Abstract
The aim of the current study was to investigate the exposure of captive wild felids to various infectious pathogens using serological and molecular methods. One hundred and fifty-nine neotropic felids and 51 exotic felids from 28 captive settings in Brazil were tested. While antibodies against Feline parvovirus and Feline coronavirus (FCoV), Feline calicivirus and Bartonella spp. were frequently detected by serologic tests, antibodies against Felid herpesvirus 1 or infection with hemotropic mycoplasmas were less prevalent. Serologic evidence of exposure to Ehrlichia spp., Feline immunodeficiency virus, and Feline leukemia virus (FeLV) was detected rarely, and infections with FeLV, Ehrlichia spp., and Cytauxzoon spp. were found infrequently. The detected Bartonella sequence was molecularly similar to B. koehlerae and B. henselae; for Cytauxzoon, the sequence resembled those from domestic cats. No Anaplasma phagocytophilum and Theileria spp. infections were detected. The positive test results varied significantly among different facilities and species. Additionally, FCoV seropositivity was more prevalent in captivity than in free-ranging populations. Results suggest that testing is appropriate prior to relocation of felids.
Most wild felid species are endangered, and the continuity of the populations often depends on protective measures. 23 Healthy captive wild felid populations provide potential candidates for breeding and reintroduction into nature programs. 19 The majority of the indigenous felids (neotropic species) kept in captivity in Brazil are animals that have been displaced from their natural range as a consequence of habitat destruction or due to illegal captures. The Brazilian captive settings are often overpopulated, and the wild felids are frequently kept in proximity to feral domestic cats (Felis catus) and rodents, and exposed to insects and arthropods, allowing intra- and interspecies transmission of infectious agents, including zoonotic pathogens.
Wild felid species are considered to be susceptible to all pathogens that infect the domestic cat, 26 including common cat viruses and vector-borne agents. Etiological diagnosis of infectious diseases and, in turn, adequate treatment, would avoid or reduce the number of die-offs in captive felids and increase the wellbeing of the animals. In addition, screening of captive wild felids for infectious agents would reveal the level of exposure to infectious agents, thus pinpointing potential problems within the populations or prior to animal movements, and would identify zoonotic risks for people in contact with the animals. Altogether, it would improve the role of zoos in animal conservation.
Thus, the goal of the current study was to determine the exposure of neotropic and exotic wild felids kept in several zoological parks, conservation, and breeding centers in Brazil to selected pathogens. The latter included 6 common domestic cat viruses: Feline leukemia virus (FeLV), Feline immunodeficiency virus (FIV), Felid herpesvirus 1 (FHV-1), Feline calicivirus (FCV), Feline parvovirus (FPV), and Feline coronavirus (FCoV), the Anaplasmataceae: Ehrlichia canis and Anaplasma phagocytophilum, the Bartonellaceae: Bartonella spp., the Mycoplasmataceae (feline hemoplasmas): Mycoplasma haemofelis, ‘Candidatus Mycoplasma haemominutum’, and ‘Candidatus Mycoplasma turicensis’, and the Theileriidae (piroplasms): Cytauxzoon felis and Theileria spp.
Between 2001 and 2004, blood, buffy coat, plasma, and serum samples were collected from 97 neotropic and 51 exotic felid specimens from São Paulo Zoological Park Foundation (SPZ) in São Paulo state, southeast region of Brazil. Additionally, 62 serum samples from neotropic felids were obtained from the serum bank of the nongovernmental organization Mata Ciliar Association (AMC), which were collected between 1996 and 2002 at 27 captive settings in Brazil, located in the Federal District and nine different states from all the five geopolitical regions of the country. The neotropic felid samples derived from 7 different species and the samples from exotic felids originated from 5 different species (Table 1). The gender and origin of the animals (wild caught vs captive born) and the region of sampling were recorded (Table 1).
Characteristic of the surveyed population for the detection of infectious agents in captive Brazilian neotropic and exotic felids.
