Abstract
A freshwater angelfish (Pterophyllum scalare) hatchery experienced variable levels of emaciation, poor growth rates, swollen coelomic cavities, anorexia, listlessness, and increased mortality within their fish. Multiple chemotherapeutic trials had been attempted without success. In affected fish, large numbers of protozoa were identified both histologically and ultrastructurally associated with the gastric mucosa. The youngest cohort of parasitized fish was the most severely affected and demonstrated the greatest morbidity and mortality. The protozoa were morphologically most consistent with Cryptosporidium. All of the protozoan life stages were identified ultrastructurally and protozoal genomic DNA was isolated from parasitized tissue viscera and sequenced. Histological, ultrastructural, genetic, and phylogenetic analyses confirmed this protozoal organism to be a novel species of Cryptosporidium.
The apicomplexan protozoal parasite Cryptosporidium has been identified within numerous vertebrate hosts, including mammals, birds, reptiles, and fish. 5,11 Cryptosporidia have a life cycle that involves both asexual and sexual reproductive cycles, which is completed within an individual host. 6 Transmission from one host to another involves direct fecal-oral transmission often involving ingestion of oocyst-contaminated water. However, recent research indicates that transmission dynamics of cryptosporidia is often more complex than previously thought. 14 Oocysts are excreted in the feces. Once ingested, sporulated oocysts excyst within the gastrointestinal tract, releasing infectious sporozoites. The sporozoites attach to and invade epithelial cells lining the alimentary tract, where they become enclosed within a parasitophorous vacuole, which results in the trophozoite stage of the parasite. Trophozoites undergo asexual proliferation by merogony. Sexual reproduction occurs by gametogony to generate both macrogamonts (female) and microgamonts (male). 11 The resultant zygotes undergo sporogony, leading to the production of sporulated oocysts containing 4 sporozoites not enclosed within a sporocyst.
Cryptosporidiosis is recognized as a serious waterborne disease in humans and other animals for which few effective chemotherapy protocols are currently available. 6,14,16 Cryptosporidium typically infects neonatal or young animals, often resulting in dysfunction of the alimentary tract. 7 Relatively little is known about the prevalence and geographic distribution of Cryptosporidium isolates that infect fish. 13 Gastric and intestinal cryptosporidiosis have been previously identified in 14 species of marine and freshwater fish. 1,2,11,18 Recently, a parasite defined as Cryptosporidium molnari was isolated from 2 marine teleosts, the gilthead sea bream (Sparus aurata L.) and the European sea bass (Dicentrarchus labrax L.). The histologic and ultrastructural features of this organism have been described in detail. 2 Although no molecular characterization of C. molnari has been published thus far by the original research group, a C. molnari-like protozoal organism isolated from a guppy has been histologically and genetically characterized. 13 A recent review of the taxonomy of cryptosporidial parasites has been published. 5
A freshwater angelfish (Pterophyllum scalare) and discus (Symphysodon sp.) hatchery in Washington state (USA) experienced variable levels of emaciation, poor growth rates, coelomic distention, anorexia, spiraling, listlessness, and increased mortality in its fish population. The clinical signs were primarily confined to the larval angelfish, and the adult angelfish were relatively unaffected. Several feed-based and water bath-based pharmaceutical trials (levamisole, erythromycin, oxytetracycline, and metronidazole) were attempted without demonstrable success. During the peak outbreak, approximately 400 angelfish died in a cohort of 500 larval fish. The aquarium system was organized in a recirculation configuration, utilized chlorinated city water, and had a total system volume of 26,500 liters, with a flow rate of 490 liters per min and a daily water exchange rate of 755 liters. Water quality was maintained with a fluidized sand filter, bubble bead biofilter, protein skimmer (foam fractionater), ozone generator, and ultraviolet sterilizers. Angelfish eggs were spawned on slate strips in the breeding tanks and transferred to the hatchery, where they were treated with 3 to 5 ml of methylene blue per gallon of water. Free-swimming “larval” fish were transferred to another tank at 2–3 weeks of age.
