Abstract
A 7-month-old mixed-breed intact female dog was presented to a private veterinarian with a 2 cm in diameter raised, pruritic, alopecic, subcutaneous, fluctuant swelling over the right eye. Cytology of the mass revealed many degenerate neutrophils, moderate numbers of eosinophils, moderate numbers of macrophages, rare mast cells, and few erythrocytes. Rare neutrophils contained a protozoal agent compatible with a Hepatozoon gamont. Real-time polymerase chain reaction of peripheral blood was positive for Hepatozoon canis. The complete sequence identity of the amplified 18S ribosomal RNA fragment from the dog’s blood confirmed H. canis and proved it was relatively distant from the corresponding fragment sequence of Hepatozoon americanum. This case is important in documenting an unusual presentation of infection with H. canis outside of the southern United States.
Hepatozoonosis is a tick-borne infection caused by an apicomplexan protozoon from the family Hepatozoidae. All species of genus Hepatozoon share a basic life cycle that includes asexual development and sporogony in a hematophagous invertebrate definitive host, and merogony followed by gamontogony in a vertebrate intermediate host. 4 Unlike most tick-borne protozoal pathogens that are transmitted via tick bites, Hepatozoon transmission takes place by ingestion of a tick containing Hepatozoon oocysts. Hepatozoon gamonts from dog isolates originating in different geographical areas are morphologically similar, and therefore it was believed, until 1997 that canine hepatozoonosis was caused by a single species. 16 Since then, research has led to the recognition of 2 distinct Hepatozoon species in dogs, H. americanum and H. canis, with very distinct clinical syndromes.2 Polymerase chain reaction (PCR) can now differentiate between the 2 distinct species based on the unique 18S ribosomal RNA (rRNA) gene sequence. 15 Although not always necessary to make a diagnosis, PCR can aid in cases that have an atypical presentation for Hepatozoon.
A 7-month-old mixed-breed intact female dog was presented to a private veterinarian with a 2 cm in diameter raised, pruritic, alopecic, subcutaneous, fluctuant swelling over the right eye. The mass had been present since adoption from a shelter in New Jersey, 3 weeks prior. No other abnormalities were found on clinical examination. Surgical removal was declined. Results of a complete blood cell count and serum chemistry panel were unremarkable with the exception of mild normocytic, normochromic anemia (hematocrit value: 33.7%, reference [ref.] interval: 36–60%), mild thrombocytosis (592 × 103/ml, ref. interval: 170–400 × 103/ml), hyperphosphatemia (10.4 mg/dl, ref. interval: 2.5–6.0 mg/dl), and mild increased activity of alkaline phosphatase (191 U/l, ref. interval 5–131 U/l).
Aspirate smears made from the lesion over the eye were stained with Wright–Giemsa. a The direct smears were highly cellular and mildly hemodilute. Smears contained many degenerate neutrophils, moderate numbers of eosinophils, moderate numbers of macrophages, rare mast cells, and few erythrocytes. Many intracellular and extracellular bacterial cocci were identified. Rare neutrophils contained an ellipsoidal structure measuring 11 µm × 4 µm that either displaced or overlaid the nucleus (Fig. 1). This structure was light blue with a variably distinct 3–4-µm round magenta-stained nucleus-like structure. Morphology was most compatible with a Hepatozoon gamont. Based on the geographical location (New Jersey) and the presence of gamonts in a tissue aspirate, a tentative diagnosis of H. americanum was made.
