidney disease—The IRIS contribution. Proceedings of the 16th annual European College of Veterinary Internal Medicine–Companion Animals Congress, pp. 51–53, Amsterdam, The Netherlands). Azotemia was defined as plasma creatinine >125 µmol/l. Dogs were considered nonproteinuric if UP/UC was <0.2, borderline if UP/UC was 0.2–0.5, and proteinuric if UP/UC >0.5 mg/mg. On the basis of the results of plasma creatinine concentration, UP/UC, and exogenous P-ClCr rate analysis, the dogs were assigned to 4 groups (Table 1). Dogs of group A (n = 8) were clinically healthy, non-azotemic, and nonproteinuric, and had P-ClCr rates >90 ml/min/m2; group B (n = 11) consisted of non-azotemic and nonproteinuric dogs with reduced P-ClCr rates (51–76 ml/min/m2); group C (n = 7) included azotemic, borderline proteinuric dogs with P-ClCr rates of 22–67 ml/min/m2; and group D (n = 6) included uremic and proteinuric dogs that were not tested for P-ClCr because of their preexisting conditions.
Median serum concentrations (range) of biochemical parameters of kidney function in 32 dogs.*
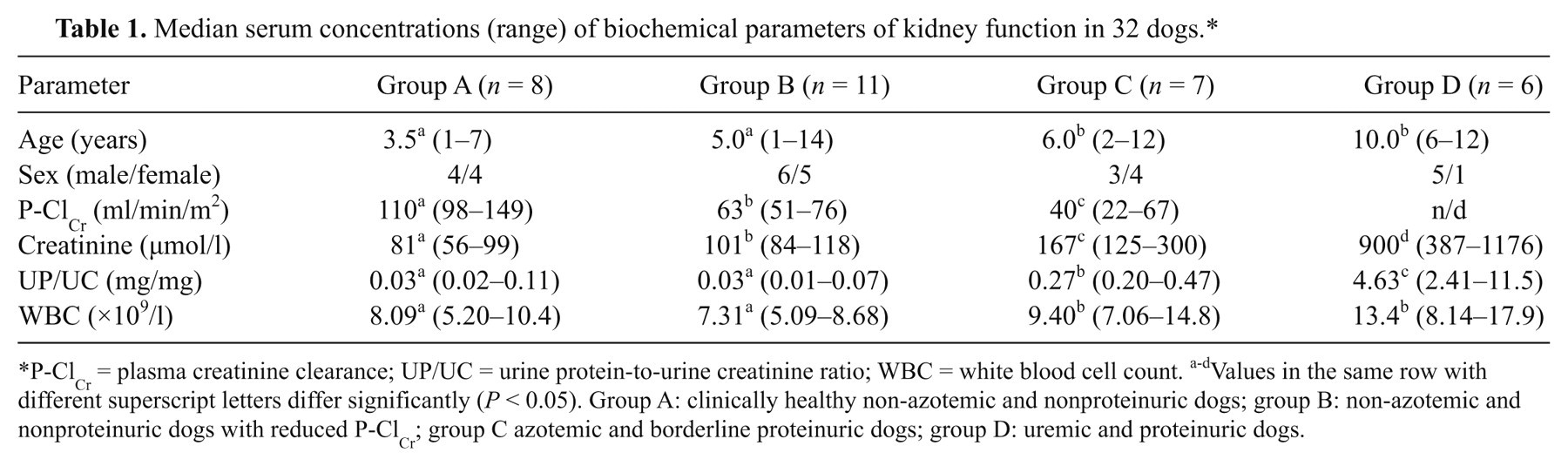
P-ClCr = plasma creatinine clearance; UP/UC = urine protein-to-urine creatinine ratio; WBC = white blood cell count. a-dValues in the same row with different superscript letters differ significantly (P < 0.05). Group A: clinically healthy non-azotemic and nonproteinuric dogs; group B: non-azotemic and nonproteinuric dogs with reduced P-ClCr; group C azotemic and borderline proteinuric dogs; group D: uremic and proteinuric dogs.
Determination of C-reactive protein
C-reactive protein was determined in serum using a solid-phase sandwich enzyme-linked immunosorbent assay f (ELISA) according to the manufacturer’s instructions. 24 The intra- and interassay coefficients of variation were 6.9% and 8.2% at CRP concentrations of 5.15 mg/l and 18.0 mg/l, respectively, according to the manufacturer’s information.
