Abstract
A voided urine sample, obtained from a 13-year-old intact male dog residing in a laboratory animal research facility, was observed to contain biflagellate protozoa 5 days following an episode of gross hematuria. The protozoa were identified as belonging to the class Kinetoplastea on the basis of light microscopic observation of Wright–Giemsa-stained urine sediment in which the kinetoplast was observed basal to 2 anterior flagella. A polymerase chain reaction (PCR) assay using primers corresponding with conserved regions within the 18S ribosomal RNA gene of representative kinetoplastid species identified nucleotide sequences with 100% identity to Parabodo caudatus. Parabodo caudatus organisms were unable to be demonstrated cytologically or by means of PCR in samples collected from the dog’s environment. The dog had a history of 50 complete urinalyses performed over the 12-year period preceding detection of P. caudatus, and none of these were noted to contain protozoa. Moreover, the gross hematuria that was documented 5 days prior to detection of P. caudatus had never before been observed in this dog. Over the ensuing 2.5 years of the dog’s life, 16 additional complete urinalyses were performed, none of which revealed the presence of protozoa. Bodonids are commonly found in soil as well as in freshwater and marine environments. However, P. caudatus, in particular, has a 150-year-long, interesting, and largely unresolved history in people as either an inhabitant or contaminant of urine. This historical conundrum is revisited in the current description of P. caudatus as recovered from the urine of a dog.
Keywords
The current report describes the microscopic observation of bi-flagellate kinetoplastid protozoa in urine voided from a dog with hematuria and the molecular identification of these protozoa as Parabodo caudatus. A majority of the species in class Kinetoplastea are free-living heterotrophs that selectively graze on bacteria and are considered important members of the microbial food web.7,18,19,21 There are, however, entire genera and individual species within genera that have evolved as obligate parasites or endo-commensals.6,9–11,17,20 This obligate parasitism or commensalism is thought to have evolved independently multiple times within the kinetoplastids. 15 In particular, P. caudatus (formerly Bodo caudatus) has an interesting, and largely unresolved history in people as either a parasite or contaminant of urine.
A 13-year-old intact male Soft Coated Wheaten Terrier × Beagle mix dog, residing in a laboratory animal research facility, was observed by kennel personnel to void urine that was described as brown-green in color and containing blood. The dog appeared otherwise clinically healthy based on physical examination. On the same day as the abnormal appearing urine was observed, a urine sample was collected by cystocentesis for a complete urinalysis and bacterial culture at 37°C in 5% CO2 in thioglycollate broth a and on MacConkey agar and Columbia agar containing 5% sheep blood. b Urine appeared brown and opaque with a specific gravity of 1.026, pH of 8, and dipstick c abnormalities of 3+ protein, >3+ blood, and trace bilirubin. Light microscopic examination of the unstained urine sediment revealed 0–5 white blood cells, >500 red blood cells, and occasional epithelial cells per high power field. There was no bacterial growth after a 72-hr duration of culture. Ultrasound examination of the urinary tract revealed a normal to slightly thickened urinary bladder, chronic degenerative changes of both kidneys, and prostate enlargement consistent with the dog’s reproductive status and age. Results of a complete blood cell count and serum biochemistry profile were within reference limits. Gross hematuria resolved within 48 hr without treatment.
A follow-up urine sample was obtained from the dog 5 days following the episode of gross hematuria. The urine sample was voided by the dog on exiting its kennel and was immediately aspirated from the floor using a sterile syringe. The concrete floor was located immediately outside of the dog’s kennel and was described as clean and dry. Urine appeared yellow and cloudy and a urinalysis revealed specific gravity of 1.034, pH of 9, and dipstick abnormalities of 3+ protein, trace blood, and trace bilirubin. Light microscopic examination of the urine sediment revealed 0–5 white blood cells, rare red blood cells, occasional epithelial cells, many triple phosphate crystals, and 0–2 flagellated motile protozoal organisms per high-power field. Bacteria were not observed. On light microscopic examination of the unstained urine sediment, the organisms were further described as each possessing 2 anterior flagella. A 50-μl volume of the urine sediment was cytocentrifuged, fixed, and stained with Wright–Giemsa. Light microscopic examination of the stained specimen under high power oil immersion (1,000× magnification) revealed occasional protozoa averaging 4–5 × 7–8 μm in size. The protozoa were generally oval in shape with a single large oval nucleus, pale basophilic cytoplasm containing numerous variably sized vacuoles (presumed to be storage granules), and a round, densely basophilic kinetoplast located basal to 2 anterior flagella (Fig. 1).