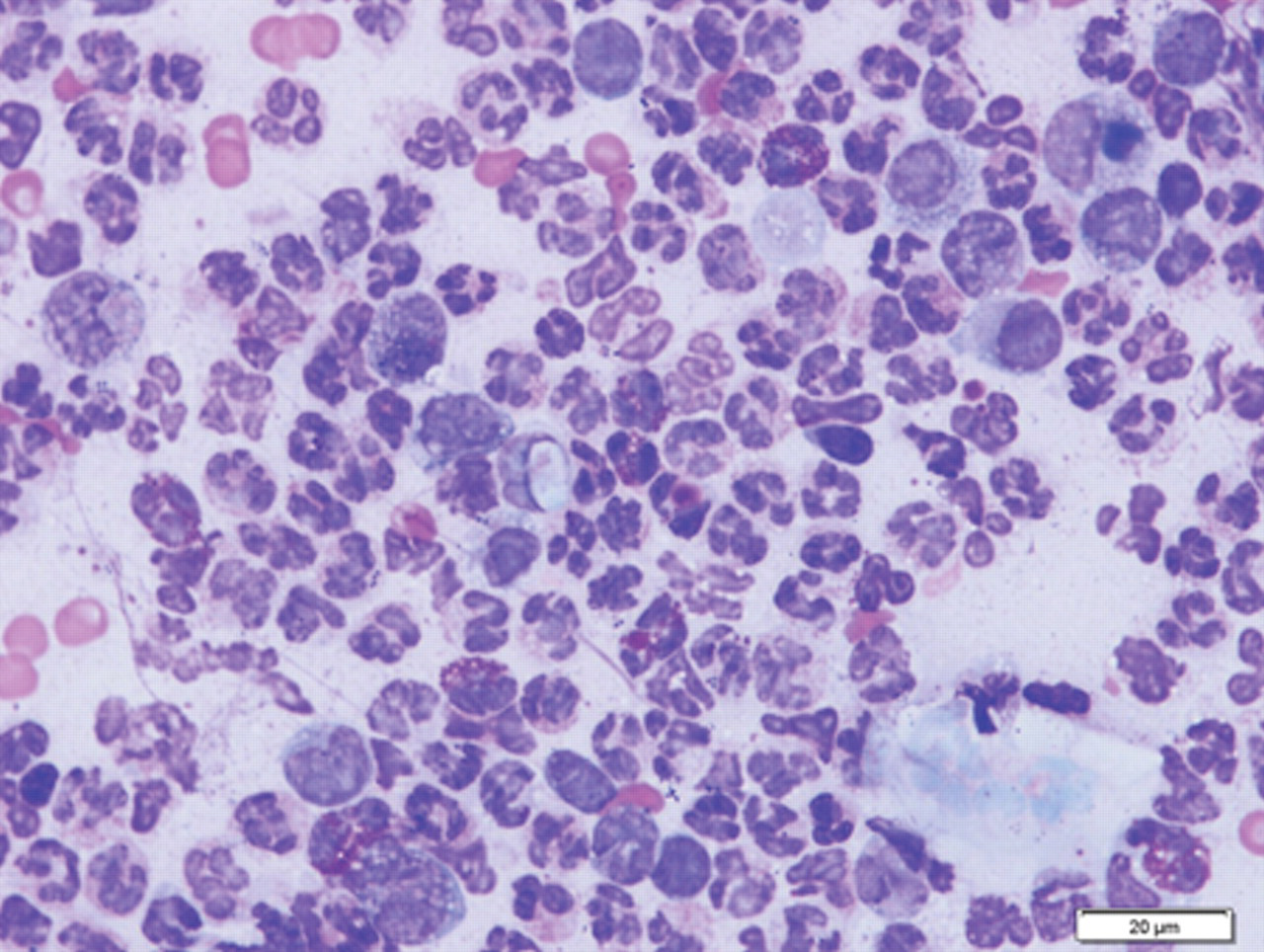
Fine-needle aspirate sample of the lesion over the eye; dog. Note neutrophilic and eosinophilic inflammation with many intracellular and extracellular cocci and a single intracellular gamont. Wright–Giemsa. Bar = 20 µm.
Lesion cultures were positive for moderate growth of Staphylococcus intermedius. Based on sensitivity testing, the initially prescribed 12.5 mg/kg of amoxicillin and clavulanic acid b was discontinued, and trimethoprim and sulfamethoxazole (15 mg/kg) and clindamycin hydrochloride drops c (10 mg/kg) were prescribed. The lesion had decreased in size by approximately 50% on amoxicillin and clavulanic acid b alone.
Upon receiving the cytology results, a peripheral blood sample was submitted to the Molecular Diagnostics Laboratory (Auburn University, Auburn, Alabama) for real-time PCR for the 18S rRNA gene of Hepatozoon. Copy numbers of the Hepatozoon spp. 18S rRNA gene were quantified, and the amplified sample was speciated via melting curve analysis. 15 Test results were a high positive, with 600 copies per µl of blood of H. canis.