Statistical analysis
The results were expressed as medians and ranges. Statistical analysis was accomplished using a statistical computer application. g The Kruskal–Wallis test was used to test for significant differences in continuous variables between the groups. If there was a significant effect, the Mann–Whitney U-rank test was performed to describe differences in proportions between case and control subjects. Spearman rank’s correlation coefficients were used to compare serum CRP concentrations and other analytes (P-ClCr, plasma creatinine, UP/UC, white blood cell count [WBC]). Values of P < 0.05 were considered significant.
Results
Azotemic dogs with borderline proteinuria (group C) and uremic dogs with overt proteinuria (group D) were older (P < 0.05) than dogs of groups A and B (Table 1). Age was significantly correlated with plasma creatinine (r = 0.55; P < 0.01), UP/UC (r = 0.37; P < 0.05), and CRP (r =0.46; P < 0.01), but it was not associated with the P-ClCr rates (r = –0.35; P = 0.10).
In non-azotemic and nonproteinuric dogs with P-ClCr rates >90 ml/min/m2 (group A), median concentrations of CRP were 3.21 mg/l (range: 2.09−8.60 mg/l). In comparison with dogs of group A, serum CRP concentrations were significantly elevated in dogs of groups B–D (Fig. 1). Median CRP concentrations in non-azotemic and nonproteinuric dogs with reduced P-ClCr rates (group B) were 17.6 mg/l (17.0−19.2 mg/l), in azotemic and borderline proteinuric dogs (group C) 24.8 mg/l (18.0−32.5 mg/l), and in uremic and