Abstract
An 11-year-old Oldenburg mare presented with a 3-month history of weight loss and swelling of the posterior right mandible. Physical examination and radiographs showed a soft-tissue mass in the right mandible with foci of mineralization, periosteal proliferation, and a retained molar. The tumor increased in size over several weeks, and the mare was euthanized. On necropsy, a 24 cm × 15 cm × 15 cm firm, white mass had obliterated the posterior right mandible. The mass was multinodular with discrete clusters of blood-filled cystic spaces. Histologically, the mass was composed of neoplastic odontogenic epithelium and pulpal mesenchyme with an accumulation of eosinophilic material resembling dentin or enamel. Microscopic and immunohistochemical staining features of the neoplasm were most consistent with an ameloblastic fibro-odontoma.
Tumors of odontogenic origin are rare in domestic animals and, due to their similar features, may present diagnostic challenges. 3,4 The simplest classification system, based on Thoma and Goldman's 1946 classification, separates these tumors into those with only odontogenic epithelium, those with only odontogenic mesenchyme, or mixed tumors. 4 Tumors of odontogenic epithelium include ameloblastoma, amyloid-producing odontogenic tumor, and canine acanthomatous ameloblastoma, and those of odontogenic mesenchyme include cementoma and cementifying fibroma. 6 Tumors of odontogenic epithelium with odontogenic mesenchyme include feline inductive odontogenic tumor, complex odontoma, compound odontoma, ameloblastic fibroma, and ameloblastic fibro-odontoma (AFO). 6 Some authors consider both complex and compound odontomas to be hamartomas and not neoplasms. 6 This report details the clinical and pathologic features of an AFO in an adult horse.
An 11-year-old Oldenburg mare presented with a 3-month history of weight loss and swelling of the posterior right mandible, was humanely euthanized, and was submitted for necropsy. Physical examination revealed a large, firm swelling over the right masseter muscle and soft tissue protruding into the oral cavity along the right mandible. Radiographs showed a soft-tissue mass with foci of mineralization, periosteal proliferation along the right mandible, and a retained molar positioned horizontally within the mass caudal to the mandibular arcade (Fig. 1). Postmortem examination revealed a 24 cm × 15 cm × 15 cm firm, white, circumscribed mass, which had obliterated the posterior right mandible and was bounded dorsally by ulcerated buccal mucosa. An intact molar was embedded within the center of the mass (Fig. 2). On cut surface, the mass was multinodular with discrete clusters of less than 1 cm blood-filled cystic spaces (Fig. 3). Differential diagnoses included fibroma, fibrosarcoma, osteoma, osteosarcoma, squamous cell carcinoma, and odontogenic tumors.
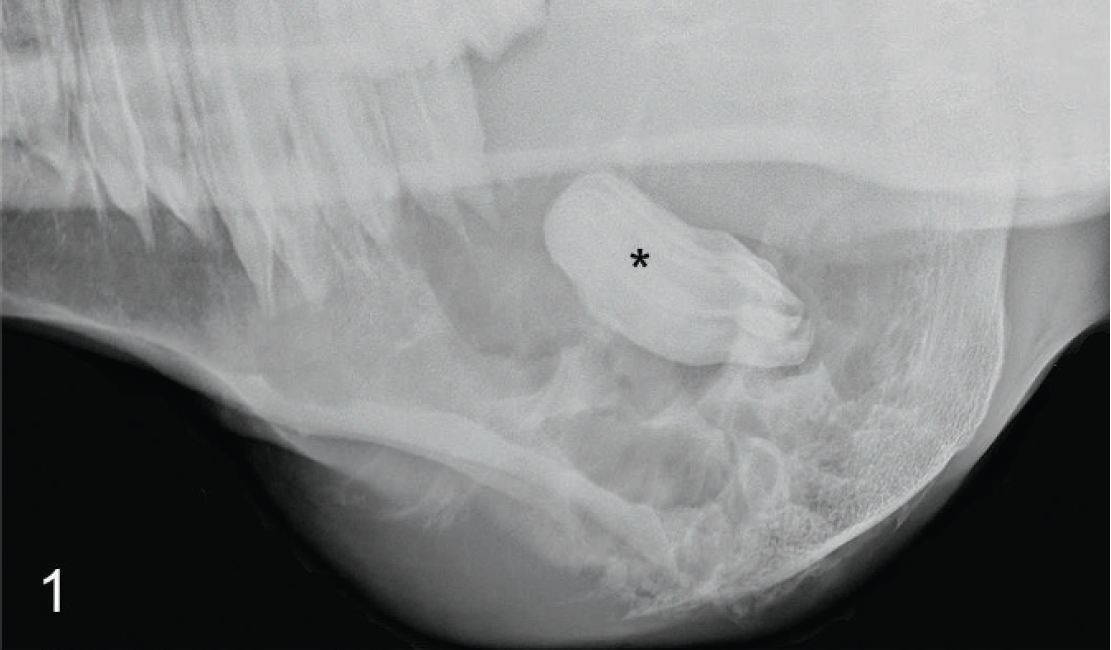
Mandibular mass; horse. Radiograph showing a soft-tissue mandibular mass with a retained molar in the center (*).

