Abstract
Diseases caused by extraintestinal pathogenic Escherichia coli (ExPEC) in wild felids are rarely reported. Although urinary tract infections are infrequently reported in domestic cats, such infections when present are commonly caused by ExPEC. The present work characterized ExPEC strains isolated from 2 adult felines, a snow leopard (Panthera uncia) and a black leopard (Panthera pardus melas), that died from secondary bacteremia associated with urinary tract infections. Isolates from both animals were classified into the B2 phylogenetic group and expressed virulence genotypes that allowed them to cause severe disease. In addition, strains from the black leopard showed multidrug resistance.
Extraintestinal pathogenic Escherichia coli (ExPEC) strains are characterized by specific virulence factors (VFs) and are related to a heterogeneous group of human disorders. Among extraintestinal diseases caused by E. coli, urinary tract infections (UTI) are the most frequently reported, along with neonatal meningitis and septicemia. 15 ExPEC strains have also been associated with animal diseases, especially in poultry and pets.12,15 There is also evidence that ExPEC strains isolated from animals present zoonotic potential.3,13 There is little published information associating this group of pathogens with diseases in wild animals. The purpose of the current study was to characterize E. coli isolates obtained from a snow leopard (Panthera uncia) and a black leopard (Panthera pardus melas) housed in a Brazilian zoological park that died due to UTIs associated with secondary bacteremia.
The first animal, an adult male snow leopard, was brought to the veterinary hospital and presented with hematuria. Urinalysis and the determination of the urea and creatinine levels in blood indicated a primary renal disease. Despite supportive therapy, death occurred within 24 hr. Necropsy revealed suppurative hemorrhagic cystitis (Fig. 1), nephritis, and suppurative hemorrhagic pyelonephritis (Fig. 2). Histopathology showed multifocal interstitial nephritis, multifocal suppurative pyelonephritis, severe necrotizing cystitis, splenic lymphocytosis, and multisystemic petechiae.
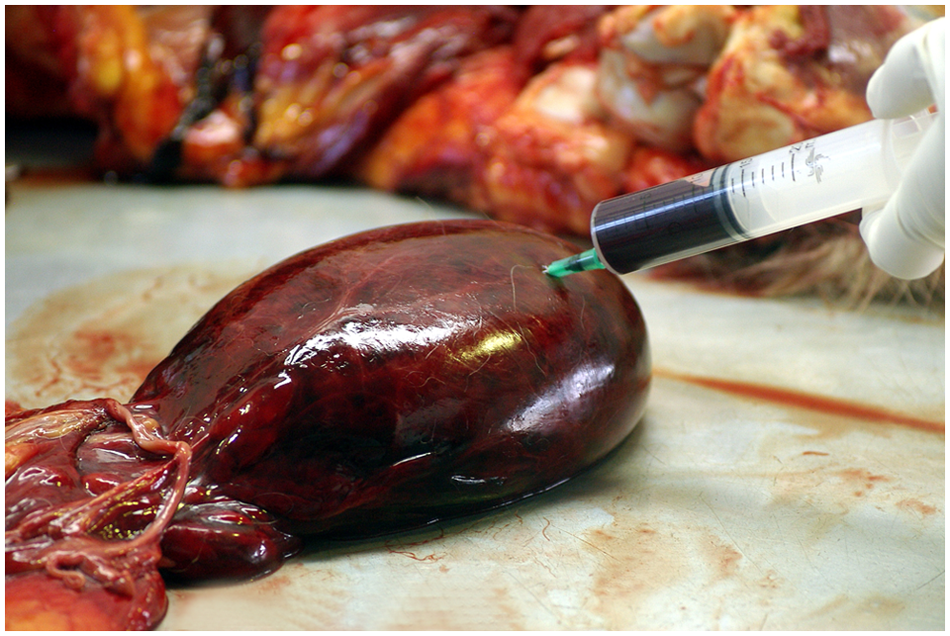
Urinary bladder; snow leopard (Panthera uncia). The urinary bladder is distended and hemorrhagic and contains a large amount of bloody urine.
