Abstract
A 33-year-old brown bear (Ursus arctos) was evaluated for chronic cough, partial anorexia, and lethargy in early fall of 2009. Radiographs revealed a generalized increase in interstitial density with focal lung field consolidation and air bronchograms more prevalent in the cranial lung lobes. Tracheal sputum and wash fluid grew mixed bacteria and 2 species of Candida on bacterial and fungal cultures, respectively. Serum was negative for antibodies to Aspergillus, Blastomyces, Coccidioides, and Histoplasma by semiquantitative radial immunodiffusion. Antimicrobial and antifungal treatment was administered. The bear died 1 month after entering hibernation. Gross necropsy revealed coalescent nodules and sheets of firm tan tissue covering pleural surfaces of the thoracic cavity and within pulmonary parenchyma, enlarged mesenteric lymph nodes, and intestinal ulcerations. Histopathology revealed granulomatous inflammation with intrahistiocytic yeast, consistent with Histoplasma organisms, in lung, diaphragm, mesenteric lymph nodes, intestine, and adrenal glands. Molecular analysis performed on DNA isolated from lung tissue, including conventional polymerase chain reaction (PCR) targeting the internal transcribed spacer region for the ribosomal RNA gene complex and real-time PCR targeting the gene encoding a unique region of M specific protein, identified the organism to be 100% identical to Histoplasma capsulatum with an average of 4.9 × 107 gene copies per gram of tissue. The present report describes histologic and molecular techniques for diagnosing histoplasmosis.
A 33-year-old female brown bear (Ursus arctos), born and housed throughout her life at the Milwaukee County Zoo (Milwaukee, Wisconsin), was immobilized for evaluation on September 29, 2009 for chronic cough of 1-month duration, partial anorexia, lethargy of 2-day duration, and worsening hindlimb stiffness that was first observed in July 2008. The bear was in good body condition and had a body weight of 288 kg at the time of immobilization. The bear was considered slightly febrile with a temperature of 38.6°C Radiographs revealed a generalized increase in interstitial density with focal lung field consolidation and air bronchograms that were more prevalent in the cranial lobes. Ventral bridging discospondylosis was observed involving multiple thoracic and lumbar vertebrae, with the lumbar lesions most severe. Blood was submitted for culture, complete blood cell count (CBC), and serum chemistry, and tracheal wash and sputum samples were submitted for aerobic bacterial and fungal cultures to Aurora Consolidated Laboratories (ACL; Milwaukee, WI). Complete blood cell count and serum chemistry showed a slightly decreased hematocrit (40.5%; International Species Inventory System [ISIS], mean +/− standard deviation: 46.8 ± 5.1) with slightly decreased mean cell volume (64.5 fl; ISIS: 73.4 ± 4.5) and mean cell hemoglobin (23.6 pg; ISIS: 37.9 ± 13.0), and mildly elevated total protein (8.6 g/dl; ISIS: 7.3 ± 0.7). No other abnormalities were identified on CBC or serum chemistry. No organisms grew in blood culture. a Mixed bacteria, considered normal oral flora, were identified on bacterial culture of the sputum using sheep blood, chocolate, MacConkey, and colistin sulfate and nalidixic acid agars. Serum and tracheal sputum samples were submitted to Marshfield Labs–Veterinary Service (MLVS; Marshfield, WI) for serology and fungal cultures, respectively. Serologic testing was negative for antibodies to Aspergillus, Blastomyces, Coccidioides, and Histoplasma by semiquantitative radial immunodiffusion assay. b Cytology performed on the sputum sample revealed moderate numbers of neutrophils, macrophages, eosinophils, and few erythrocytes; no fungal or bacterial elements were identified. Fungal cultures were held for 4 weeks on Sabouraud dextrose agar, brain–heart infusion agar, and mycobiotic agar at both laboratories. Fungal organisms identified by culture were Candida lipolytica at ACL and Candida albicans at MLVS. Antimicrobial and antifungal treatment was initiated at the time of immobilization with a single intravenous dose of 6,000 mg of cefazolin c administered while the bear was under anesthesia followed by 6,000 mg of oral cephalexin d given twice daily for 17 days. With no resolution of coughing after 10 days of cephalexin treatment, oral enrofloxacin e was added for 10 days (1,547 mg once per day), and the cough was reported to have resolved by the 6th day of treatment. Once the fungal culture results were known, antibiotic therapy was followed by fluconazole f given orally for 10 days (800 mg per day). Anti-inflammatory medication, meloxicam, g was also given for the spinal arthritis (22.7 mg orally once daily). The bear’s cough remained resolved until she went into hibernation in late November. On January 14, 2010 she was found dead. Body weight at the time of death was 182 kg with a 36.8% loss of body weight since initial evaluation 3 months prior to death.