An additional sample was then submitted to the Hebrew University School of Veterinary Medicine in Israel for additional genetic characterization. Polymerase chain reaction for the amplification of a fragment of the Hepatozoon 18S rRNA gene was carried out as previously described, 20 and the 352 base pairs amplicon was sequenced at the Alexander Silberman Institute of Life Sciences at the Hebrew University of Jerusalem using a commercial sequencing kit d and genetic analyzer. e Sequencing was performed from products of both the reverse and forward primers. Obtained sequences were evaluated with the ChromasPro software version 1.33 f and compared to sequence data available from GenBank using the BLAST 2.2.9 program (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). Analysis of the partial 18S rRNA gene sequence determined that the dog was infected with H. canis with 100% similarity to a H. canis sequence from Spain (GenBank AY150067). 8 The closest H. americanum GenBank accession (no. AF176836) had only a 90% similarity to the amplified sequence. 18
Hepatozoon canis was first described in the blood of dogs in India in 1905 (Christophers S: 1906, Leucocytozoon canis. Scientific Memoirs by the Officers of the Medical and Sanitary Departments of the Government of India. Office of the Superintendent of Government Printing, Calcutta, India. Available at: http://books.google.com/books?id=3jTlAAAAMAAJ&pg=PA23&dq=Scientific+Memoirs+by+the+Officers+of+the+Medical+and+Sanitary+Departments+of+the+Government+of+India+1907&hl=en&ei=OAAmTbuQDoT78Aak7_WwAQ&sa=X&oi=book_result&ct=result&resnum=1&ved=0CCMQ6AEwAA#v=onepage&q&f=false. Accessed on January 6, 2011) and is well documented in tropical, subtropical, and temperate regions all over the world. 10,11,13 The main vector of H. canis is the brown dog tick (Rhipicephalus sanguineus). 3 Although this tick is commonly found throughout the United States, H. canis was only recently identified in dogs in the southern United States. 1 In addition to ingestion of infected ticks, horizontal transmission through the uterus from the dam to its offspring has also been demonstrated in H. canis. 19 The majority of dogs infected with H. canis develop subclinical infections (less than 5% neutrophils infected) and only rarely does infection cause severe disease. The dogs that develop high parasitemia (90–100% of neutrophils infected) can exhibit pyrexia, lethargy, anemia, and emaciation. 6 High parasitemia can result in extreme neutrophilia with greater than 50,000 gamonts per µl of blood. Other clinical laboratory findings associated with H. canis include mild anemia, thrombocytopenia, hyperglobulinemia, and increased creatine kinase and alkaline phosphatase activities. 3
Hepatozoon americanum, a well-documented pathogen that causes American canine hepatozoonosis (ACH) in the southern United States, was first recognized in Texas, but the disease has since been reported in dogs from several other states, including Louisiana, Alabama, Georgia, Florida, Tennessee, and Oklahoma. 9 Dogs become infected with H. americanum when they ingest the vector tick, Amblyomma maculatum, containing oocysts. The infection is proposed to occur during grooming, ingestion of tick-infested prey, 10 or ingestion of cystozoites in rodent paratenic hosts. 14 Amblyomma maculatum is primarily found along the Gulf Coast, but has spread to Oklahoma and Kansas. 7 Hepatozoon americanum causes severe clinical signs such as high pyrexia, cachexia, depression, muscle atrophy, anemia, generalized pain, and weakness. These clinical signs have been attributed to merogonous cysts in skeletal muscle and bony proliferative lesions. 16 A marked leukocytosis ranging from 20,000 to 200,000 leukocytes/µl of blood is typically found with parasitemia not usually exceeding 0.1% of circulating leukocytes. 3 Unlike the subclinical course common to H. canis, ACH is often fatal if left untreated. Immunodeficient dogs, such as those with immature immune systems, congenital immunodeficiencies, and/or concurrent infections (toxoplasmosis, leishmaniasis, babesiosis, or ehrlichiosis), are more susceptible to infection with H. canis, whereas H. americanum can cause severe clinical signs in immunocompetent dogs. 3
The present case is unusual in that the diagnosis of H. canis was made on a tissue lesion from an otherwise asymptomatic dog, and thus emphasizes the importance of a patient’s travel history, since H. canis is not endemic to New Jersey. The complete sequence identity of the amplified 18S rRNA fragment from the dog’s blood with H. canis (GenBank accession no. AY150067) and the fact that it was distant from the corresponding fragment sequence of H. americanum (GenBank accession no. AF176836) confirmed the diagnosis of H. canis infection. Hepatozoon canis is found primarily in hemolymphatic tissues, whereas H. americanum infects mainly muscular tissues. 3 Before the advent of a species-specific PCR, the most common method of diagnosing H. canis infection was demonstration of gamonts in a blood smear. Less frequently, H. canis meronts can also be detected in histopathologic specimens or in cytologic preparations made from aspirates of impression smears of hemolymphatic tissues. Since H. americanum infects mainly muscular tissue, diagnosis typically relies on unique “onion skin” cyst lesions in muscle biopsies. 3 An enzyme-linked immunosorbent assay is available for detection of antibodies reactive with gamont (H. canis) or sporozoite antigens (H. americanum) in sera; however, it is mainly utilized for epidemiological studies. 12,17 In more recent studies in the United States, diagnosis of H. canis has been based on positive PCR results in asymptomatic dogs or those suspected of having H. americanum. In a study published in 2008 of Hepatozoon species in the United States, 1 2 out of 200 randomly sampled asymptomatic shelter dogs in Payne County, Oklahoma were positive for H. canis. In addition, a review of PCR results on 274 blood samples submitted to the Molecular Diagnostic Laboratory at Auburn University revealed H. americanum, H. canis, and a mixture of H. americanum and H. canis in 68 (24.8%), 2 (0.7%), and 7 (2.6%), respectively, of 77 canine blood samples found positive for Hepatozoon spp. 1 In another study, 15 examination of 614 ethylenediamine tetra-acetic acid blood samples from dogs with suspected hepatozoonosis identified H. americanum in 167 samples (27.2%), H. canis in 14 (2.3%), and a coinfection of the 2 species in 14 (2.3%) cases. Hepatozoon was not identified in the remaining 68.2% of cases. Although hepatozoonosis was thought to be only endemic in the southeastern United States, the aforementioned study also documented Hepatozoon spp. DNA in samples from California, Nebraska, Vermont, Virginia, and Washington State. These studies confirmed H. canis is present in North America and that coinfections with H. americanum do occur, despite different tick vectors.