Two separate shipments of moribund fish, approximately 3 weeks apart, were shipped live from the hatchery to the diagnostic laboratory. Affected fish included individually bagged cohorts of younger than 60 days, 150 days old, and older than one year. Upon arrival at the diagnostic laboratory, the fish were observed and humanely euthanized with a buffered solution of MS222 anesthetic, a dosed to affect. Each euthanized cohort was divided into 4 lots: frozen at −80°C, fixed with 10% buffered formalin, fixed with 2.5% gluteraldehyde, or submitted fresh for microbial culture of the visceral tissues. A clinically normal adult plecostomus fish (Hypostomus plecostomus) housed in the same tank as the affected angelfish for the purpose of algae control was humanely euthanized, as previously described. The plecostomus was necropsied, and the tissues were examined histologically.
The tissues were fixed in 10% buffered formalin and decalcified b for 1–4 hr before being embedded in paraffin. Longitudinal sections were routinely processed and stained with hematoxylin and eosin. All of the tissue sections were examined by veterinary pathologists (BGM, KRS, TFW). Gastric protozoa were measured from multiple individual fish and from different cohorts with an ocular micrometer at a magnification of 1,000x. For transmission electron microscopy, small pieces of gastric tissue (<1 mm 3 ) were fixed in 2.5% (v/v) glutaraldehyde and routinely processed for electron microscopy. Ultrathin sections were cut with a microtome and examined with a transmission electron microscope. c Black and white photographs were taken with film and scanned into a digital format.
Frozen fish viscera were minced, and DNA was extracted and purified from both frozen tissue and formalin-fixed, paraffin-embedded sections utilizing a DNA extraction kit. d Polymerase chain reaction was utilized to amplify a 695-nucleotide region of the cryptosporidial 18s ribosomal RNA (rRNA) gene using 18S forward primer (5′-AGTCATAGTCTTGTCTCAAAGATT-3′) and 18S reverse primer (5′-TTAACAAATCTAAGAATTTCACC-3′), as previously described. 9 Extracted sterile diethylpyrocarbonate-treated water was used as a negative control, and water was used as a no-template control for all polymerase chain reactions. The polymerase chain reaction amplicons were visualized on 1.5% agarose gels containing ethidium bromide and purified with the use of a DNA gel extraction kit. e The amplicon DNA was sequenced by a local vendor f using a commercial sequencing kit, g with analysis on a DNA sequencer. h Sequencing reactions were done in duplicate, and sequences were confirmed by the sequencing of both strands. Nucleotide sequences were aligned using the AlignX function of Vector NTIi. Align X calculations for generating the guide tree dendrogram are based on the Clustal W algorithm. 17 The nucleotide sequence of the 18s rRNA polymerase chain reaction product of the angelfish Cryptosporidium isolate has been deposited in GenBank under the accession number FJ769050.1.
Grossly evident visceral lesions were not apparent in any fish. Small numbers of aerobic bacterial organisms were isolated from the kidneys of approximately 2 fish per cohort, including Aeromonas hydrophilia, Pseudomonas putrefaciens, Sphingomonas paucimobilis, and Micrococcus spp. Severe intestinal capillariasis was diagnosed in a single large, moribund angelfish (older than 1 year).
Thirty angelfish were examined histologically in multiple, stepped sagittal sections. Large numbers of Cryptosporidium organisms were identified via light microscopy within the gastric mucosal epithelium and adjacent gastric lumina of multiple fish (Fig. 1). Myriad cryptosporidial organisms ranging from 1 to 4.1 μm in diameter were identified along the apical surface of the gastric epithelium. In severely affected individuals, more than 100 gastric Cryptosporidium organisms could be identified per 1,000x field (200-μm length of mucosal epithelium). Both sporulated and unsporulated oocysts were identified embedded deep within the cytoplasm of the gastric epithelial cells. The sporulated oocysts were nearly spherical and measured 3.4 μm + 0.4 μm (diameter, n = 64). Sporulated oocysts contained basophilic sporozoites. The oocysts occurred singly and in clusters of 4–10 within cytoplasmic vacuoles surrounded by zones of clearing in gastric epithelial cells.