Mandibular mass; horse. Firm, white, multinodular mass obliterating the posterior mandible. Intact molar (*) embedded within the mass.

Mandibular mass; horse. Cross-section of mandibular mass showing clusters of blood-filled cystic spaces.

Mandibular mass; horse. Follicular odontogenic epithelium associated with odontogenic mesenchyme. Hematoxylin and eosin. Bar = 200 μm.

Mandibular mass; horse. Mesenchyme composed of interlacing spindled to stellate cells resembling dental pulp. Hematoxylin and eosin. Bar = 50 μm.

Mandibular mass; horse. Odontogenic epithelium composed of polygonal cells with moderately abundant eosinophilic cytoplasm and round to oval to fusiform nuclei. Hematoxylin and eosin. Bar = 50 μm.

Mandibular mass; horse. Eosinophilic fibrillar material with basophilic tubular-like structures (*) between and surrounding epithelial cells. Hematoxylin and eosin. Bar = 20 μm.

Mandibular mass; horse. Follicular odontogenic epithelium (red) associated with odontogenic mesenchyme (blue). Masson trichrome. Bar = 200 μm.
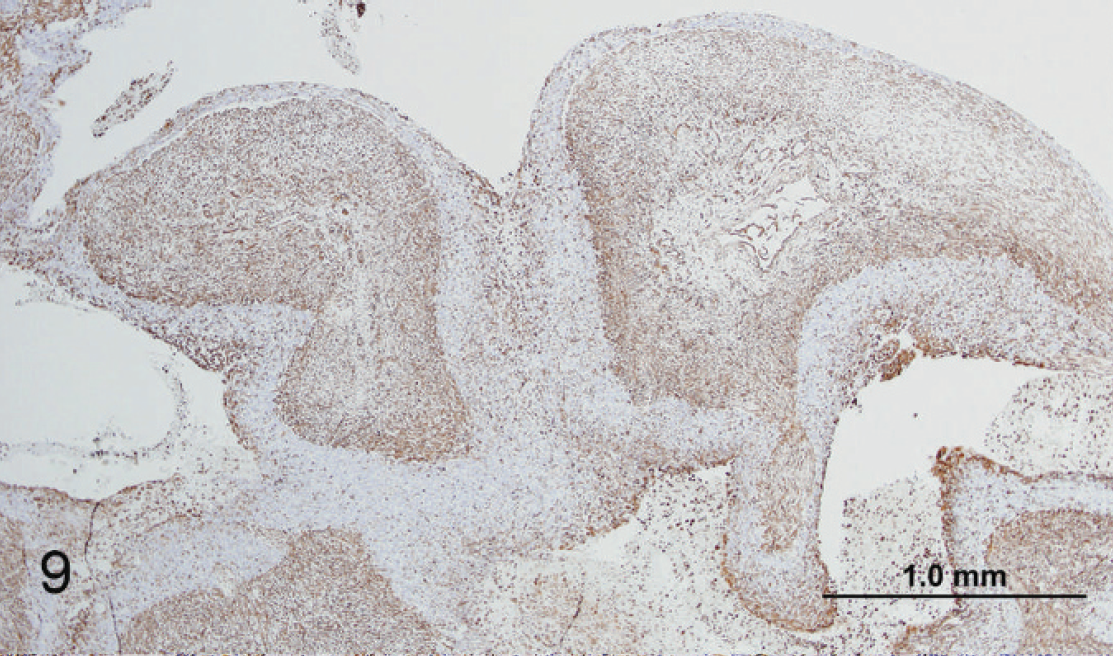
Mandibular mass; horse. Low magnification showing intracytoplasmic staining for vimentin (brown) of mesenchymal cells. 3,3′-diaminobenzidine chromogen, hematoxylin counterstain. Bar = 1 mm.