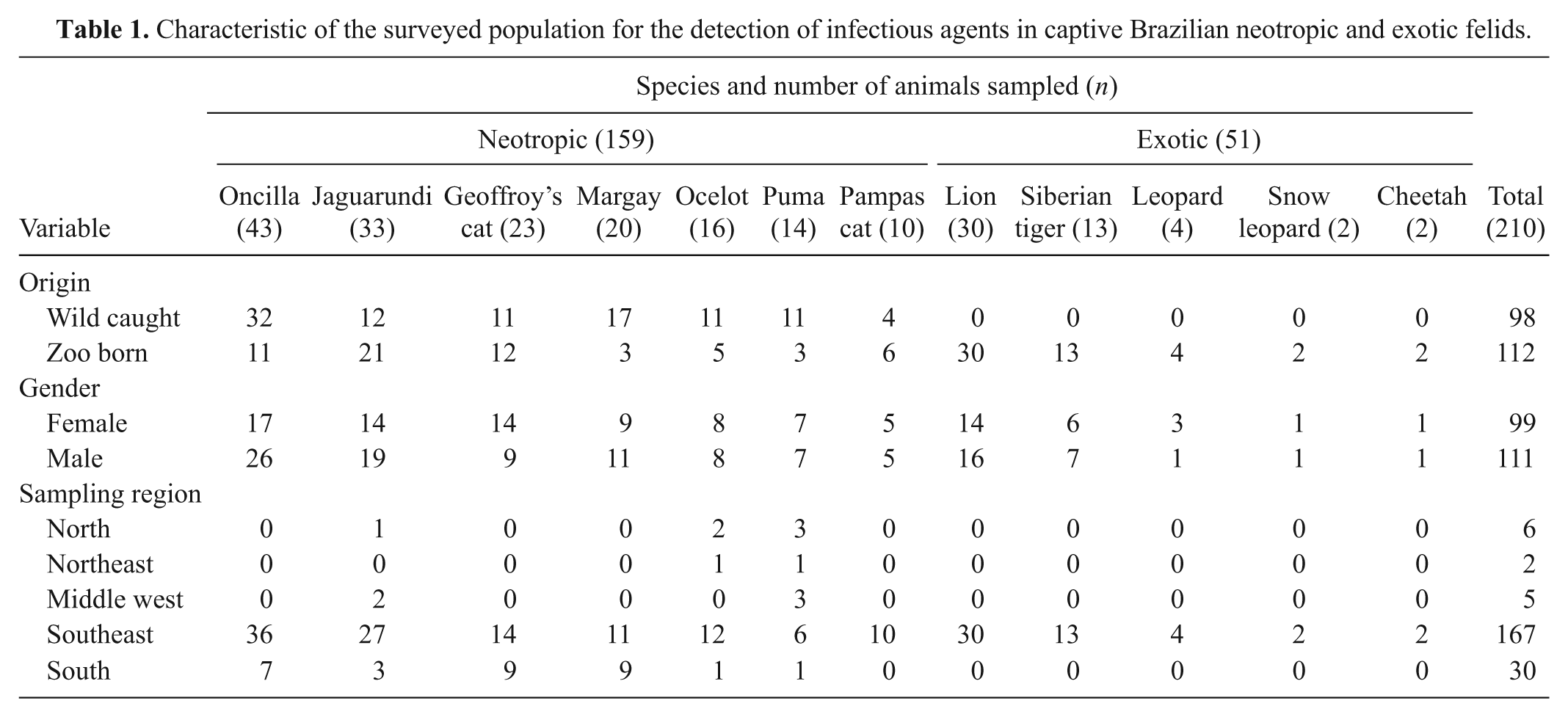
For sample collection, the animals were captured and anesthetized as previously described 4 or using a combination of tiletamine hydrochloride and zolazepam hydrochloride. a Blood was drawn by venipuncture using glass potassium salt ethylenediamine tetra-acetic acid (K2 EDTA) evacuated blood collection tubes for obtaining whole blood and plasma, and plain tubes for obtaining serum. b Serum and blood samples collected at SPZ were stored at −80°C, and serum samples obtained from AMC at −20°C. The sampling and international transportation procedures were approved by the Bioethic Commission of the Faculty of Veterinary Medicine and Zootechny of the University of São Paulo (protocol no. 256/2003), the Convention on International Trade in Endangered Species (permit nos. 0112928BR and 1562/04), the regulations of the Genetic Heritage Management Council, the Agriculture Ministry (CSI 1530/2004), and International Air Transport Association packing instruction 650.