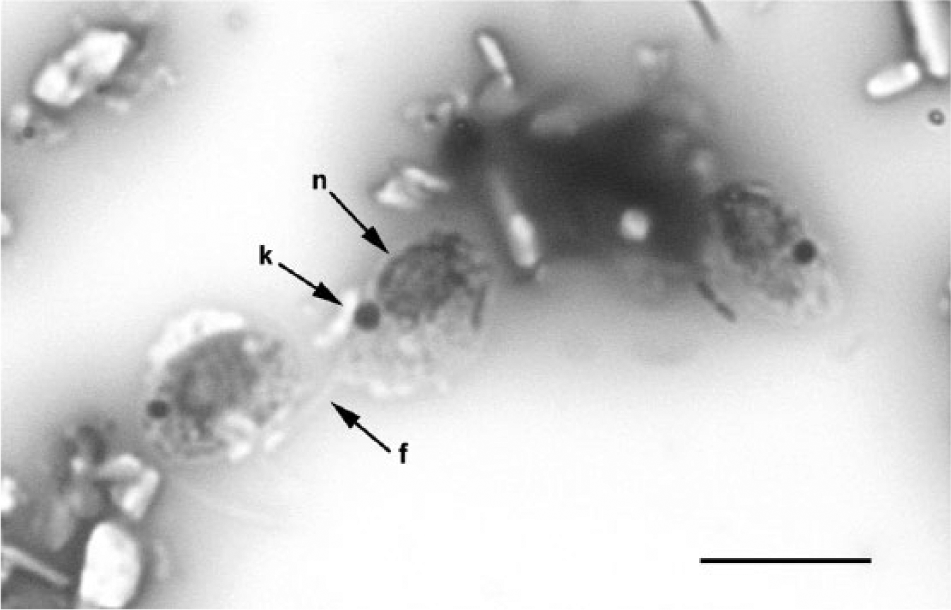
Cytocentrifuged urine specimen; dog. Protozoal organisms appear oval in shape with a single large oval nucleus (n, closed arrow), pale basophilic cytoplasm containing numerous variably sized vacuoles, and a round, densely basophilic kinetoplast (k, closed arrow). Flagella are difficult to discern in this image; however, 2 anterior flagella per organism could be visualized. The arrow (f) indicates 1 negatively staining refractile flagellum in the plane of focus of this image. Wright–Giemsa stain. Bar = approximately 10 μm.
In an effort to ascertain if the protozoa were contaminants, additional samples were collected from the dog’s environment. A sample of water from the sipper tube in the dog’s kennel, a sample of effluent trapped in the drain behind the dog’s kennel, and a sample of sterile saline that was introduced onto and then aspirated from the floor where the dog had previously voided were obtained. The samples were concentrated by centrifugation, stained with Wright–Giemsa, and examined by light microscopy for the presence of the protozoa; however, none were observed.
Based on observation of the kinetoplast, it was surmised that the protozoa were members of the class Kinetoplastea (http://www.marinespecies.org/aphia.php?p=taxdetails&id=562565). Members of the subclass Metakinetoplastina are divided into the exclusively parasitic order Trypanosomatida, which includes the Trypanosoma and Leishmania species, and the orders Neobodonida, Parabodonida, and Eubodonida, which includes a variety of parasitic, endocommensal, and free-living species.1,5,9,19 A bodonid was considered likely in the dog in the current study because trypanosomatids have a single flagellum and a small, compact kinetoplast, while the bodonids have 2 flagella.1,5 Based on this rationale, further studies were undertaken to identify the protozoa by means of polymerase chain reaction (PCR) using primers corresponding with conserved regions within the 18S ribosomal RNA (rRNA) gene of representative kinetoplastid species. 3 Accordingly, an approximately 300-μl aliquot of the dog’s urine was centrifuged at 35,060 RCF to concentrate the flagellates for DNA extraction using a commercial kit. d Primers KinSSUF1 (5′-GGTTGATTCTGCCAGTACTC-3′) and KinSSUseqR2 (5′-TCAACTACGAACCCTTTAAC-3′) were used in the PCR to amplify an approximate 650-bp product from the 5′-end of the 18S rRNA gene. The PCR amplification reaction contained 20 mM Tris–HCl (pH 8.4), 3 mM MgCl2, 50 mM KCl, 25 pmol of each primer, 2.5 mM of each deoxynucleoside triphosphate, 0.2 units of Taq polymerase, e and 1 µl of the urine sediment DNA extract (concentration not determined) in a total reaction volume of 50 μl. The PCR cycling conditions were 2 min at 95°C, 35 cycles of 30-sec denaturation at 95°C, 30-sec annealing at 60°C, and 30-sec extension at 72°C, and a final 5-min extension at 72°C. A 4-µl aliquot of each PCR reaction was examined for the presence of a specific amplification product by agarose gel electrophoresis (2% Tris–acetate/ethylenediamine tetra-acetic acid, 100 V) and ethidium bromide staining. The specific products from the PCR reactions were cloned into the commercial plasmid vector using a commercial cloning kit. f Plasmids were isolated and purified g and then sequenced using a commercial sequencer h and kit. i The DNA templates were sequenced in both directions using primers M13F (5′-GTAAAACGACGGCCAG-3′) and M13R (5′-CAGGAAACAGCTATGAC-3′). The partial 18S rRNA gene sequence was assembled using commercial software. j Comparison of the resulting DNA sequence with those in GenBank showed a 100% identity to the kinetoplastid protozoa P. caudatus (formerly Bodo caudatus; Dujardin, 1841). 12
Eighteen days after the demonstration of the kinetoplastid protozoa, voided and cystocentesis urine samples and a saline prepucial wash specimen were obtained from the dog for light microscopic examination of unstained and Wright–Geimsa-stained preparations. Cytological examination of these samples did not reveal the presence of protozoa. Attempts to PCR amplify the partial 18S rRNA gene sequence of P. caudatus from samples of urine sediment and drain effluent were also negative. Over the ensuing 2.5 years of the dog’s life, 16 additional complete urinalyses were performed; none of the urinalyses revealed the presence of protozoa.