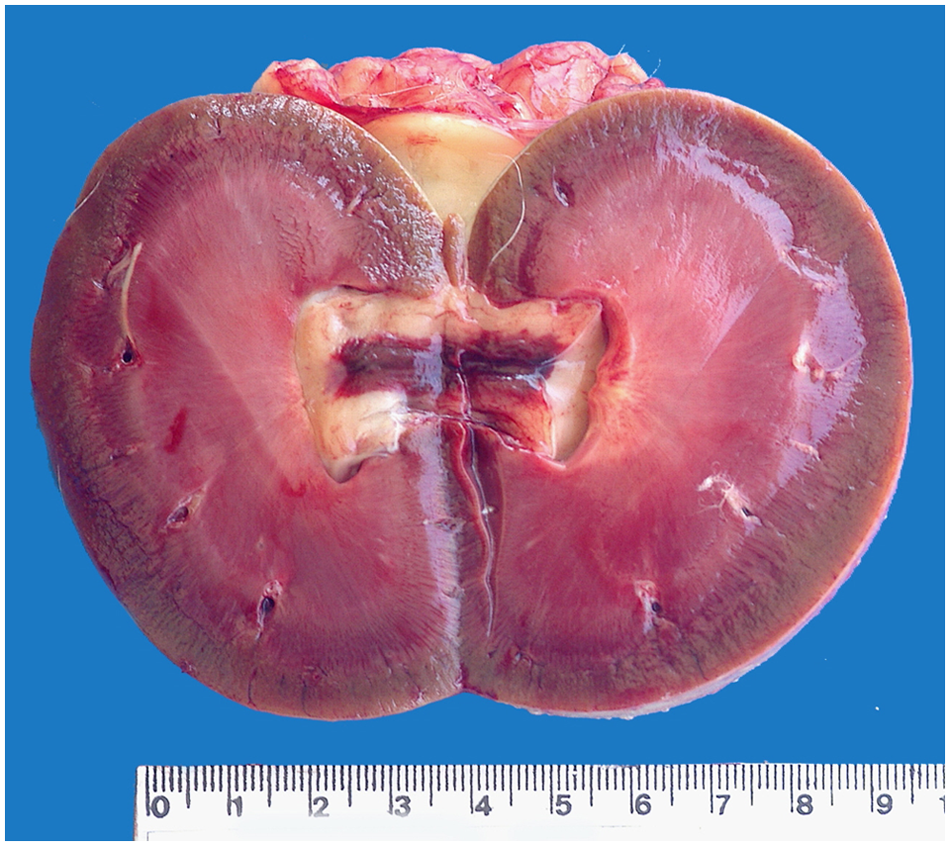
Kidney; snow leopard (Panthera uncia). The medulla shows congestion and diffuse hemorrhages associated with cortical-medullar white-tan striations and hemorrhagic pyelonephritis.
The second case was a 17-year-old female black leopard that had a prior history of recurring UTIs, without etiological diagnosis, that started 9 months before death. On the last UTI manifestation, 7 days of enrofloxacin treatment was adopted with apparent success, according to pre- and post-treatment urinalysis. The animal was brought to the hospital exhibiting lethargy and pale mucous membranes. Complementary exams revealed azotemia and moderate neutrophilia, compatible with infection. The animal died 48 hr after presentation. Chronic pyelonephritis, multifocal cardiac petechiae, and severe pulmonary edema were the pathological lesions.
The animals were necropsied immediately after death. Heart blood samples from both animals and a vesical urine sample from the snow leopard were aseptically collected and cultivated in brain–heart infusion broth, a blood agar, a and MacConkey agar. a Pure E. coli strains from the 3 samples were isolated and identified using a commercial system. b
Serotyping was performed at a reference center for E. coli serotyping (Instituto Adolfo Lutz, São Paulo, SP, Brazil), according to standard international procedures of tube agglutination test 10 and using currently available O (O1–O181) and H (H1–H56) antisera. The molecular characterization of the strains was performed by polymerase chain reaction (PCR), following protocols 12 previously reported for putative genes encoding VFs, including adhesins (papA, papEF, sfa, afaI, fimH, iha), siderophores (iucD, fyuA), toxins (cnf-1, hlyA), protectins (traT, cvaC), invasive factor (ibeA), and a pathogenicity island (malX). The phylogenetic group of isolates, as well as hemolysin expression, were determined according to methods previously described.4,6 The susceptibility to antibiotics was determined according to Clinical and Laboratory Standards Institute guidelines8,9 using the E. coli strain ATCC 25922 as a control and the following antibiotic disks c : ampicillin (10 μg), amoxicillin (30 μg), cephalexin (30 μg), cefoxitin (30 μg), ceftiofur (30 μg), gentamicin (10 μg), streptomycin (10 μg), enrofloxacin (5 μg), ciprofloxacin (5 μg), chloramphenicol (30 μg), tetracycline (30 μg), nitrofurantoin (300 μg), and trimethoprim–sulfamethoxazole (25 μg).