A commercial immunoassay c for FeLV antigen detection and FIV antibody detection was performed for 145 felids at the SPZ at the moment of sampling using fresh blood or serum following the manufacturer’s recommendations. These 145 felids, except for 1 ocelot for which no additional serum or plasma was available, plus 2 pumas and 1 lion (total n = 147), were also tested for the presence of FeLV p27 by a sandwich enzyme-linked immunosorbent assay (ELISA), as previously described. 16 ELISA-positive samples were retested in the presence of mouse serum to exclude cross-linking of the monoclonal mouse anti-FeLV antibodies by feline anti-mouse antibodies. Serum or plasma antibodies to FeLV p45 were measured through an ELISA, as described, 15 for 209 of the sampled animals; for 1 ocelot, no sample was available for this analysis. Results for FeLV antibodies were confirmed in 28 selected samples (24 jaguarundis, 1 oncilla, 1 margay, 1 Geoffroy’s cat, and 1 Pampas cat) by Western blotting (WB) technique, as described. 9 Samples containing antibodies to FeLV gp70, p58, p27, p15, and p12 were classified as positive. Similarly, FIV antibody results were confirmed in 10 selected samples (9 lions and 1 Geoffroy’s cat) by FIV WB, as described. 17 Samples containing antibodies to FIV p24 and p15 were classified as positive. Antibody titers to FHV-1, FCV, and FPV were determined in the serum samples from the unvaccinated animals (n = 63) by the use of an fluorescent antibody test (FAT), as described3,10; felids with a history of vaccination were excluded from testing for these agents. The samples included all 7 neotropic felid species tested; the serum samples were derived from the AMC serum bank (n = 62) and from 1 Pampas cat at the SPZ. Sera were screened at a dilution of 1:20; positive and questionable results were titrated in twofold serial dilutions until endpoint. Antibody titers to FCoV were determined in serum or plasma samples (n = 209) by FAT, as described 10 ; positive samples were titrated using 1:25, 1:100, 1:400, and 1:1,600 dilutions. Exposure to E. canis, or closely related agents, was evaluated by FAT d using 1:80 dilutions of the serum or plasma samples (n = 209). Serum or plasma samples (n = 147) from SPZ felids were assayed for antibodies to B. henselae by FAT, as described, 7 using dilutions of 1:64 and 1:128; titers of ≥ 1:64 were considered positive and positive serum samples were titrated until endpoint by 2-fold serial dilutions. For quality control of all the FAT slides, aliquots of the cell cultures or scrapings from the slides were tested for presence of unwanted antigens, as previously described. 5
Total nucleic acid (TNA) was extracted e from 100 µl of EDTA-anticoagulated blood or buffy coat samples collected from 109 felids housed at the SPZ. To monitor for cross-contamination, negative controls consisting of 100 µl of phosphate buffered saline were concurrently prepared with each batch of 15 samples. Samples were analyzed by real-time polymerase chain reaction (PCR), f as described previously for FeLV provirus, 25 E. canis, 6 A. phagocytophilum, 24 Bartonella spp., 22 M. haemofelis, ‘Candidatus M. haemominutum’, and ‘Candidatus M. turicensis’. 28 In addition, samples were tested as described by conventional PCR for C. felis 20 and Theileria spp. 11 In all PCR assays, water was used as a negative control. All negative extraction and PCR controls tested negative.