Gross necropsy was performed revealing coalescent proliferate nodules and plaques of firm tan tissue covering >90% of the parietal and pleural surfaces of the thoracic cavity, tightly adhering the lungs to the thoracic walls, to the markedly expanded and effaced mediastinum, and to the surface of the pericardial sac. Along the ventral and ventrolateral surfaces of the lung lobes and mediastinum were approximately 50, up to 2 cm in diameter, cystic structures that had transparent walls and contained clear light yellow watery fluid (Fig. 1). Numerous firm, tan, poorly demarcated, and irregularly shaped firm to soft, tan masses ranging in size from pinpoint to 3 cm in diameter, were scattered throughout the otherwise diffusely dark red and wet pulmonary parenchyma. Multifocally expanding the mucosa of the small intestine, most concentrated in the duodenum and proximal one-third of the jejunum, were >200, up to 1 cm in diameter, often centrally ulcerated, firm tan nodules (Fig. 2). Several mesenteric lymph nodes were enlarged, with the largest located in the root of the mesentery and being 8 cm in greatest dimension; enlarged lymph nodes were tan and firm with soft centers and bulged on cut section. Focally within the right adrenal gland, at the corticomedullary junction, was a 0.5 cm in diameter, poorly demarcated area of grayish tan, mildly firm tissue that bulged on cut section. Additional significant findings included severe chronic bilateral osteoarthritis of the shoulders, elbows, and stifles along with mild articular cartilage erosions in the hips.
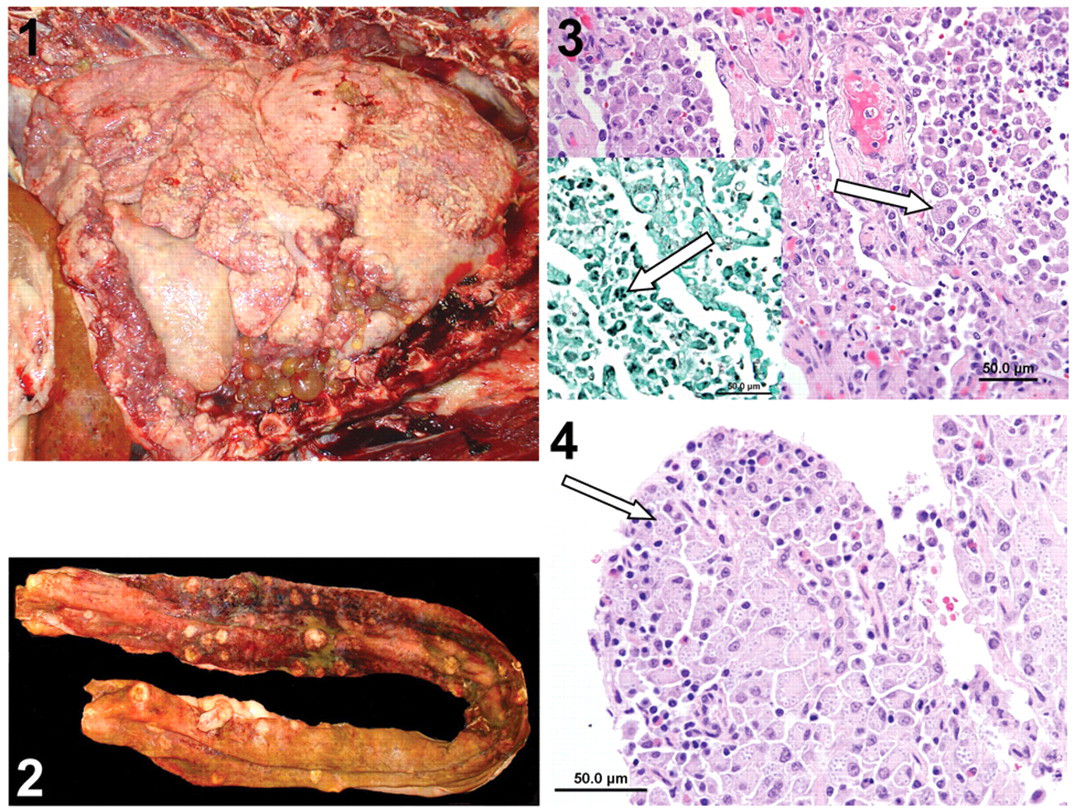
Thoracic cavity; brown bear (Ursus arctos). Coalescent proliferative nodules and plaques of firm tan tissue covering >90% of the parietal and pleural surfaces of the thoracic cavity and markedly expanded and effaced mediastinum. Up to 2 cm in diameter cystic structures having transparent walls and contained clear light yellow watery fluid were seen along the ventral and ventrolateral surfaces of the lung lobes and mediastinum.
