Abstract
Rabbit hemorrhagic disease virus 2 (RHDV2) has spread across the United States infecting and causing death in domestic and wild rabbits. Immunohistochemistry (IHC) would be a useful tool for the detection of RHDV2 antigen in tissues as it is inexpensive and readily achievable in most diagnostic laboratories. However, there is no readily available antibody for this purpose. To fill this void, we generated an RHDV2 capsid protein VP60–specific antibody in chicken eggs and validated the antibody using formalin-fixed tissues from 5 domestic rabbits naturally infected with RHDV2. Viral antigen was detected immunohistochemically in various tissues, most prominently in hepatocytes and macrophages in liver, and in macrophages in spleen and cecal lymphoid tissue. Intravascular mononuclear cells in lung and renal tubular and biliary epithelium also were immunolabeled. Both nuclear and cytoplasmic immunolabeling were observed. This peptide-generated antibody is a potentially useful tool as an adjunct to reverse-transcription PCR or in situ hybridization for detection of RHDV2 in tissues.
Keywords
Rabbit hemorrhagic disease virus 2 (RHDV2;
To date, the diagnosis of RHVD2 infection has relied largely on reverse-transcription PCR (RT-PCR) and antigen ELISA (Ag-ELISA). In situ hybridization has also been used for the detection of RHDV2 RNA in tissues of naturally and experimentally infected rabbits,7,8 although this technique is expensive and less adaptable to a diagnostic laboratory setting. Others have used immunohistochemistry (IHC) to detect the virus in tissues of both natural and experimental cases; however, the antibodies used were sourced from the OIE Reference Laboratory for RHD (Italy) and are not readily available.1,5 In a 2021 review, IHC was suggested as a method for RHDV2 detection without citing the specific antibody to be used. 1 To address the lack of a readily available antibody for RHDV2 immunodetection, including IHC, our objectives were to produce a polyclonal antibody for RHDV2 IHC using a synthetic peptide and to validate the antibody using tissue specimens derived from natural cases of RHD.
For the production of RHDV2-specific polyclonal antibody, the relevant nucleotide (nt) sequences within the VP60 capsid protein gene of RHDV2 (syn. RHDV2, RHDVb, and Lagovirus GI.2) were obtained from NCBI GenBank. Each gene within the ~7,400-bp genome was aligned in MAFFT 1 using default settings. Initial alignments revealed that isolates shared ≥95% nt homology across the VP60 capsid protein gene, thus making it the most favorable candidate for peptide design. A standard protein BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi) search confirmed that the peptide was not homologous to any rabbit host sequence and shared 83.3% homology to RHDV1. Subsequently, the complete 1,737-nt capsid sequence from a 2020 Texas isolate 2 (GenBank MT506233), representative of RHDV2 isolates from North America, was submitted for peptide design and subsequent polyclonal antibody production (Thermo Fisher). A polyclonal antibody with specificity to an 18–amino acid portion of the synthesized peptide starting at position 544 of the RHDV2 VP60 (LIDLSELVDIRPVGPRPS) was chosen for production in chicken eggs. Following verification of cross-reactivity of the unpurified antibody using liver from a jackrabbit confirmed to be infected with RHDV2 (kindly provided by Karen Fox, Colorado Parks and Wildlife), the antibody was purified by affinity chromatography.
To standardize and validate an IHC protocol using our polyclonal antibody as a potential detection tool, we used tissue specimens derived from 5 recently dead Lionhead rabbits (
During autopsies, tissue specimens, including but not limited to liver, spleen, lung, trachea, heart, brain, kidney, gastrointestinal tract, thymus, and lymph node, were collected in 10% neutral-buffered formalin for histopathology and IHC for RHDV2. In addition, fresh samples of liver, lung, and spleen from 3 of the rabbits were submitted to the Foreign Animal Disease Diagnostic Laboratory (APHIS/NVSL) for Ag-ELISA and RT-PCR testing for RHDV2. For all 3 animals, tissues were positive for RHDV2 by Ag-ELISA, and RHDV2 was detected by RT-PCR.