Samples that tested PCR positive for Bartonella spp., feline hemoplasmas, or C. felis were further characterized by sequencing. For Bartonella spp., the partial ftsZ gene was subjected to direct sequencing, as described, 27 using primers Bfp1 and Bfp2. 29 The 16s ribosomal RNA (rRNA) gene of M. haemofelis, ‘Candidatus M. haemominutum’, and ‘Candidatus M. turicensis’ were sequenced, as described. 28 For C. felis, the 222–base pair (bp) PCR product was subjected to direct sequencing, as described. 27 Furthermore, the nearly complete 18S rRNA gene was amplified and sequenced using the primers: CytIblynx.23f: 5’-GCCATGCATGTCTAAGTATAAGC-3’ (23 bp) and CytIblynx.1659r: 5’-CGCGCCTAACGAATTAGAAG-3’ (20 bp). Briefly, 2.5 µl of TNA was amplified in a 25-μl reaction containing 1× phusion buffer, g 500 nM of each primer, 200 nM of each deoxyribonucleotide triphosphate, h and 1 U DNA polymerase i using a thermocycler. j The thermal program comprised 98°C for 3 min, 35 cycles of 98°C for 10 sec, 56°C for 30 sec, and 72°C for 1 min, and finally 72°C for 10 min. PCR products were purified k and vector cloned l using a cloning kit for sequencing. m Plasmid DNA was purified from positive clones. n The 3 clones were subjected to direct sequencing as described 27 with vector-specific M13 and an internal primer (Cytlynx.1352r: 5’-TTATAACCGCAAAGTCCCTCTAAGA-3’ [25 bp]). The frequency of seropositive animals in different groups were compared with the Fisher’s exact test (cell frequencies of ≤5; pF) and the chi-square test (cell frequencies of >5; pchi2). o
Of the 147 samples tested for FeLV p27, 2 samples from zoo-born jaguarundis (CAD 20761 and 21810) were positive; one of them (CAD 20761) was also positive by the commercial immunoassay test. The blood samples of both jaguarundis were FeLV provirus positive, but negative for antibodies to FeLV. High antibody levels to FeLV (94% of the positive control in ELISA, positivity confirmed by WB) were detected in another jaguarundi (CAD 20762). Some additional samples showed low ELISA reactivity and 2 animals (jaguarundi CAD 27689 and a Pampas cat CAD 20034) were confirmed to be positive by WB. None of these animals showed evidence of relevant diseases at the time of sampling. The clinical importance of FeLV infections in jaguarundis will need further attention. Recently, FeLV was suspected to have a major impact on another wild felid species, the Iberian lynx. 21
Among the 145 felids from the SPZ tested for FIV using the commercial immunoassay test, samples from 5 clinically healthy lions from SPZ were positive and one sample was inconclusive. Four of these 6 lions (CAD 290801, 20748, 26281, 15856) were confirmed to be positive using WB, but 2 showed inconclusive results. Wild felids may be infected with FIV-related lentiviruses and not exhibit overt clinical signs. Much older lentiviruses related to the emergent FIV have so far been detected in African lions and North American pumas and long-term co-evolutionary processes might have provided asymptomatic host–virus relationships. 2 In addition to the lions (CAD 290801, 20748, 26281, 15856), a Geoffroy’s cat (CAD 28022) was found positive for FIV by WB.