Fresh lung tissue was collected and frozen at –20°C. A standard representative set of visceral tissues and the eyes were collected and fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4–5 µm, and stained with hematoxylin and eosin. Additional sections of lung were stained with Grocott–Gomori methenamine silver (GMS), periodic acid–Schiff (PAS), and mucicarmine stains. All examined sections of lung tissue were affected and effaced to varying extents by inflammation, necrosis, and fibrosis. Inflammation was composed of numerous epithelioid macrophages and few to moderate multinucleated giant cells that formed dense sheets that blended into large, often-mineralized centers of necrosis. Moderate numbers of lymphocytes, plasma cells, and fewer neutrophils were scattered within and at the periphery of the described granulomatous inflammation. Remaining airways were often filled with mixed inflammatory cells, predominantly macrophages, and varying amounts of mucous, and few to moderate free erythrocytes. Within the cytoplasm of most of the macrophages and multinucleated giant cells, and occasionally extracellularly, were numerous round to oval, 2–4 µm diameter, yeast organisms that had basophilic centers and peripheral clear zones enclosed by an indistinct outer cell wall that stained with both GMS (Fig. 3) and PAS but did not stain with mucicarmine stain. Histomorphology and staining characteristics were consistent with Histoplasma capsulatum. Alveolar walls within viable regions of the lungs were moderately to markedly thickened by fibrous connective tissue, and sheets of fibrovascular connective tissue (fibrosis) replaced large areas of pulmonary parenchyma. The small intestinal nodules were composed of dense sheets of granulomatous inflammation that contained numerous intracytoplasmic yeast organisms, similar to that identified in the lung (Fig. 4). Similar inflammation with intracytoplasmic yeast organisms regionally effaced both adrenal glands. Incidental findings included a focal thyroid cystadenoma and bilateral ovarian changes including subsurface epithelial structures, cystadenomas, and arteriosclerosis with vascular mineralization and osseous metaplasia. The eyes were submitted to the Comparative Ocular Pathology Laboratory of Wisconsin at the University of Wisconsin School of Veterinary Medicine and were considered grossly and histologically normal.
Formalin-fixed, paraffin-embedded (FFPE) and fresh-frozen lung tissues were sent to the Mycology Laboratory at the New York State Department of Health (Albany, New York) for molecular characterization and confirmation of the pathogenic fungal organism identified on histopathology. The FFPE and fresh-frozen lung tissues were processed for DNA extraction using a commercial kit h per manufacturer’s instructions, with the following minor modification: after the tissue lysis step, glass beads were added, and the mixtures were disrupted in a cell disrupter for 20 min. Genomic DNA from 1 standard strain of H. capsulatum (M619) was prepared as described. 2 The internal transcribed spacer (ITS) region (ITS1-5.8S-ITS2) of the ribosomal gene was amplified using primer set V1827 (ITS5) 5’-GGAAGTAAAAGTCGTAACAAGG-3’ and V50 (ITS4) 5’-TCCTCCGCTTATTGATATGC-3’. Polymerase chain reaction (PCR) was performed in 25 µl of a reaction mixture containing 2 µl of DNA, 1× buffer containing 1.5 mM of MgCl2, 0.02% bovine serum albumin, 0.2 mM of each deoxyribonucleotide triphosphate, 0.4 µM of each primer, and 0.5 U DNA polymerase. i The PCR set-up included initial denaturation at 94°C for 1 min, annealing at 55°C for 1 min, and extension at 68°C for 2 min, and a final extension step of 68°C for 3 min. The PCR products were electrophoresed on 2% agarose in Tris–borate–ethylenediamine tetra-acetic acid–ethidium bromide buffer (pH 8.3) and photographed with an imaging system. j Amplicon was purified k and sequenced with the same primers used for PCR.