In severely parasitized fish, the gastric lumen was filled with large numbers of desquamated, degenerate epithelial cells; macrophages; sporulated oocysts; and granular acellular debris. Scattered, parasitized gastric epithelial cells had pyknotic nuclei, cytoplasmic hypereosinophilia, and vacuolation (epithelial cell degeneration). The subjacent lamina propria was often multifocally infiltrated by moderate numbers of lymphocytes, macrophages, and eosinophilic granular cells (Fig. 1). Cryptosporidium organisms were not identified within the intestine, esophagus, or pharynx in any of the examined fish. A clinically normal plecostomus fish housed in the same tank as the affected angelfish had no histologic evidence of alimentary cryptosporidiosis.
All of the cryptosporidial life stages were identified ultrastructurally. 2 Extracytoplasmic trophozoites were located in apical epithelial cell-derived parasitophorous vacuoles with basilar feeder organelles (Fig. 2). Epithelial cell surfaces were covered by remnants of microvilli. Meronts containing either 4 (type II) or 8 (type I, Fig. 3) merozoites were also detected. Apical to luminal macrogamonts were distinguished from trophozoites by the numerous cytoplasmic amylopectin (polysaccharide) granules (Fig. 4). Microgamonts were rarely identified relative to the other developmental stages, as has been found previously with C. molnari. 2 Both sporulated and unsporulated oocysts were embedded deep within the epithelial cell cytoplasm, often occurred in clusters, and also contained cytoplasmic amylopectin granules (Fig. 5). Sporulated oocysts contained 4 sporozoites and a residual body filled with amylopectin granules. The sporulated oocysts were naked (lacked a sporocyst), as has been described previously for C. molnari-infected fish. 2
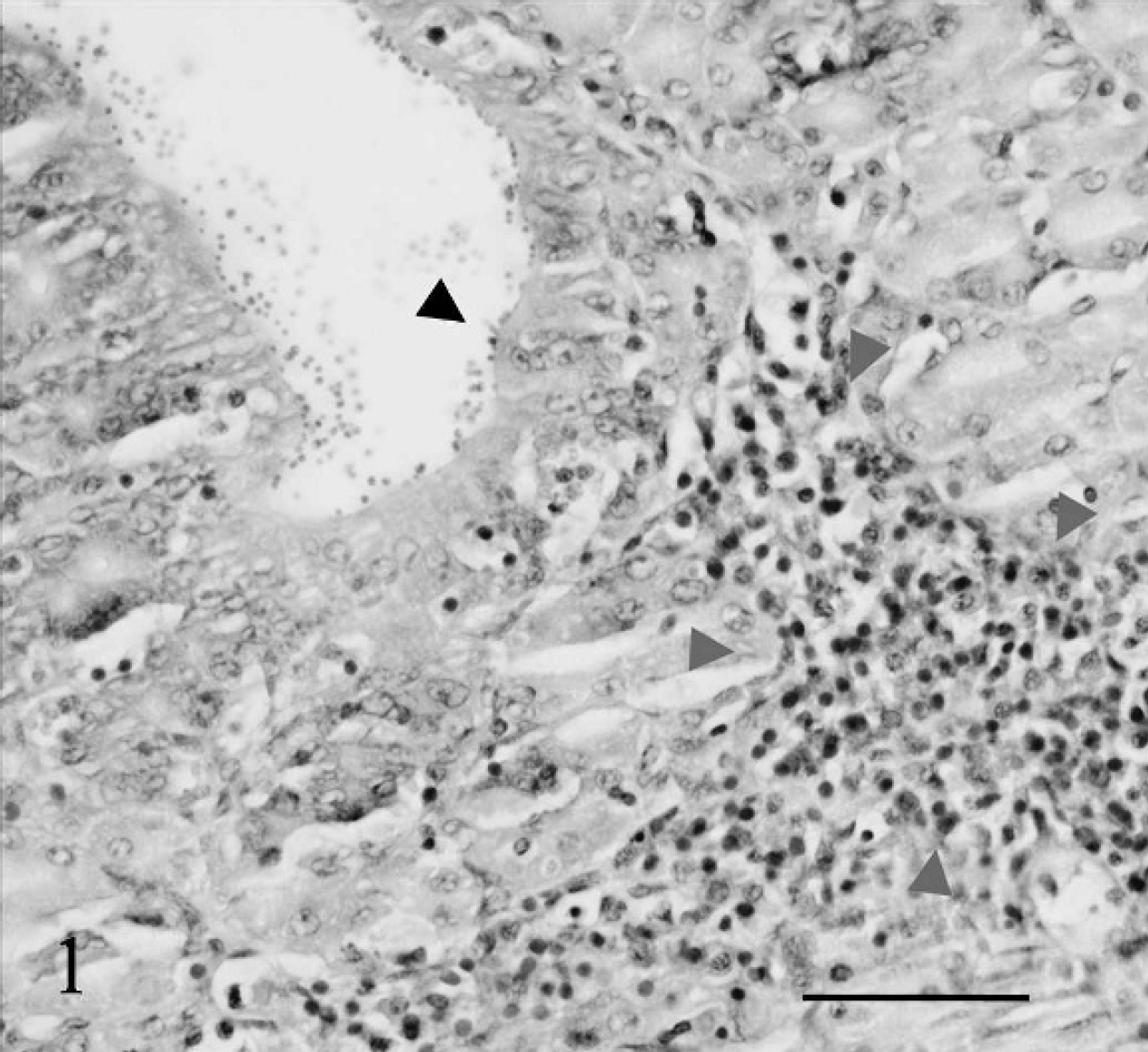