The frequency of seropositive animals among the 63 unvaccinated neotropic cats as determined by FAT were 19%, 51%, and 70% for FHV-1, FCV, and FPV, respectively (Table 2). Evidence of FHV-1, FCV, or FPV infections were found in all neotropic species tested, with the exception of the Pampas cat, for which only 1 sample from a kitten was available. Antibodies to FPV were significantly more frequent than antibodies to FCV (pchi2 = 0.0289), and antibodies to FPV and FCV were both more frequent than antibodies to FHV-1 (pchi2 ≤ 0.0002). Nine animals were seronegative for FHV-1, FCV and FPV; the negative felids originated significantly more frequently from zoos (6/11 zoo-born unvaccinated animals; 55%) than from the wild (3/52 wild-caught unvaccinated animals; 6%; pF = 0.0009). The results of the 63 unvaccinated felids kept at several captive settings in Brazil suggested a widespread exposure to FHV-1, FCV, and FPV. An important source of infection may be the wild caught animals introduced to the captive settings. Although comparisons with other studies may be difficult due to differences in the tested sample it can be noted that the current sample prevalence for FHV-1 and FCV were not markedly different from those reported earlier for free-ranging Brazilian felids using identical methods. 5 Moreover, the FHV-1 sample seroprevalence for captive felids was similar to that previously obtained in 250 samples from captive Brazilian felids from 6 neotropic species. 1 The higher seropositivity rates found for FCV compared to FHV-1 in captive animals in the present study may be related to a higher environmental resistance of FCV compared to FHV-1 or to differences in the latency phase of the infection: while FHV-1 latently infected felids are intermittent virus shedders, FCV seropositive felids may be active carriers continuously shedding viruses for several months to years.12,18 The very high environmental resistance of parvoviruses and the fecal–oral infection route may explain the high antibody frequency found for FPV in captivity (70%). In the present study, it was found that FPV or antigenic similar viruses were ubiquitous in captive settings in Brazil as well as in the wild. 5
Fluorescent antibody test results for Felid herpesvirus 1, Feline calicivirus, Feline parvovirus, and the vector-borne bacteria Ehrlichia canis and Bartonella henselae in captive Brazilian neotropic and exotic felids*
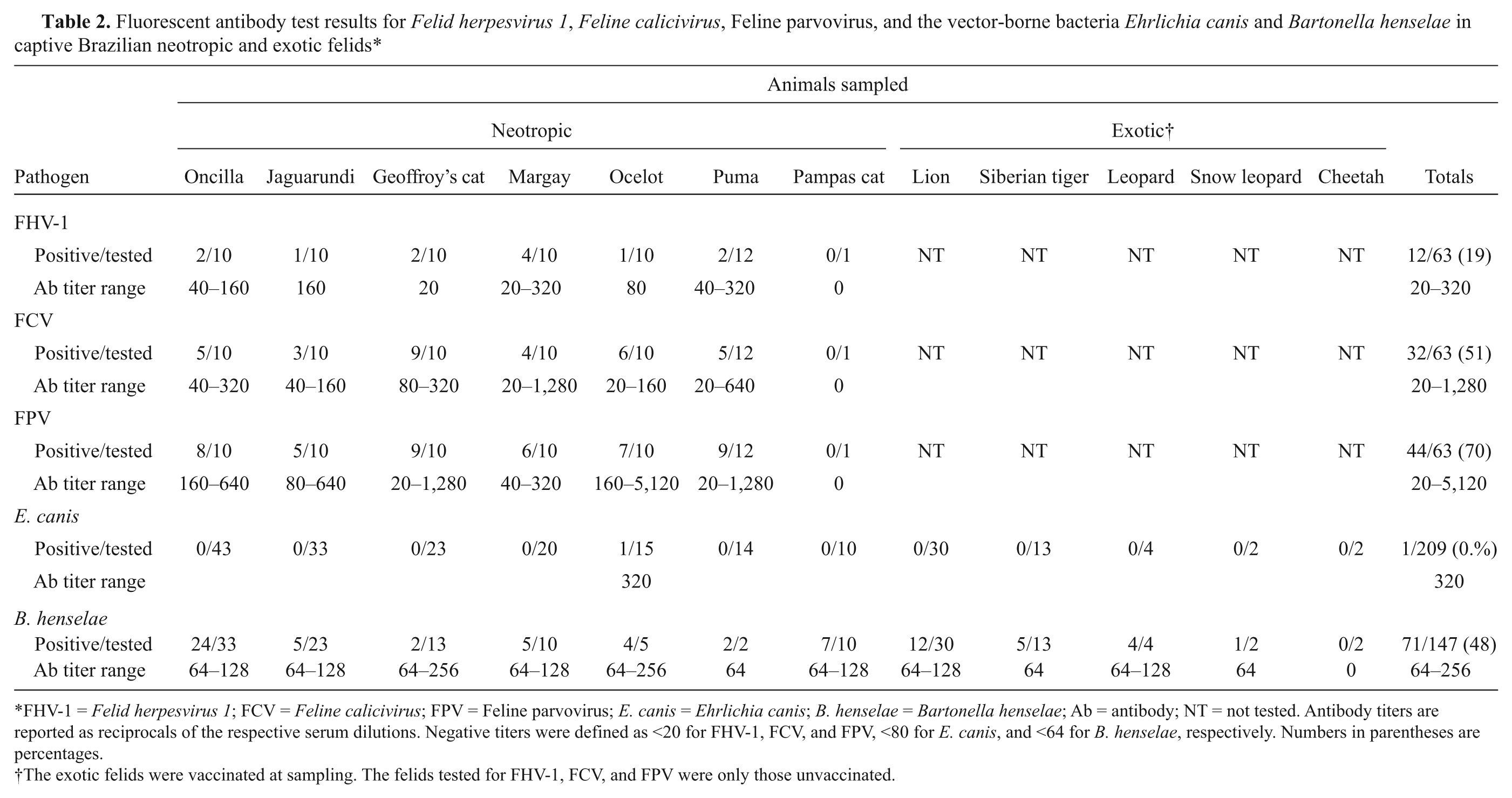
FHV-1 = Felid herpesvirus 1; FCV = Feline calicivirus; FPV = Feline parvovirus; E. canis = Ehrlichia canis; B. henselae = Bartonella henselae; Ab = antibody; NT = not tested. Antibody titers are reported as reciprocals of the respective serum dilutions. Negative titers were defined as <20 for FHV-1, FCV, and FPV, <80 for E. canis, and <64 for B. henselae, respectively. Numbers in parentheses are percentages.
The exotic felids were vaccinated at sampling. The felids tested for FHV-1, FCV, and FPV were only those unvaccinated.
As for FCoV, 65% of the samples tested positive by FAT (Table 3). All neotropic and exotic felid species (with the exception of the cheetah) had individuals that tested positive for FCoV. The FCoV antibody titers ranged from 25 to 1,600. In the jaguarundi population 30% of the animals had a titer of 1,600 (Table 3), which was significantly more frequent than in the remaining wild cats (3.4% with a titer of 1,600; pchi2 < 0.0001). Nine out of 10 jaguarundis with FCoV titer of 1,600 were housed at the SPZ. High titers of 400 and 1,600 were only detected in neotropic wild felids with the exception of one male leopard (CAD 14753) at the SPZ. Wild cats housed at the SPZ zoo were significantly more frequently FCoV seropositive (72%) than wild cats from other facilities (47%; pchi2 = 0.0005).