DNA extracted from the fresh-frozen lung tissue produced a 700-bp amplicon against ITS4/5 primer set, which was identical to the size produced by genomic DNA extracted from the control strain of H. capsulatum (Fig. 5). Sequencing and BLAST search using 2 databases, GenBank (www.ncbi.nih.gov.genbank/index/html) and Centraalbureau voor Schimmelcultures (www.fungalbiodiversitycentre.com/yeast/BioloMICSSequences.aspx), identified the PCR amplicon to be 100% identical to H. capsulatum. The ITS gene sequence of this amplicon was deposited in GenBank (accession no. HM439693). The DNA extracted from FFPE tissues did not yield any amplicon.
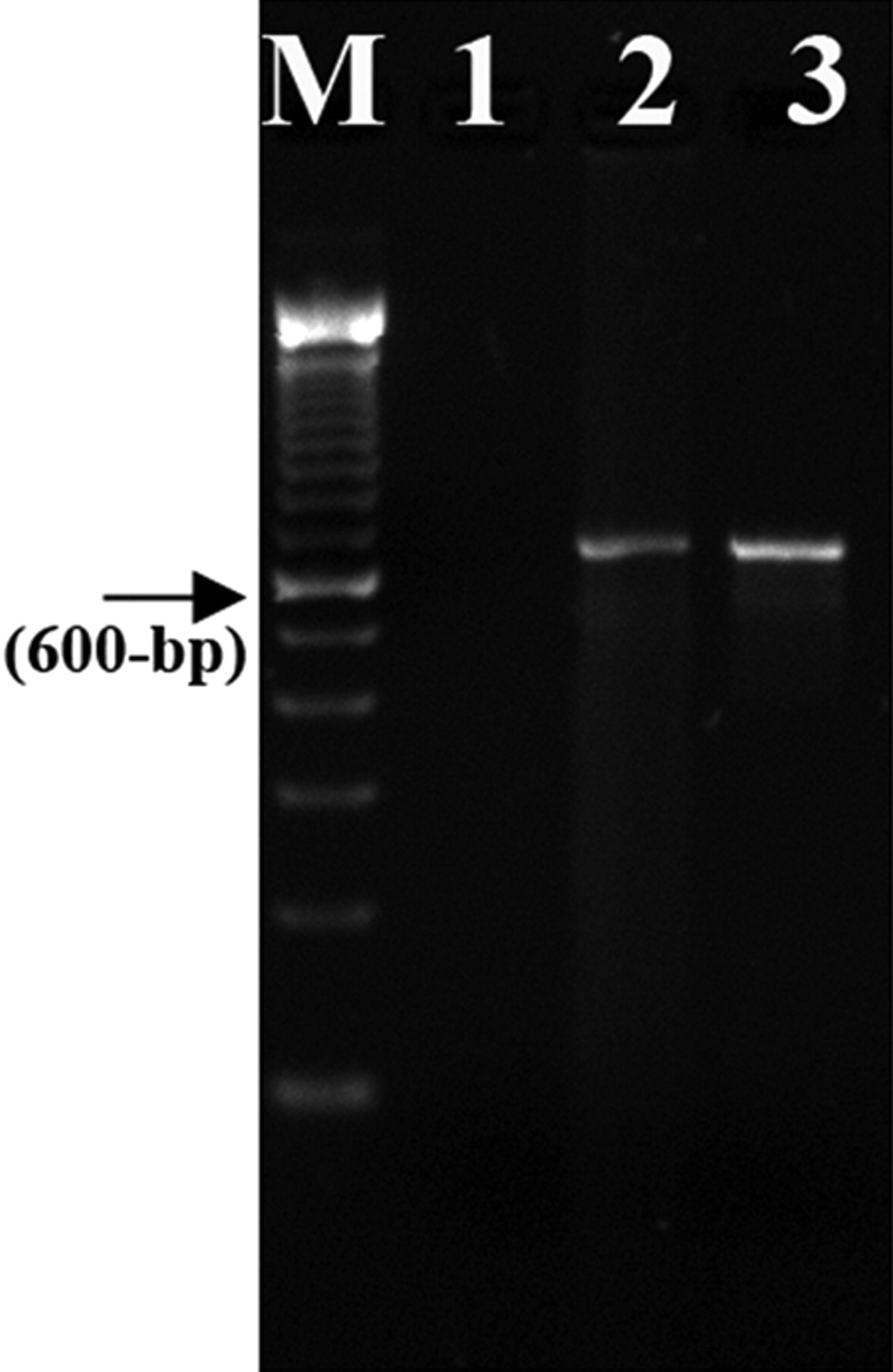
DNA extracted from fresh-frozen lung tissue produced a 700-bp amplicon against ITS4/5 primer set, which was identical to the size produced by genomic DNA extracted from the control strain of Histoplasma capsulatum. Lane M: 100-bp DNA ladder; lane 1: no template control; lane 2: brown bear (Ursus arctos) tissue DNA (fresh-frozen lung); lane 3: H. capsulatum DNA (positive control).