Diseases caused by ExPEC in wild felids are rarely described. Although UTIs are infrequently reported in domestic cats, such infections when present are commonly caused by ExPEC. 2 In previous reports,5,16 cases of necrotizing pneumonia have been associated with ExPEC in both domestic cats and tiger cubs. Regarding nondomestic cats, a report of pyometra involving E. coli was described in a lioness, 1 and colisepticemia was reported in a tiger. 14 However, VFs were not characterized in either of those cases. The present report describes the occurrence of bacteremia in 2 captive wild cats that were associated with ExPEC infections of urinary tract origin. The results obtained regarding the investigated VFs, serotyping, and phylogenetic classification of the isolates are presented in Table 1.
Strain origin, serotype, phylogenetic group, virulence-associated genes, and antibiotic susceptibility of extraintestinal pathogenic Escherichia coli isolated from captive wild felids with bacteremia of urinary tract origin.*
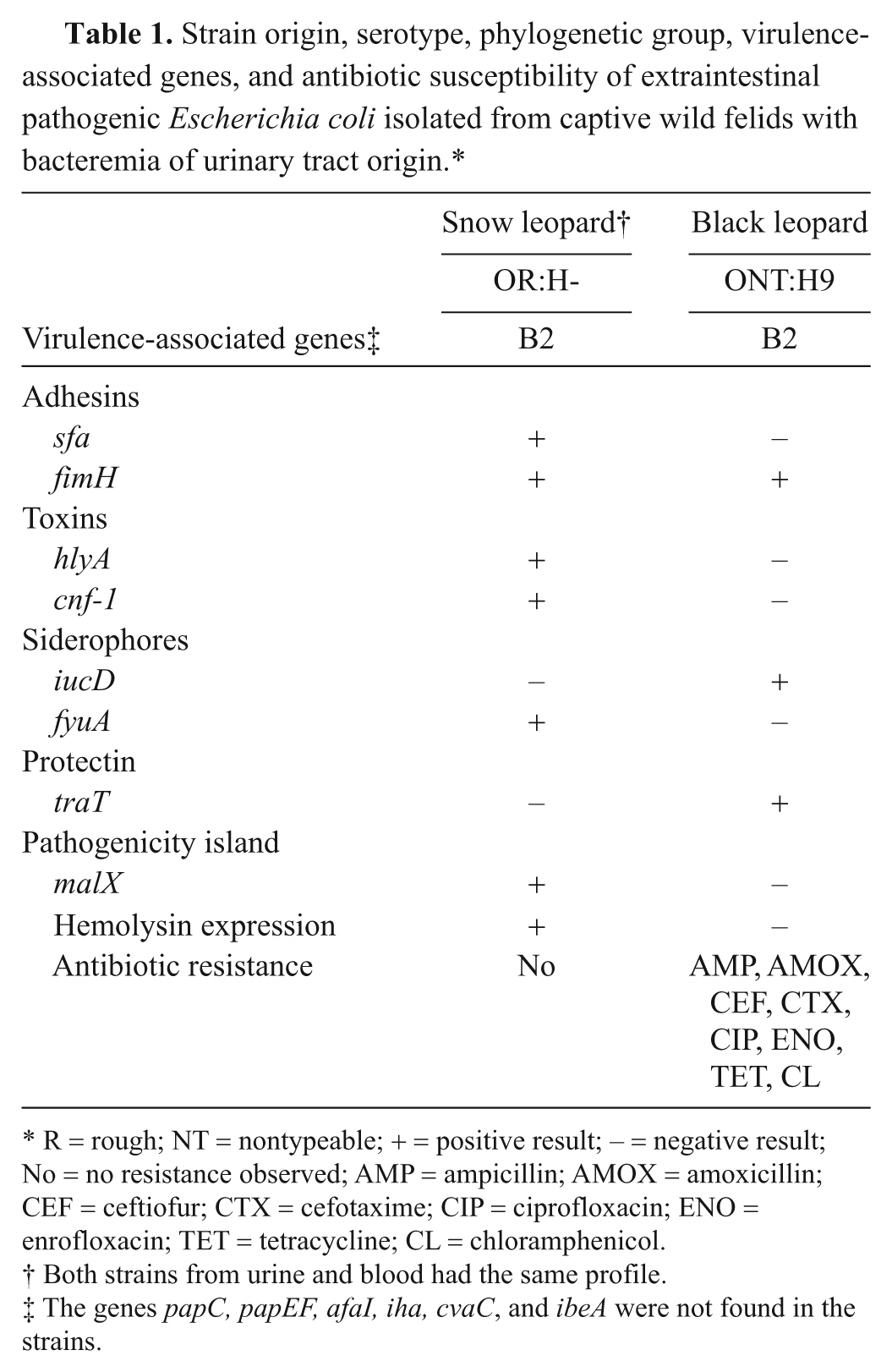
R = rough; NT = nontypeable; + = positive result; – = negative result; No = no resistance observed; AMP = ampicillin; AMOX = amoxicillin; CEF = ceftiofur; CTX = cefotaxime; CIP = ciprofloxacin; ENO = enrofloxacin; TET = tetracycline; CL = chloramphenicol.
Both strains from urine and blood had the same profile.
The genes papC, papEF, afaI, iha, cvaC, and ibeA were not found in the strains.
In both cases, although the isolates could not be associated with the classical ExPEC serogroups, the presence of virulence markers linked with ExPEC 15 was demonstrated. There were, however, interesting differences between the isolates. The strains isolated from blood and urine of the snow leopard presented the same phylogenetic group and virulence gene profile and were susceptible to all of the antibiotics tested, showing a great range of VFs, which included those classically related to ExPEC, such as S fimbriae (sfa), the yersiniabactin receptor (fyuA), and genes for CNF-1 (cnf-1) and hemolysin (hlyA) toxins; the expression of the latter was demonstrated in a phenotype test. These strains also had the gene malX, which was linked to the pathogenicity island of urosepticemic human E. coli (CFT073). Some reviews of the literature indicate a close relationship between this island and high virulence potential.11,15