Mandibular mass; horse. Higher magnification showing intracytoplasmic staining for vimentin (brown) of mesenchymal cells. 3,3′-diaminobenzidine chromogen, hematoxylin counterstain. Bar = 50 μm.

Mandibular mass; horse. Low magnification showing intracytoplasmic staining for pancytokeratin (brown) of odontogenic epithelial cells. 3,3′-diaminobenzidine chromogen, hematoxylin counterstain. Bar = 200 μm.

Mandibular mass; horse. Higher magnification showing intracytoplasmic staining for pancytokeratin (brown) of odontogenic epithelial cells. 3,3′-diaminobenzidine chromogen, hematoxylin counterstain. Bar = 50 μm.
Tissues were fixed in neutral buffered, 10% formalin solution, decalcified in Kristensen solution (equal parts 8 N formic acid and 1 N sodium formate), processed routinely, embedded in paraffin, sectioned at approximately 4 μm, and stained with hematoxylin and eosin. Additional sections were stained with Masson trichrome, periodic acid-Schiff (PAS), and Congo red. Immunohistochemical evaluation was performed by applying the following primary antibodies to additional sections: anti-pancytokeratin (mouse monoclonal, 1:100), a anti-cytokeratin cocktail [AE1/AE3] (mouse monoclonal 1:200), b and anti-vimentin [V9] (mouse monoclonal 1:600). b For positive controls, canine skin was used with both anti-cytokeratin antibodies and canine intestine was used with anti-vimentin antibodies. The substrate chromogen system 3,3′-diaminobenzidine a was used to visualize sites of primary antibody localization.
Histologically, a highly cellular, circumscribed, unencapsulated, infiltrative odontogenic neoplasm extended from the ulcerated mucosa and replaced the posterior mandible. The neoplasm was composed of odontogenic epithelium predominantly arranged in follicular and plexiform patterns associated with odontogenic mesenchyme consisting of abundant spindled to stellate cells, which resembled dental pulp (Figs. 4, 5). Follicular and plexiform structures were composed of closely apposed, pleomorphic polygonal cells with distinct cell borders; moderately abundant, pale eosinophilic cytoplasm; irregularly round to oval to fusiform nuclei with stippled chromatin; and 1–3 nucleoli (Fig. 6). Between and occasionally surrounding cells, irregular islands, and trabeculae was an eosinophilic fibrillar material, which in some foci was mineralized or contained granular to tubular-like basophilic structures, suggestive of dentin (Fig. 7). There were moderate anisocytosis and anisokaryosis. Mitoses were 12 per 10 high-powered fields at 400X. Throughout the neoplasm were multiple foci of hemorrhage and necrosis. There was no evidence of vascular invasion.
With a Masson trichrome stain, the cytoplasm of spindled and stellate cells stained blue, indicating connective tissue origin, and epithelial cells stained red, indicating the presence of keratin (Fig. 8). The eosinophilic fibrillar material stained negative for both carbohydrates and amyloid with PAS and Congo red stains, respectively (not shown). Spindled and stellate cells stained positive for vimentin, indicating mesenchymal origin (Figs. 9, 10). Polygonal cells arranged in follicular and plexiform structures stained positive for pancytokeratin, indicating epithelial origin (Figs. 11, 12), and negative for cytokeratin AE1/AE3, indicating the absence of these proteins (not shown). In the present case, history and gross and microscopic findings along with the results of immunohistochemistry were most characteristic of an AFO, which was associated with a retained molar.