To quantify the DNA concentration of H. capsulatum in the bear lung tissue samples, a quantitative real-time PCR (real-time qPCR) assay was performed using primers and probes designed from a unique region of the gene encoding M antigen specific for H. capsulatum. 8 The real-time qPCR assay was conducted in commercial system. j Duplicates of H. capsulatum genomic DNA standard (0.1 pg to 10,000 pg/µl) and diluted tissue samples (10–1,000 fold) were run including 1 negative control (no template control [NTC]). The average threshold cycle (Ct) values were used for the calculation of the fungal load. The real-time qPCR conditions included a single denaturation cycle of 95°C for 10 min followed by 40 cycles at 95°C for 15 sec and annealing at 60°C for 1 min each. During the annealing–extension step, the PCR system j monitored real-time PCR amplification by quantitatively analyzing the fluorescence emissions. The specificity of primers and probes were extensively investigated against other dimorphic and common molds prior to use in the current study (Chaturvedi S, Peck M, Sidamonidze K: 2009, Development of real time PCR assay for Histoplasma capsulatum based on M antigen. In: Proceedings of the 109th General Meeting of the American Society of Microbiology, abstract no. C-072:96, Philadelphia, PA).
A standard curve was generated by amplification of a 10-fold dilution series of genomic DNA from 1 standard strain of H. capsulatum. The correlation between Ct values and the target DNA concentration was high (R 2 = 0.985). Also, the standard curve obtained demonstrated that the selected primer and probe set were highly accurate over a linear range of at least 5 orders of magnitude (Fig. 6). To quantify H. capsulatum load in the fresh-frozen lung tissue, DNA extracted from 200–300 mg of lung tissue from 4 randomly excised sites were diluted 10–1,000 folds, and 2 µl from each dilution was used in the PCR assay in duplicate. All tissue specimens were positive for H. capsulatum DNA with Ct values ranging from 19 to 32, while NTC did not yield detectable fluorescence signal (Table 1). Based on 28-Mb haploid genome with a single copy of the target gene, the average copy number calculated was 4.9 × 107 copies/g of tissue (Table 1). DNA extracted from paraffin embedded tissue was also positive for H. capsulatum DNA (Ct = 28.30 ± 0.683) confirming the sensitivity of the PCR assay.