In contrast, the strain isolated from the black leopard showed multidrug resistance to 8 different active compounds (5 different classes) but a narrower range of virulence markers, including fimH, iucD, and traT. Data from a published report 11 has mentioned the negative association involving antimicrobial resistance and VFs and/or phylogenetic background, ascribing this phenomenon to the balance between host compromise and bacterial fitness advantage. However, hybrid plasmids codifying multiple VFs along with multidrug resistance has already been found in avian pathogenic E. coli strains with zoonotic potential 13 The finding of multidrug resistance and VFs in strains isolated from captive animals is worrisome because the selective pressure exerted by extensive antibiotic use may lead to the selection of more pathogenic strains, which may threaten the animals’ survival.
Although in lower number, all 3 virulence markers found in the black leopard isolate indicate potential for the establishment of systemic infections. Type 1 fimbriae (fimH), in addition to having an adhesion function, also has a role in invasion. The siderophore encoded by the iucD gene, although not very prevalent in isolates from domestic animals, has been detected in high percentages of human isolates from patients with pyelonephritis. 11 Finally, the strains in the current study were classified into the phylogenetic group B2, which is known to include the ExPEC strains responsible for the most severe and invasive syndromes in animals and human beings.7,11 Considering the zoonotic potential of ExPEC, as well as the severity of the reported cases, it is believed that the characterization of such conditions is important to improve clinical approaches for wild animals kept in captivity.
Footnotes
Acknowledgements
The authors thank LAB&VET Diagnostics and Veterinarian Consultation Ltd. for providing bacterial strains, Dr. James R. Johnson for kindly providing the controls strains, and Glória Jafet for the assistance with the picture.
a.
Difco Laboratories Inc., Sparks, MD.
b.
API, bioMérieux, Marcy l’Etoile, France.
c.
Cefar, Jurubatuba, SP, Brazil.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was made by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP; grant #06/54343-2).