Serological investigation of Feline coronavirus infection in captive Brazilian neotropic and exotic felids by the use of fluorescent antibody test.*
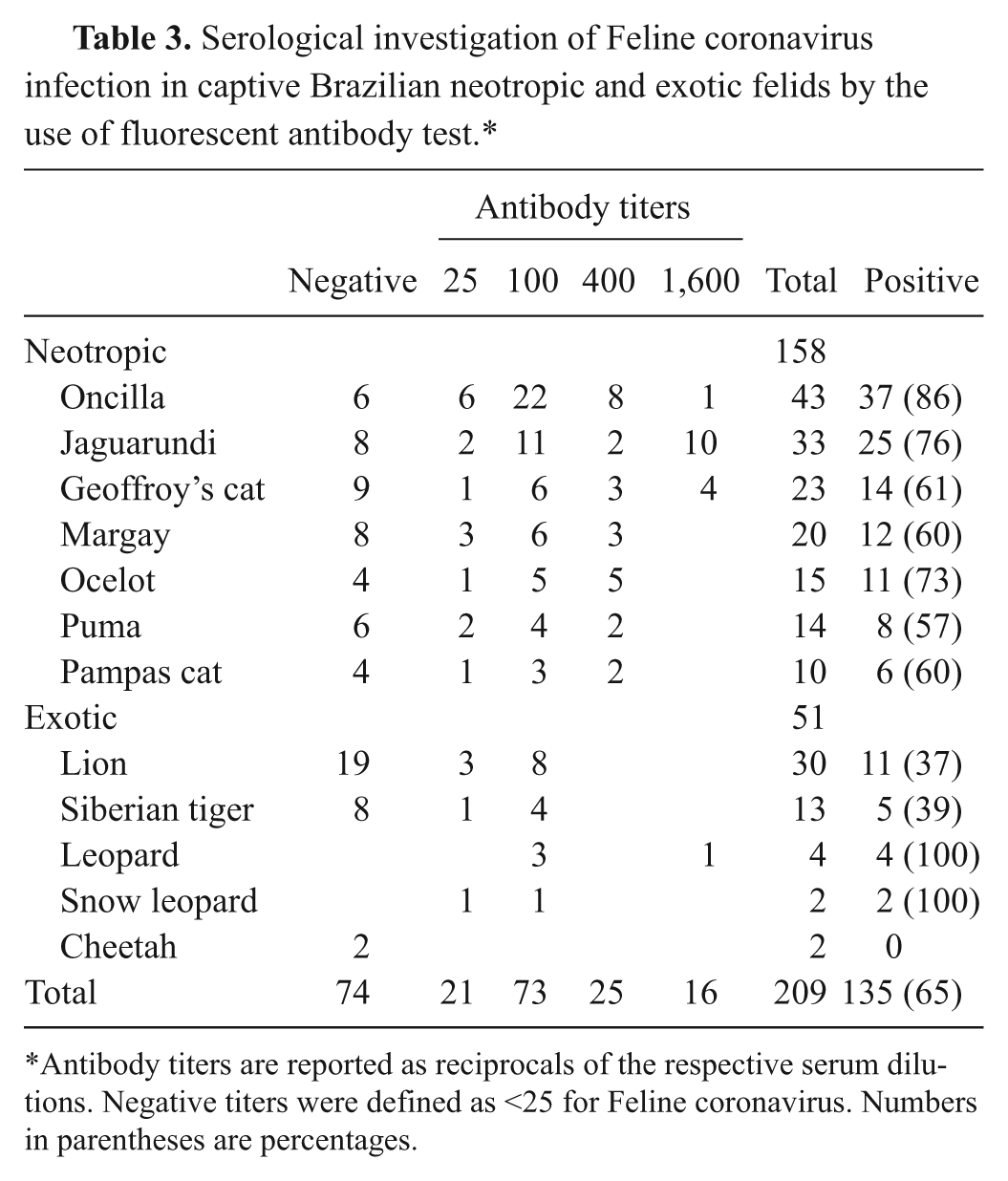
Antibody titers are reported as reciprocals of the respective serum dilutions. Negative titers were defined as <25 for Feline coronavirus. Numbers in parentheses are percentages.