Angelfish gastric mucosa. Mucosal cryptosporidia (black arrowhead) and associated infiltration of the lamina propria by lymphocytes, macrophages, and eosinophilic granular cells (grey arrowheads). Light microscopy, hematoxylin and eosin-stained histologic sections, 600x magnification. Bar = 50 μm.

Angelfish gastric mucosa. Multiple sections of apical trophozoites within parasitophorous vacuoles. Note the feeder organelle (arrows) and remnant microvilli (arrowhead). Transmission electron micrograph, 6,000x magnification. Bar = 1 μm.

Angelfish gastric mucosa. Luminal type I meront containing 8 merozoites (a) adjacent to apical trophozoite (b). Transmission electron micrograph, 6,000x magnification. Bar = 1 μm.

Angelfish gastric mucosa. Multiple luminal macrogamonts (arrowheads) and numerous apical trophozoites. Transmission electron micrograph, 3,000x magnification. Bar = 2 μm.

Angelfish gastric mucosa. A cluster of sporulated (white arrowhead) and unsporulated (black arrowhead) oocysts embedded within the cytoplasm of a gastric epithelial cell. Transmission electron micrograph, 2,000x magnification. Bar = 3.5 μm.
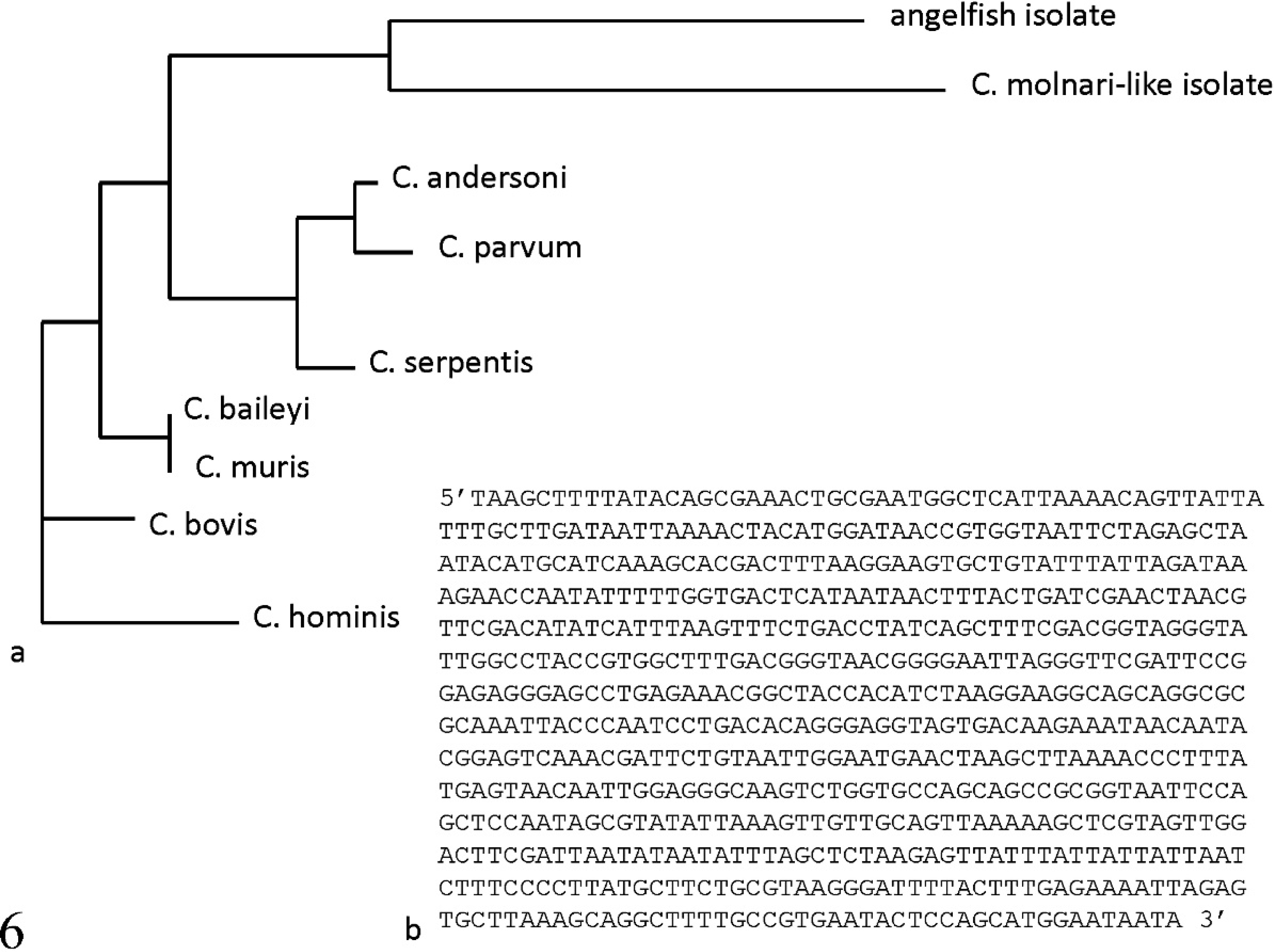
Phylogenetic dendrogram for sequences of 18s rRNA from various cryptosporidial isolates (
The 18s rRNA sequence was compared with published sequences for a C. molnari-like organism (GenBank accession no. AY524773) using Vector NTI software. i Phylogenetic analysis of the 18s rRNA locus for a variety of cryptosporidial species (C. molnari-like, C. serpentis, C. muris, C. andersoni, C. parvum, C. baileyi, C. bovis, and C. hominis) determined the angelfish isolate to be most closely related to, although distinct from, the C. molnari-like isolate (87% similarity; Fig. 6).