proteinuric dogs (group D) 59.7 mg/l (17.7−123 mg/l). Serum CRP was significantly correlated with P-ClCr (r = −0.83; P < 0.001), plasma creatinine (r = 0.81; P < 0.001), UP/UC (r = 0.70; P < 0.001), and WBC (r = 0.49; P < 0.01; Table 2).
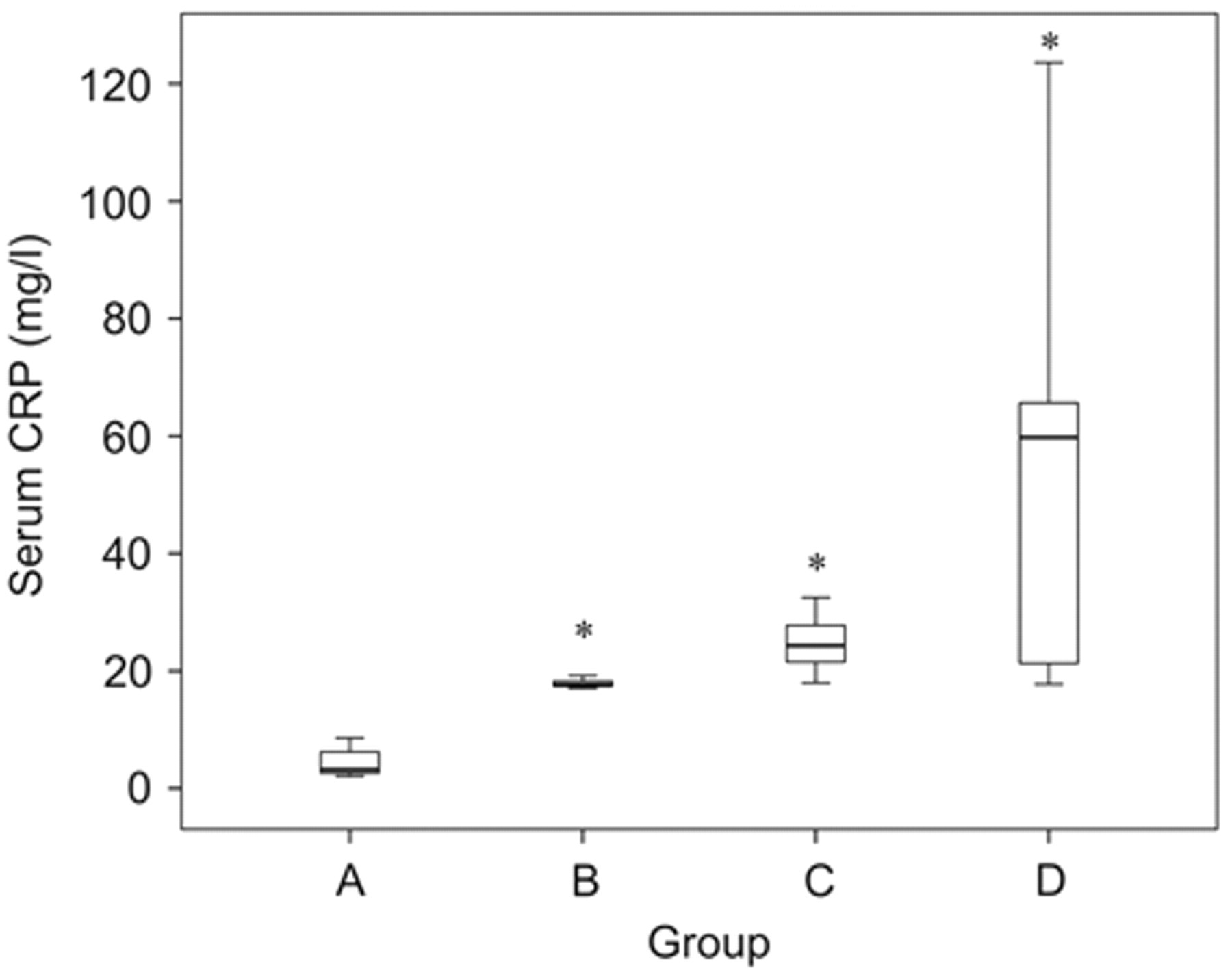
Box plots of serum C-reactive protein (CRP) concentrations in clinically healthy non-azotemic and nonproteinuric dogs (group A), non-azotemic and nonproteinuric dogs with reduced P-ClCr (group B), azotemic and borderline proteinuric dogs (group C), and uremic and proteinuric dogs (group D). The box represents the interquartile range (i.e., 25–75% range), the horizontal bar represents the median value, and the T-bars represent the range of the data. Asterisk indicates significance (P < 0.01) when compared against group A.
Correlation matrix of C-reactive protein (CRP) with other variables in 32 dogs.*
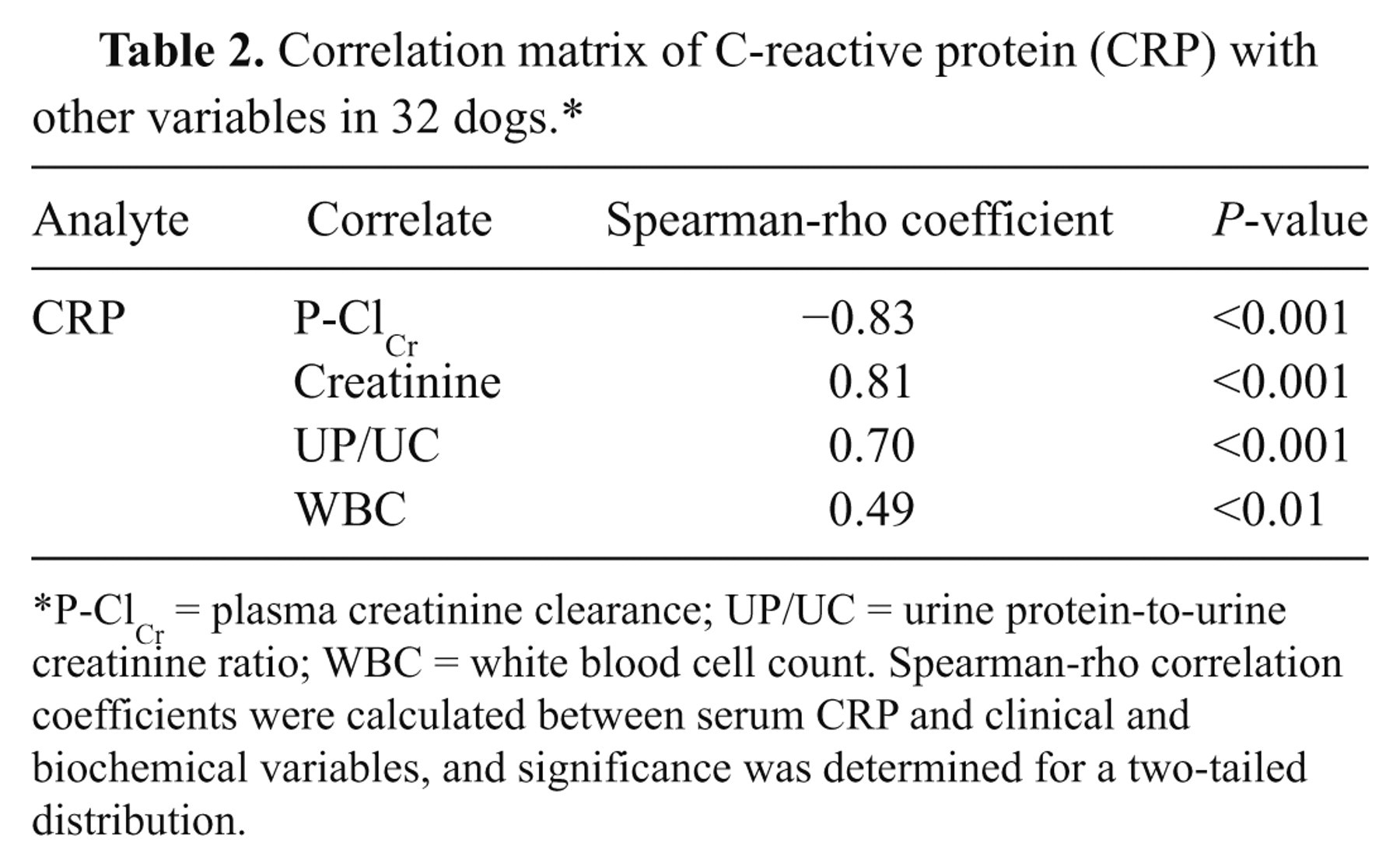
P-ClCr = plasma creatinine clearance; UP/UC = urine protein-to-urine creatinine ratio; WBC = white blood cell count. Spearman-rho correlation coefficients were calculated between serum CRP and clinical and biochemical variables, and significance was determined for a two-tailed distribution.
Discussion
It is well established that the concentration of serum CRP can increase significantly in many infectious and inflammatory conditions and thus, might provide important diagnostic information to clinicians in veterinary medicine. 