Whether or not P. caudatus was actually present in the urinary tract of the dog in the current study prior to voiding or subsequently contaminated the urine sample after it was voided is unknown. The dog spent his entire life in the same laboratory animal research facility where he participated in a longitudinal study of breed-associated protein-losing enteropathy and nephropathy. As part of that study, the dog had exactly 50 complete urinalyses performed over a 12-year period preceding detection of P. caudatus. It is compelling that 48 of these urine samples were recorded as having been voided or collected off of the floor, and none of these were noted to contain protozoa. Moreover, the gross hematuria that was documented 5 days prior to detection of P. caudatus had never before been observed in this dog. The source of the P. caudatus found in the urine of this dog is unknown as the organisms could not be demonstrated in the dog’s environment, and all urine samples were analyzed on the same day as voiding.
The general opinion is that members of the genus Parabodo are free-living2,3 and occur commonly in air, water, and soil. The literature on P. caudatus is sparse, but remarkable for an association of this organism with urine. The P. caudatus isolate deposited in the American Type Culture Collection (ATCC 30906) was reportedly obtained from a human urine sample in Prague, Czechoslovakia in 1950 but the case history pertaining to this particular isolate can no longer be located (J. Kulda, personal communication, 2010). Whether or not P. caudatus is a true inhabitant of urine or merely a contaminant is a conundrum that can be traced back to the late 1800s. The earliest report of Bodo-like flagellates in urine was in 1859 when flagellates were observed in samples of urine from a number of different patients, some of whom were diagnosed with albuminuria. 8 The urine was described as alkaline or weakly acidic, and the flagellates appeared only after the urine had stood for several days. The organism was named Bodo urinarius. 8 In 1918, Bodo urinarius flagellates were reported in urine sediment collected on 2 different occasions from a Mexican sailor at the Royal Southern Hospital in Liverpool. 16 The urine specimens were 6 hr and 24 hr old, neutral to alkaline, and, in both cases, contained albumin and abundant casts. The organisms could not be demonstrated when urine was obtained under aseptic conditions. It was noted that the organisms die rapidly at temperatures of 37°C and require the presence of bacteria for successful cultivation. In 1920, a Bodo-like flagellate was obtained from human urine with the aid of a urinary catheter. 14
In all of these historical accounts, a compelling case can be made that the Bodo-like flagellates were likely a contaminant of urine. The most convincing case for P. caudatus being a true inhabitant of the urinary tract is the 1920 report in which a Bodo-like flagellate was documented repeatedly over a period of 5 years in urine samples that were drawn aseptically from the bladder of a Parsi clerk in India. 13 In contrast to the prior reports, the flagellates generally died within 8 hr after the urine was passed. The repeated demonstration in 1938 of a Bodo-like flagellate in the feces of a patient in Calcutta that thrived under culture conditions at 37°C has also been reported. 4 It remains unclear whether these historical reports of Bodo-like flagellates all represent descriptions of the same organism or encompass a complex of closely related cryptic species (i.e., one or more of which is parasitic and the rest free-living). Such variability would be consistent with the multiple transitions to obligate parasitism observed within closely related Kinetoplastea species.9,11
In the current report of kinetoplastid flagellates in urine from a dog, these organisms have been molecularly identified as P. caudatus. As with historic accounts of Bodo-like flagellates in the urine of people, whether this organism was an inhabitant or contaminant of urine from this dog remains unclear.
Footnotes
Acknowledgements
The authors thank Tonya Harris and the clinical pathology laboratory, College of Veterinary Medicine, North Carolina State University for excellent technical assistance, Dr. Amy B. Lehman Etingüe for help with German translation, and Dr. Jaroslav Kulda for unique historical insight on Bodo caudatus.
a.
Remel fluid thioglycollate medium, Thermo Fisher Scientific Inc., Waltham, MA.
b.
BBL, BD Biosciences, Sparks, MD.
c.
Chemstrip 10UA urine test strips, Roche Diagnostics Corp., Indianapolis, IN.
d.
Ultraclean soil DNA isolation kit, Mo Bio Laboratories, Solana Beach, CA.
e.
Invitrogen Platinum Taq DNA polymerase, Life Technologies, Grand Island, NY.
f.
Invitrogen Plasmid vector pCR2.1, TOPO TA cloning kit; Life Technologies, Carlsbad, CA.
g.
Wizard Plus minipreps DNA purification system, Promega Corp., Madison, WI.
h.
ABI 3730xl DNA sequencer, Life Technologies, Grand Island, NY.
i.
Applied Biosystems Deoxy Terminator cycle sequencing kit, Life Technologies, Grand Island, NY.
j.
Invitrogen Vector NTI Advance 11 program, Life Technologies, Grand Island, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