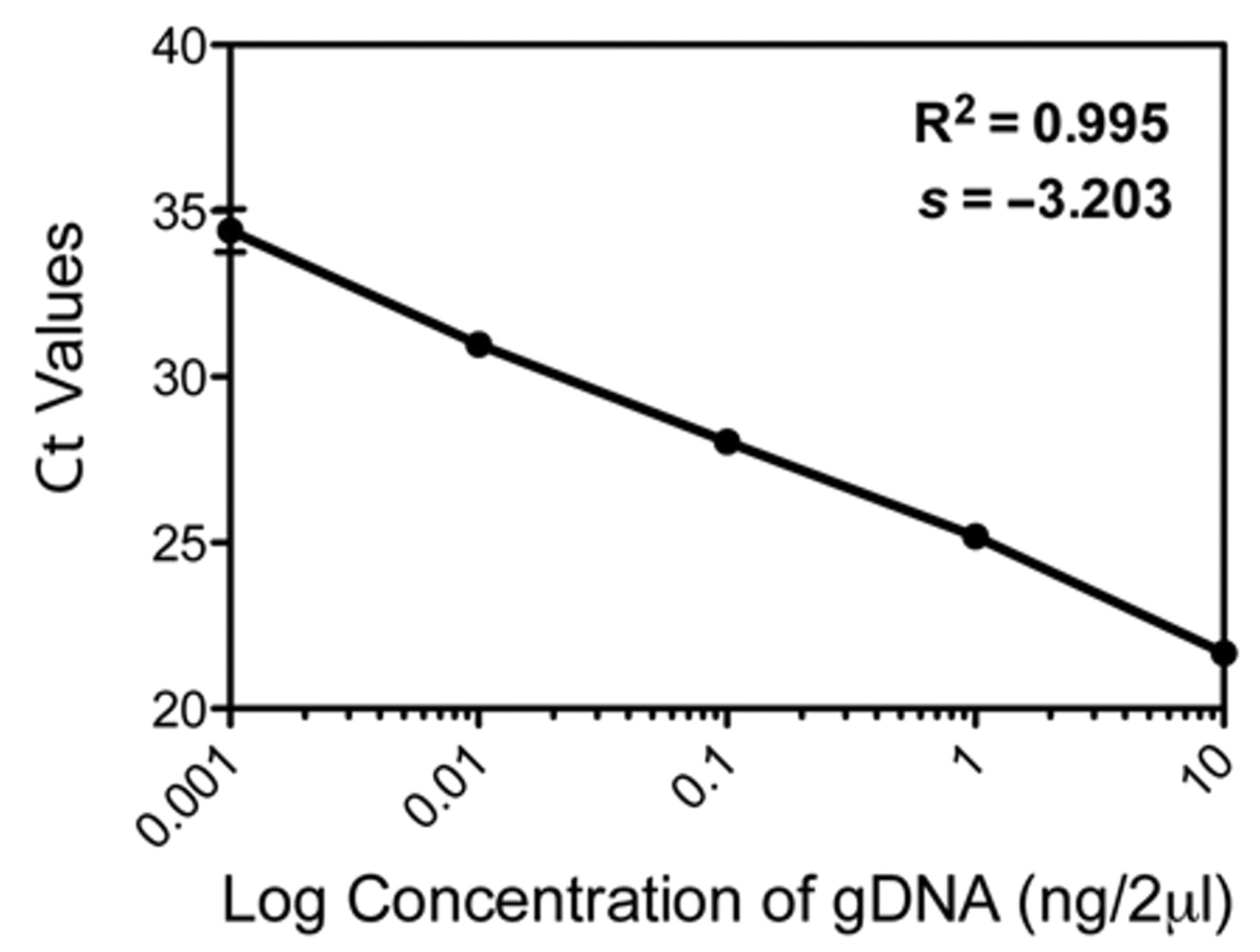
Standard curve obtained demonstrated that the selected primer and probe set were highly accurate over a linear range of at least 5 orders of magnitude. Ct = threshold cycle; gDNA = genomic DNA.
Quantitative real-time polymerase chain reaction quantification of Histoplasma capsulatum genomic DNA in bear lung tissue samples from the current study.*
Ct = threshold cycle; SD = standard deviation; NTC = no template control; NA = not applicable; – = outside detection limit of standard curve.
Histoplasma capsulatum infections in veterinary medicine have been reported in multiple species, most commonly in dogs and cats, 4,7,25 with fewer reports in horses, 18 and a few case reports in other species including captive maras (or Patagonian cavies, Dolichotis patagonum), 16 an Atlantic bottlenose dolphin (Tursiops truncates), 9 a sea otter (Enhydra lutris), 14 an owl monkey (or Ma’s night monkey, Aotus nancymai), 23 and badgers (Meles meles). 24 Fungal infection with H. capsulatum is also recognized as an important human pathogen, particularly in immunocompromised individuals. 17 Histoplasma capsulatum has a worldwide distribution and is typically found in temperate and subtropical climates. In the United States, the majority of reported cases of histoplasmosis occur in regions along the Ohio, Missouri, and Mississippi Rivers, although favorable environmental conditions such as nitrogen-rich soil and environments contaminated with bird or bat excrement can provide a suitable niche for survival of H. capsulatum within or outside of the recognized endemic areas. 1,13,25 The source of infection in the bear in the current report remains uncertain, as the bear was born and lived its entire life in southeastern Wisconsin at the Milwaukee County Zoo, did not live on natural substrate, and there were no bats or accumulations of bird excrement noted in or around the bear dens. Studies indicate that hibernation suppresses both the innate and the adaptive immune systems, therefore leading to increased infection risk. 3 The effects of hibernation on the immune system are very well illustrated by the current emerging, fungus-associated white nose syndrome in hibernating bats. 6 Although the bear in the present report was ill and its immune status unknown prior to entering hibernation, immune suppression secondary to hibernation may have allowed rapid progression of the H. capsulatum infection.