Formalin-fixed tissues from the 5 rabbits were processed routinely, embedded in paraffin, and 4-µm sections stained routinely with H&E. Additional unstained 4-µm sections were placed on charged slides for IHC as needed. Sections of the following tissues were evaluated: liver, spleen, kidney, lung, trachea, heart, skeletal muscle, adrenal gland, brain, and small and large intestine (5 of 5 rabbits); pancreas and cecal lymphoid tissue (4 of 5 rabbits); bone marrow, thyroid gland, and esophagus (3 of 5 rabbits); lymph node, stomach, sciatic nerve, salivary gland, urinary bladder, and thymus (2 of 5 rabbits); and gallbladder (1 of 5 rabbits).
Using the purified chicken anti-RHDV2 antibody that was generated using a synthetic peptide, the IHC protocol was standardized using formalin-fixed, paraffin-embedded liver from one of the rabbits confirmed by RT-PCR to be infected with RHDV2. Briefly, 4-µm liver sections were deparaffinized and rehydrated through 75%, 95%, and 100% graded ethanols. Subsequent to this step, antigen retrieval was performed in pH 6 citrate buffer (Antigen Retrieval Citra; BioGenex) for 15 min at 110°C in a commercial decloaking chamber (Decloaker; Biocare). The remaining steps of the IHC protocol were performed at room temperature (RT) in an automated tissue stainer (intelliPath; Biocare) starting with 3% H202 for 5 min followed by a protein blocking (Power Block; BioGenex) step for 5 min. Starting with a concentration of 0.19 mg/mL, the chicken anti-RHDV2 polyclonal antibody was diluted 1:350 in a commercial antibody diluent (Renaissance background reduction diluent; Biocare) and applied to the slides for 60 min at RT followed by biotinylated goat anti-chicken antibody (Vector) diluted 1:100 in a commercial antibody diluent (Renaissance background reduction diluent; Biocare) for 10 min. For a negative control, primary antibody was replaced with chicken IgY control serum (Vector). Alkaline phosphatase–labeled streptavidin (4plus streptavidin AP label; Biocare) was then applied to the slides for 10 min, followed by a fast red chromogen (Warp Red; BioCare) for 10 min. Sections were counterstained with hematoxylin, coverslipped, and later examined under a light microscope.
Histologically, severe periportal-to-midzonal or massive hepatocellular necrosis with disruption of hepatic architecture and sometimes associated hemorrhage was noted in 4 rabbits (Table 1; Suppl. Figs. 2–4). Hepatic inflammation in these 4 rabbits was mild and limited to a few lymphocytes, plasma cells, macrophages, and/or heterophils within portal areas. These 4 rabbits also had severe fibrinonecrotizing splenitis. Other than severe splenic congestion, the fifth rabbit did not have any other remarkable hepatic or splenic changes. Cecal lymphoid changes included depletion of germinal centers with accumulation of necrotic cellular debris. All rabbits had variable pulmonary edema and congestion, sometimes with small, multifocal areas of hemorrhage, and moderate-to-marked vascular congestion throughout the trachea, often accompanied by diffuse, submucosal edema. Four rabbits had occasional glomerular fibrin thrombi, 2 had small areas of hemorrhage throughout the ventricular myocardium, and 1 rabbit had fibrin thrombi within small blood vessels at the villus tips in the small intestine.
Signalments, manner of death, rabbit hemorrhagic disease virus 2 (RHDV2) test results, and comparison of histologic lesions observed on H&E staining and immunohistochemical labeling with a polyclonal antibody to RHDV2 nonstructural protein in 5 domestic rabbits from an outbreak of RHDV2 in a single household.

A = adult; F = female; J = juvenile; M = male; ND = testing not done; NE = not examined with H&E staining. RHDV2 testing refers to the results of Ag-ELISA and RT-PCR testing for RHDV2.
+ = histologic lesions observed with H&E staining; – = no histologic lesions observed with H&E staining.
++ = prominent immunolabeling for RHDV; + = minimal or rare immunolabeling for RHDV; – = no immunolabeling for RHDV.
Mucosa was artifactually detached, precluding full evaluation of tissue.