The presence of antibodies to FCoV in 65% of the captive felids should be a matter of concern. Seropositive felids were more frequently encountered in the captive settings than reported earlier for free-ranging Brazilian felids using identical methods (5%; pchi2 < 0.0001). 5 The present study relied on Transmissible gastroenteritis virus (Purdue strain) 10 as an antigen for FAT; it had been reported that for serodiagnosis of FCoV infection this antigen should be used to attain the highest diagnostic efficiency. 14 Nonetheless, it cannot be excluded that antibodies to some immunologically distinct strains of coronaviruses, which may infect felids, 13 were not detected. Exposure to FCoV may be higher when many animals are kept in close proximity, as may be the case in captivity. In the SPZ, the high titers of 400 and 1,600 were almost exclusively detected in neotropic wild felids. This could be explained by the disposition and structure of the enclosures and their accessibility to domestic cats; the latter are a potential source of FCoV infection. The control of FCoV in zoological settings is challenging. Identification of virus shedders by molecular methods followed by segregation of active shedders is very difficult to achieve due to logistic limitations. Control should then rely on a series of measures, including partition of domestic cats, use of solid physical barriers, frequent replacing of soil substrate, and diligent cleaning and disinfection of water and food receptacles and other fomites.
The sample seroprevalence of B. henselae was 48% (Table 2). The seroprevalence was significantly higher in the wild caught (28/45 animals; 62%) than in the zoo born felids (43/102 animals; 42%; pchi2 = 0.0248). Using Bartonella spp. real-time PCR, 1 male margay (CAD 17336) from the SPZ was positive. The animal was born in the wild but lived for 15 years in captivity prior to sampling, thus it is not possible to ascertain if the infection was acquired in the wild or in captivity. Compatible with bartonellosis, however, the animal had shown two previous episodes of accentuated weight loss, dullness, dehydration, and anemia five and three years before sampling. Using sequencing analysis, the Bartonella sp. detected in this margay (GenBank accession no. GU903912) revealed the highest identity with B. koehlerae (AF467755; 99%) and B. henselae (AF061746; 95%). A second female margay (CAD 20766) also from the SPZ was weakly PCR-positive for Bartonella sp.; the load was too low for sequencing analysis. The antibody frequency for B. henselae (48%) indicated a high exposure rate of captive felids from SPZ to this pathogen, although significantly lower than the 95% frequency reported for free-ranging Brazilian felids using identical methods (pchi2 < 0.0001). 5 While only 2 margays out of 109 tested felids were positive for Bartonella spp. by real-time PCR, another study 8 detected 15% of the 67 Brazilian captive neotropic felids positive to Bartonella spp. in a conventional PCR. Such difference in the percentage of PCR-positive animals may be due to the specificity and sensitivity of the PCR assays. Moreover, the sample under investigation may have influenced the outcome. In a previous study, mainly felids from a single captive setting in the southern region were investigated, 8 while in the present study, all animals tested by molecular methods for Bartonella spp. originated from SPZ in the Southeast Region. In addition, in the current study jaguars (Panthera onca) were not tested, while in the other study, 8 15% of the felids evaluated were jaguars. Bartonella henselae has a zoonotic potential and may cause cat scratch disease in human beings. Thus, safety procedures should be put in place when handling potentially positive felids or sample material from them.
Using real-time PCR assays specific for feline hemoplasmas, 10 of the 109 tested wild cats were PCR-positive for ‘Candidatus M. haemominutum’ (9.2%). This included 1 lion and 9 neotropic wild felids. All 10 positive wild cats had been housed at the SPZ, and 3 of them had also been born in the zoo. A wild caught male margay (CAD 28257) was co-infected with ‘Candidatus M. haemominutum’ and M. haemofelis. No animal was positive for ‘Candidatus M. turicensis’. The ‘Candidatus M. haemominutum’ amplicons from an oncilla and the margay, as well as the M. haemofelis amplicon from the margay were sequenced. The M. haemofelis 16S rRNA gene sequence of the margay (GenBank accession no. DQ825438) showed >99% identity when aligned with a M. haemofelis 16S rRNA sequence from a domestic cat (DQ157160). 28 The 2 ‘Candidatus M. haemominutum’ 16S rRNA sequences (DQ825439, DQ825440) exhibited 99% identity to each other. They were most closely related to ‘Candidatus M. haemominutum’ sequences from a domestic cat from Israel (AY150974; 100% identity), an Iberian lynx from Spain (DQ825445; 99% identity) and a sequence originating from a domestic dog from the United States of America (AY297712; 99% identity). The detection of ‘Candidatus M. haemominutum’ and M. haemofelis was conclusive for the presence of these hemoplasma species in the nondomestic felid population of the SPZ, as published elsewhere. 28 Sequence comparisons revealed that the M. haemofelis amplicon from the margay was very closely related to hemotropic mycoplasmas from domestic cats. The ‘Candidatus M. haemominutum’ amplicons from the margay and the ocelot had formed a separate group, when compared to amplicons from other wild and domestic cats. 28 Nonetheless, they were now found to be very closely related to feline and canine sequences from a very wide geographic region.