7,8,13,15,18,30 However, the contribution of chronic or recurring inflammation in the pathogenesis of renal disease in dogs is not well understood. Furthermore, it is not known whether serum CRP can be used as a diagnostic or prognostic marker of canine renal dysfunction. Therefore, the concentrations of CRP were determined in the serum of clinically healthy dogs with normal P-ClCr (group A) and compared with CRP values obtained from clinically healthy dogs with reduced P-ClCr (group B), azotemic and borderline proteinuric dogs with reduced P-ClCr (group C), and uremic and proteinuric dogs (group D).
In the serum of healthy control dogs (group A), CRP concentrations were less than 10 mg/l, which is in accordance with the results reported by other investigators, 21,24,39 who also used the solid-phase sandwich ELISA f for CRP determination. In several other studies that also used this assay, higher CRP levels were reported in clinically healthy dogs, which ranged from 9 mg/l to 31 mg/l, 14 2.5–23.1 mg/l, 7 or were 10.2 ± 2.9 mg/l (mean ± standard deviation). 1 All results demonstrate a physiological variation of serum CRP in dogs under healthy conditions. Moreover, various assay methods are currently available for CRP measurement including electroimmunoassay, 5 single radial immunodiffusion, 8 ELISA, 11,44 and turbidimetric immunoassay, 10 so that the concentrations of serum CRP in dogs is dependent on the methodology used. Therefore, further research is needed to assess reference values and especially upper limits for CRP in dogs under healthy conditions.