The hepatocytes in all rabbits had moderate-to-strong immunolabeling of RHDV2 viral protein, including the animal that did not have histologic lesions in the liver. Hepatocyte immunolabeling was diffuse, granular in the cytoplasm, and frequently intense in the nucleus (Fig. 1; Suppl. Fig. 5). Two rabbits had cytoplasmic and/or nuclear labeling of biliary ductular epithelium. All rabbits had granular, cytoplasmic labeling within macrophages in both the red and white pulp of the spleen, which was strong in 4 rabbits and minimal in 1 rabbit (Fig. 2). Cecal lymphoid tissue macrophages also had mild-to-strong labeling for RHDV2 in cecal tissue available from 4 rabbits (Fig. 3). Thymus available from 2 rabbits had mild labeling of medullary macrophages. The IHC findings in the lungs were variable: 3 rabbits had no labeling, other than rare intravascular monocytes, and the remaining 2 had rare-to-occasional labeling of macrophages and monocytes within alveoli, alveolar septa, and blood vessels (Fig. 4). Rare-to-occasional cytoplasmic and nuclear labeling was present in renal glomeruli and tubular epithelial cells (Suppl. Fig. 6). In other organs, a few intravascular monocytes sometimes had moderate-to-strong cytoplasmic labeling. Additional staining was seen in scattered macrophages in the thymus and bone marrow of 2 rabbits. The stomach of one rabbit and affected ceca had occasional mucosal epithelial cells with weak-to-moderate labeling. No RHDV2 viral antigen was detected immunohistochemically in the endothelial cells in any of the rabbits.
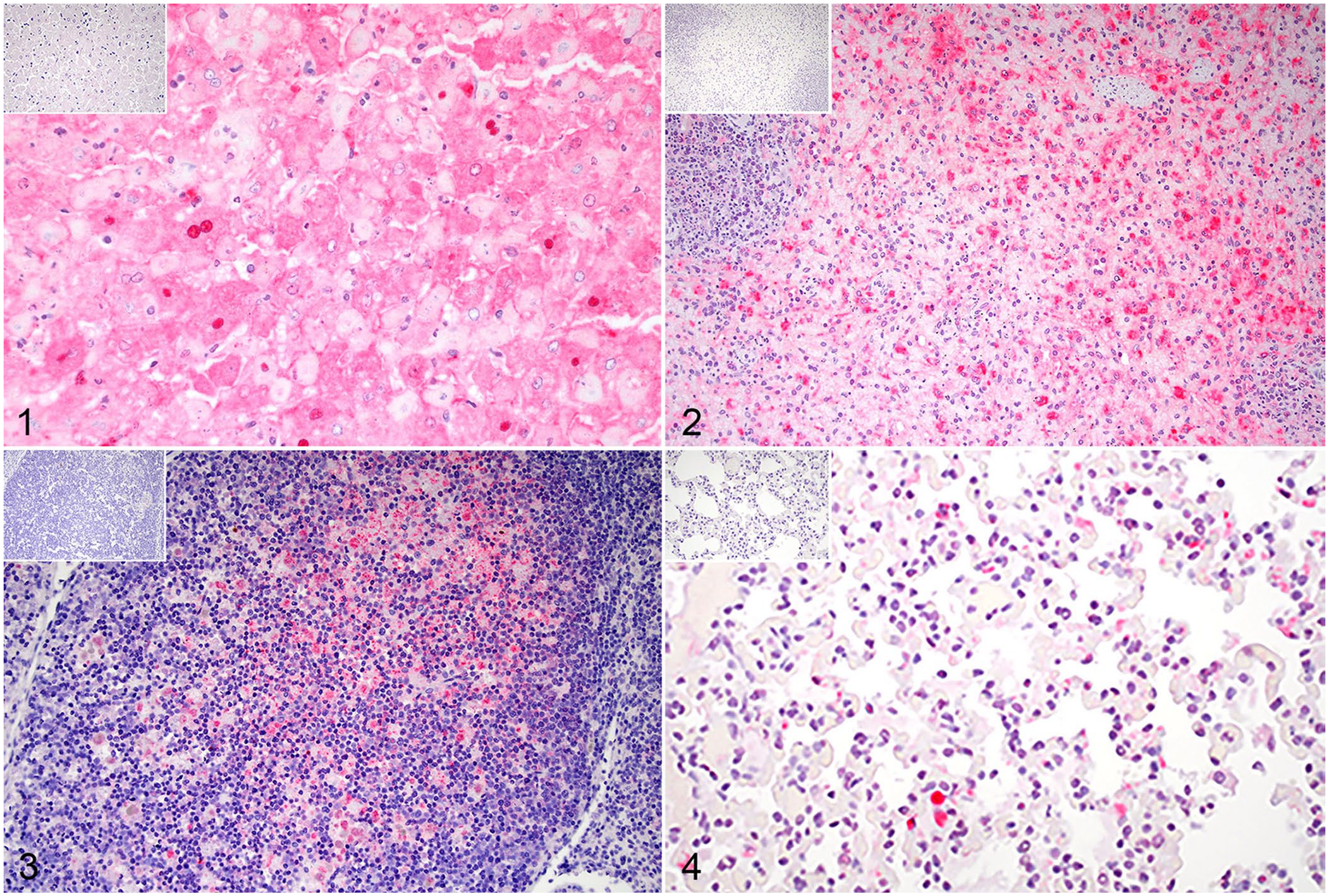
Immunohistochemistry using peptide-generated polyclonal antibody for rabbit hemorrhagic disease virus 2 (RHDV2) capsid protein VP60 on tissues from domestic rabbits naturally infected with RHDV2. Red chromogen and hematoxylin counterstain. Insets are at the same magnification as the corresponding tissue.
The polyclonal antibody that we raised in chicken eggs against a synthetic peptide corresponding to the VP60 gene product of RHDV2 was used successfully for immunohistochemical detection of RHDV2 antigen in rabbit tissues. Among RHDV genes, this gene has a high degree of observed homology across RHDV2 strains even after recombination events. 10 Due to its highly conserved nature, VP60 has been used successfully as the major antigen for development of several subunit vaccines, and we considered it to be a favorable candidate for assays designed to detect all RHDV2 variants. 6 Although additional studies may be needed to further evaluate this antibody, its potential properties for the immunodetection of RHDV1 and RHDV2 variants are anticipated based on our data.