After the first description of C. muris in 1907, 8 multiple protozoa of this genus were named after the host in which they were found. 4 Recent morphologic, genetic, and cross-transmission studies have subsequently invalidated many of these species. At present, there is disagreement among taxonomists as to the precise number of species within the genus Cryptosporidium. To resolve these controversies, some taxonomists have argued the need for polyphasic typing (i.e., an integrated approach using standardized morphologic, biologic, and molecular methods [e.g., genetic methods] for describing Cryptosporidium species). 3 Currently, C. molnari is the only recognized species that parasitizes fish accepted as a valid cryptosporidial species. 14 Until more molecular and biologic data become available for cryptosporidia of piscine species, some researchers feel that there is insufficient data for establishing valid names for those cryptosporidia-like parasites in fish. 5
Many of the previously described Cryptosporidium infections in fish have not been associated with clinical signs of disease. 11 However, evidence of acute cellular injury has been associated with infections with C. molnari in fingerlings and juvenile marine fish 2 and Cryptosporidium-infected
cichlids from a lake in Israel. 12 Subacute to chronic cellular injury has been identified in a C. molnari-like infection in guppies. 13 Cryptosporidium molnari has been associated with mortalities in some stocks of cultured gilthead sea bream. More recently, coinfections with bacteria (Vibrio harveyi) and C. molnari resulted in greater histopathologic gastric damage relative to infections with C. molnari alone. 15 In the study described herein, small numbers of bacterial organisms were inconsistently isolated from the viscera of multiple fish. This finding, along with the lack of histologic lesions consistent with bacterial infection (e.g., necrosis, granulomas), suggests that bacterial coinfection did not play an important role in fish morbidity. In the present study, histopathologic gastric changes were most pronounced in the larval and juvenile angelfish. Clinically, this cohort of fish demonstrated the greatest morbidity and mortality.