Of the differential diagnoses considered, the observed mass is most consistent with a neoplasm of odontogenic origin. Unlike squamous cell carcinomas, which are composed of epithelial cells and associated with keratin formation, the observed neoplasm had a distinct mesenchymal and epithelial component. Fibromas, fibrosarcomas, osteomas, and osteosarcomas are mesenchymal neoplasms that lack an associated epithelial component. Thus, the observed neoplasm is most likely of odontogenic origin with both an epithelial and a mesenchymal component.
Compound and complex odontomas, as well as ameloblastic fibromas and AFOs, are odontogenic neoplasms with both an epithelial and a mesenchymal component. Compound odontomas represent the most complete odontogenic maturation of all the odontogenic neoplasms and often contain fully differentiated tooth-like structures. 10 They are composed primarily of dense collagenous and vascular connective tissue in which there are cords of odontogenic epithelium. 10 Complex odontomas contain dentin, enamel matrix, odontogenic epithelium, and cementum in horses and rodents but do not form tooth-like structures. 10 Ameloblastic fibromas are the least differentiated odontogenic neoplasm that has both odontogenic epithelium and mesenchyme. 10 Histologically, they consist of cords of well-differentiated odontogenic epithelium within a matrix of primitive mesenchyme, which resembles dental pulp. 3,6 The islands and cords of epithelium resemble the enamel organ with a basal lamina between the epithelium and the connective tissue. 10 In an AFO, there is further maturation of odontoblasts than in ameloblastic fibromas, evidenced by the presence of dentin and enamel matrix. 10 Based on the lack of fully differentiated dental components or tooth-like structures and the presence of dentin, a diagnosis of an AFO was made.
Ameloblastic fibroma and AFO are rare in all domestic species but are the most commonly diagnosed odontogenic neoplasm in the mandibles of young cattle. 3,5 Ameloblastic odontomas, originally diagnosed in human beings, are now recognized in the veterinary literature as being ameloblastic fibro-odontomas, odonto-ameloblastomas, or developing odontomas. 4 Ameloblastic odontomas, occurring within the maxilla, have been reported from a 6-week-old Arabian foal, a 4-month-old mixed Arab foal, and a 1-year-old Shetland pony. 7,8
Although there is no site predilection in animals, AFOs usually occur in the body or ramus of the mandible in humans. 1 Ameloblastic fibro-odontomas are slow-growing neoplasms, 4 and although some authors suggest their behavior is noninvasive, as seen with ameloblastic fibromas, others equate their behavior with invasive ameloblas-tomas. 2,4 These tumors are generally reported as being locally destructive. However, metastases to the lymph nodes, lungs, liver, and orbital cavity have been reported in a dog, and infiltration of adjacent soft tissues has been reported in a cow. 5,6,9 Radiographically, osteolysis is evident in ameloblastic fibromas, and foci of mineralization within the tumor have been reported. 3 Both odontomas and AFOs contain tooth substance and may be associated with a retained tooth, as seen in the current case. 1 The recommended treatment of mixed odontogenic neoplasms is surgical excision followed by curettage of the bony lesion. 1 If the neoplasm recurs, en bloc resection may be necessary. 1
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
BioGenex Laboratories Inc., San Ramon, CA.