The tissue samples that we examined from naturally infected rabbits had gross and histologic lesions similar to those reported by others who studied natural and experimental RHDV2 infections in rabbits.3–5 Livers were typically pale, and there were hemorrhages within various tissues. Hepatic necrosis and fibrinonecrotizing splenitis were most consistent histologically, but not always present. Similar to other studies that used an antibody provided by the OIE Reference Laboratory for RHD (Italy) for IHC, both nuclear and cytoplasmic immunoreactivity were observed in IHC slides immunostained with our antibody.2,5 Although the number of studied animals are few, the liver, spleen, and cecal lymphoid tissue were found to be the best tissues for the detection of virus by IHC. Interestingly, immunostaining was also present in a liver without histologic changes, underscoring the utility of our IHC protocol for the detection of the target virus.
While hepatocytes were a common target of the virus, other tissues (bile duct, renal tubules, gastrointestinal epithelium) were sometimes immunoreactive. In many tissues, however, immunoreactivity was confined to monocytes and macrophages. This is similar to reports of in situ hybridization for RHDV2 in rabbits naturally infected with RHDV2. 8 It is noteworthy that tropism of RHDV1 for macrophages has also been reported. 9 Microvascular fibrin thrombi and small hemorrhages were seen in some tissues, but direct viral damage of associated endothelium was not likely the cause as we did not note immunoreactivity for RHDV2 in endothelial cells. The possibility of other mechanisms of thrombosis and hemorrhage, such as disseminated intravascular coagulation, need to be explored as likely causes in RHDV2-infected rabbits.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241265370 – Supplemental material for Development of a peptide-generated antibody to rabbit hemorrhagic disease virus 2 VP60 and its immunohistochemical application in natural cases
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241265370 for Development of a peptide-generated antibody to rabbit hemorrhagic disease virus 2 VP60 and its immunohistochemical application in natural cases by Alisia A.W. Weyna, Natalie K. Stilwell, Rachel Anders, Brittany McHale, Rita McManamon and Elizabeth W. Howerth in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the technicians in the Histology Laboratory, Department of Pathology, College of Veterinary Medicine, University of Georgia for producing beautiful H&E and IHC slides.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