Identification and organ load determination of dimorphic fungi such as Histoplasma spp. are typically performed using culture and colony forming unit techniques. Artifact due to sample handling may result in false-negative results, and culture creates a biohazard risk for laboratory workers. Storage of tissue at freezing temperatures is known to be detrimental to H. capsulatum culture recovery, and room temperature storage for as little as 24 hr postcollection greatly decreases to completely inhibits culture viability of H. capsulatum from sputum or tracheobronchial wash fluid. 21 Candida albicans, frequently cultured from human sputum, and the only fungus identified by tracheobronchial wash culture in the present case, is known to be a potent inhibitor of H. capsulatum growth on Sabouraud dextrose agar due to decreased pH. 10 However, naturally buffered media, such as blood heart infusion agar, allow for Histoplasma growth even in the presence of other yeasts, such as Candida spp. 5,20 In the present case, the sputum sample and tracheal wash may have contained too few Histoplasma yeasts for culture or sample handling may have hindered growth. Similar to the finding in the present case, other publications report false-negative serological results by agar gel radial immunodiffusion in animals with active fungal infections, indicating this technique to be of questionable reliability and perhaps of limited use as a diagnostic tool. 15,22,25
Histoplasma spp. are dimorphic fungi that live in the mycelial or mold form and produce microconidia and macroconidia in soil and at 30°C or room temperature culture conditions. Infection typically occurs following inhalation of aerosolized microconidia with subsequent conversion to the yeast form, budding reproduction, and local and/or disseminated disease in the host. Disease depends on both immune status and the number of inhaled microconidia or infective dose. 11 The culture-based methods available currently are designed to isolate the mold form of the fungus, as the yeast form is highly sensitive to both cycloheximide and chloramphenicol, which are used to inhibit the fast growing molds and bacteria present in clinical specimens. 12 Exposure to microconidia during culture thus poses a biohazard risk to laboratory workers.
A 2010 study has shown that real-time PCR provides better sensitivity than culture for identifying H. capsulatum from human sputum samples. 19 The real-time qPCR assay enables more rapid enumeration of H. capsulatum compared with the conventional serial dilution plating, greatly decreases the risk of human exposure by eliminating the need for culture, and as shown in the current report, can be used with both fresh-frozen or FFPE tissue whereas standard PCR was only of diagnostic value in fresh-frozen tissue. In addition to the increased diagnostic sensitivity that PCR provides, sequencing of the amplified product allows for speciation of the pathogen, which is important for assessing geographical distribution and range expansion and early identification of the emergence of potential new pathogens.
Footnotes
Acknowledgements
The authors thank Dr. Richard Dubielzig of the Comparative Ocular Pathology Laboratory of Wisconsin for histologic evaluation of the ocular tissues from this case. The authors also acknowledge Tanya Victor for data analysis and Molecular Genetic Core for DNA sequencing.
a.
BacT/ALERT®, bioMérieux Inc., Durham, NC.
b.
ID Fungal Antibody System, Immuno-Mycologics Inc., Norman, OK.
c.
West-Ward Pharmaceutical Corp., Eatontown, NJ.
d.
World-Gen LLC, Paramus, NJ.
e.
Bayer Health Care LLC, Animal Health Division, Shawnee Mission, KS.
f.
IVAX Pharmaceuticals Inc., Miami, FL.
g.
Unichem Laboratories Ltd., Pilerne, Bardez, Goa, India.
h.
QIAamp®, Qiagen Inc., Valencia, CA.
i.
JumpStart™ KlenTaq® LA, Sigma-Aldrich, St. Louis, MO.
j.
iQ™5, Bio-Rad Laboratories, Hercules, CA.
k.
ExoSAP-IT, USB Corp., Cleveland, OH.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