Cryptosporidiosis has previously been described in plecostomus fish. 10 For this reason, a plecostomus fish from a tank with the affected juvenile fish was euthanized and examined both grossly and histologically. No evidence of alimentary cryptosporidiosis was identified in this animal. The mechanism of transmission between angelfish tanks was not determined but is likely to be the result of fecal-contaminated water passing through multiple tanks. The recirculation system lacked a system for in-line particulate filtration before the water reached the various ultraviolet sterilizers. As a result, the particulate size was most likely greater than the optimal 20–50 μm recommended for adequate ultraviolet exposure and kill rates for bacterial and protozoan pathogens.
No previously published report describing cryptosporidiosis in angelfish was identified in a search of the veterinary peer-reviewed literature. In addition, review articles defining piscine cryptosporidiosis do not reference any such cases. 2,11 Several features of the Cryptosporidium organisms described in the current study are consistent with the previously described C. molnari. As with C. molnari, these organisms are fish parasites, occur within the gastric mucosa, are associated with gastric epithelial cell degeneration, and have similar ultrastructural features. The feeder organelle is thought to facilitate the uptake of nutrients by the parasite from the host cell. 11 The presence of the feeder organelle is one of the key ultrastructural features that currently defines the genus and family. 18 In addition, the merogonial and gamogonial stages are located along the apical surface of the gastric epithelial cells (extracytoplasmic portion), whereas oogonial and sporogonial stages are deep within the gastric epithelium. 2 The size of the oocysts described in the present study (3.0–3.8 μm) is somewhat less than previously described for C. molnari. 2,13 It is possible that the oocyst measurements in the present study may not be accurate due to the effects of formalin fixation.
Although the research group originally describing C. molnari has yet to publish any gene sequences, a C. molnari-like organism isolated from a guppy has been sequenced. 13 Sequencing of the 18s rRNA gene is considered to be one of the best ways to discriminate between various cryptosporidial species (Aaron R. Jex, The University of Melbourne, Australia, personal communication, 2009). An 87% sequence similarity was identified in a comparison of the 18s rRNA sequences between the C. molnari-like parasite and the freshwater angelfish isolate described in the current study. A Basic Local Alignment Search Tool (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) search, as well as a guide tree dendrogram generated for 18s rRNA gene sequences from a variety of cryptosporidial species, indicate that the C. molnari-like isolate is most closely related to the angelfish isolate. An 87% similarity between the 2 isolates suggests that the angelfish isolate described in the present study is a new species of Cryptosporidium that shares many common morphologic and niche features with C. molnari. Whether this angelfish isolate warrants designation as a new species awaits sequencing results from cryptosporidial organisms recently described in other piscine hosts.
Acknowledgements. The authors thank Ruth Brown for her expert assistance with the ultrastructural components of this study. The authors also acknowledge and are grateful to Cary Swanson for submitting this interesting case.
Footnotes
a.
Tricaine methanesulfonate, Argent Chemical Laboratories, Redmond, WA.
b.
Decal Stat, Decal Chemical Corporation, Tallman, NY.
c.
Hitachi H-600, Hitachi Medical Systems, Twinsburg, OH.
d.
QIAamp, Qiagen, Valencia, CA.
e.
Freeze n'Squeeze Kit, Bio-Rad, Hercules, CA.
f.
Amplicon Express, Pullman, WA.
g.
Amersham DYEnamic ET Terminator Cycle Sequencing Kit, GE Healthcare, Stockholm, Sweden.
h.
ABI 373 DNA Sequencer, Applied Biosystems, Foster City, CA.
i.
Invitrogen, Carlsbad, CA.