Since the dog in the current report was otherwise healthy, it is presumed that gamonts were identified in the skin lesion due to extravasation of infected circulating neutrophils to the site of the bacterial infection. No gamonts were reported by a technician on routine review of the blood smear. Unfortunately, the peripheral blood smear was no longer available for pathologist review when requested. Since H. canis has not been documented to preferentially infect subcutaneous tissue, it seems unlikely that H. canis was the initiating cause of the lesion. Additionally, the lesion resolved with routine antibiotic treatment. Additional diagnostics (tick-borne disease titers or PCR, biopsy of the lesion) and treatment with imidocarb dipropionate were declined by the owners, due to the otherwise healthy status of the patient. At the time of publication, the patient was reported as healthy with no clinical signs of disease.
Based on the geographic location of this patient (New Jersey), further inquiries revealed that the puppy had been whelped in a Texas shelter and subsequently transported to a shelter in New Jersey for adoption services. The dam or littermates could not be located for additional diagnostics. Infection in this dog could have occurred via vertical transmission from the dam or ingestion of a tick, likely in the Texas shelter environment. The transportation timeline for this animal was not available, but it is unlikely infection occurred by ingestion of a tick in New Jersey since naturally occurring infections have not yet been confirmed in this state. In an experimental transmission study, H. canis gamonts appeared in the peripheral blood 28 days postinoculation. 5
The hyperphosphatemia and increased alkaline phosphatase were attributed to bone growth in the young dog in the present study, particularly in the absence of skeletal lesions. The mild normocytic–normochromic anemia was attributed to the age of the patient and perhaps anemia of inflammatory disease, despite the lack of an inflammatory leukogram. Thrombocytosis could be attributed to epinephrine-mediated splenic contraction, underlying inflammatory disease, and/or erythropoietin-induced thrombopoiesis due to a decreased erythropoietic state.
The current case is important in documenting an unusual presentation of infection with H. canis outside of the southern United States. Veterinarians and diagnosticians all over the United States should be aware of the unique differences between H. canis and H. americanum. The diagnosis of canine hepatozoonosis is typically accomplished by the identification of gamonts in blood smears or of “onion skin” cysts in muscle biopsies. 11 Although the clinical presentation and the aforementioned diagnostic techniques are usually adequate in differentiating between the 2 infections, PCR should be pursued if the clinical presentation is unusual. Dogs with a low H. canis parasitemia generally have a good prognosis if treated with the recommended protocols, but the prognosis for dogs with high parasitemia is guarded. 3,6 Treatment for H. americanum is more difficult since infected dogs are more debilitated at the time of diagnosis and because there is no effective therapy to eliminate the tissue stages of this organism. 4
Footnotes
Acknowledgements
The authors thank Dr. Onesios at the Hoboken Animal Hospital in Hoboken, NJ, for her clinical assistance and Dr. Susan Little, Department of Veterinary Pathobiology, Center for Veterinary Health Sciences, Oklahoma State University, Stillwater, OK, for her molecular expertise and advice.
a.
Caligor Inc., Greenville, PA.
b.
Clavamox®, Pfizer Inc., New York, NY.
c.
Antirobe Aquadrops®, Pfizer, New York, NY.
d.
BigDye® Terminator v3.1 Cycle Sequencing Kit, Applied Biosystems, Foster City, CA.
e.
ABI PRISM 3100 Genetic Analyzer, Applied Biosystems, Foster City, CA.
f.
© Technelysium Pty Ltd., Digital River Inc., Eden Prairie, MN.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