The important finding of the present study was that dogs with naturally occurring renal disease had significantly higher serum CRP concentrations compared to healthy control dogs. The median levels of CRP were highest in dogs with uremia and overt proteinuria, and were associated with a lower P-ClCr as well as increases in plasma creatinine, UP/UC, and WBC counts. These results suggest that an activation of the acute phase response and thus a low-grade inflammation might play a role in the pathogenesis of renal disease in dogs; but the detailed molecular mechanisms underlying this relation remain elusive. Hepatocytes produce circulating CRP in response to proinflammatory mediators, predominantly IL-6 and TNF-α. 32 But CRP may also be synthesized extrahepatically as described for human peripheral blood mononuclear cells that express a transcript of the CRP gene. 29 Studies with iodinated CRP in human beings have shown that approximately 70% of the protein is estimated intravascular, 40 and that CRP is excreted in the urine of mice. 28 Therefore, a retention of plasma CRP through a diminished glomerular filtration and reduced excretion of CRP can be proposed to explain higher CRP concentrations in renal disease. 43 However, canine CRP is a high molecular weight plasma protein that is restricted from the glomerular filtration and is not detectable in the urine of healthy dogs. 5,37 Therefore, the elevated CRP levels observed in dogs with renal disease cannot be explained by a retention of CRP due to impaired glomerular filtration. Only severe damage of the glomerular filtration membrane allows the filtration of high molecular weight proteins, and thus an excretion of CRP in the urine of dogs. 37 Although CRP in urine was not measured in the current study, results showed that CRP was positively associated with UP/UC, and that despite the presence of severe proteinuria (UP/UC > 2.0), serum CRP was still elevated in dogs with uremia. A second explanation for higher CRP concentrations in renal disease might be the reduced clearance of inflammatory cytokines as reported in animal models of renal failure. 2,33 Proinflammatory cytokines (e.g., IL-6 and TNF-α) play a central role in the activation of the acute phase response reflected in the production of CRP and other acute phase reactants in the liver. 32 Even though concentrations of IL-6 were not determined in the present study, it was recently reported that the concentration of IL-6 is invariably elevated in dogs with end-stage renal disease. 46 Therefore, the mechanism of a reduced renal cytokine clearance should be investigated in further studies. Third, renal disease is accompanied by increased oxidative stress resulting in an increased production of advanced oxidation products that may stimulate CRP production by hepatocytes, either directly or indirectly through the interaction with monocytes. 3,27 Fourth, cell culture studies have shown that both mesangial cells and tubular epithelial cells synthesize pentraxins in response to inflammatory stimuli. 4,31 CRP messenger RNA transcripts and CRP protein expression were reported in the kidneys, suggesting that the kidneys contribute to a local immune response. 22
In the present study, the highest CRP values were observed in dogs with uremia and proteinuria (group D). Uremia is often accompanied by additional inflammatory conditions, such as uremic gastroenteritis and stomatitis, which may further stimulate the acute phase response and with it serum CRP levels. 1 The stimulation of the acute phase response might also explain the higher WBC counts compared to non-azotemic dogs. Moreover, a recent study showed that the kidneys of dogs with end-stage renal disease contain a large number of infiltrated mononuclear inflammatory cells, which contribute to local inflammation and renal fibrosis in dogs with uremia. 46 Another finding of the present study was that CRP is significantly positive related to UP/UC. In this regard, several experimental studies have shown that increased protein filtration and subsequent endocytosis by proximal tubular cells induce the synthesis of proinflammatory cytokines. 36,41,48 Proinflammatory cytokines may trigger the development of interstitial inflammation, which is thought to be responsible for progression of renal disease in proteinuric states, 35 suggesting that inflammation may be an important mechanism of proteinuria in dogs.
The present study has several limitations that should be addressed in future studies. First, a relationship between serum CRP and biochemical markers in the serum and urine of dogs with renal disease was observed. Therefore, it was not possible to determine whether serum CRP is a cause or consequence of renal dysfunction. Second, most dogs were only examined once in the current study. Therefore, the causality between serum levels of CRP and the progression of renal disease should be evaluated in a follow-up study, which should also include the histopathological investigation of renal biopsy or necropsy specimens in order to correlate the type of renal lesion with serum CRP in affected dogs.
In summary, the present study showed that an activated acute phase response is related to renal function as demonstrated by a significant correlation between serum CRP and reduced P-ClCr, increased serum creatinine, and elevated UP/UC. The findings deserve further investigations in dogs with renal disease to characterize the diagnostic and predictive value of CRP in this condition.
Footnotes
Acknowledgements
Some data were published previously (Raila J, Brunnberg L, Schweigert FJ, Kohn B: 2010, Influence of kidney function on the urinary excretion of albumin and retinol-binding protein in dogs with naturally occurring renal disease. Am J Vet Res 71:1387–1394).
a.
S-Monovette®, Sarstedt AG & Co., Nümbrecht, Germany.
b.
CELL-DYN®, Abbott GmbH & Co. KG, Ludwigshafen, Germany.
c.
Konelab 30i, Thermo Fisher Scientific GmbH, Dreieich, Germany.
d.
Bradford Protein-Assay, Bio-Rad Laboratories GmbH, Munich, Germany.
e.
Merck KGaA, Darmstadt, Germany.
f.
Phase Range™ canine CRP ELISA, Tridelta Development Ltd., Kildare, United Kingdom.
g.
SPSS statistical package, version 15.0 for windows, IBM Deutschland GmbH, IBM Business Analytics, Munich, Germany.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