None of the 109 animals tested positive for Theileria spp. by PCR. One sample tested PCR-positive for C. felis. The sample originated from an asymptomatic wild born female ocelot (CAD 22028) that had lived at least nine years in captivity prior to sampling. Sequencing of a 1,598-bp section of the 18S rRNA gene of this C. felis amplicon (GenBank accession no. GU903911) revealed a very high identify (>99%) with that from domestic cats from the United States and South Africa (e.g., AF399930, L19080), but less identity (96%) with Cytauxzoon spp. from Pallas cats from Mongolia (e.g., AY485690, AF531418), and Iberian lynxes from Spain (e.g., AY496273, EF094468). It was 99% identical over a 1,204 bp stretch that was known of a C. felis sequence from an oncilla from Brazil (DQ382277). A short 404-bp stretch sequenced from Brazilian ocelots, pumas, and one jaguar (EU376525, EU376526, and EU376527) showed 97–98% identity to the C. felis amplicon detected. Although it is not possible to know whether the animal had been infected in the wild or in captivity, the high identity of this C. felis sequence with detected sequences of domestic cats in comparison to other sequences of nondomestic felids, and the long period spent in captivity suggests that domestic cats might have acted as source of infection for the wild felids.
Serological evidence of E. canis by FAT was found in 1 (SB014) out of 209 tested animals. Such finding suggests that infections with this pathogen are not a major problem in Brazilian captive settings. This is in accordance to what has been previously reported for free-ranging Brazilian felids. 5
Adopting a unified and efficient strategy for controlling felid infectious diseases in Brazilian captive settings would greatly improve the role of Brazilian zoos in the conservation of wild felid species and reduce the risks of transmission of zoonotic pathogens. Priority should be given to the improvement of control measures, such as effective routine disinfection of enclosures and facilities, vaccination of felids against common feline viruses, avoidance of contact with domestic cats, and reduction of exposure to arthropod vectors. Testing for pathogens should be considered before moving or regrouping of animals, for diagnostic and clinical purposes, and for those individuals aimed for breeding and ex situ reintroduction programs.
Footnotes
Acknowledgements
The authors express their sincere appreciation to all veterinarians and staff of the zoological parks and other captive settings throughout the country. Laboratory work was performed with logistical support of the Center for Clinical Studies at the Vetsuisse Faculty, University of Zurich. The authors are also indebted to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Dean of Graduate Studies from University of São Paulo and the International Relations Office, University of Zurich, Switzerland. José Luiz Catão-Dias is a recipient of a scholarship by the National Council for the Scientific and Technological Development (301517/2006-1). The current study was conducted by Claudia Filoni as partial fulfillment of the requirements for a doctorate degree at the Department of Pathology, Faculty of Veterinary Medicine and Zootechny, University of São Paulo.
a.
Zoletil®, Virbac do Brasil, São Paulo, Brazil.
b.
BD Vacutainer® Tubes, BD, Franklin Lakes, NJ.
c.
Snap™ Combo FeLV Antigen/FIV Antibody Test Kit, IDEXX Laboratories Inc., Westbrook, ME.
d.
Mega Screen Fluoehrlichia c. slides, MegaCor Diagnostik GmbH, Hörbranz, Austria.
e.
MagNaPure LC TNA isolation kit, Roche Diagnostics, Rotkreuz, Switzerland.
f.
TaqMan® PCR, ABI Prism 7700 Sequence Detection System, Applied Biosystems, Foster City, CA.
g.
Phusion HF Buffer, Finnzymes, Espoo, Finland.
h.
Sigma-Aldrich, Buchs, Switzerland.
i.
Phusion™ DNA Polymerase, Finnzymes, Espoo, Finland.
j.
T-personal 48 Thermocycler, Biometra GmbH, Goettingen, Germany.
k.
MinElute Gel Extraction Kit, Qiagen, Hombrechtikon, Switzerland.
l.
pCR®4-TOPO®, Invitrogen, Basel, Switzerland.
m.
TOPO TA Cloning® Kit for Sequencing, Invitrogen, Basel, Switzerland.
n.
QIAprep Spin Miniprep Kit Qiagen, Hombrechtikon, Switzerland.
o.
Analyse-it Software, Leeds, United Kingdom.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received financial support from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Dean of Graduate Studies from University of São Paulo, and Fundação de Amparo à Pesquisa do Estado de São Paulo (2008/54338-4).
